Abstract
The current COVID-19 pandemic, characterized by a highly contagious severe acute respiratory syndrome, led us to look for medicinal plants as an alternative to obtain new drugs, especially those with immunomodulatory abilities, capable of acting against the pulmonary infection caused by coronavirus 2 (SARS-CoV-2). Despite medical advances with COVID-19 drugs and vaccines, plant-based compounds could provide an array of suitable candidates to test against this virus, or at the very least, to alleviate some symptoms. Therefore, this review explores some plants widely used in Peru that show immunomodulatory properties or, even more, contain phytoconstituents potentially useful to prevent or alleviate the COVID-19 infection. More interestingly, the present review highlights relevant information from those plants to support the development of new drugs to boost the immune system. We used three criteria to choose nine vegetal species, and a descriptive search was then conducted from 1978 to 2021 on different databases, using keywords focused on the immune system that included information such as pharmacological properties, phytochemical, botanical, ethnobotanical uses, and some clinical trials. From these literature data, our results displayed considerable immunomodulation activity along with anti-inflammatory, antiviral, antioxidant, and antitumoral activities. Noticeably, these pharmacological activities are related with a wide variety of bioactive phytoconstituents (mixtures or isolated compounds) which may be beneficial in modulating the overt inflammatory response in severe COVID-19. Further scientific research on the pharmacological activities and clinical utilization of these potential plants are warranted.
Graphical Abstract

Similar content being viewed by others
Introduction
Throughout human history, human beings have been subject to numerous infectious diseases, despite the existence of an immune defense system capable of fighting pathogenic agents, such as bacteria, viruses, fungi, and protozoa (Jantan et al. 2015; Babich et al. 2020). Since ancient times, human beings have learned to use natural resources (for example, plants), with the aim of alleviating symptoms or even curing the most varied ills (Hardy 2021). A healthy immune system is capable of responding to or facing any infection, maintaining a stable physiological internal environment (Jantan et al. 2015; Babich et al. 2020). The defense system comprises a complex network integrated by cells, tissues, organs, proteins, and soluble mediators, and is capable of protecting against foreign entities or counteracting the invasion of harmful native cells. The factors that trigger immunity include previous infection (bacterial or viral), immunization, and various external stimuli, with the immune system being capable of distinguishing between the self (own proteins/cells) and the non-self (foreign molecules or microbes from the environment). Thus, once the foreign agent is identified, an immune response is triggered to eliminate or neutralize it through a collective and coordinated response of specific cells and mediators (Jantan et al. 2015; Catanzaro et al. (2018). A brief classification is described in Fig. 1, consisting of the innate immune system (non-specific immune system) and the adaptive immune system (specific or acquired immune system). Both systems work together in a coordinated manner (Jantan et al. 2015; Catanzaro et al. 2018).
Innate immunity comprises physical and biochemical barriers alongside non-specific cells, such as phagocytic cells (monocytes, macrophages, and neutrophils), and granulocytes (basophils and eosinophils). The latter, being the most abundant, are capable of engulfing and destroying all kinds of pathogens. Meanwhile, adaptive immunity has memory cells when the body acquires immunity. Some examples are the lymphocytic cells, such as T cells, B cells, T helper (Th) cells, dendritic cells, and natural killer cells, which help to maintain the immune homeostasis of the human body (Catanzaro et al. 2018). The term immunomodulation arises from these considerations. It means changes in the immune system’s response by the action of agents capable of activating or suppressing the immune system, maintaining the homeostasis of the organism (Di Sotto et al. 2020). In this sense, immunomodulators are molecules of synthetic or biological origin that help to regulate these effects on the immune system. They are able to modulate, suppress, and stimulate the pathophysiological processes within the body (Jantan et al. 2015). According to how they influence the efficiency of the immune system, they can be categorized as immunosuppressants, immunostimulants, and immunoadjuvants (Fig. 1) (Catanzaro et al. 2018; Behl et al. 2021).
Literature and documentation of traditional uses have shown that the consumption of plants may have a modulatory role on the immune system. For instance, one of the best known immunostimulant species is Echinacea purpurea (L.) Moench, Asteraceae, along with other species of the same family. This came through millenary knowledge of folk medicine practiced by North American ethnic groups (Di Sotto et al. 2020). On the other side of the continent, the ancestral knowledge of the native communities of South America contributed to the pharmacopoeia with the bark of the Cinchona tree (Cinchona officinalis L., Rubiaceae), whose quinine metabolite inspired the development of hydroxychloroquine. Another example is coca (Erythroxylum coca Lam., Erythroxylaceae), whose metabolite cocaine led to the discovery of local anesthetics such as lidocaine (Lock et al. 2016). Few medicinal plants in Peru have been well-documented in phytochemical and pharmacological studies, while many others have only been the subject of ethnobotanical studies (Lock et al. 2016). Some of these plants have been commercialized globally and have a deep history of medicinal use in Peruvian communities. Unfortunately, many others have been neglected, or even ignored, by modern medicine and scientific communities, despite being used in popular medicine. Only in the last two decades have some medicinal plants been listed in the “Formulario Nacional de Recursos Naturales y Afines” (National Form of Natural and Related Resources) of the Peruvian Social Security System (ESSALUD, Peru) with the resolution no. 037-GCS-ESSALUD-2002, and later in another document called Floristic Catalog of Peruvian Medicinal Plants (2013) belonging to the National Institute of Health from Peru.
With this review, we aimed to highlight several herbal medicines from Peruvian communities as immunomodulatory agents in such a way that they can serve as a co-adjuvant treatment applied to integrative complementary medicine or to improve health-related quality of life. Similarly, this review highlights some medicinal plants widely used in Peru that can be explored for their therapeutic potential in the prevention or treatment of COVID-19, based on their immunomodulatory properties. With this aim, relevant information has been briefly summarized from ethnobotanical, phytochemical, and pharmacological approaches focused on immunological properties, along with anti-inflammatory and antioxidant properties strongly related to immune system regulation. Furthermore, cytotoxicity and antiviral properties have been included for their relevant importance in this pandemic situation. Medicinal plants traditionally used in Peru could be an alternAgrodataperuative source of phytomedicines to regulate the human immune system.
Search Strategy
Nine medicinal plants have been selected based on three criteria: (1) native plants with high demand in local and international markets. We included some plants from “The list of the category of medicinal plants and parfum-2015” on the agro-exportation website Agrodataperu (AGRODATA 2015); (2) deep-rooted traditional use in Peruvian culture, being well-known and widely used in traditional Peruvian medicine and/or cuisine (Lock et al. 2016); (3) background of scientific evidence, taking into account the keywords within the scope.
From 86 plants in the category of medicinal plants and parfum exported in 2015, non-native species were excluded and then followed the steps 2 and 3 mentioned above, to reach nine plants. These plants are presented in alphabetic order as follows: Chenopodium quinoa Willd., Amaranthaceae (quinoa); Chuquiraga spinosa Less., Asteraceae (huamanpinta); Croton lechleri Müll. Arg., Euphorbiaceae (dragon’s blood); Lepidium meyenii Walp., Brassicaceae (maca); Mauritia flexuosa L.f., Arecaceae (aguaje); Maytenus macrocarpa Briq., Celastraceae (chuchuhuasi); Plukenetia volubilis L., Euphorbiaceae (sacha inchi); Physalis peruviana L., Solanaceae (aguaymanto); Uncaria tomentosa DC., Rubiaceae (cat’s claw). The bibliographic survey started in August 2020, covering a wide time period from 1978 to 2021, utilizing databases such as PubMed, Embase, ScienceDirect, and Google Scholar, and using the following keywords: plant’s scientific or synonym names (according to The World Flora Online), plant’s popular name, immunomodulators, immunomodulatory, immunomodulation, immunostimulation, immunoadjuvants, immunosuppressors, immunosuppression, immune system, and/or immunity. Moreover, additional keywords were included to support this search, such as anti-inflammatory, antioxidant, cytotoxicity, and antiviral, due to their strong relationship with the immune function. The inclusion criteria limited the search to full text from research and review articles in English, Portuguese, and Spanish languages. Conversely, literature that was in neither the English nor Portuguese nor Spanish language was excluded, and dissertations, theses, and articles without accessibility to the complete text were also excluded.
Discussion
Immunomodulation and SARS-CoV-2
Could plants with immunomodulation activity be useful in the treatment of SARS-CoV-2 infection?
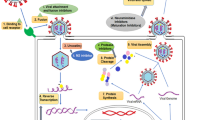
SARS-CoV-2 belongs to the order Nidovirales, family Coronaviridae, and genus Betacoronavirus. This infectious entity is an enveloped virus of spherical shape (80–160 nm in length) with a single-stranded RNA genome (27–32 kb in length) in the nucleus. Its virion surface comprises mainly proteins such as a spike protein, envelope protein, small membrane protein, and nucleocapsid protein (Bhuiyan et al. 2020; Pal et al. 2020). SARS-CoV-2 has reached all continents, affected more than 430 million people, and caused almost 6 million deaths worldwide (WHO 2020). The human–human transmission of this virus occurs through close contact by coughing, sneezing, and respiratory droplets. It is classified as an asymptomatic infection (patients with high nasopharyngeal viral levels); a mild illness with characteristics such as fever, dry cough, shortness of breath, systemic fatigue, dyspnea, anosmia, ageusia, and reduction in oxygen saturation <94%; a moderate illness with prostration, severe asthenia, fever >38 °C, or persistent cough; a severe illness with lung lesion >50%, respiratory rate ≥30 breaths/min, oxygen saturation ≤92% at a rest state, and aerial partial pressure of oxygen (PaO2)/inspired oxygen fraction (FiO2) ≥300; and a critical illness (Gandhi et al. 2020). Severely infected SARS-CoV-2 patients are associated with increased cytokine levels called a “cytokine storm,” such as interleukins IL-2, IL-4, IL-5, IL-6, and IL-10, tumor necrosis factor-α (TNF-α), ferritin, macrophage inflammatory protein-1a, and D-dimer, lymphopenia (CD4 + and CD8 + T cells), and decreased IFN-γ expression in CD4 + T cells, C-reactive protein, and erythrocyte sedimentation rate. This triggers a hyperinflammatory pathological state, which consequently leads to overwhelming systematic inflammation, exacerbating viral pathogenesis and causing sepsis, acute respiratory distress syndrome, and multi-organ dysfunction or failure (Bhuiyan et al. 2020; Dutta et al. 2020). With this in mind, there is the possibility of finding potential therapeutic targets against some of these symptoms in COVID-19 patients.
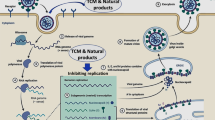
Palliative drugs and vaccines against COVID-19 have been developed (Babich et al. 2020; Barbosa and Nunes de Carvalho 2021). The UK National Health Service offers treatments to people with coronavirus (COVID-19) who are at the highest risk of becoming seriously ill. The treatments available are as follows: (1) antiviral synthetic medicines, such as nirmatrelvir and ritonavir (Paxlovid or Bexovid), remdesivir (Veklury), and molnupiravir (Lagevrio); and (2) a neutralizing monoclonal antibody, sotrovimab (Veklury). However, the behavior of SARS-CoV-2 is not fully known, but mutations in new strains have caused new symptoms and unexpected effects. Herbal medicines may provide effective treatments to alleviate this serious health situation, either as a main therapy or as a secondary therapy combined with other medicines (Catanzaro et al. 2018; Babich et al. 2020). Nowadays, immunomodulators based on medicinal plants, such as Echinacea purpurea (L.) Moench, Asteraceae, or Curcuma longa L., Zingiberaceae, have become an alternative medicine for integrative therapies such as complementary medicine, probably because they are mostly considered to have fewer side effects based on popular beliefs (Di Sotto et al. 2020). A good alternative would therefore be to look for new plant species as a therapy against immune system dysfunctions, as their chemical components can act on the immune system, affecting the functions of immune cells directly (adaptive and innate immunity) or indirectly by modulating the function of non-immune cells, reducing inflammation, scavenging free radicals, and influencing the secretion of cytokines or modulating angiogenesis. We cannot rule out the possibility of direct antimicrobial and antiviral action from these chemical compounds (Babich et al. 2020; Behl et al. 2021).
Natural immunomodulators have a wide range of chemical structures and can act directly or indirectly in modulating the immune response. Plants have been demonstrated to be a rich source of immunomodulating agents such as alkaloids, terpenoids, polyphenols (e.g., phenolic acids and flavonoids), and sesquiterpene lactones. They also present immunomodulator agents originating from a plant’s primary metabolism, such as polysaccharides, glycoproteins and lectins, fatty acids, and other organic compounds (e.g., aldehydes, and primary/secondary alcohols) (Alhazmi et al. 2021; Barbosa and Nunes de Carvalho 2021; Behl et al. 2021).
