Abstract
There is a gap in knowledge about the biodiversity of living foraminifera (protozoa) in Brazil, particularly in coastal environments. In Sepetiba Bay (SE Brazil), a coastal system highly impacted by anthropogenic activities, several previous studies have been carried out on foraminifera based on total (living + dead) assemblages and sub-fossil records. Thus, this study intends to analyze for the first time the species richness of living foraminifera (stained with Rose Bengal) in Sepetiba Bay. Based on the morphological characteristics, 214 living species were identified in 50 samples collected in Sepetiba Bay in May 2022. The number of living specimens was counted as 6548. Living foraminifera density was < 252 specimens per gram of sediment and the species richness (S) ranged from 15 to 61 (mean 16.9 ± 15.5) in the analyzed stations. The main taxonomic classes found were Globothalamea and Tubothalamea. The main Globothalamea taxa belonged to the genus Ammonia, with Ammonia tepida, Ammonia buzasi, and Ammonia rolshauseni standing out, as well as the Bolivina, Buliminella, and Elphidium, represented mainly by Bolivina striatula, Buliminella elegantissima, and Elphidium excavatum. A non-Metric Multidimensional Scaling (nMDS) and cluster analysis based on a presence-absence matrix and Bray–Curtis similarity index allowed the identification of two main groups of stations: 1. Located in the innermost areas of Sepetiba Bay and surrounding the mainland and Marambaia Barrier Island, at shallower depths, with lower diversity; 2. an outermost group of stations with higher diversity in areas with greater oceanic influence. Compared with previous studies, the results of this work suggest an enlargement of the “innermost” zone with less diversity towards the oceanic region of Sepetiba Bay. This work shows that even using a matrix of presence/absence of living foraminifera and species richness, it is possible to distinguish environments in coastal systems. Data based on the living foraminifera is needed to understand current biodiversity to better characterize coastal environments and carry out biomonitoring studies.
Similar content being viewed by others
1 Introduction
The pollution and degradation of coastal and transitional waters have been the focus of many studies in recent decades, with the main causes being anthropogenic activities such as mining, the operation of industrial complexes, the burning of fossil fuels, the emission of domestic liquid effluents and port activities (Baptista Filho et al., 2019; Duleba et al., 2019; Potratz et al., 2019). In response to concerns about environmental degradation, many nations have promulgated legislation to monitor the problems caused by the impact of anthropogenic pollution and study remediation measures, such as the Clean Water Act (CWA) or Oceans Act in the USA, Australia or Canada, the Water Framework Directive (WFD, 2000/60/EC) and the Marine Strategy Framework Directive, 2008/56/EC, in Europe. In Brazil, the resolution of CONAMA No 454/2012, for example, establishes the general guidelines and reference procedures for managing material to be dredged in waters under national jurisdiction.
Brazil has several highly impacted coastal regions, such as the coast of São Paulo (Duleba et al., 2019) and Guanabara Bay (Nunes et al., 2023). However, one of the most highly impacted regions in SE Brazil is Sepetiba Bay (Díaz Morales et al., 2019; Silva et al., 2022a, 2022b a, b). Pollution problems in Sepetiba Bay after the 1950s have been very problematic (Barcellos et al., 1997; Copeland et al., 2003; Freret-Meurer et al., 2010; Ribeiro et al., 2013; Ferreira & Moreira, 2015; Araújo et al., 2017; Alves Martins et al., 2019a, 2019b; Díaz Morales et al., 2019; Castelo et al., 2021a, 2021b a, b; Quaresma et al., 2021; Souza et al., 2021). Therefore, it is important to monitor these regions to be able to assess and follow the evolution of the state of ecological quality of coastal systems, particularly Sepetiba Bay.
A bioindicator can be defined as an organism or set of organisms, or biological components, such as cells, biochemical processes, structures, or biological functions, which make it possible to characterize the state of an ecosystem and show the first natural and/or anthropogenic modifications (Zaghloul et al., 2020). In coastal and transitional waters, some organisms have been considered useful bioindicators for assessing environmental quality, including macroalgae, phytoplankton, seagrass (Ballesteros et al., 2007), fish (Coates et al., 2007) and benthic macrofauna (Rincón et al., 2008). Among the organisms recognized as bioindicators, benthic foraminifera (meiofaunal protozoa) have been increasingly used to recognize degraded environments (e.g., Alve, 1995; Bouchet et al., 2018; Francescangeli et al., 2020). Due to their rapid response to environmental stimuli (e.g., increase in organic matter, oxygen shortage, changes in the quality of organic matter, pollution), they reflect the environmental conditions of the area where they live (Martins et al., 2015a, 2015b; Murray, 2006 a). These organisms are sensitive to variations in the environment (Charrieau et al., 2018; Mendes et al., 2010), to sedimentary change (Koho et al., 2007; Rostami et al., 2023), to low concentrations of dissolved oxygen and pH changing (Jorissen et al., 1995; Sen Gupta & Machain-Castillo, 1993), the quantity and quality of available food (Dessandier et al., 2015, 2016; Koho et al., 2008; Martins et al., 2015a) and pollution by metals (Martins et al., 2010, 2013) as well as by organic compounds (Frontalini et al., 2020). Benthic foraminiferal assemblages change, registering alterations in the composition and structure of their communities 1–2 months after the change in the environment occurs (Gooday, 1988).
Benthic foraminifera are easily found in ocean sediments and have the advantage over other organisms of having great fossilization potential (Kucera & Kennet, 2000; Kawagata, 2001). Their fossil record is generally abundant in oceanic and transitional environments (Arz et al., 1999). The study of dead assemblages, subfossils and fossils provides important information that allows past environmental reconstructions (Alve, 1991; Debenay & Fernandez, 2009; Hayward et al., 2004). In addition, they become excellent paleoenvironmental indicators in the sedimentary record, since their tests have the potential to be preserved in the sedimentary record (Francescangeli et al., 2018). In polluted coastal areas, they have been successfully used to distinguish pre-impacted periods (reference conditions) from those subjected to high environmental stress (Francescangeli et al., 2016; Hess et al., 2020; Jesus et al., 2020).
However, the knowledge of the living fauna of foraminifera is still very limited in tropical environments, especially in coastal environments in Brazil. Most of the work in Sepetiba Bay has been based on studying the total (living + dead) or subrecent/fossil fauna. Examples include the work of Tinoco (1965), Zaninetti et al. (1976, 1977), Suguio et al. (1979), Zaninetti (1979), Moura et al. (1982), Brönnimann et al. (1981a, 1981b, 1981c a, b, c), Beurlen and Hiltermann (1983), Brönnimann and Zaninetti (1984), Dias-Brito et al. (1988), Tinoco (1995), Laut et al. (2006, 2012), Laut and Rodrigues (2011). Brönnimann et al. (1981a), based on the total assemblages of foraminifera (living plus dead) in 176 samples from Sepetiba Bay distinguished four sectors: Normal Sea, Normal Marine Lagoon, Transitional Zone 1, Mixo-haline Lagoon A and Mixo-haline Lagoon B (Figure S1).
The total assemblages’ analysis (living + dead) may include tests transported from oceanic areas as bottom or suspended load (Schönfeld et al., 2012). As a result, it is not yet known exactly what foraminiferal fauna is living in Sepetiba Bay. Thus, the main aim of this study is to analyze the biodiversity of living benthic foraminifera (stained with Rose Bengal) in this coastal system. This work is important because it provides a species checklist database that can be used in studies to assess environmental quality in Sepetiba Bay and in other coastal systems and to reconstruct and understand the fluctuations in paleoclimate and sea level.