Promising Plants
A total of 116 papers were included for the nine selected plants, summarizing botanical, ethnomedicinal, phytochemical, pharmacological, and clinical applications. Information on the immunomodulatory effects and their anti-inflammatory, antioxidant, cytotoxic, and antiviral activities were extracted from the full-text articles included. These are presented descriptively in table 1S.
Chenopodium quinoa
Chenopodium quinoa Willd., Amaranthaceae (popular name: “quinoa”), is considered a pseudo-cereal and has been cultivated as a food for centuries by native communities from the Andean highland (Peru, Bolivia, Ecuador, Chile, Argentina, and Colombia). Quinoa seed has been consumed in a similar way to rice, in soup, in breakfast cereals, or ground into flour to produce other food alternatives. Interestingly, fermented quinoa seeds are also used in alcoholic beverages for traditional ceremonies. Finally, quinoa leaves are eaten like spinach (Graf et al. 2015).
The plant usually reaches 1–2 m in height, branching with large panicles, and produces large to small flat grains, which are oval and usually pale-yellow in color, but can vary from pink to black, depending on the variety. Its main characteristic, compared with other grains, is its ability to grow in the most adverse climatic conditions (great flexibility) such as extreme temperatures (~4 to 38 °C), frost, very low rainfall, nutrient-poor soils with pH ranging from 6 to 8.5, and high salinity (40 mS/cm) (mainly Peru and Bolivia) (Graf et al. 2015); consequently, its cultivation has recently expanded to other countries, such as Canada, US, Australia, China, India, and England, among others (Pereira et al. 2019). According to the Food and Agriculture Organization (FAO 2022), quinoa is considered to be a promising plant for humanity.
Chemically, quinoa seeds contain carbohydrates (59–74%), constituted mainly by polysaccharides. Experimental studies have extracted homo- and heteropolysaccharide fractions (codified fractions such as CQP, QWP, QAP, and QPS1) with unit sugars such as d-mannose, d-xylose, rhamnose, maltose, arabinose, fructose, and glycose (Yao et al. 2014a; Graf et al. 2015; Hu et al. 2017; Fan et al. 2019); proteins (10–18%) were constituted by globulins (37% constituted by chenopodin, a globulin 11S-type protein), albumins (35%, a 2S-type protein), and traces of prolamins (0.5–7.0%) (Verza et al. 2012; Zevallos et al. 2012; Yao et al. 2014b; Capraro et al. 2020); lipids (4–10%); minerals (3–4%); vitamins (E, B group, and C); and carotenoids (lutein, zeaxanthin, and neochrome) (Graf et al. 2015).
The molecular richness coming from primary metabolism could be responsible for their pharmacological properties in the immune system. Nevertheless, the secondary metabolites identified in C. quinoa could also be responsible for its ascribed immunological properties. Quinoa triterpenoids-saponins with a tetracyclic or pentacyclic core are the most studied metabolites (Lin et al. 2019). El Hazzam et al. (2020) gathered information about quinoa seed saponins, highlighting oleanolic acid derivatives, such as hederagenin acid, spergulagenic acid, serjanic acid, phytolaccagenic acid, gypsogenin acid, and 3β-hydroxy-27-oxo-olean-12-en-28-oic acid. Other molecules, such as tocopherols, tocotrienols, steroids (e.g., ∆7-stigmastenol (51.3%), sitosterol (27.2%), and ∆7-avenasterol (8.7%)), polyphenols (e.g., benzoic acid derivatives such as gallic acid), cinnamic acid derivatives (predominantly ferulic acid and derivatives, as well as vanillic acid), and flavonoids (mainly kaempferol and quercetin and derivatives, as well as acacetin, myricetin, and daidzein) were also the focus in experimental studies (Gómez-Caravaca et al. 2011; Navruz-Varli and Sanlier 2016; Lin et al. 2019; El Hazzam et al. 2020).
The nutritional and pharmacological properties of quinoa have also been reported in research and review papers. Although numerous studies on vegetal species are focused on secondary metabolites, the primary metabolites of the quinoa seed reveal strongly pharmacological properties related to the immune system, anti-inflammatory, and inclusive, cytotoxic properties, which are the mainly target in the polysaccharide and protein fractions of some research (Verza et al. 2012; Zevallos et al. 2012; Yao et al. 2014a; Hu et al. 2017; Fan et al. 2019; Capraro et al. 2020). For instance, an early study by Zevallos et al. (2012) evaluated a protein fraction rich in prolamin on patients with celiac problems (an ex vitro method using an organ culture of a celiac duodenal biopsy sample) and showed not only its safety but also activation of the innate immune response, stimulating T cell lines and the secretion of some cytokines (IFN-γ and IL-15). Two years later, Yao et al. (2014a) observed an increasing production of IL-6, TNF-α, and nitric oxide (NO) in a dose-dependent manner (max. concentration at 200 μg/ml) using water-extractable polysaccharide fractions from quinoa. Likewise, a new quinoa polysaccharide promoted the proliferation of RAW264.7 macrophages, while suppressing NO production in a dose- and time-dependent manner (Hu et al. 2017). This study was corroborated by Fan et al. (2019), wherein quinoa crude polysaccharides (QPS1) successfully improved the levels of IFN-γ, IL-6, IFN-ɑ, IgM, and lysozymes in serum, enhancing the phagocytic function of mononuclear macrophages and reducing the allergic reaction in mice (Fan et al. 2019). Recently, Capraro et al. (2020) described the potential immunomodulation capacity and anti-inflammatory effects of fractions rich in chenopodin, the major protein of quinoa seeds. Chenopodin may exert biological effects on intestinal cell models, activating the canonical nuclear factor kappa B (NF-ƘB) signaling pathway and decreasing IL-8 expression.
Facing these findings, molecules belonging to secondary metabolism were also studied and ascribed immunomodulatory and anti-inflammatory properties (Lin et al. 2019). In quinoa, for example, Verza et al. (2012) observed a significant enhancement of humoral and cellular immune responses to ovalbumin in mice, promoted by two quinoa saponin fractions, a mixture of hederagenin (1), phytolaccagenic acid (2), serjanic acid (3), and oleanolic acid (4) glycosylated saponin derivatives. A decrease in the production of inflammatory mediators such as NO, TNF-α, and IL-6 was observed in lipopolysaccharide-induced RAW264.7 cells using other fractions of quinoa saponin at different concentrations (Yao et al. 2014b). Moreover, Lozano et al. (2013) reported significant anti-inflammatory effects for extracts of saponin and its isolated compounds, such as oleanolic acid, methyl oleanate (5), hederagenin, and phytolaccagenic acid, in ear and paw edema assays. This anti-inflammatory action could be complemented with phytosterols present in quinoa seeds, such as β-sitosterol (Lin et al. 2019). On the other hand, studies with polyphenols, e.g., ferulic acid (6), ferulic acid 4-O-glucoside (7), isoferulic acid (8), kaempferol (9), kaempferol 3-O-glucoside (10), and kaempferol 3,7-di-O-α-l-rhamnoside (11), are occasionally mentioned. The review article by Lin et al. (2019) highlighted some polyphenols in quinoa seeds and leaves that showed several pharmacological properties, including antioxidant and anti-inflammatory activity.


Chuquiraga spinosa
Chuquiraga spinosa Less., Asteraceae (popular name: “huamanpinta”), is an Andean evergreen shrub whose growth is mostly restricted to areas with elevations of ~2500–4000 m (Peru, Bolivia, and Argentina). Its branched stems are semi-woody at the base and herbaceous in the aerial parts. The shrub can reach up to 1.5 m in height and shows sessile small leaves with axial hooks, and inflorescence like a calix, with numerous flowers of yellow-orange color. Infusions and decoctions of the stems and leaves have been widely used in folk medicine by Andean communities, basically for inflammation and infection of the genitourinary regions, problems related to the reproductive system, and respiratory diseases (Sotelo-Córdova 1998; Casado et al. 2011). Other plants, e.g., Bixa orellana L., Bixaceae, and Plantago major L., Plantaginaceae, are also reported to be added to C. spinosa infusion preparations (Perez-Chauca et al. 2020).
Despite its presence in national and international markets, and in addition to being included in the list of medicinal plants of the Formulario Nacional de Recursos Naturales y Afines (ESSALUD, Lima, Peru), phytochemical and pharmacological studies are scarce. The first studies were done by Casado et al. (2011) and Senatore (1996, 1999), and glycosylated flavonoids (quercetin-3-O-rutinoside, kaempferol-3-O-rutinoside, and kaempferol-3-O-glucoside), in addition to an acetophenone (p-hydroxyacetophenone), were isolated from hydroalcoholic extracts of the aerial parts (Senatore 1999). Years later, other flavonoids (e.g., kaempferol, quercetin, isorhamnetin, and some derivatives) were reported (Landa et al. 2009). Furthermore, seventy compounds, mainly carbonyl compounds, such as p-methoxyacetophenone and p-hydroxyacetophenone, were characterized from the essential oil of C. spinosa leaves, which include sesquiterpenes (β-humulene, ar-curcumene, cuparene, and spathulenol), monoterpenes, and phenylpropanoids (apiol), among others (Senatore 1996).
Only one study related to immunomodulation has been reported for this medicinal plant, in which the anti-inflammatory and antioxidant properties were related to flavonoids, such as 5,6,7-trihydroxy-4′-methoxyflavone (12), 3′,5,6,7-tetrahydroxy-4′-methoxyflavanone (13), 4′,5,7,8-tetrahydroxyflavone (14), and 5,7,8-trihydroxy-4′-methoxyflavone (15) (Ramírez et al. 2014). The administration of a chloroform extract of C. spinosa leaves to rats, at 200 mg/kg (48.23%) and 300 mg/kg (46.76%), resulted in an increase of the phagocytic activity of the macrophages, which was comparable to the positive control isoprinosine (59.9%). The anti-inflammatory activity of hydroalcoholic extracts was evaluated in paw edema induced by carrageenan, with results being comparable to ibuprofen (Ramírez et al. 2014). The results were corroborated by Casado et al. (2011) and Sotelo-Córdova (1998) using hydroalcoholic extracts from aerial parts, with indomethacin as the positive control. Both authors suggested that flavonoids and acetophenones, mainly p-hydroxyacetophenone (16), should be responsible for the anti-inflammatory action. Additionally, the hydroethanolic extract from aerial parts showed potent antioxidant activity and cytotoxicity properties with IC50 values ranging from 5 to 10 μg/ml, suggesting that kaempferol (10), quercetin (17), and rutin (18) glycosides could be the bioactive compounds (Herrera-Calderon et al. 2017).

Croton lechleri
Croton lechleri Müll.Arg., Euphorbiaceae (popular name: “sangre de grado”), is an Amazonian tree that grows up to 20 m in height. Its trunk has a diameter of 30 cm and shows a white or gray bark that exudes clear red and viscous sap when lacerated (Jones 2003; Rossi et al. 2011). This species occurs mainly in the Amazonian basin (Peru and Ecuador). Croton lechleri has been a well-known medicinal plant for centuries, reputed for its red latex-bearing sap. Products based on C. lechleri extracts are widely used by Amazonian communities, being included in the list of medicinal plants of ESSALUD and have been exported in recent decades as raw materials (Lock et al. 2016). Its ethnomedicinal uses include healing wounds, and inflammatory and septic processes (mainly skin conditions). This plant is also used to treat gastrointestinal ulcers, pyorrhea, menstrual cramps, fevers of digestive causes, and bleeding after childbirth; information collected through community use indicates that ca. 8 drops are administrated in almost all folk medicine uses, although this dose could reach 20–30 drops in an infusion combined with other aromatic plants (Jones 2003; Rossi et al. 2011; Lock et al. 2016). The sap obtained from the stem bark of C. lechleri has received great attention from scientists due to its use in folk medicine. Cai et al. (1991) suggested that abundant polyphenols could be participating in the regulation of the immune system. These polyphenols are mainly composed of proanthocyanidins (oligomerics and polymerics of flavan-3-ols), for instance, SP-303 (19), a proanthocyanidin heterogeneous oligomer. These chemical constituents are predominance in aqueous fractions from sap’s bark (Cai et al. 1991). Meanwhile, other molecules, such as catechin/epicatechin (20 and 21) or gallocatechin/epigallocatechin (22 and 23), were identified in low quantities (Cai et al. 1991). Two years later, the same research group identified clerodane-type diterpenes, such as korberin A (24) and B (25), from the bark (Cai et al. 1993). These and other chemical groups, such as flavonols (quercitrin, 26), lignans (3’,4-O-dimethylcedrusin from the sap, 27), and alkaloids such as sinoacutine (28) and glaucine (29) in the leaves, and taspine (30) (>1% dry wt. from sap) were highlighted by Jones (2003) in a review article. Meanwhile, other researchers also found 74 substances in the essential oil of C. lechleri (Rossi et al. 2011).

The chemical peculiarity of this plant led us to suppose that it could be a source of chemical agents capable of acting on multiple pharmacological targets in interaction with the immune system. However, only one immunological study has been performed (Risco et al. 2003), where it was demonstrated the stimulation of phagocytosis in human monocytes by the bark’s sap at 5–20 µg/ml and observed the inhibition of phagocytosis activity in human blood monocytes at a higher concentration (50 µg/ml). The immunomodulatory activity of the plant extract was attributed to catechins (monomeric flavan-3-ols) and proanthocyanidins (19–23). Recently, Shakoor et al. (2021) suggested that polyphenols such as catechins (monomeric flavan-3-ols) and proanthocyanidins could be directly or indirectly involved in the stimulation of the immune response. These molecules would also be strongly related to antioxidant effects, compared with quercetin and ascorbic acid (De Marino et al. 2008; Risco et al. 2003). In addition, proanthocyanidin fractions revealed a significant inhibitory activity against DNA and RNA viruses, for example, respiratory syncytial virus, influenza A virus, parainfluenza virus, herpesvirus types 1 and 2, and hepatitis A and B virus (Barnard et al. 1993; Ubillas et al. 1994).
This plant also showed a potent anti-inflammatory effect that was attributed to alkaloids, especially to taspine (30). This alkaloid showed significant activity at 20 mg/kg/day when compared with the positive control (indomethacin; 1 mg/kg/day) in a carrageenan-induced edema assay (Perdue et al. 1979). Meanwhile, the sap from C. lechleri at 5 mg/kg i.p. exhibited potent activity during the first 4 h, in comparison with naproxen at 20 mg/kg i.p. (Risco et al. 2003). 3′,4-O-dimethylcedrusin (27) was related to healing, due to its stimulatory actions in wound repair (De Marino et al. 2008). These investigations clearly indicate its therapeutic potential against post-COVID-19 symptoms.
Lepidium meyenii
Lepidium meyenii Walp., Brassicaceae (popular name: “maca”), is an herbaceous biennial plant with an edible underground storage organ (root) that is ca. 20 cm in circumference and shows different shapes (spherical or oval) and colors (yellow, purple, or cream). The aerial plant (including leaves, stems, and inflorescence) can reach a height of 10–20 cm (Wang and Zhu 2019). This plant occurs in non-forested area between 3700 and 4500 m above sea level, mainly in the Peruvian and Bolivian Central Andes. The favorable climatic conditions for this plant are characterized by strong winds, high UV radiation, low oxygen levels, and low temperature (~10 °C) (Lock et al. 2016). However, this species has been adapted in other parts of the world where it is cultivated (Wang and Zhu 2019).
According to Peruvian history, people of the Andean highlands have an ancient tradition of growing maca in large areas, among other foods. Its harvest was widely spread throughout the Inca Empire (10,000 years ago). Since then, its energizing power has been recognized to improve mental capacity, as well as strengthening the immune system. Its consumables are mainly sweet and flavored hypocotyls (root type). Typically, they are crushed and then mixed with milk until they reach a porridge-like consistency (Lock et al. 2016). This is reflected in publications related to the rich content of nutritional constituents: carbohydrate (59–73%), starch (37–77%), protein (10–18%), total dietary fiber (15–26%), and lipids (0.59–2.2%), in addition to the mineral content (Wang and Zhu 2019).
Recent studies reported that molecules from the primary metabolism of L. meyenii are a pivotal part of some pharmacological properties. Polysaccharide fractions from maca leaf (LMPL, 58.43 kDa) (Li et al. 2017), root (MC-1, 11.3 kDa; MC-2, 9.83 kDa, LPM-1) (Zhang et al. 2017, 2016; Zha et al. 2018), and MP21 (Unknown) (Wang et al. 2016; Li et al. 2017) possess immunomodulatory activity, increasing phagocytosis and the release of NO and cytokines, such as IL-6, IL-10, and TNF-α, in a dose-dependent manner. On the other hand, the MPI protein (maca protein isolate, 22 kDa) obtained from the root caused a significant increase in the levels of TNF-α, IL-6, and, NO in macrophage cells (RAW264.7) at 62.5 μg/ml (Wu et al. 2019). Furthermore, Fei et al. (2020) suggested that oral doses (0.75–1.5 mg/kg) of the aqueous extract of maca root administered in mice enhanced cell-mediated and humoral immune responses by increasing the secretion of T1 and decreasing T2 cytokines. In addition, they improved the proliferation and transformation of splenic T lymphocytes. These in vitro and in vivo immunomodulatory studies were corroborated in a clinical trial, when Ren (2019) demonstrated that the consumption of dry tablets of maca (250 mg/capsule) for 2 weeks in boxing athletes can effectively improve their immune function by raising the level of white blood cells and the immunoglobulins A, M, and G levels.
In the context of secondary metabolites, glucosinolates, e.g., benzylglucosinolate (31) and m-methoxybenzylglucosinolate (32), are the most abundant chemical group in the root. Interestingly, they possess strong antioxidant activity, as summarized in the review article by Carvalho and Ribeiro (2019). In addition, flavonoids, mainly catechins and its epimers (20–23), have been identified in L. meyenii extracts; these molecules have also been involved in antioxidant activity (Carvalho and Ribeiro 2019). Studies have also shown that lignans such as ( +)-epi-pinoresinol (33) exert anti-inflammatory activity on lipopolysaccharide (LPS)-stimulated RAW264.7 cells by inhibiting IL-6 production in a dose-dependent manner (Zhou et al. 2021), and some macamides, as N-benzyl-9-oxo-10E,12E-octadecadienamide (34) and N-benzyl-9-oxo-10E,12Z-octadecadienamide (35), exhibited relevant cytotoxic activity against the tumor cell line HT-29 (Carvalho and Ribeiro 2019). Similar pharmacological action was shown by alkaloids such as lepidiline A (36) and lepidiline B (37) against human pancreatic adenocarcinoma (PACA2), human bladder carcinoma (UMUC3), and human ovarian carcinoma (FDIGROV). Furthermore, Mendoza et al. (2014) reported that methanol extracts from the root demonstrated inhibitory effects on Flu-A (IC50 5.40 μg/ml) and Flu-B (IC50 7.69 μg/ml) virus replication, with a selectivity index (SI) of 157.4 and 110.5, respectively. It is important to note that the traditional use of L. meyenii encompasses many other relevant biological activities, including sexual performance enhancement, immunomodulatory properties, the reduction of osteoporosis, ultraviolet radiation protection, and hepatoprotective effects, where other chemical groups could be involved (phytosterols, saponins, and phenolic acids) (Carvalho and Ribeiro 2019; Wang and Zhu 2019). Like other plants, maca root has been shown to have multiple pharmacological activities, such as anti-inflammatory, anti-fatigue, improving reproductive health, antioxidant, neuroprotective, hepatoprotective, and antiviral, some of which suggest that this plant is promising for the treatment of COVID-19 (Mendoza et al. 2014; Wang and Zhu 2019).

Mauritia flexuosa
Mauritia flexuosa L.f., Arecaceae (popular name: “aguaje”), is a native palm tree occurring in the lowlands of the Amazon biome, being considered one of the most abundant palm trees. In Brazil, it is known as buriti, while the local name in Peru is aguaje. This palm can have 30–40 m height, with a diameter between 30 and 60 cm, and large composite leaves. The edible and highly nutritious fruits are oval drupoids exhibiting a yellow-orange to dark reddish color, measuring about 5–7 cm in length and 4–5 cm in width, and weighing 40–85 g (Pacheco-Santos 2005; Koolen et al. 2018). This species has been domesticated since pre-Columbian times and occurs in low-lying areas of the Amazon rainforest. Its pulp has a particular flavor (bittersweet taste) and aroma, and is usually consumed as jams, sweets, ice creams, and wines. In recent decades, consumption of the pulp and oil of M. flexuosa has expanded in the global market, offering considerable socioeconomic potential (Cândido et al. 2015; Koolen et al. 2018; Cruz et al. 2020).
The fruit pulp is a rich source of macro and micronutrients. Carbohydrates are composed mainly of arabinan-rich pectic and linear polysaccharides (e.g., (1 → 5)-α-l-arabinan, (1 → 3)-(1 → 4)-α-d-glucan, and (1 → 4)-β-d-xylan) (Cordeiro et al. 2015). Moreover, its fruit has a rich fat content (20% w/w of the crude), which is comprised of oleic acid (76.7%), palmitic acid (18.7%), stearic acid (1.5%), linoleic acid (1.5%), linolenic acid (0.7%), and arachidic acid (0.5%). It is also highly rich in carotenoids, mainly β-carotenes (e.g., 13-cis-β-carotene, 9-cis-β-carotene), followed by tocopherols (1169 μg/g of dry matter of M. flexuosa pulp) (e.g., β-tocopherol, α-tocopherol, and δ-tocopherol). Secondary metabolites such as phenolic compounds, mainly quinic acid and derivatives, protocatechuic, chlorogenic, and caffeic acid; and flavonoids (catechin, epicatechin, naringenin, luteolin, quercetin, and their glycosylated derivatives, and glycosylated anthocyanins such as cyanidin-3-glucoside and cyanidin-3-rutinoside) were also identified (Koolen et al. 2012; 2018; De Oliveira et al. 2013; Bataglion et al. 2014; Abreu-Naranjo et al. 2020). Aguaje oil has phytosterols and high levels of carotenoids (β-carotenes, vitamin A). Some glycosyl flavonoids, such as quercetin-3-O-rutinoside (rutin) (18), luteolin-8-C-glucoside (orientin) (38), and luteolin-6-C-glucoside (isoorientin) (39), were isolated in hydroalcoholic extracts from the leaves and roots and reported to show anti-inflammatory and antioxidant properties in studies conducted with other plant species (Koolen et al. 2012; De Oliveira et al. 2013; Lin et al. 2019).

Despite being one of the most iconic plants of the Amazon rainforest, scientific studies on this species are scarce. Recently, Cruz et al. (2020) evaluated the immunomodulatory effect of M. flexuosa oil against Escherichia coli, showing a 47% increase in the phagocytosis index when compared with the untreated group. However, the most remarkable property of aguaje fruit is its antioxidant capacity, which has been evaluated using different approaches (DPPH, TBARS or FRAP assays) (Cândido et al. 2015; Abreu-Naranjo et al. 2020) in addition to an in vivo antioxidant test using young rats to determine the catalase non-protein sulfhydryl groups in the liver and quantify malondialdehyde in the plasma and tissues (Romero et al. 2015). Furthermore, molecules such as 13-cis-β-carotene (40), 9-cis-β-carotene (41), and mainly α-carotene (42) were shown to be promissory in the in silico assay of molecular docking and molecular dynamics stimulations using a 2GTB peptidase complex (the main peptidase of SARS-CoV) (Costa et al. 2020). Thus, we see an interesting opportunity to continue more experimental studies on the immunomodulatory effects of promising molecules.

Maytenus macrocarpa
Maytenus macrocarpa Briq., Celastraceae (popular name: “chuchuhuasi”), is native to lowland tropical rainforests, exclusively in the Amazon region. It is a tree that can grow up to 30 m tall. Traditional medicine of the Peruvian Amazon communities mainly uses the stem bark and the root in preparations by decoction or maceration. There are popular reports that hydroalcoholic extracts of the bark or root (prepared from sugar cane) are mainly used for rheumatic, anti-inflammatory, anti-diarrheal, antimalarial, and antileishmanial treatments, or to improve wound healing. There are also reports of stem bark decoctions being used in the treatment of bronchitis and colds, to improve the immune system, and as a muscle relaxant (Rommel et al. 2016; Malaník et al. 2019).