2 Study area
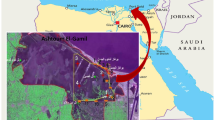
Sepetiba Bay covering ≈300 km2, has a semicircular configuration partially enclosed to the south by a sandy strip, the Marambaia Barrirer Island (known as Restinga da Marambaia). This barrier island was shaped after the last glaciation and separates the bay from the Atlantic Ocean. The semi-enclosed configuration of Sepetiba Bay influences the hydrodynamic conditions, isolating the inner part of the bay and protecting this environment from the direct influence of marine processes (Paraquetti et al., 2004).
The inflow of seawater and the freshwater supplied by the rivers have a significant effect on the distribution of salinity within the bay. It is connected to the ocean via two channels (each about 2 km wide) located in opposite parts of the system. The first channel, in the western part of the inlet, is natural, while the second, in the eastern sector, is artificial and connects the Barra de Guaratiba region to the ocean.
With an area of 2,165 km2, the hydrographical basin of Sepetiba Bay receives fresh water from nine main rivers, including the Guandu, Guandu-Mirim and Guarda rivers (Dourado et al., 2012). These rivers contribute an annual freshwater flow of approximately 7.6 million m3 (Lacerda et al., 2001). The Guandu River supplies most of the drinking water for the metropolitan region of Rio de Janeiro (Lacerda et al., 2004).
The hydrodynamics of Sepetiba Bay are controlled by the fluvial contribution and by the wind and tidal action. The main mechanism responsible for generating currents in Sepetiba Bay is the tide (Signorini, 1980), which overrides the influence of the winds. The effect of the tide on currents is quite evident, with maximum speeds of around 1 m/s. The cold and dense ocean water enters Sepetiba Bay as a bottom current and becomes warmer and less dense in the inner region, rising and leaving the bay as a surface current (Borges & Nittrouer, 2016). The water renewal time in the inner region of Sepetiba Bay is around 100 h (Paraquetti et al., 2004).
The bay is considered shallow, given its average depth of 6 m, except in the channels, where the depth is greater and can reach 31 m between Guaíba Island and Marambaia Island (GTZ, 2001).
The sedimentation process in Sepetiba Bay is governed by a mixture of fluvial and marine materials; however, it also receives the contribution of material transported by coastal drift (Barcellos et al., 1997). Sediments of fluvial origin are predominant in the eastern inner region, while the marine contribution to the coastal deposits occurs mainly in the western part (Barcellos et al., 1997). In addition, Sepetiba Bay receives domestic and industrial effluents, especially heavy metals from urban discharges and the metallurgical industry (Amado Filho et al., 1999).
3 Materials and methods
This work is based on studying the species found in the living assemblages of 50 samples collected in Sepetiba Bay in May 2022 (Fig. 1). The sites sampled were georeferenced using a GPS (GPSMAP® 78S model). WGS84 coordinates were used (Table 1; Appendix 1). The depth of the sites studied was estimated using an echosounder.
The samples for the study of foraminifera were stained and preserved with a solution of alcohol and rose Bengal (2 g of dye per 1 of 90º ethyl alcohol). Once in the laboratory, the samples were washed with a 63 µm sieve to remove the fine fraction and excess dye. Foraminifera were analyzed in the sedimentary fraction > 125 µm. Whenever possible, we tried to sort at least 100 specimens of living foraminifera at each sampling station. However, at a few stations, less than 100 living specimens were found. The species richness (number of species per sample) was determined. The density (or abundance) of living foraminifera per sample was calculated per gram of bulk sediment.
The species found were identified using references such as Loeblich and Tappan (1987), Boltovskoy et al. (1980), Jones (1994), Yassini and Jones (1995), Alves Martins et al., 2019a, 2019b), and Disaró et al. (2022). The species names and taxonomy were updated according to the World Marine Species Database (Hayward et al., 2023). Some main species were described and photographed using a scanning electron microscope.
3.1 Statistical analysis
A presence-absence matrix based on the living foraminifera species found at the sampling stations was analyzed by non-Metric Multidimensional Scaling (nMDS) and cluster analysis (with the complete linkage method) using the Bray–Curtis similarity index. The nMDS ordinated the stations according to their similarity concerning the presence/absence of species. The cluster analysis (CA) grouped the stations most similar in terms of the species' presence/absence. These statistical analyses were performed using Primer software (version 6.1.13, Plymouth; Clarke & Gorley, 2006). The groups of stations established by the nMDS and CA (Figs. 2, 3) were mapped (Figs. 4 and 5).
Cluster analysis based on the presence-absence matrix of the living foraminifera species found in Sepetiba Bay (Appendix 1)
Map of distribution of stations included in Zone A (in red), and Zone B (in blue) resulting from the ordering carried out by non-Metric Multidimensional Scaling (nMDS; Fig. 2)
Map of distribution of stations included in Group 1 (in blue), and Group 2 (in red) resulting from the Cluster Analysis (Fig. 3)
4 Results
Foraminifera density was: less than 10 specimens per gram of bulk sediment at most stations (31 stations; 62% of the analyzed sites); varied between 10 and 30 specimens per gram of bulk sediment at 12 stations (24% of the analyzed sites) and; ranges between 30 and 65 specimens per gram of bulk sediment at 6 stations (12%). Only in one station was found 252 specimens per gram of bulk sediment.
Two hundred and fourteen living species were identified in the studied samples (Appendices 1 and 2). It should be noted that foraminifers belong to the Kingdom Chromista, Subkingdom Harosa, Infrakingdom Rhizaria, and Phylum Foraminifera. The main taxonomic classes found in Sepetiba Bay are Globothalamea and Tubothalamea (Appendix 2). The main Globothalamea taxa belonged to the Ammonia genus, with Ammonia tepida (found in 50 stations), Ammonia rolshauseni (found in 34 stations) and Ammonia buzasi (found in 19 stations) and standing out, as well as to the Bolivina, Buliminella and Elphidium genera, represented above all by Bolivina striatula (found in 23 stations), Buliminella elegantissima (found in 33 stations) and Elphidium excavatum (found in 27 stations).
Compared to Globothalamea, the Tubothalamea class has a lower representation in Sepetiba Bay. It has Quinqueloculina as its main representative genus, where the most representative species are Quinqueloculina seminulum (found in 21 stations), Quinqueloculina bosciana (found in 18 stations), and Quinqueloculina lamarckiana (found in 9 stations).
Species richness (S) ranged from 15 to 61 (mean 16.9 ± 15.5). The highest S values (> 50) were found at stations located in the outer sector of Sepetiba Bay (SP5, SP50 and SP8). S values between 43–18 were found at 16 stations located in the external and central areas of the bay (near and to the west of Itacuruça Island). S values < 15 were found in (31) stations located near the mainland, namely near Madeira Island and Itacuruça, in front of the mouths of the rivers, flanking the Marambaia Barrier Island and Ponta da Pombeba.
4.1 Statistical results
The non-Metric Multidimensional Scaling (nMDS) analysis allowed us to identify two main zones in Sepetiba Bay (Fig. 2). The nMDS-Zone A includes stations located in the inner region of the bay and flanking the mainland (Figs. 2, 4). The nMDS-Zone B encompasses external and centrally-located stations (Figs. 2, 4). The stations of the nMDS-Zone B have the greatest species richness. Specimens from Quinqueloculina, Trifarina, Rosalina and Nonionella are commonly found in this zone.