Few phytochemical reports are found for this species. Its chemical composition comprises mainly terpenoids (triterpenoids and dihydro-β-agarofuran sesquiterpenes) isolated from the bark and leaves of M. macrocarpa. These compounds include dihydro-β-agarofurane sesquiterpenes obtained from the leaves, tetracyclic dammarane triterpenes from stem bark and exudate, lupane triterpenes from the bark, pentacyclic triterpenes (e.g., friedelin, scutione, pristimerin, ilicifoline) from stem bark and exudate, pentacyclic triterpenes (macrocarpine A-D) from the root, p-hydroxybenzoic acid derivatives, 3,4,5-trimethoxybenzyl alcohol, and noreugenin (Chávez et al. 1998, 1999; Piacente et al. 2006; Torpocco et al. 2007; Veloso et al. 2017; Malaník et al. 2019).
Chemical constituents present in stem bark extracts from M. macrocarpa, such as celastrol, lupeol, 3-E-caffeoyl-betulin, and friedelin, would act in different inflammatory processes (Malaník et al. 2019). Moreover, M. macrocarpa leaves displayed an anti-inflammatory effect at 1250 mg/kg, which was higher than the effects with diclofenac (74.14% vs 58.62%, one-way ANOVA, p < 0.05) (Luján-Carpio et al. 2019). Additionally, M. macrocarpa leaves were tested along with 34 other Amazonian plants for antiviral activity against HCV virus, with promising results. At 25 µg/ml, they inhibited the infection by >50% (Roumy et al. 2020). Furthermore, the leaf extract and isolated substances, such as pristimerin (43), macrocarpins A-D (44–47), betulin (48) derivatives, 28-hydroxyfriedelane-1,3-dione (49), scutione (50), vitideasin (51), 6β,8β,15-triacetoxy-1α,9α-dibenzoyloxy-4β-hydroxy-β-dihydroagarofuran (52), and 1α,6β,8β,15-tetraacetoxy-9α-(benzoyloxy)-4β-hydroxy-β-dihydroagarofuran (53), act against different tumoral cell lines, demonstrating its potent antitumoral activity (Malaník et al. 2019; Roumy et al. 2020). These findings, obtained in preliminary pharmacological studies, although not directly related to the immune system, may feature promising active compounds, considering that the ethnomedicinal uses are related to strengthen the immune system and relieve pulmonary COVID-19 symptoms (Malaník et al. 2019; Roumy et al. 2020). Undoubtedly, more studies should be carried out to understand the medicinal value of this plant.

Plukenetia volubilis
Plukenetia volubilis L., Euphorbiaceae (popular name: “sacha inchi”) is a climbing, monoecious, and perennial plant that grows up to 2 m in height. It occurs preferably in warm tropical climates, at high altitude (>2000 m above sea level), from the Andean rainforest to the Peruvian Amazon lowlands, where it has been cultivated as an edible nut by native people since pre-Columbian times (populations from Mochica and Chimú). As popular as C. quinoa, the cultivation of this plant has expanded to other American and Asian countries. The peanut-like fruit of P. volubilis is shaped like a star, with a capsule (3–5 cm) containing the edible oval seeds, which change from green to dark brown when ripe. Its seeds are valued for their polyunsaturated acid oils and protein content (Alayón and Echeverri 2016; Li et al. 2018).
The most studied part of the plant is the seeds of the fruits. The oil extracted from the seeds has been extensively studied regarding its chemical composition. The fruit seeds are a rich source of lipids (35–60%) and proteins (25–30%), and also contain heat-labile substances with a bitter taste. In particular, the seed oil is characterized predominantly by high levels of essential fatty acids, namely, C18:3 omega 3 (cis,cis,cis-9,12,15-octadecatrienoic acid, syn. α-linolenic acid) (54) and C18:2 omega 6 (cis,-cis-9,12-octadecadienoic acid, syn. α-linoleic acid) (55), other fatty acids, representing about 82% of the total oil content, according to the seed variety (Fanali et al. 2011; Chirinos et al. 2013). In addition, the amino-acid profile of the protein fraction shows a relatively high level of cysteine, tyrosine, threonine, and tryptophan, compared to other seed oil sources, and a large quantity of tocopherols (γ- and δ-tocopherols) (56 and 57), and phytosterols (β-sitosterol and stigmasterol), as well as phenolic compounds like ferulic acid (7) (Fanali et al. 2011; Chirinos et al. 2013; Ramos-Escudero et al. 2021). On the other hand, there is little information about the chemical composition of the seed shell and leaves of sacha inchi when compared to the seed. This was highlighted in a review article by Wang et al. (2018), where the antioxidant and anticancer properties of the seed shell and leaves are reported.

It is well known that antioxidant supplementation with fruit seed oils can improve the immune response; therefore, it is important to consider the chemical diversity not only of the seed oil, but also of the seed shell and leaves of P. volubilis (Chirinos et al. 2013; Nascimento et al. 2013; Cisneros et al. 2014). In addition to the antioxidant property, hydroalcoholic extracts of leaves (250 µg/ml) were able to reduce the proliferation of HeLa cancer cells by ~50% (Nascimento et al. 2013). A few studies have been carried out focusing on the immunological activity of fractions of polysaccharide and albumin extracted from sacha inchi seeds (Li et al. 2018; Tian et al. 2020). In one study, the albumin fraction exhibited immunomodulatory activity by stimulating the proliferation and enhancing the TNF-α secretion of splenic lymphocytes, increasing the cellular lysosomal enzyme and pinocytic activities, and moderately promoting the NO and H2O2 production of RAW264.7 cells (Li et al. 2018). In a second study, the water-soluble polysaccharide fraction-1 (PVLP-1) enhanced the expression of inflammatory cytokines IL-6, TNF-α, and IL-1β in RAW264.7 cells. This fraction could be used as a functional food or adjuvant drug to improve the biological immunity of immunodeficiency diseases and hypo-immunity (Tian et al. 2020). The results of a clinical trial involving patients who consumed a high-fat breakfast with sacha inchi oil revealed a decrease in IL-6 expression in serum (Alayón et al. 2019). Therefore, this species deserves further in-depth study.
Physalis peruviana
Physalis peruviana L., Solanaceae (popular name: “aguaymanto”) is a perennial hairy shrub reaching between 1 and 1.5 m in height. Currently, it grows in tropical and sub-tropical regions around the world, but it is native to the Andes of South America, where agricultural areas of this species have been known since before the Inca Empire. Its edible fruits are similar to small tomatoes (4–10 g) of a bright yellow color, containing about 100–200 seeds, and protected by a calix. The ripe and fresh fruit is traditionally consumed as a juice or infused to treat colds, asthma, cough, pharyngitis, scurvy (stomatitis), and jaundice (or conditions related to the kidney). Infusions and decoctions of the leaves are used for oropharyngeal treatment and to purify the blood. P. peruviana became famous worldwide due to its fruits, which have a pleasant, refreshing, and sweet-tart taste (Ramadan 2011; Lock et al. 2016).
Despite P. peruviana fruit being highly valued internationally, there are few studies about its phytochemical composition. Recent studies (Etzbach et al. 2018; El-Beltagi et al. 2019; Yu et al. 2019) have shown that P. peruviana has a high health value, especially due to its high content of carotenoids (e.g., carotenes 1.6–2 mg %), lutein esters, and several unesterified carotenoids, as well as xanthophyll esters, zeaxanthin esters, and β-cryptoxanthin (Puente et al. 2011; Etzbach et al. 2018). It also contains vitamin C (38–43 mg %), vitamin B complex (thiamine, niacin, and riboflavin), tocopherols (mainly γ- and α-), and vitamin K, along with a high content of polyunsaturated fatty acids (γ-linolenic acid 18.8%, linoleic acid, oleic acid, and palmitic acid) and minerals (e.g., calcium, copper, iron, zinc, and phosphorus) (Ramadan 2011; Yu et al. 2019).
As indicated in some review articles (Puente et al. 2011; Ramadan 2011; Singh et al. 2019), this plant possesses phenolic acids, e.g., p-coumaric (58), caffeic (59), ferulic (7), gallic (60), and chlorogenic acid (61), flavonoids, e.g., catechin/epicatechin (21 and 22), rutin (19), myricetin (62), and steroid lactones such as withanolides, e.g., withanolide E (63), 4β-hydroxywithanolide E (64), and physapruin A (65) that can be the subject of additional studies. Finally, more than 100 compounds associated with flavor, including volatiles and non-volatiles, have been identified in the fresh fruits by means of gas chromatography (Singh et al. 2019).

Nutritionally, P. peruviana plays an important role in applied food sciences (Yu et al. 2019). Its berries have been shown to provide significant health benefits, especially due to their high antioxidant activity (Ramadan 2011). Moreover, precursors of vitamin A, such as α- and β-carotene (66 and 67), as well as β-cryptoxanthin (68), are claimed to be rich in these berries and could be ascribed to boost the immune function, as well as the normal growth and development of the body (Wen et al. 2020).

Supercritical fluid extracts of P. physalis leaves have been shown to be an effective inhibitor of LPS-induced NO generation and PGE2 production, as well as pro-inflammatory enzymes (inducible nitric oxide synthase protein (iNOS) and COX-2 expression) in RAW246.7 cells (Wu et al. 2006). In addition, these extracts proved to be potent superoxide anions scavengers and xanthine oxidase inhibitors, with IC50 values of 6.78 µg/ml and 7.48 µg/ml, respectively (Wu et al. 2006). Physalis peruviana extracts presenting a high content of flavonoids and other phenolics were reported to have promising anti-inflammatory and antioxidant effects (Wu et al. 2006). Franco et al. (2007) showed a significant inhibition of acute ear edema induced by 12-O-tetradecanoylforbol-13-acetate in a dose-dependent manner. Later, Martínez et al. (2010) observed a reduction of proinflammatory cytokines such as TNF-α, monocyte chemoattractant protein-1 (MCP-1), and IL-6 in a study using an in vitro phagocytosis model (Leishmania panamensis, Trypanosomatidae, infection in murine macrophages). Moreover, this finding would be strongly related to an immunosuppressive activity (a decrease in pro-inflammatory cytokines). Two years later, the protective effect of P. peruviana leaf extract was evaluated in murine models of cigarette smoke-induced and LPS-induced lung inflammation. The results showed a reduction in the influx of inflammatory cells in the bronchoalveolar lavage fluid (BALF). It is noteworthy that reactive oxygen species and pro-inflammatory cytokines (TNF-α and IL-6) were reduced in BALF, and the activation of MPC-1 was attenuated. On the other hand, the lung activation of extracellular signal-regulated kinase, nuclear factor erythroid 2–related factor 2 (Nrf2), and heme oxygenase-1 (HO-1) expression was observed (Park et al. 2017). Therefore, these findings would be strongly correlated with immunosuppressive activity, suggesting that these extracts may have an adjuvant role in inflammatory airway and lung diseases. This immunomodulator profile was reinforced by Mier-Giraldo et al. (2017), when they demonstrated that hydroalcoholic extracts from P. peruviana reduced the release of IL-6, IL-8, and MCP-1 in a dose-dependent manner. According to the authors, the immunomodulator activity could be associated with the presence of rosmarinic acid (69) and ursolic acid (70) from the alcoholic extract of ripe P. peruviana fruits. Meanwhile, catechin, gallic acid, and epicatechin gallate from isopropanol extract could be related to the antioxidant activity (Mier-Giraldo et al. 2017). Furthermore, withanolides (63–65) isolated from alcoholic extracts of whole plants of P. peruviana showed NO inhibitory effects (IC50 < 8 μM) and a strong affinity with iNOS, indicating their anti-inflammatory potential (Dong et al. 2019). Similarly, Park et al. (2019) evaluated methanolic extracts from leaves of P. peruviana, with promising results. A reduction in the number of inflammatory cells (Th2 cytokines) in BALF, including eosinophils and macrophages, and a significant relief of IL-3, IL-4, IL-5, and serum IgE levels was observed in a model of murine OVA-induced allergic asthma. There was also a potent inhibition of MCP-1 production in LPS-stimulated RAW264.7 macrophages (Park et al. 2019).

Uncaria tomentosa
Uncaria tomentosa DC., Rubiaceae (popular name: “uña de gato” or cat’s claw) is a large woody vine geographically distributed in Central America and the Amazon tropical forest. It is one of the most studied and representative medicinal plant of the Amazon Peruvian culture, and its inner bark, roots, and leaves are extensively used for medicinal and ceremonial purposes by Amazon healers (“ashaninkas”) (Domingues et al. 2011; Shen et al. 2018). The ethnomedicinal uses for the bark (most commonly used) and roots (less commonly used) are for the treatment of rheumatic ailments, prostate inflammation, cancer inflammation, and respiratory and digestive diseases; fresh bark in snakebites; leaf infusions for measles (viral infection), inflammation processes, and allergies; and macerations (alcoholic beverages from sugar cane) from bark or root to boost the immune system (Lock et al. 2016).
Extensive research has been carried out on U. tomentosa leaves, the inner part of the bark and the root in the last 50 years, due to its use in folk medicine or complementary therapies for its immunological and antiviral properties. Since then, its medicinal formulations have been chemically explored (Domingues et al. 2011). Substances such as tetracyclic oxindole alkaloids, e.g., rhynchophylline (71), corynoxeine (72), isorhynchophylline (73), and isocorynoxeine (74); pentacyclic oxindole alkaloids, e.g., mitraphylline (75), pterodine (76), uncarine F (77), speciophylline (78), isopteropodine (79), and isomitraphylline (80); quinovic acid glycosides, e.g., quinovic acid 3β-O-β-d-quinovopyranoside (81), quinovic acid 3β-O-β-d-fucopyranosyl-(27 → 1)-d-glucopyranoylester (82), and quinovic acid 3β-O-[β-d-glucopyranosil-(1 → 3)-β-d-fucopyranosyl]-(27 → 1)-β-d-glucopyranosylester (83), in addition to procyanidins, e.g., proanthocyanidin B1-B4 (84–87), B5 (88), and C1 (89), could be strongly related to the regulation of the immune system. Furthermore, it is suggested that its concentration of carboxylalkyl esters of quinic acids (e.g., 3,4-O-dicaffeoylquinic acid) (8–20%), chlorogenic acids, triterpenoids (lupeol, ursolic, and oleanolic acids), and vaccenic (omega-7 unsaturated fatty acid) would confer potent pharmacological action.


It is noteworthy that U. tomentosa contains pentacyclic and tetracyclic indole/oxindole alkaloids, which would lead to two different chemotypes, according to Keplinger et al. (1998): chemotype I (tetracyclic oxindole alkaloids) and chemotype II (tetracyclic oxindole alkaloids). Both types of alkaloids showed an antagonistic effect on each other; therefore, the medicinal use of these two alkaloid mixtures has been inaccurately reported. In order to better understand the behavior of these alkaloids, Heitzman et al. (2005) evaluated fractions obtained from U. tomentosa and found that the pentacyclic oxindole alkaloid-enriched fractions caused an increase in phagocytosis of human granulocytes and macrophages, blocking the proliferation of myeloid cell lines and suppressing TNF-α synthesis. Conversely, tetracyclic oxindole alkaloids act antagonistically on the release of these factors. Nevertheless, the most relevant, immune-stimulating pentacyclic oxindole alkaloid-alkaloid, such as pteropodine and isopteropodine, have positive modulatory effects on the brain (Heitzman et al. 2005). In another study, Peñaloza et al. (2015) corroborated not two chemotypes, but three: chemotype I (pteropodine, isopteropodine, speciophylline, and uncarine F), chemotype II (mitraphylline and its isomer isomitraphylline), and chemotype III (rhynchophylline and its isomer isorhynchophylline).
Furthermore, extracts from U. tomentosa stem bark stimulated the production of IL-6 and IL-1 by rat macrophages in a dose-dependent manner in the range of 0.025–0.1 mg/ml (Lemaire et al. 1999). Later, Winkler et al. (2004) suggested that acidified aqueous and ethanolic extracts of U. tomentosa interfere with immunopathogenetic pathways involving Th-1 type cytokines (IFN-ɣ), while Sandoval et al. (2000) suggested they were a remarkably potent inhibitor of TNF-α. In addition, the alkaloid fraction exhibited a strong immunomodulatory effect, with decreased levels of TNF-α and IFN-α and the modulation of IL-10 in DENV-infected monocytes (Reis et al. 2008). Lima-Junior et al. (2013) treated a human lineage of dermal microvascular endothelial cells infected with DENV-2 using an alkaloidal fraction, demonstrating its immunomodulatory activity. In another in vitro experiment, Domingues et al. (2011) observed the immunomodulatory effects of a pentacyclic oxindole alkaloid-enriched extracts from the bark. An increase of T helper lymphocytes and B lymphocytes was observed; however, the extracts inhibited the T lymphocyte proliferation at high concentrations. Interestingly, Lenzi et al. (2013) evaluated the effects of an aqueous extract rich in polyphenols (86%) and proteins, observing an increase of TNF-α and IL-6 levels and a reduction of IL-1β levels in an in vitro immunostimulant screening assay in a macrophage cell culture. In this study, other chemical classes rather than alkaloids stood out in relation to the immunomodulatory properties of this species.
In a clinical trial with human volunteers, cat’s claw aqueous extract (C-Med-100) at doses of 250 and 350 mg/day increased the number of lymphocytes and leukocytes, which could be ascribed to carboxyl alkyl esters of quinic acids (8–10% w/w) (Sheng et al. 2001). Considering this evidence, new studies were launched related to molecules capable of modulating the immune system or acting against the SARS-CoV-2 virus. Ferreira et al. (2020) explored 12 therapeutic agents, including Miodesin®, which contains U. tomentosa as an ingredient. They assumed that the presence of oxindole alkaloids would play a role in immunoregulation, while triterpenoid alkaloids and quinovic acid glycosides could inhibit some DNA and RNA viruses. Furthermore, plant polyphenols could contribute to reducing the oxidative stress by enhancing phagocytosis. This research suggested that the components of Miodesin® (U. tomentosa, Endopleura uchi (Huber) Cuatrec., Humiriaceae, and astaxanthin, a keto-carotenoid) may act synergistically for the prophylaxis or improvement of symptoms associated with COVID-19. Meanwhile, Yepes-Perez et al. (2021) evaluated aqueous extract and alkaloid-enriched fractions obtained from the plant stem bark against the SARS-CoV-2 virus. At 25 μg/ml, these fractions inhibited SARS-CoV-2 replication on Vero E6 cells by 92.7% (EC50 6.6 µg/ml) in a plate test.
The anti-inflammatory efficacy of this plant has also been tested in various experimental studies using extracts and enriched fractions. Allen-Hall et al. (2010) and Azevedo et al. (2018) suggested that the anti-inflammatory effects of the stem bark ethanolic extract are due to the regulation of the NF-ƘB and the activator protein-1 (AP-1) transcription factors. Once these transcription factors are activated, pro-inflammatory cytokines such as TNF-α and IL-1β are expressed. Consequently, these extracts may be promising applications in anti-inflammatory therapies.
Perspective and Future Directions
In this review, we present plants that show evidence of immunomodulatory action in experimental studies, as well as antiviral properties in several in vitro and in vivo assays. In addition, some studies focused on the evaluation of anti-inflammatory, antioxidant, and cytotoxic activities, complemented with additional experiments to better assess their potential as a possible source of immunomodulatory drugs for COVID-19. In this context, the immunomodulatory properties of medicinal plants widely used in Peru by native communities have been little studied, except for C. quinoa, L. meyenii, and U. tomentosa, which have aroused the interest of researchers only in the last two decades. Additionally, few plants have been submitted to pre-clinical and clinical trials, probably due to the colossal challenge of fulfilling all the rigorous requirements to obtain a phytomedicine.
Crude extracts and isolated chemical constituents have been studied for their beneficial effects, as reported from traditional knowledge. The results of these studies reveal that many of them exert significant immunomodulatory effects. Both polysaccharides and proteins, as well as saponins, alkaloids, and flavonoids can act in an immunomodulatory way by inhibiting the release of inflammatory cytokines, such as TNF-α, blocking the activation of NF-ƘB, and downregulating NO production in macrophages, IL-6 production, or MCP-1 expression. The immunomodulatory profile of these extracts and isolated components gives them significant potential in the treatment of COVID-19.
Another aspect to be discussed is that, despite the passing of many centuries, pre-Columbian traditional medicine in South America has not been able to be integrated into the modern medicinal system, contrary to that observed with traditional Chinese medicine. Only at the beginning of this century were a few more representative medicinal plants, both native and exotic, included in the Peruvian health system. Some of these plants have been the subject of incipient scientific studies. It is noteworthy that the barrier between raw material and herbal product is influenced by multiple factors, among them, the plasticity of plants, because they change both physiologically and metabolically according to environmental and genetic factors, with consequences on the qualitative and quantitative profile of the plant’s extracts. It is important to point out that plant crude extracts have not been standardized due to the lack of adequate chemical markers and process with ecologically correct extraction processes with enough green chemistry concepts are needed. In addition, the lack of a complete knowledge of the chemical and pharmacological/toxicological profiles of the extracts, as well as their performance in clinical trials, does not allow them to reach their therapeutic relevance according to the requirements of standardized contemporary medicine. Some medicinal plants are exported as raw materials or supplementary foods, while many others remain regrettably understudied. It is urgent to improve and protect the traditional knowledge of medicinal plants that could be a source of new medicines in the future, since they constitute a good, flexible, and cheap alternative for the treatment of COVID-19.
Native communities from pre-Columbian civilizations already had medicinal knowledge based mainly on plants. It is possible that some plants have already played an important role in pandemic situations and recurrent diseases faced throughout the history of these people. The bark of the cinchona tree (Cinchona officinalis), used in traditional medicine against malaria, was the first effective treatment at the time. In recent decades, there has been a growing interest in validating traditional medicine, with its holistic and systematic approach, through scientific research that proves its real effectiveness and, consequently, supports its use in conventional medicine. However, there is still a large gap between traditional medicine and the experimental evidence required in conventional medicine. The use of traditional medicinal plants showing an immunomodulatory profile could provide a therapeutic alternative. In the current scenario, with the COVID-19 pandemic far from over, some plants or plant-based remedies have been popularly used to alleviate some symptoms of the disease.
In addition, the present review highlights the importance of the pharmacological information (Table S1) from nine native Peruvian plants, which could potentially provide new alternatives for the treatment of COVID-19 addressed to the strength of the immune system, the fight against viral infection, hyperinflammation, or post-COVID-19 complications through different mechanisms associated with the SARS-CoV-2 virus. However, further experimental studies need to be completed.
Conclusions
In this review, the medicinal species Chenopodium quinoa, Chuquiraga spinosa, Croton lechleri, Lepidium meyenii, Mauritia flexuosa, Maytenus macrocarpa, Plukenetia volubilis, Physalis peruviana, and Uncaria tomentosa stood out in terms of the biological benefits resulting from their wide variety of metabolites. Thus, it is possible to suggest some therapeutic hypotheses relating to the immune system or directly to the infection caused by SARS-CoV-2. The first reports are already available with regard to the species U. tomentosa and M. flexuosa, pointing to promising molecules for the development of a new drug to combat COVID-19. We can suppose that herbal formulations to relieve symptoms such as fever, cough, lung inflammation, and fatigue could be developed, reducing the probability of patients developing severe conditions. We should highlight the lack of standardization in sample preparation, extraction, and characterization, as well as in the enriched fractions of bioactive molecules in most of the studies related to medicinal plants, which makes it very unlikely that the immunomodulatory activity can be attributed to a single compound. Despite the many unknowns concerning the role of these compounds in the body’s immune response, they can be considered potential immunomodulatory agents. In addition, other pharmacological benefits, including antiviral, antioxidant, and anti-inflammatory activities, would significantly increase the protective effect of these plant preparations. It is also important to note that some of the plants discussed here are already commercialized and exported to many countries (e.g., the USA) as supplements for the human diet, supporting the use of these medicinal plants for therapeutic purposes in the near future. Finally, this review highlights the scientific evidence for nine plants that are rich sources of valuable potential therapeutic agents to modulate the immune system. However, new studies need to be carried out to validate the use of these plants as immunomodulatory agents before assessing their potential beneficial effect in the treatment of patients with COVID-19.