Two main groups of stations were also identified by Cluster analysis (CA) based on the presence-absence matrix of the living foraminifera species found in Sepetiba Bay (Appendix 1; Fig. 3). The CA-Group 1 includes stations that are present in the nMDS-Zone B and are located in the external and central region of Sepetiba Bay (Figs. 3, 5). The CA-Group 2 encompasses the innermost stations of Sepetiba Bay and contains the stations of the nMDS-Zone A (Figs. 3, 5), with the lowest species richness.
4.2 Systematics and classification of the most frequent species
Some of the most abundant species in the study area are mentioned and described below. Plates 1 and 2 contain photos of the main species found during the sampling period (May 2022).
Photos of the main species of foraminifera found in Sepetiba Bay. 1: Ammonia buzazi; 2: Ammoniatepida; 3: Amonia roshauseni; 4: Bolivina ordinaria; 5: Bolivina striatula; 6: Bolivina striatula (Side/vertical view); 7: Bulimina aculeata; 8: Buliminella elegantissima; 9: Cancris auricula; 10: Criboelphidium poeyanum; 11-12: Elphidium excavatum; 13: Hopkinsina pacifica; 14: Nonion fabum; 15: Nonionella auris
List of the main species of foraminifera found in SB. 1: Quinqueloculina bosciana; 2: Quinqueloculina lamarckiana; 3: Quinqueloculina seminulum; 4: Rosalina globularis; 5: Rosalina bradyi; 6: Rosalina williansoni; 7: Siphonaperta aspera; 8: Spirillina vivipara; 9: Textularia earlandi; 10: Trochammina hadai; 11: Amobaculites exiguus; 12: Rectocibicides miocenicus (R. miocenicus specimen attached to a quartz grain)
Ammobaculites exiguus Cushman & Brönnimann, 1948
Cushman, J. A., Brönnimann, P., 1948. Additional new species of arenaceous Foraminifera from shallow waters of Trinidad. Contribution to Laboratory for Foraminiferal Research, 24(2), 37–42.
Description: Agglutinating species, initially planispiral, later becoming uniserial. The planispiral portion is involute, with 3 or more chambers visible externally. The uniseriate portion extends from the initial coiling; it is rectilinear, with a rounded periphery. The sutures are straight and depressed on the uniseriate part. It has a single terminal aperture without a lip. Wall with poorly sorted, medium to coarse grains, including quartz and other minerals.
Ecology notes: Ammobaculites exiguus has been recorded in transitional environments (Ishiwada, 1958).
Ammonia buzasi Hayward and Holzmann, 2021 (in Hayward et al., 2021)
Hayward, B.W., Holzmann, M., Pawlowski, J., Parker, J.H., Kaushik, T., Toyofuku, M.S., Tsuchiya, M., 2021. Molecular and morphological taxonomy of living Ammonia and related taxa Foraminifera and their biogeography. Micropaleontology, 67, 109–313.
Description: The test is trochospiral and globose, with a relatively high convex lateral profile and rounded periphery. Spire with 3 or 4 turns. The slightly inflated chambers gradually increase, all visible on the dorsal side. On the ventral side, only the chambers of the last turn are visible; on this side, the chambers irregularly project towards the umbilical region; the central umbilical area is filled with numerous small tubercles and sometimes with pustules. The dorsal side may have limbate sutures slightly elevated above the surface of the test; on the umbilical face, the sutures are excavated and depressed. The aperture is a simple extra-umbilical interiomarginal slit.
Ecology notes: Ammonia buzasi was recently described in Cuba (Hayward et al., 2021). This study reports its occurrence in Brazil for the first time.
Ammonia rolshauseni (Cushman & Bermúdez, 1946)
Original name: Rotalia rolshauseni Cushman & Bermúdez, 1946
Cushman, J.A., Bermúdez, P.J. 1946. A new genus, Cribropyrgo, and a new species of Rotalia. Contr. Cushman Lab. Foram. Res. 22, 119, 120.
Synonymized with: Asterorotalia rolshauseni (Cushman & Bermúdez, 1946), Rolshausenia rolshauseni (Cushman & Bermúdez, 1946).
Description: The test is free, biconvex, trochospiral, with contour lobate. The spiral has 3 or 4 turns. Sutures depressed, slightly curved and retroverted; calcareous wall, finely perforated. The aperture is interiomarginal.
Ecology notes: Occurrence in less saline and nutrient-rich waters (Yamashita, 2020).
Ammonia tepida (Cushman, 1926)
Nome original: Rotalia beccarii var tepida Cushman, 1926.
Cushman, J.A. 1926 Description: Agglutinating species of the Carnegie Institution of Washington, 342, 73–84.
Description: The test is trochospiral, globose, and biconvex and has a rounded periphery. Spire with 3 or 4 turns. The slightly inflated chambers gradually increase and are all visible on the dorsal side. On the ventral side, only the chambers of the last turn are visible. The dorsal side may have limbate sutures; on the umbilical side, the sutures are excavated and depressed. The umbilicus is open and depressed, partially covered with extensions of the chambers; sometimes occupied by a small carbonated protrusion. The aperture is a simple extra-umbilical interiomarginal slit.
Ecology notes: It is a frequent species in transitional environments due to its markedly euryhaline behavior (Boltovskoy et al., 1980). It tolerates pollution and oxygen scarcity (Alves Martins et al., 2020b; Raposo et al., 2016, 2018).
Bolivina ordinaria (Phleger & Parker, 1952)
Pheleger & Parker; Pheleger, F. B. & Parker, F. L., Ecology of foraminifera northwest Gulf of Mexico: Part II – Foraminifera species. Geol. Soc. Amer. Mem., New York, n.º 46, pt. 2, p. 14, pl. 7, figs. 4-6.
Bolivina ordinaria Pheleger & Parker; Boltovskoy et al., 1980, p. 18, pl. 3, figs. 1-3.
Bolivina ordinaria Pheleger & Parker; Denne & Sen Gupta, 1991, p. 176, pl. 1, fig. 6.
Description: The test, small and compressed, is elliptical in cross-section, rounded at the oral end and acute at the apical end. Its maximum length is along the midline, as is its greatest thickness. It has 7 to 10 pairs of narrow, elongated and arched chambers. The wall is translucent to opaque. The sutures are depressed, wide, and curved and make an angle of 50-60º with the growth axis. The aperture is narrow and small; it extends from the basal suture and has a lip.
Ecology notes: It is common on the continental shelf of Argentina and southern Brazil (South Atlantic; Boltovskoy et al., 1980). It is a species tolerant to oxygen scarcity (Hermelin & Shimmield, 1990).
Bolivina striatula Cushman, 1922
Cushman, J. A., 1922. Shallow water foraminifera of the Tortugas region. Carnegie Inst. Washington, Publ. n.º 311, (Dept. Marine Biol., Papers, vol. 17), Washington, D. C., U. S. A., p. 27, pl. 3, fig. 10.
Bolivina striatula Cushman; Boltovskoy et al., 1980, p. 18, pl. 3, figs. 9-13.
Bolivina striatula Cushman; Jannink, 2001, p. 36-37, pl. 2, figs. 1, 2.
Description: B. striatula has a compressed, elongated test with a subangular peripheral margin and a broad, rounded oral termination. Numerous thin, longitudinal ribs emerge from the acute aboral termination, which are eventually reduced to striations; these reach only half or less than half of the test. The acute apical termination often has a spine. The test can consist of 7 to 10 pairs of chambers. The translucent wall is finely perforated. The depressed sutures form an angle of around 60º with the growth axis. A lip surrounds the oval aperture.
Ecology notes: It is a frequent species in oxygen-poor environments rich in organic matter (Lutze & Coulbourn, 1984).
Bulimina aculeata d’ Orbigny, 1826
Orbigny, A. D. d’, 1826. Tableau méthodique de la classe des Céphalopodes. Ann. Sci. Nat., Paris, France, 1826, sér. 1, tome 1, p. 269. Figure in: Parker, Jones & Brady, Ann. Mag. Nat. Hist., London, England, 1871, ser. 4, vol. 8, pl. 11, Fig. 128.
Bulimina aculeata d’Orbigny; Boltovskoy et al., 1980, p. 19, pl. 5, figs, 1–3
Bulimina aculeata d’Orbigny; Jones, 1994, p. 56, pl. 51, figs. 7–9
Description: The test consists of a spiral with 4 to 5 turns. The chambers of the last turns are inflated and occupy most of the test, with a protruding lower edge marked by small thick spines; the aboral end of the test is sharp and carries one or more terminal spines; the transparent, white and shiny wall is finely perforated; the sutures are depressed, and the aperture is relatively large.
Ecology notes: B. aculeata is an infaunal species (Schmiedl et al., 2000). It is associated with calm sedimentation environments and a high organic matter flow (Mackensen et al., 1990).
Buliminella elegantissima (d’ Orbigny, 1839)
Original name: Bulimina elegantissima d’Orbigny, 1839
Orbigny, 1839. A. d’, Voyage dans l’Ámérique Méridionale; Foraminiféres. Strasbourg, France, Levrault, 1839, tome 5, pt. 5, p. 51, pl. 7, figs. 13–14. Buliminella elegantissima (d’Orbigny); Boltovskoy et al., 1980, p. 21, pl. 6, figs. 7–10.
Description: The fusiform test is obtuse at its oral end and pointed at its initial end; the spiracle has two to three turns. The elongated and curved chambers are narrow and tilted obliquely to the axis of elongation. The smooth, finely perforated wall has depressed sutures; the small, rounded aperture has a slight depression on the oral face.
Ecology notes: This species is tolerant to anthropogenic pollution (Snyder, 1990).
Cancris auricula (Fichtel & Moll, 1798)
Original name: Nautilus auriculus Fichtel & Moll, 1798
Synonymized with: Cancris auriculatus de Monfort, 1808
Fichtel, L. von & Moll, J. P. C. von, 1798. Testacea microscopica aliaque minuta ex generibus Argonauta et Nautilus (Microscopische und andere kleine Schalthiere aus den Geschlechteern Argonaute und Schiffer). Vienna: Anton Pichler, p. 108.
Cancris auricula (Fichtel & Moll); Loeblich & Tappan, 1987, p. 545, pl. 591, figs. 1-4.
Description: The inequilateral and biconvex test is generally elongated. The trochospiral winding consists of two or more turns. The spiral side is evolute. The umbilical side may have an open umbilicus. It has 7 to 8 chambers in the last turn; these grow rapidly, and the last chamber can occupy two-thirds of the test. The sutures on the spiral side are backward and flat, and on the ventral side, they are depressed, radial, rectilinear, or slightly arched. The umbilical-extraumbilical aperture is located below a wide, imperforate lip projecting over the umbilicus. The peripheral margin is carinate. The semi-hyaline wall is perforated, especially in the outermost area of the chambers.
Ecology notes: It is a common species in oceanic environments of the Western South Atlantic (Disaró et al., 2022).
Cribroelphidium poeyanum (d'Orbigny, 1839)
Original name: Polystomella poeyana d’Orbigny, 1826
Orbigny, A. D. d'. (1839). Foraminifères, in de la Sagra R., Histoire physique, politique et naturelle de l'ile de Cuba. A. Bertrand. 1–224.
Synonymized with: Cribrononion miyakoense Ujiié & Kusukawa, 1969, Elphidium subgranulosum Asano, 1938.
Description: The test is involute planispiral and with a rounded periphery. The umbilical region is slightly depressed; the chambers grow rapidly as they are added. The sutures are slightly depressed and have short, narrow septal bridges. The wall is finely perforated carbonate. The areal aperture consists of basal and scattered pores on the oral face.
Ecology notes: The species is found in various coastal environments in Brazil, from restricted environments such as the Saquarema Lagoon (Debenay et al., 2001) to continental shelf environments between 4.5 and 58 m deep, in places with 0.7% to 13.9% organic matter (Disaró et al., 2022).
Deuterammina discorbis (Earland, 1934)
Original name: Trochammina discorbis Earland, 1934
Earland, A. (1934). Foraminifera. Part III. The Falklands sector of the Antarctic (excluding South Georgia). Discovery Reports, University Press, Cambridge, 10 (1935): p 1–208 (p. 104, Figs. 28–31).
Description: Trochospiral test, rounded in outline, made up of 3–4 turns, with four or five chambers in the last turn. All the turns can be seen on the dorsal side. The ventral side shows the chambers of the last turn and a deep umbilicus. The sutures are depressed and recurved on the dorsal side and straight on the ventral side. The peripheral edge is rounded but compressed. The aperture on the ventral side is a slit at the end of the last chamber. The wall is finely agglutinated with a lot of cement. The test has a rusty brown color and a diameter of around 60 µm.
Ecology notes: Reported by Alves Martins et al. (2019b) for the inner shelf of Aveiro as a species of low recurrence.
Deuterammina (Deuterammina) minuta Brönnimann and Whittaker, 1988
Brönnimann, P., Whittaker, J. E. (1988). The Trochamminacea of the Discovery Reports. A Review of the Trochamminacea (Protozoa: Foraminiferida) described from South Atlantic and Antarctic Waters by Heron-Allen and Earland (1932) and Earland (1933, 1934, 1936). British Museum (Natural History), 152 p.
Description: Trochospiral test, rounded in outline, made up of 3–4 turns, with 5 to 6 chambers in the last turn. All the turns can be observed on the dorsal side. The ventral side shows the chambers of the last turn and a deep umbilicus. The aperture on the ventral side is an interiomarginal slit at the end of the last chamber. The wall is finely agglutinated with a lot of cement and is made of lithic and biogenic materials. The test has a ferruginous brown color and a diameter of ≈160 µm.
Ecology notes: In the Western South Atlantic, this species is associated with environments impoverished in organic matter; it was found between 2 and 58 m deep (Disaró et al., 2022). It is more frequent in lithoclastic and lithobioclastic sediments and moderately to poorly sorted sands (Disaró et al., 2022).
Elphidium excavatum (Terquem, 1875)
Original name: Polystomella excavata Terquem, 1875
Terquem, O. (1875). Essai sur le classement des animaux qui vivent sur le plage et dans les environs de Dunkerque (premier fascicule). Paris.
Description: Test circular in outline, slightly compressed, peripheral margin rounded and slightly lobed; it has 7–10 inflated chambers in the final turn; it displays a smooth wall with gently curved, depressed sutures, often covered with pustules; it exhibits dimples of variable size and shape; concave umbilical region, covered with pustules; aperture formed by a series of holes at the base of the oral face.