References
Abreu-Naranjo R, Paredes-Moreta JG, Granda-Albuja G, Iturralde G, González-Paramás AM, Alvarez-Suarez JM (2020) Bioactive compounds, phenolic profile, antioxidant capacity and effectiveness against lipid peroxidation of cell membranes of Mauritia flexuosa L. fruit extracts from three biomes in the Ecuadorian Amazon. Heliyon 6:E05211. https://doi.org/10.1016/j.heliyon.2020.e05211
AGRODATA (2015) Plantas y semillas de medicina y perfumería: demas plantas, partes de plantas, semillas e frutos utilizados Homepage. https://www.agrodataperu.com/2015/06/plantas-y-semillas-de-medicina-y-perfumeria.html. Accessed 12 Sept 2020
Alayón NA, Echeverri JI (2016) Sacha inchi (Plukenetia volubilis Linneo): ¿una experiencia ancestral desaprovechada? Evidencias clínicas asociadas a su consumo. Rev Chil Nutr 43:167–171. https://doi.org/10.4067/S0717-75182016000200009
Alayón AN, Ortega Ávila JG, Echeverri Jiménez I (2019) Metabolic status is related to the effects of adding of sacha inchi (Plukenetia volubilis L.) oil on postprandial inflammation and lipid profile: randomized, crossover clinical trial. J Food Biochem 43:e12703. https://doi.org/10.1111/jfbc.12703
Alhazmi HA, Najmi A, Javed SA, Sultana S, Al Bratty M, Makeen HA, Meraya AM, Ahsan W, Mohan S, Taha MME, Khalid A (2021) Medicinal plants and isolated molecules demonstrating immunomodulation activity as potential alternative therapies for viral diseases including COVID-19. Front Immunol 12:637553. https://doi.org/10.3389/fimmu.2021.637553
Allen-Hall L, Cano P, Arnason JT, Rojas R, Lock O, Lafreine RM (2007) Treatment of THP-1 cells with Uncaria tomentosa extracts differentially regulates the expression if IL-1β and TNF-α. J Ethnopharmacol 109:312–317. https://doi.org/10.1016/j.jep.2006.07.039
Allen-Hall L, Arnasond JT, Cano P, Lafrenie RM (2010) Uncaria tomentosa acts as a potent TNF-alpha inhibitor through NF-kappaB. J Ethnopharmacol 127:685–693. https://doi.org/10.1016/j.jep.2009.12.004
Azevedo BC, Morel LJF, Carmona F, Cunha TM, Contini SHT, Delprete PG, Ramalho FS, Crevelin E, Bertoni BW, França SC, Borges MC, Pereira AMS (2018) Aqueous extracts from Uncaria tomentosa (Willd. ex Schult.) DC. reduce bronchial hyperresponsiveness and inflammation in a murine model of asthma. J Ethnopharmacol 218:76–89. https://doi.org/10.1016/j.jep.2018.02.013
Azevedo L, de Araujo Ribeiro PF, de Carvalho Oliveira JA, Correia MG, Ramos FM, de Oliveira EB, Barros F, Stringheta PC (2019) Camu-camu (Myrciaria dubia) from commercial cultivation has higher levels of bioactive compounds than native cultivation (Amazon forest) and presents antimutagenic effects in vivo. J Sci Food Agric 99:624–631. https://doi.org/10.1002/jsfa.9224
Babich O, Sukhikh S, Prosekov A, Asyakina L, Ivanova S (2020) Review: medicinal plants to strengthen immunity during a pandemic. Pharmaceuticals 13:313. https://doi.org/10.3390/ph13100313
Barbosa J, Nunes de Carvalho J (2021) Polysaccharides obtained from natural edible sources and their role in modulating the immune system: biologically active potential that can be exploited against COVID-19. Trends Food Sci Technol 108:223–235. https://doi.org/10.1016/j.tifs.2020.12.026
Barnard DL, Smee DF, Huffman JH, Meyerson LR, Sidwell RW (1993) Antiherpesvirus activity and mode of action of SP-303, a novel plant flavonoid. Chemotherapy 39:203–211. https://doi.org/10.1159/000239127
Bataglion GA, Da Silva FMA, Eberlin MN, Koolen HHF (2014) Simultaneous quantification of phenolic compounds in buriti fruit (Mauritia flexuosa L.f.) by ultra-high performance liquid chromatography coupled to tandem mass spectrometry. Food Res Int 66:396–400. https://doi.org/10.1016/j.foodres.2014.09.035
Behl T, Kumar K, Brisc C, Rus M, Nistor-Cseppento DC, Bustea C, Aron RAC, Pantis C, Zengin G, Sehgal A, Kaur R, Kumar A, Arora S, Setia D, Chandel D, Bungau S (2021) Exploring the multifocal role of phytochemicals as immunomodulators. Biomed Pharmacother 133:110959. https://doi.org/10.1016/j.biopha.2020.110959
Bhuiyan FR, Howlader S, Raihan T, Hasan M (2020) Plants metabolites: possibility of natural therapeutics against the COVID-19 pandemic. Front Med 7:444. https://doi.org/10.3389/fmed.2020.00444
Cai Y, Evns FJ, Roberts MF, Phillipson JD, Zenk MH, Gleba YY (1991) Polyphenolic compounds from Croton lechleri. Phytochemistry 30:2033–2040. https://doi.org/10.1016/0031-9422(91)85063-6
Cai Y, Chen ZP, Phillipson JD (1993) Clerodane diterpenoids from Croton lechleri. Phytochemistry 34:265–268. https://doi.org/10.1016/S0031-9422(00)90816-1
Cândido TLN, Silva MR, Agostini-Costa TS (2015) Bioactive compounds and antioxidant capacity of buriti (Mauritia flexuosa L.f.) from the Cerrado and Amazon biomes. Food Chem 177:313–319. https://doi.org/10.1016/j.foodchem.2015.01.041
Capraro J, de Benedetti S, di Dio M, Bona E, Abate A, Corsetto PA, Scarafoni A (2020) Characterization of chenopodin isoforms from quinoa seeds and assessment of their potential anti-inflammatory activity in caco-2 cells. Biomolecules 10:795. https://doi.org/10.3390/biom10050795
Carvalho FV, Ribeiro PR (2019) Structural diversity, biosynthetic aspects, and LC-HRMS data compilation for the identification of bioactive compounds of Lepidium meyenii. Food Res Int 125:108615. https://doi.org/10.1016/j.foodres.2019.108615
Casado R, Landa A, Calvo J, García-Mina JM, Marston A, Hostettmann K, Calvo MI (2011) Anti-inflammatory, antioxidant and antifungal activity of Chuquiraga spinosa. Pharm Biol 49:620–626. https://doi.org/10.3109/13880209.2011.577436
Catanzaro M, Corsini E, Rosini M, Racchi M, Lanni C (2018) Immunomodulators inspired by nature: a review on curcumin and Echinacea. Molecules 23:2778. https://doi.org/10.3390/molecules23112778
Chavéz H, Estévez-Braun A, Ravelo AG, Gonzaléz G (1998) Friedelane triterpenoids from Maytenus macrocarpa. J Nat Prod 61:82–85. https://doi.org/10.1021/np970232k
Chávez H, Callo N, Estévez-Braun A, Ravelo AG, Gonzalez AG (1999) Sesquiterpene polyol esters from the leaves of Maytenus macrocarpa. J Nat Prod 62:1576–1577. https://doi.org/10.1021/np990232f
Chirinos R, Zuloeta G, Pedreschi R, Mignolet E, Larondelle Y, Campos D (2013) Sacha inchi (Plukenetia volubilis): a seed source of polyunsaturated fatty acids, tocopherols, phytosterols, phenolic compounds and antioxidant capacity. Food Chem 141:1732–1739. https://doi.org/10.1016/j.foodchem.2013.04.078
Cisneros FH, Paredes D, Arana A, Cisneros-Zevallos L (2014) Chemical composition, oxidative stability and antioxidant capacity of oil extracted from roasted seeds of Sacha-inchi (Plukenetia volubilis L.). J Agric Food Chem 62:5191–5197. https://doi.org/10.1021/jf500936j
Cordeiro LMC, De Almeida CP, Iacomini M (2015) Unusual linear polysaccharides: (1→5)-α-L-Arabinan, (1→3)-(1→4)-α-D-glucan and (1→4)-β-D-xylan from pulp of buriti (Mauritia flexuosa), an edible palm fruit from the Amazon region. Food Chem 173:141–146. https://doi.org/10.1016/j.foodchem.2014.10.020
Costa AN, de Sá ÉRA, Bezerra RDS, Souza JL, Lima FDCA (2020) Constituents of buriti oil (Mauritia flexuosa L.) like inhibitors of the SARS-Coronavirus main peptidase: an investigation by docking and molecular dynamics. J Biomol Struct Dyn 39:4610–4617. https://doi.org/10.1080/07391102.2020.1778538
Cruz MB, Oliveira W da S, Araújo RL, Honório França AC, Pertuzatti PB (2020) Buriti (Mauritia flexuosa L.) pulp oil as an immunomodulator against enteropathogenic Escherichia coli. Ind Crops Prod 149:112330. https://doi.org/10.1016/j.indcrop.2020.112330
De Marino S, Gala F, Zollo F, Vitalini S, Fico G, Visioli F, Iorizzi M (2008) Identification of minor secondary metabolites from the latex of Croton lechleri (Muell-Arg) and evaluation of their antioxidant activity. Molecules 13:1219–1229. https://doi.org/10.3390/molecules13061219
De Oliveira DM, Siqueira EP, Nunes YRF, Cota BB (2013) Flavonoids from leaves of Mauritia flexuosa. Rev Bras Farmacogn 23:614–620. https://doi.org/10.1590/S0102-695X2013005000061
Di Sotto A, Vitalone A, Di Giacomo S (2020) Plant-derived nutraceuticals and immune system modulation: an evidence-based overview. Vaccines 8:468. https://doi.org/10.3390/vaccines8030468
Domingues A, Sartori A, Valente LMM, Golim MA, Siani AC, Viero RM (2011) Uncaria tomentosa aqueous-ethanol extract triggers an immunomodulation toward a Th2 cytokine profile. Phytother Res 25:1229–1235. https://doi.org/10.1002/ptr.3549
Dong B, An L, Yang X, Zhang X, Zhang J, Tuerhong M, Jin DQ, Ohizumi Y, Lee D, Xu J, Guo Y (2019) Withanolides from Physalis peruviana showing nitric oxide inhibitory effects and affinities with iNOS. Bioorg Chem 87:585–593. https://doi.org/10.1016/j.bioorg.2019.03.051
Dutta A, Roy A, Roy L, Chattopadhyay S, Chatterjee S (2020) Immune response and possible therapeutics in COVID-19. RSC Adv 11:960–977. https://doi.org/10.1039/d0ra08901j
El Hazzam K, Hafsa J, Sobeh M, Mhada M, Taourirte M, Kacimi KEL, Yasri A (2020) An insight into saponins from Quinoa (Chenopodium quinoa Willd): a review. Molecules 25:1059. https://doi.org/10.3390/molecules25051059
El-Beltagi HS, Mohamed HI, Safwat G (2019) Chemical composition and biological activity of Physalis peruviana L. Gesunde Pflanzen 71:113–122. https://doi.org/10.1007/s10343-019-00456-8
Etzbach L, Pfeiffer A, Weber F, Schieber A (2018) Characterization of carotenoid profiles in goldenberry (Physalis peruviana L.) fruits at various ripening stages and in different plant tissues by HPLC-DAD-APCI-MSn. Food Chem 245:508–517. https://doi.org/10.1016/j.foodchem.2017.10.120
Fan S, Li J, Bai B (2019) Purification, structural elucidation and in vivo immunity-enhancing activity of polysaccharides from quinoa (Chenopodium quinoa Willd.) seeds. Biosci Biotechnol Biochem 83:2334–2344. https://doi.org/10.1080/09168451.2019.1650635
Fanali C, Dugo L, Cacciola F, Beccaria M, Grasso S, Dachà M, Dugo P, Mondello L (2011) Chemical characterization of sacha inchi (Plukenetia volubilis L.) oil. J Agric Food Chem 59:13043–13049. https://doi.org/10.1021/jf203184y
FAO (2022) Food and agriculture organization of the United Nations: Quinoa homepage. https://www.fao.org/quinoa/en/?fb_locale%3Dar_ar=. Accessed 15 Aug 2021
Fei W, Hou Y, Yue N, Zhou X, Wang Y, Wang L, Li A, Zhang J (2020) The effects of aqueous extract of maca on energy metabolism and immunoregulation. Eur J Med Res 25:1–8. https://doi.org/10.1186/s40001-020-00420-7
Ferreira AO, Polonini HC, Dijkers ECF (2020) Postulated adjuvant therapeutic strategies for COVID-19. J Pers Med 10:1–33. https://doi.org/10.3390/jpm10030080