Ecology notes: This cosmopolitan is found in various parts of the world ranging from low to high latitudes due to its good adaptation to a high range of temperature (± -5° to 30 °C) and salinity (20 to 35) (Pregnolato, et al., 2018; Hawkes, 2014; Raposo et al., 2016). It has been reported in several bathymetries but found more abundantly in transitional environments (Alves Martins et al., 2020b). The species' feeding is conditioned by the amount of light available in the environment where it is found (Lintner et al., 2021) and by the quality of the organic matter present in the environment (Alves Martins et al., 2020b).
Hanzawaia concentrica (Cushman, 1918)
Original name: Truncatulina concêntrica Cushman, 1918
Cushman, J. A. (1918). Some Pliocene and Miocene Foraminifera of the Coastal Plain of the United States. U.S. Geological Survey, Bulletin, 676, 1–100.
Description: The trochospiral test is compressed and has an oval or subcircular outline; it is covered in small pores, which give it a rough appearance. The peripheral margin is rounded and lobed. The spiral side is flat. The ventral side is convex, with the umbilicus slightly depressed. The chambers of the last turn are curved and inflated, with an arched flap on the inner edge and on the umbilical side, which partially covers the previous turns. The sutures are depressed. The main aperture begins in a peripheral position, extending into 3 or more chambers along the spiral suture.
Ecology notes: It is an infaunal species; it has been reported at depths ranging from 35 to 200 m, comprising the inner shelf of various regions around the world (Subba Rao et al., 1979; Uchimura et al., 2017; Harikrishnan & Nathan, 2023), in areas with low carbonate content; it occurs both in muddy and sandy sediments (Disaró et al., 2022). Hanzawaia concentrica is indicative of areas with high productivity, such as the resurgence zones that occur in the vicinity of the Cabo Frio region (Disaró et al., 2017), as well as well-oxygenated environments with high hydrodynamics (Teodoro et al., 2010). Laut et al. (2022) reported the positive association of this species with high concentrations of biopolymers.
Hopkinsina pacifica Cushman, 1933
Cushman, J. A. (1933). Some new Recent Foraminifera from the tropical Pacific. Contributions from the Cushman Laboratory for foraminiferal research, 9(4): 77–95.
Description: Small, elongated, fusiform test, slightly twisted and laterally compressed. The cross-section is oval to subcircular. The first stage of coiling is triserial, later becoming biserial. The biserial chambers are inflated, and tall and rapidly increase as they are added. The sutures are depressed, straight and slightly oblique. Carbonate wall with very fine pores. The aperture is located in a projection of the last chamber; it is oval and has a tooth.
Ecology notes: An infaunal species typical of shallow inner shelf environments under the influence of rivers (Duijnstee et al., 2003; Ernst et al., 2005; Langlet et al., 2014). It tends to occupy the first few cm of sediment but makes upward migratory movements according to the availability of organic matter during times of anoxia (Ernst et al., 2005; Langlet et al., 2014). In Brazil, it has been identified in areas with low carbonate content (both muddy and sandy sediments). It has been reported as an indicator species of high productivity, being frequent in the Cabo Frio resurgence region (Disaró et al., 2017). Langlet et al. (2014) point out that H. pacifica prefers fresh food and has opportunistic behavior, being one of the most resistant species to oxygen scarcity.
Nonion faba (Fichtel & Moll, 1798)
Original name: Nautilus faba Fichtel & Moll, 1798.
Fichtel, L. von & Moll, J. P. C., von, 1798. Testacea microscópica aliaque minuta ex generibus Argonauta et Nautilus (Microscopische und andere kleine Schalthiere aus den Geschlechtern Argonaute und Schiffer). Vienne: Anton Pichler, 1798, p. 103, pl. 19.
Nonion fabum (Fichtel & Moll); Loeblich & Tappan, 1987 p. 617, pl. 690, figs. 1–7.
Nonion fabum (Fichtel & Moll); Jones, 1994, p. 108, pl. 109, figs. 12–13.
Description: The test is planispiral, bilaterally symmetrical, involute and displays a depressed umbilicus. The chambers rapidly increase in length as they are added. The distinct sutures are depressed near the umbilical zone and complanate and limbate on the periphery of the test. The wall, calcareous and finely perforated, is mostly smooth, except in the umbilical region and also along the sutures, as well as in the central area of the test, where it has pustules. The narrow, interiomarginal aperture is located at the base of the oral face and is partially covered with pustules.
Some authors have also designated this species as Nonion boueanum (d'Orbigny), and as Nonion asterizans (Fichtel & Moll). Jones (1994) and Levy et al. (1995) discussed the synonymy of this species.
Ecology notes: Infaunal species, highly adaptable to spring blooms, able to migrate and settle between surface and subsurface due to its high capacity to support different microhabitats, being able to feed both on fresh phytodetritus, but also on other types of food (Fontanier et al., 2023).
Nonionella auris (d'Orbigny, 1839)
Original name: Valvulina auris d'Orbigny, 1839
Orbigny, A. D. d'. (1839). Voyage dans l'Amérique Méridionale. Foraminifères. t. 5 pt. 5: 1–86.
Description: Large, robust test, ovate on their outer contour; peripheral margin subcarinate, slightly lobed; with 9–11 inflated chambers, slightly curved in the last turn. They have lobed and digitate extensions above the umbilicus, on the involute side; spiral side partially evolute, flattened; wall smooth, white, thin and densely perforated; sutures very depressed, especially near the umbilicus; apertural face broad, triangular, convex; with a distinct aperture.
Ecology notes: Infaunal species, with a preference for muddy sediments; it is a detritivorous, free-living taxon that inhabits depths ranging from 10 to 1000 m; it can settle in areas with normal salinity, but also in environments with lower salinities (Bernasconi & Cusminsky, 2015).
Pararotalia sarmientoi (Redmond, 1953)
Original name: Rotalia sarmientoi Redmond, 1953
Redmond, C. D., 1953. Miocene Foraminifera from the Tubara Beds of Northern Colombia. Journal of Paleontology, 27(5), 708–733.
Description: The test has a low trochospiral shape and comprises 2 to 3 turns and 6 to 9 chambers in the last turn. The chambers have slightly depressed, radial to oblique and curved sutures. The chambers’ size increases as they are added. The test has a subcircular outline, a lobed margin and a subacute to carinate periphery. The umbilicus is open, with a knob; the umbilical side has triangular chambers with projections over the umbilical region. Carbonate wall, opaque, porous, with small papillae. The test has an interiomarginal aperture with a lip.
Notes on the ecology: It is common between 2 and 45 m deep in sediments with variable amounts of organic matter (1%-13.9%; Disaró et al., 2022). It is common in the Western South Atlantic and the Caribbean Sea (Disaró et al., 2022).
Pyrgo williamsoni (Silvestri 1923)
Original name: Biloculina williamsoni Silvestri, 1923.
Silvestri, A., 1923. Microfauna pliocenica rizopodi reticolari di Capocolle presso Forli. Atti della Pontificia Accademia della Scienze Nuovi Lincei, Roma (1922–1923). 76: 70–77.
Synonym of Biloculina ringens (Lamarck, 1804).
Description: Bilocular and biconvex test with an elongated oval outline and a rounded peripheral margin. There is a small gap between the last 2 chambers, the only ones visible externally. Sutures are depressed. The wall is shiny, smooth without pores, and composed of porcelain carbonate. Oval-shaped aperture at the end of the last chamber, with a flat and bifid tooth.
Ecology notes: Epifaunal species typical of oxygenated environments, with a significant supply of organic matter (De & Gupta, 2010; Murray, 1991).
Quinqueloculina bosciana d'Orbigny, 1839
Orbigny, A. D. d'., 1839. Foraminifères, in de la Sagra R., Histoire physique, politique et naturelle de l'Ile de Cuba. A. Bertrand. 1–224.
Description: Quinqueloculine test, oval, elongated, 2 to 2.5 times longer than wide, compressed, truncated at the oral end and rounded at the aboral end, with rounded periphery; chambers half a turn long, convex, not inflated, elongated, slightly protruding at the oral end and expanded at the aboral end, depressed sutures. Porcelain carbonate wall, without pores, smooth and shiny. Terminal aperture oval to circular at the protruding end of the last chamber, with a thin lip not evident externally and with a short, simple tooth.
Ecology notes: Species associated with environments recorded between 2 and 168 m deep in places with relatively high levels of organic matter (between 1.5% and 13.9%; Disaró et al., 2022). It is common in the Western South Atlantic, Caribbean Sea and Gulf of Mexico (Disaró et al., 2022).
Quinqueloculina lamarckiana, d’Orbigny, 1839
Orbigny, A. D. d'. (1839). Foraminifères, in de la Sagra R., Histoire physique, politique et naturelle de l'ile de Cuba. A. Bertrand. 1–224.
Description: Large quinqueloculine test. The margin of the chambers is compressed into a keel. The aperture is a rounded hole with a bifid tooth. The carbonated wall is smooth, imperforate and porcelain.
Ecology notes: Recorded in various locations worldwide (Hofker, 1971); it is frequently found in restricted lagoon and continental shelf environments (Javaux and Scott, 2003).
Quinqueloculina seminulum (Linnaeus, 1758)
Original name: Serpula seminulum Linnaeus, 1758.
Linnaeus, C., 1758. Systema Naturae per regna tria naturae, secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Editio decima, reformata [10th revised edition], vol. 1, 824 pp. Laurentius Salvius: Holmiae.
Description: Elongated quinqueloculine test with a rounded periphery. The aperture at the end of the last chamber is a large, elongated hole with a simple, elongated tooth. The carbonate wall is imperforate and porcelain, with a smooth surface.
Ecology notes: It is a frequent species in coastal environments (Damasio et al., 2020; Martins et al., 2015b, 2016), tolerating high levels of organic matter (Belart et al., 2019; Laut et al., 2022). This species tolerates relatively high concentrations of metals, such as Zn, Cd and Pb, as observed in Guanabara Bay (Alves Martins et al., 2020b).
Rectocibicides miocenicus Cushman and Ponton, 1932
Cushman, J. A. & Ponton, G. M., 1932. Some interesting new Foraminifera from the Miocene of Florida. Contributions from the Cushman Laboratory for Foraminiferal Research, 8(1), 1–4.
Description: Encrusting compressed test; the first stage of development is spiral, later becoming uniserial; at this stage, the chambers may form one or two lineaments. The chambers are wider than long; they are separated by sutures, limbed and slightly depressed. Hyaline, translucent carbonate wall with numerous pores. The multiple apertures, located at the base of the oral face, are composed of subcircular holes surrounded by a rim.
Ecology notes: This species is common on the inner shelf and is frequent in shallow waters (Uchio, 1960) and in environments with high hydrodynamics (Disaró, 2014).
Rosalina bradyi (Cushman, 1915)
Original name: Discorbis globularis var. bradyi Cushman, 1915
Cushman, J. A., 1915. A monograph of the Foraminifera of the North Pacific Ocean. Part V. Rotaliidae. Bulletin of the United States National Museum, 71 (5), 1–87.
Description: Low trochospiral test, with an approximately circular outer contour; the chambers of the last turn are all visible on the spiral side; they rapidly increase in size as they are added. The chambers are inflated. The aperture is located on the ventral side of the test. Carbonaceous wall is perforated and smooth.
Ecology notes: This species is commonly found in shallow environments rich in organic matter (Dubois et al., 2021) but also in rocky environments, algal substrates and coral reefs (Hayward et al., 1999; Murray, 2006).
Rosalina globularis d'Orbigny, 1826
Loeblich, A. R., Tappan, H. N., 1994: Foraminifera of the Sahul Shelf and Timor Sea. Cushman Foundation for Foraminiferal Research, Special Publication 31. 661 pp. Plate 286, Figs. 13–15.
Description: The vitreous shell is slightly trochoid, with a circular peripheral contour, more or less lobed. The margin is rounded or slightly acute. Spirally arranged chambers with perforated walls form the dorsal side. On the dorsal side, the sutures are flat in the early stages of the test and slightly depressed in the adult portion. The initial chamber is visible on the dorsal side; the ventral side is flat, with radial sutures. Extensions of the chamber wall partially cover the open umbilicus. The aperture is a slit situated along the suture of the last chamber; on the umbilical side, apertures of the chambers of the last turn can be seen. The innermost turns, on the evolute side, are characteristically reddish-brick or reddish-pink. This species can be more or less irregular as the individuals adapt during growth to the shape of the substrate to which they adhere.
Ecology notes: It is an epiphytic species (Coppa & Di Tuoro, 1995) that shows predatory activity when there is a food shortage (Murray, 2006). Its distribution is related to the availability of nutrients, often occurring on algal beds (Sliter, 1965).
Rosalina williamsoni (Chapman and Parr, 1932)
Original name: Discorbis williamsoni Chapman and Parr, 1932
Williamson, W. C., 1958. On the recent foraminifera of Great Britain. Ray Soc., London, England, 1858, p. 54, pl. 4, figs. 106-108.
Parr, W. J. (1932). Victorian and South Australian shallow-water foraminifera. Part II. Proceedings of the Royal Society of Victoria, 44, 218-234
Discorbis williamsoni Chapman & Parr; Boltovskoy et al., 1980, p. 28, pl. 12, figs. 5-12.
Description: The test is plano-convex, with a carinated subacute peripheral margin and a circular outer contour. The spiral side shows a winding with two or three turns. On the ventral side, the chambers are triangular with a rounded umbilical margin. The chambers are well-defined but not globose. The smooth, transparent vitreous wall may or may not be perforated. The sutures are slightly depressed, radial and curved on the umbilical side. The open umbilicus is covered by extensions of the chambers. The aperture is an interiomarginal slit.
Ecology notes: Rosalina williamsoni is an epifaunal species (Corliss, 1991). The distribution of this species is directly associated with dynamic environments (Teodoro et al., 2010).
Siphonaperta aspera (d'Orbigny, 1826)
Original name: Quinqueloculina aspera d'Orbigny, 1826
Orbigny, A. D. d'., 1826. Tableau méthodique de la classe des Céphalopodes. Annales des Sciences Naturelles, 7, 96–169, 245–314.
Description: The arrangement of the chambers in the test is of the "quinqueloculine" type. The wall contains agglutinated materials and is imperforate. The test is oval in lateral view. The chambers are elongated longitudinally and subrounded in cross-section. The aperture is oval, delimited by a non-agglutinated edge, and provided with a relatively long bifid tooth. The wall is agglutinated with fine-grained material.
Ecology notes: it is common in the infralittoral zone, where the sediment is composed of medium sand (Magno et al., 2012), but is rarely associated with seaweed (Di Bella et al., 2012).