Franco LA, Matiz GE, Calle J, Pinzón R, Ospina LF (2007) Actividad antinflamatoria de extractos y fracciones obtenidas de cálices de Physalis peruviana L. Biomedica 27:110–115. https://doi.org/10.7705/biomedica.v27i1.237
Gandhi RT, Lynch JB, del Rio C (2020) Mild or moderate Covid-19. N Engl J Med 383:1757–1766. https://doi.org/10.1056/nejmcp2009249
Gómez-Caravaca AM, Segura-Carretero A, Fernández-Gutiérrez A, Caboni MF (2011) Simultaneous determination of phenolic compounds and saponins in quinoa (Chenopodium quinoa Willd) by a liquid chromatography-diode array detection-electrospray ionization-time-of-flight mass spectrometry methodology. J Agric Food Chem 59:10815–10825. https://doi.org/10.1021/jf202224j
Graf BL, Rojas-Silva P, Rojo LE, Delatorre-Herrera J, Baldeón ME, Raskin I (2015) Innovations in health value and functional food development of quinoa (Chenopodium quinoa Willd). Compr Rev Food Sci Food Saf 14:431–445. https://doi.org/10.1111/1541-4337.12135
Hardy K (2021) Paleomedicine and the evolutionary context of medicinal plant use. Rev Bras Farmacogn 31:1–15. https://doi.org/10.1007/s43450-020-00107-4
Heitzman ME, Neto CC, Winiarz E, Vaisberg AJ, Hammond GB (2005) Ethnobotany, phytochemistry and pharmacology of Uncaria (Rubiaceae). Phytochemistry 66:5–29. https://doi.org/10.1016/j.phytochem.2004.10.022
Herrera-Calderon O, Tinco-Jayo JA, Franco-Quino C, Chumpitaz- Cerrate V, Castro-Pari W, Pari-Olarte B, Castillo-Romero, P, Arroyo-Acevedo JL (2017) Antioxidant activity and cytotoxic profile of Chuquiraga spinosa Lessing on human tumor cell lines: a promissory plant from Peruvian flora. Asian Pac J Trop Dis 7:304–308. https://doi.org/10.12980/apjtd.7.2017D6-436
Hu Y, Zhang J, Zou L, Fu C, Li P, Zhao G (2017) Chemical characterization, antioxidant, immune-regulating and anticancer activities of a novel bioactive polysaccharide from Chenopodium quinoa seeds. Int J Biol Macromol 99:622–629. https://doi.org/10.1016/j.ijbiomac.2017.03.019
Jantan I, Ahmad W, Bukhari SNA (2015) Plant-derived immunomodulators: an insight on their preclinical evaluation and clinical trials. Front Plant Sci 6:1–18. https://doi.org/10.3389/fpls.2015.00655
Jones K (2003) Review of sangre de drago (Croton lechleri), a South American tree sap in the treatment of diarrhea, inflammation, insect bites, viral infections, and wounds: traditional uses to clinical research. J Altern Complement Med 9:877–896. https://doi.org/10.1089/107555303771952235
Keplinger K, Laus G, Wurm M, Dierich MP, Teppner H (1998) Uncaria tomentosa (Willd.) DC. Ethnomedicinal use and new pharmacological, toxicological and botanical results. J Ethnopharmacol 64:23–34. https://doi.org/10.1016/S0378-8741(98)00096-8
Koolen HHF, Soares ER, Da Silva FMA, da Souza AQL, Filho ER, De Souza ADL (2012) Triterpenes and flavonoids from the roots of Mauritia flexuosa. Rev Bras Farmacogn 22:189–192. https://doi.org/10.1590/S0102-695X2011005000201
Koolen HHF, da Silva FMA, da Silva VSV, Paz WHP, Bataglion GA (2018) Buriti fruit— Mauritia flexuosa. Exotic Fruits 2:61–67. https://doi.org/10.1016/B978-0-12-803138-4.00004-6
Landa A, Casado R, Calvo MI (2009) Identification and quantification of flavonoids from Chuquiraga spinosa (Asteraceae). Nat Prod Commun 4:1353–1355. https://doi.org/10.1177/1934578x0900401008
Lemaire I, Assinewe V, Cano P, Awang DVC, Arnason JT (1999) Stimulation of interleukin-1 and -6 production in alveolar macrophages by the neotropical liana, Uncaria tomentosa (uña de gato). J Ethnopharmacol 64:109–115. https://doi.org/10.1016/S0378-8741(98)00113-5
Lenzi RM, Campestrini LH, Okumura LM, Bertol G, Kaiser S, Ortega GG, Gomes EM, Bovo F, Zawadzki-Baggio SF, Stevan-Hancke FR, Maurer JBB (2013) Effects of aqueous fractions of Uncaria tomentosa (Willd.) D.C. on macrophage modulatory activities. Food Res Int 53:767–779. https://doi.org/10.1016/j.foodres.2013.02.042
Li S, Hao L, Kang Q, Cui Y, Jiang H, Liu X, Lu J (2017) Purification, characterization and biological activities of a polysaccharide from Lepidium meyenii leaves. Int J Biol Macromol 103:1302–1310. https://doi.org/10.1016/j.ijbiomac.2017.05.165
Li P, Wen J, Ma X, Lin F, Jiang Z, Du B (2018) Structural, functional properties and immunomodulatory activity of isolated Inca peanut (Plukenetia volubilis L.) seed albumin fraction. Int J Biol Macromol 118:1931–1941. https://doi.org/10.1016/j.ijbiomac.2018.07.046
Lima-Junior RS, da Silva MC, Kubelka CF, Siani AC, Valente LMM (2013) Uncaria tomentosa alkaloidal fraction reduces paracellular permeability, il-8 and ns1 production on human microvascular endothelial cells infected with dengue virus. Nat Prod Commun 8:1547–1550. https://doi.org/10.1177/1934578x1300801112
Lin M, Han P, Li Y, Wang W, Lai D, Zhou L (2019) Quinoa secondary metabolites and their biological activities or functions. Molecules 24:1–47. https://doi.org/10.3390/molecules24132512
Lock O, Perez E, Villar M, Flores D, Rojas R (2016) Bioactive compounds from plants used in peruvian traditional medicine. Nat Prod Commun 11:315–337
Lozano M, Gonzales E, Flores Y, Almanza G.R (2013) Effect in acute inflammation of sapogenin extract and isolated sapogenins from quinoa waste (Chenopodium quinoa willd). Rev Bol Quim 30:115–121. https://www.redalyc.org/comocitar.oa?id=426339680002. Accessed on 14 May 2021
Luján-Carpio E, Medina-Salazar H, Mayor-Vega A, Medrano-Canchari K, Mazuelos-Rivas M, Lizarraga-Castañeda Z, Pante-Medina C, Salazar-Granara A (2019) Anti-inflammatory and neurobehavioral effects of the leaves from Maytenus macrocarpa (Ruiz and Pavón) briquet in mice. Pharmacogn J 11:75–80. https://doi.org/10.5530/pj.2019.1.14
Malaník M, Treml J, Rjašková V, Tížková K, Kaucká P, Kokoška L, Kubatka P, Šmejkal K (2019) Maytenus macrocarpa (Ruiz & Pav.) Briq.: phytochemistry and pharmacological activity. Molecules 24:1–22. https://doi.org/10.3390/molecules24122288
Martínez W, Ospina LF, Granados D, Delgado G (2010) In vitro studies on the relationship between the anti-inflammatory activity of Physalis peruviana extracts and the phagocytic process. Immunopharmacol Immunotoxicol 32:63–73. https://doi.org/10.3109/08923970903143957
Mendoza JV, Pumarola T, Gonzales LA, Del Valle LJ (2014) Antiviral activity of maca (Lepidium meyenii) against human influenza virus. Asian Pac J Trop Med 7:S415–S420. https://doi.org/10.1016/S1995-7645(14)60268-6
Mier-Giraldo H, Díaz-Barrera LE, Delgado-Murcia LG, Valero-Valdivieso MF, Cáez-Ramírez G (2017) Cytotoxic and immunomodulatory potential activity of Physalis peruviana fruit extracts on cervical cancer (HeLa) and fibroblast (L929) cells. J Evid Based Complementary Altern Med 22:777–787. https://doi.org/10.1177/2156587217718751
Nascimento AKL, Melo-Silveira RF, Dantas-Santos N, Fernandes JM, Zucolotto SM, Rocha HAO, Scortecci KC (2013) Antioxidant and antiproliferative activities of leaf extracts from Plukenetia volubilis Linneo (Euphorbiaceae). Evid-Based Complement Altern Med 2013: 950272. https://doi.org/10.1155/2013/950272
Navarro M, Arnaez E, Moreira I, Hurtado A, Monge D, Monagas M (2019) Polyphenolic composition and antioxidant activity of Uncaria tomentosa commercial bark products. Antioxidants 8:339. https://doi.org/10.3390/antiox8090339
Navruz-Varli S, Sanlier N (2016) Nutritional and health benefits of quinoa (Chenopodium quinoa Willd.). J Cereal Sci 69:371–376. https://doi.org/10.1016/j.jcs.2016.05.004
Pacheco-Santos LM (2005) Nutritional and ecological aspects of buriti or aguaje (Mauritia flexuosa Linnaeus filius): a carotene-rich palm fruit from Latin America. Ecol Food Nutr 44:345–358. https://doi.org/10.1080/03670240500253369
Pal M, Berhanu G, Desalegn C, Kandi V (2020) Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2): an update. Cureus 12:e7423. https://doi.org/10.7759/cureus.7423
Park HA, Kwon OK, Ryu HW, Min JH, Park MW, Park MH, Paik JH, Choi S, Paryanto I, Yuniato P, Oh SR, Ahn KS, Lee JW (2019) Physalis peruviana L. inhibits ovalbumin-induced airway inflammation by attenuating the activation of NF-κB and inflammatory molecules. Int J Mol Med 43:1830–1838. https://doi.org/10.3892/ijmm.2019.4110
Park HA, Lee JW, Kwon OK, LEE G, Lim Y, Kim JH, Paik JH, Choi S, Paryanto I, Yuniato P, Kim DY, Ryu HW, Oh SR, Lee SJ, Ahn KS (2017) Physalis peruviana L. inhibits airway inflammation induced by cigarette smoke and lipopolysaccharide through inhibition of extracellular signal-regulated kinase and induction of heme oxygenase-1. Int J Mol Med 40:1557-1565. https://doi.org/10.3892/ijmm.2017.3139
Peñaloza EMC, Kaiser S, de Resende PE, Pittol V, Carvalho ÂR, Ortega GG (2015) Chemical composition variability in the Uncaria tomentosa (cat’s claw) wild population. Quim Nova 38:378–386. https://doi.org/10.5935/0100-4042.20150007
Perdue GP, Blomster RN, Blake DA, Farnsworth NR (1979) South american plants II: Taspine isolation and anti-inflammatory activity. J Pharm Sci 68:124–126. https://doi.org/10.1002/jps.2600680145
Pereira E, Encina-Zelada C, Barros L, Gonzalez-Barron U, Cadavez V, Ferreira ICFR (2019) Chemical and nutritional characterization of Chenopodium quinoa Willd (quinoa) grains: a good alternative to nutritious food. Food Chem 280:110–114. https://doi.org/10.1016/j.foodchem.2018.12.068
Perez-Chauca E, Saldaña-Bobadilla V, Michán-Herrera P (2020) Ethnobotany, pharmacology, phytochemistry and medicinal uses of Huamanpinta – Chuquiraga spinosa Less. (Asteraceae) in Peru. Ethnobot Res Appl 19:1–13. https://doi.org/10.32859/era.19.22.1-13
Piacente S, dos Santos LC, Mahmood N, Pizza C (2006) Triterpenes from Maytenus macrocarpa and evaluation of their anti-HIV activity. Nat Prod Commun 1:1073–1078. https://doi.org/10.1177/1934578x0600101201
Puente LA, Pinto-Muñoz CA, Castro ES, Cortés M (2011) Physalis peruviana Linnaeus, the multiple properties of a highly functional fruit: a review. Food Res Int 44:1733–1740. https://doi.org/10.1016/j.foodres.2010.09.034
Ramadan MF (2011) Bioactive phytochemicals, nutritional value, and functional properties of cape gooseberry (Physalis peruviana): an overview. Food Res Int 44:1830–1836. https://doi.org/10.1016/j.foodres.2010.12.042
Ramírez E, Bonilla P, Suarez S, Choquesillo F, Castro A (2014) Actividad antioxidante, antiinflamatoria e inmunomoduladora del extracto clorofórmico de las hojas de Chuquiraga lessing “Huamanpinta”. Cienc investig 17:37–42. https://doi.org/10.15381/ci.v17i1.11116