Spirillina vivipara Ehrenberg, 1843
Ehrenberg, C.G. (1843). Verbreitung und Einfluss des mikroskopischen Lebens in Süd-und Nord-Amerika. Abhandlungen der Königlichen Akademie der Wissenschaften zu Berlin, 1841, 291–445, 4 pls.
Description: The carbonaceous and hyaline test is formed by a simple proloculus and an elongated tubular chamber, spirally coiled, which gradually grows in size; this chamber has no septa and in cross-section is subrounded; the spiral suture is slightly depressed. A simple aperture is located at the end of the tubular chamber.
Ecology notes: It has a wide distribution in neritic environments, preferring waters up to 26 °C (Murray, 2013), and can survive in hypersaline environments (Murray, 2006).
Textularia earlandi Parker, 1952
Parker, F. L., 1952. Foraminiferal distribution in the Long Island Sound – Buzzards Bay area. Harvard Coll., Mus. Comp. Zoöl., Bull., vol. 106 (1951-1952), nº 10, p. 458
Synonymized with: Textularia tenuissima Earland, 1933 and Textularia elegans Lacroix, 1932.
Textularia earlandi Parker; Alve & Murray, 1994, pp. 22, 24, pl. 1, fig. 14.
Description: The test is much longer than wide, with an initial planispiral coiling followed by a biseriate arrangement in which the chambers are distributed in opposite pairs; the chambers gradually increase in size as they are added. The peripheral margin is rounded and lobed. The thin, pale-yellow wall is made up of particles of varying sizes. The depressed, narrow sutures make a 90º angle with the growth axis. The aperture consists of a small interiomarginal arch on the convex oral surface.
Ecology notes: It is a eurythermal and euryhaline species, frequent in neritic and bathyal environments; it can withstand large variations in salinity and temperature (Alve and Nagy, 1990), showing resilience and the potential to survive environmental stress (Alve & Goldstein, 2010). It is common in the South Atlantic continental shelf (Boltovskoy and Totah, 1985).
Trochammina hadai Uchio, 1962
Uchio, T., 1962. Influence of the River Shinano on foraminifera and sediment grain size distribution. Publications of the Seto Marine Biological Laboratory, 10, 363–392.
Description: Trochospiral test, essentially made up of poorly sorted quartz grains, agglutinated with brownish cement. The test is biconvex, with inflated chambers, all visible on the spiral side; the chambers of the last turn are visible on the dorsal side; the last chambers grow significantly and are quite inflated, especially the last one, sometimes giving the test morphological diversity. The aperture is umbilical.
Ecology notes: Trochammina hadai has been found in the Santos and Campos basins and the Flamengo Cove, in the latter as an invasive species in recent sediments (Sousa et al., 2017).
5 Discussion
This study found a smaller number of foraminifera (214) than previous studies carried out in the Guaratiba / Sepetiba Bay mangrove complex, in which 264 species of benthic foraminifera were recorded (Laut et al., 2012). However, the current study does not include the Guaratiba mangrove swamp or dead foraminifera, which contains a record of past faunas and specimens transported from other areas (Schönfeld et al., 2012).
It can, therefore, be considered that the diversity of foraminifera in Sepetiba Bay is relatively high when compared to other Brazilian coastal systems. For example, Belert et al. (2018) only found 8 living species in the Saquarema Lagoon System, Filippos et al. (2023) found 23 species in the area surrounding the diffusers of a domestic sewage outfall at Cigarras Beach; Raposo et al. (2022) found 49 living species in the Cachoeira Estuary; Rodrigues et al. (2014) found 88 taxa in Flamengo Inlet, Ubatuba, São Paulo State; Alves Martins et al. (2020b) identified 52 living benthic foraminifera in the NE region of Guanabara Bay and Nunes et al. (2023) identified 94 living foraminiferal taxa in other regions of this bay.
The same can be observed when comparing the number of species identified in Sepetiba Bay with those recorded in other regions of the world. For example, Al-Enezi et al. (2022) identified (45 stations) in Kuwait Bay, Arabian Gulf 107 species of living foraminifera Dimiza et al. (2016) identified (22 stations) in Elefsis Bay and Saronikos Gulf, Greece, Eastern Mediterranean, 55 species of living foraminifera; Frontalini et al. (2020) identified in 32 stations surrounding three offshore gas platforms (Armida – AA, Garibaldi A—GA and Agostino B—AB) in front of the Ravenna coast, Adriatic Sea, 32 species of living foraminifera; Martins et al. (2015a) identified in 53 stations located along the channels of Aveiro Lagoon (Portugal) 76 species of living foraminifera.
The most common species in Sepetiba Bay found in this work are Ammonia tepida, Ammonia rolshauseni, Buliminella elegantissima, Elphidium excavatum, Bolivina striatula, Textularia earlandi, Quinqueloculina seminulum, Trochammina hadai and Ammonia buzasi. Most of these species have been found in other Brazilian coastal systems, such as South Bay, Florianópolis (Rudorff et al. (2012), Flamengo Inlet (Eichler et al., 2018), Ubatuba Bay, Flamengo (Rodrigues et al., 2014), São Sebastião Channel (Duleba et al., 2018) Cigarras Beach (Filippos et al., 2023), Guanabara Bay (Alves Martins et al., 2020b; Nunes et al., 2023), Itaipu Lagoon (Raposo et al., 2018), Saquarema Lagoon (Belart et al., 2018, 2019), Vermelha Lagoon (Laut et al., 2022), Almada Estuary (Laut et al., 2021), and Cachoeira Estuary (Raposo et al., 2022). Trochammina hadai is an invasive species that was recently found in the Flamengo Cove (Eichler et al., 2018; Sousa et al., 2017). Ammonia buzasi was recently identified through genetic analysis in Sepetiba Bay, having not been identified in Brazil before (Damasceno et al., 2024).
Considering the analyzed data and the statistical results (by CA and nMDS), two main zones can be considered in Sepetiba Bay. The stations of CA-Group 1 (Figs. 3) and zone B of the nMDS (Fig. 2) correspond to the central and outermost stations of Sepetiba Bay (Figs. 4, 5), which are characterized by higher species richness (S), indicating greater diversity. In this region the marine influence is higher, the water renewal by tidal currents is effective (Kjerfve et al., 2021), the hydrodynamics is stronger (Cunha et al., 2006; Santos et al., 2018) and therefore the sediments are rich in oxygen (Schratzberger & Ingels, 2018). In this outer sector of Sepetiba Bay can be found species that are typical of transitional environments, such as that belonging to Ammonia genus (Holzmann & Pawlowski, 2000), as well as species typical of neritic environments such as Rosalina williamsoni, Rosalina bradyi, Quinqueloculina bosciana, Nonionella auris, Globocassidulina crassa and Hanzawaia concentrica (Disaró, 2014, 2017, 2022). These species prefer oxygenated substrates with organic matter of high environmental quality (Martins et al., 2015a) under higher marine influence.