Ramos-Escudero F, Morales MT, Ramos-Escudero M, Muñoz AM, Cancino-Chavéz K, Asuero AG (2021) Assessment of phenolic and volatile compounds of commercial sacha inchi oils and sensory evaluation. Food Res Int 140:110022. https://doi.org/10.1016/j.foodres.2020.110022
Reis SRIN, Valente LMM, Sampaio AL, Siani AC, Gandini M, Azeredo EL, D’Avila LA, Mazzei JL, Henriques M das GM, Kubelka CF (2008) Immunomodulating and antiviral activities of Uncaria tomentosa on human monocytes infected with dengue virus-2. Int Immunopharmacol 8:468–476. https://doi.org/10.1016/j.intimp.2007.11.010
Ren H (2019) Effect of Lepidium meyenii Walp. on the immune function of boxers. Prog Nutr 21:882–888. https://doi.org/10.23751/pn.v21i4.8596
Risco E, Ghia F, Vila R, Iglesias J, Álvarez E, Cañigueral S (2003) Immunomodulatory activity and chemical characterisation of sangre de drago (dragon’s blood) from Croton lechleri. Planta Med 69:785–794. https://doi.org/10.1055/s-2003-43208
Romero AB da R, Martins MDCDC, Nunes PHM, Ferreira NRT, Brito AK da S, da Cunha PFM, de Lima A, de Assis RC, Araújo EM (2015) La actividad antioxidante in vitro e in vivo de la fruta burití (Mauritia Flexuosa L.F). Nutr Hosp 32:2153-2161. https://doi.org/10.3305/nh.2015.32.5.9603
Rommel ZH, Alexander ZH, Karelya WP, Jorge GB, Tiara VB, Dayana ZG, José MC, Alberto SG (2016) Study on the temperature, respiratory rate, heart rate and electrocardiogram of concomitant administration of Maytenus macrocarpa “Chuchuhuasi” and propranolol in escalating doses. Pharmacogn Commun 6:64–71. https://doi.org/10.5530/pc.2016.2.3
Rossi D, Guerrini A, Maietti S, Bruni R, Paganetto G, Poli F, Scalvenzi L, Radice M, Saro K, Sacchetti G (2011) Chemical fingerprinting and bioactivity of Amazonian Ecuador Croton lechleri Müll. Arg. (Euphorbiaceae) stem bark essential oil: A new functional food ingredient? Food Chem 126:837–848. https://doi.org/10.1016/j.foodchem.2010.11.042
Roumy V, Ruiz L, Ruiz Macedo JC, Gutierrez-Choquevilca AL, Samaillie J, Encinas LA, Mesia WR, Ricopa Cotrina HE, Rivière C, Sahpaz S, Bordage S, Garçon G, Dubuisson J, Anthérieu S, Seron K, Hennebelle T (2020) Viral hepatitis in the Peruvian Amazon: ethnomedical context and phytomedical resource. J Ethnopharmacol 255:112735. https://doi.org/10.1016/j.jep.2020.112735
Sandoval M, Charbonnet RM, Okuhama NN, Roberts J, Krenova K, Trentacosti AM, Miller MJS (2000) Cat’s claw inhibits TNFa production and scavenges free. Free Radic Biol Med 29:71–78. https://doi.org/10.1016/S0891-5849(00)00327-0
Sandoval M, Okuhama NN, Zhang XJ, Condezo LA, Lao J, Angeles FM, Musah RA, Bobrowski P, Miller MJS (2002) Anti-inflammatory and antioxidant activities of cat’s claw (Uncaria tomentosa and Uncaria guianensis) are independent of their alkaloid content. Phytomedicine 9:325–337. https://doi.org/10.1078/0944-7113-00117
Senatore F (1999) Flavonol glycosides and p-hydroxyacetophenone from Chuquiraga spinosa. Flavour Fragr J 37:366–368. https://doi.org/10.1076/phbi.37.5.366.6050
Senatore F (1996) Composition of the essential oil of Chuquiraga spinosa (R. et P.) D. Don. Pharm Biol 11:215–217. https://doi.org/10.1002/(SICI)1099-1026(199607)11:4%3C215::AID-FFJ574%3E3.0.CO;2-L
Shakoor H, Feehan J, Apostolopoulos V, Platat C, Dhaheri AS, Ali HI, Ismail LC, Bosevski M, Stojanovska L (2021) Immunomodulatory effects of dietary polyphenols. Nutrients 13:1–18. https://doi.org/10.3390/nu13030728
Shen J, Shalom J, Cock IE (2018) The antiproliferative properties of Uncaria tomentosa Willd. DC. extracts against Caco2 and HeLa Cancer Cell Lines. Pharmacogn Commun 8:8–14. https://doi.org/10.5530/pc.2018.1.3
Sheng Y, Li L, Holmgren K, Pero RW (2001) DNA repair enhancement of aqueous extracts of Uncaria tomentosa in a human volunteer study. Phytomedicine 8:275–282. https://doi.org/10.1078/0944-7113-00045
Singh N, Singh S, Maurya P, Arya M, Khan F, Dwivedi DH, Saraf SA (2019) An updated review on Physalis peruviana fruit: cultivational, nutraceutical and pharmaceutical aspects. Indian J Nat Prod Resour 10:97–110. http://nopr.niscpr.res.in/handle/123456789/52785. Accessed 13 May 2021
Sotelo-Córdova SJ (1998) Atividade antiinflamatória da Chuquiraga spinosa Subsp. Huamanpinta em ratos e camundongos. Dissertation, Universidade Estadual de Campinas, UNICAMP – Brazil. https://doi.org/10.47749/T/UNICAMP.1998.123329
Tian W, Xiao N, Yang Y, Xiao J, Zeng R, Xie L, Qiu Z, Li P, Du B (2020) Structure, antioxidant and immunomodulatory activity of a polysaccharide extracted from sacha inchi seeds. Int J Biol Macromol 162:116–126. https://doi.org/10.1016/j.ijbiomac.2020.06.150
Torpocco V, Chavéz H, Estevéz-Braun A, Ravelo AG (2007) New Dammarane Triterpenes from Maytenus macrocarpa. Chem Pharm Bull 55:812–814. https://doi.org/10.1248/cpb.55.812
Ubillas R, Jolad SD, Bruening RC, Kernan MR, King SR, Sesin DF, Barrett M, Stoddart CA, Flaster T, Kuo J, Ayala F, Meza E, Castañel M, Mcmeekin D, Rozhon E, Tempesta MS, Barnard D, Huffman J, Smee D, Sidwell R, Soike K, Brazier A, Safrin S, Orlando R, Kenny PTM, Berova N, Nakanishi K (1994) SP-303, an antiviral oligomeric proanthocyanidin from the latex of Croton lechleri (sangre de drago). Phytomedicine 1:77–106. https://doi.org/10.1016/S0944-7113(11)80026-7
Veloso CC, Soares GL, Perez AC, Rodrigues VG, Silva FC (2017) Pharmacological potential of Maytenus species and isolated constituents, especially tingenone, for treatment of painful inflammatory diseases. Rev Bras Farmacogn 27:533–540. https://doi.org/10.1016/j.bjp.2017.02.006
Verza SG, Silveira F, Cibulski S, Kaiser S, Ferreira F, Gosmann G, Roehe PM, Ortega GG (2012) Immunoadjuvant activity, toxicity assays, and determination by UPLC/Q-TOF-MS of triterpenic saponins from Chenopodium quinoa seeds. J Agric Food Chem 60:3113–3118. https://doi.org/10.1021/jf205010c
Wang S, Zhu F (2019) Chemical composition and health effects of maca (Lepidium meyenii). Food Chem 288:422–443. https://doi.org/10.1016/j.foodchem.2019.02.071
Wang W, Zou Y, Li Q, Mao R, Shao X, Jin D, Zheng D, Zhao T, Zhu H, Zhang L, Yang L, Wu X (2016) Immunomodulatory effects of a polysaccharide purified from Lepidium meyenii Walp. on macrophages. Process Biochem 51:542–553. https://doi.org/10.1016/j.procbio.2016.01.003
Wang S, Zhu F, Kakuda Y (2018) Sacha inchi (Plukenetia volubilis L.): Nutritional composition, biological activity, and uses. Food Chem 265:316–328. https://doi.org/10.1016/j.foodchem.2018.05.055
Wen X, Heller A, Wang K, Han Q, Ni Y, Carle R, Schweiggert R (2020) Carotenogenesis and chromoplast development during ripening of yellow, orange and red colored Physalis fruit. Planta 251:1–14. https://doi.org/10.1007/s00425-020-03383-5
WHO (2020) WHO Coronavirus (COVID-19) Dashboard: WHO (COVID-19). https://covid19.who.int/. Accessed 14 Feb 2022
Winkler C, Wirleitner B, Schroecksnadel K, Schennach H, Mur E, Fuchs D (2004) In vitro effects of two extracts and two pure alkaloid preparations of Uncaria tomentosa on peripheral blood mononuclear cells. Planta Med 70:205–210. https://doi.org/10.1055/s-2004-815536
Wu SJ, Tsai JY, Chang SP, Lin DL, Wang SS, Huang SN, Ng LT (2006) Supercritical carbon dioxide extract exhibits enhanced antioxidant and anti-inflammatory activities of Physalis peruviana. J Ethnopharmacol 108:407–413. https://doi.org/10.1016/j.jep.2006.05.027
Wu L, Zhang M, Xin X, Lai F, Wu H (2019) Physicochemical and functional properties of a protein isolate from maca (Lepidium meyenii) and the secondary structure and immunomodulatory activity of its major protein component. Food Funct 10:2894–2905. https://doi.org/10.1039/c8fo02490a
Yao Y, Shi Z, Ren G (2014a) Antioxidant and immunoregulatory activity of polysaccharides from quinoa (Chenopodium quinoa Willd.). Int J Mol Sci 15:19307–19318. https://doi.org/10.3390/ijms151019307
Yao Y, Yang X, Shi Z, Ren G (2014b) Anti-inflammatory activity of saponins from quinoa (Chenopodium quinoa Willd.) seeds in lipopolysaccharide-stimulated RAW 264.7 macrophages cells. J Food Sci 79:H1018–H1023. https://doi.org/10.1111/1750-3841.12425
Yepes-Perez AF, Herrera-Calderón O, Oliveros CA, Flórez-Álvarez L, Zapata-Cardona MI, Yepes L, Aguilar-Jimenez W, Rugeles MT, Zapata W (2021) The Hydroalcoholic Extract of Uncaria tomentosa (cat’s claw) inhibits the infection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in vitro. J Evid-Based Complementary Altern Med 2021:1–11. https://doi.org/10.1155/2021/6679761
Yu Y, Chen X, Zheng Q (2019) Metabolomic profiling of carotenoid constituents in Physalis peruviana during different growth stages by LC-MS/MS technology. J Food Sci 84:3608–3613. https://doi.org/10.1111/1750-3841.14916
Zevallos VF, Ellis HJ, Šuligoj T, Herencia LI, Ciclitira PJ (2012) Variable activation of immune response by quinoa (Chenopodium quinoa Willd.) prolamins in celiac disease. Am J Clin Nutr 96:337–344. https://doi.org/10.3945/ajcn.111.030684
Zha Z, Wang SY, Chu W, Lv Y, Kan H, Chen Q, Zhong L, Yue L, Xiao J, Wang Y, Hongping Y (2018) Isolation, purification, structural characterization and immunostimulatory activity of water-soluble polysaccharides from Lepidium meyenii. Phytochemistry 147:184–193. https://doi.org/10.1016/j.phytochem.2018.01.006
Zhang M, Wang G, Lai F, Wu H (2016) Structural characterization and immunomodulatory activity of a novel polysaccharide from Lepidium meyenii. J Agric Food Chem 64:1921–1931. https://doi.org/10.1021/acs.jafc.5b05610
Zhang M, Wu W, Ren Y, Li X, Tang Y, Min T, Lai F, Wu H (2017) Structural characterization of a novel polysaccharide from Lepidium meyenii (Maca) and analysis of its regulatory function in macrophage polarization in vitro. J Agric Food Chem 65:1146–1157. https://doi.org/10.1021/acs.jafc.6b05218
Zhou Y-Y, Wang D-W, Yan Y-M, Cheng Y-X (2021) Lignans from Lepidium meyenii and their anti-inflammatory activities. Chem Biodivers 18:2–9. https://doi.org/10.1002/cbdv.202100231
Author information
Authors and Affiliations
Contributions
EMCP: conceptualization, methodology, bibliographic survey, choice of the medicinal plants, writing, formatting, reviewing, and editing; SSC: writing, reviewing, and editing; OHC: reviewing and editing. All authors have read and approved the final submitted version.
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peñaloza, E.M.C., Costa, S.S. & Herrera-Calderon, O. Medicinal Plants in Peru as a Source of Immunomodulatory Drugs Potentially Useful Against COVID-19. Rev. Bras. Farmacogn. 33, 237–258 (2023). https://doi.org/10.1007/s43450-023-00367-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43450-023-00367-w