The stations of CA-Group 2 (Figs. 3, 5), and zone A the nMDS (Figs. 2, 4) are located in the inner region and close to the margins of Sepetiba Bay, with lower species richness (S). In this region the waters have lower salinity due to the greater fluvial influence, especially from the Guandú River (Molisani et al., 2006). This region is relatively more confined since the Marambaia Barrier Island and several islands reduce communication with the ocean. Pollution has greater expression in this zone of the bay (Carvalho et al., 2022; Lacerda et al., 2004; Magalhães et al., 2001; Molisani et al., 2004; Rodrigues et al., 2020). The significant reduction in the number of living species in the stations of the CA-Group 2 (Figs. 3, 5; Appendix 1) is probably related to the high environmental stress caused by the variability of physicochemical parameters (Kjerfve et al., 2021). The discharge of domestic sewage into the inner region of the bay (Molisani et al., 2004) can contribute to the decrease in the sediment pore-water pH, hindering the calcification of the tests of various species (Dias et al., 2010; Dong et al., 2020). In addition, the large accumulation of organic matter on the bottom can stimulate the proliferation of decomposer organisms and contribute to a reduction in the availability of oxygen in the bottom sediment, allowing colonization only by taxa resistant to lower oxygen conditions, such as Ammonia and Buliminella (Alves Martins et al., 2019a). These genera are essentially composed of opportunistic species that survive in environments with high levels of organic matter and withstand a sharp decrease in oxygen and variations in salinity (Donnici et al., 2012; Koho et al., 2018).
Several authors have tried to distinguish different environments in Sepetiba Bay based on the distribution of foraminifera (e.g., Brönnimann et al., 1981a; Oliveira-Silva, 2003; Suguio et al., 1979). Suguio et al. (1979) identified a biotope (C), located mainly between the islands of Itacuruçá and Jaguanúm, the Marambaia inlet and the transitional portion to the outer sector of the bay, corresponding to a large extent of what in this study was called the nMDS-Zone B, the zone of greatest diversity. Although the zones identified in this work, based on the biodiversity of living foraminifera, cannot be strictly compared with the zones defined by Brönnimann et al. (1981a) and Oliveira-Silva (2003), also based on variations in foraminiferal diversity (but on living + dead specimens; Figs. S1 and S2), it can be deduced that there has been an expansion of the most "inner" zone (nMDS-Zone A; Figs. 4, 5), encompassing the transitional Zone 1, mixohaline lagoon A and mixohaline lagoon B, of Brönnimann et al., (1981a; Fig. S1).
6 Conclusion
This study, based on the analysis of 50 sampling stations in Sepetiba Bay (May 2022), shows that the biodiversity of living foraminifera is significant (in 6548 counted specimens were found 214 living species). Most of the foraminifera belonged to the Class Globothalamea, with emphasis on the genera Ammonia, namely Ammonia tepida, Ammonia buzasi, and Ammonia rolshauseni, and the genera Bolivina, Buliminella, and Elphidium, represented mainly by Bolivina striatula, Buliminella elegantissima, and Elphidium excavatum. A comparison of the number of species found in Sepetiba Bay with those found in other Brazilian and global coastal systems shows that the diversity in this bay is relatively high.
Based on a species presence-absence matrix and multivariate statistical analyses, two main zones were identified in Sepetiba Bay: the "inner" zone with the lowest species richness and the "central outer" zone, located in the region most closely connected to the ocean, with the highest diversity. The results of this work, compared with previous (performed in the 1980s) foraminiferal biofacies mappings, suggest an extension of the “internal” zone with less diversity towards the oceanic region of Sepetiba Bay.
Data based on the living assemblages of foraminifera is important for knowing the effective biodiversity and distinguishing different environments, influenced, for instance, by water depth, salinity, water renovation, sediment characteristics, and eutrophication processes, among others.
Availability of data and materials
The data supporting this work are available in tables and as supplementary materials.
References
Al-Enezi, E., Francescangeli, F., Balassi, E., Borderie, S., Al-Hazeem, S., Al-Salameen, F., Anwar, A. B., Pawlowski, J., & Frontalini, F. (2022). Benthic foraminifera as proxies for the environmental quality assessment of the Kuwait Bay Kuwait, Arabian Gulf: Morphological and metabarcoding approaches. Science of the Total Environment, 833, 155093. https://doi.org/10.1016/j.scitotenv.2022.155093
Alve, E. (1991). Benthic foraminifera in sediment cores reflecting heavy metal pollution in Sørfjord, Western Norway. Journal of Foraminiferal Research, 21, 1–19.
Alve, E. (1995). Benthic foraminiferal distribution and recolonization of formerly anoxic environments in Drammensfjord, southern Norway. Marine Micropaleontology, 25, 169–186. https://doi.org/10.1016/0377-8398(95)00007-N
Alve, E., & Goldstein, S. T. (2010). Dispersal, survival and delayed growth of benthic foraminiferal propagules. Journal of Sea Research, 63(1), 36–51. https://doi.org/10.1016/j.seares.2009.09.003
Alve, E., & Murray, J. W. (1994). Ecology and taphonomy of benthic foraminifera in a temperate mesotidal inlet. Journal of Foraminiferal Research, 24(1), 18–27.
Alve, E., & Nagy, J. (1990). Main features of foraminiferal distribution reflecting estuarine hydrography in Oslo Fjord. Marine Micropalaeontology, 16, 181–206. https://doi.org/10.1016/0377-8398(90)90003-5
Alves Martins, M. V., Hohenegger, J., Frontalini, F., Sequeira, C., Miranda, P., Rodrigues, M. A. C., et al. (2019b). b). Foraminifera checklist and the main species distribution in the Aveiro Lagoon and Adjacent Continental Shelf (Portugal). Journal of Sedimentary Environments, 4, 1–52. https://doi.org/10.12957/jse.2019.39308
Alves Martins, M. V., Hohenegger, J., Martínez-Colón, M., Frontalini, F., Bergamashi, S., Laut, L., Belart, P., Mahiques, M., Pereira, E., Rene Rodrigues, R., Terroso, D., Miranda, P., Geraldes, M. C., Villena, H. H., Reis, T., Socorro, O. A. A., Sousa, S. H. M., Yamashita, C., & Rocha, F. (2020b). Ecological quality status of the NE sector of the Guanabara Bay (Brazil): A case of living benthic foraminiferal resilience. Marine Pollution Bulletin, 158, 111–449. https://doi.org/10.1016/j.marpolbul.2020.111449
Alves Martins, M. V., Pereira, E., Figueira, R. C. L., Oliveira, T., Pinto Simon, A. F. S., Terroso, D., Ramalho, J. C. M., Silva, L., Ferreira, C., Geraldes, M. C., Duleba, W., Rocha, F., & Rodrigues, M. A. (2019a). Impact of eutrophication on benthic foraminifera in Sepetiba Bay (Rio de Janeiro State, SE Brazil). Journal of Sedimentary Environments, 4(4), 480–500. https://doi.org/10.12957/jse.2019.47327
Alves Martins, M. V., Pinto, A. F. S., Borghi, L., Carelli, T. G., Morloye, M., Rey, D., & Rodrigues, M. A. (2020a). Influence of the Holocene relative sea level on the coastal plain of Sepetiba Bay (Southeast Brazil). Journal of Sedimentary Environments, 5(1), 35–39. https://doi.org/10.1007/s43217-019-00002-6
Amado Filho, G. N., Rezende, C. E., & Lacerda, L. D. (1999). Poluição da Bacia de Sepetiba já ameaça outras áreas. Ciência Hoje, Rio De Janeiro, 25, 46–48.
Araújo, D. F., Peres, L. G. M., Yepez, S., Mulholland, D. S., Machado, W., Tonhá, M., et al. (2017). Assessing man-induced environmental changes in the Sepetiba Bay (Southeastern Brazil) with geochemical and satellite data. Comptes Rendus Geoscience, 349, 290–298. https://doi.org/10.1016/j.crte.2017.09.007
Arz, H. W., Patzold, J., & Weffer, G. (1999). The deglacial history on the western tropical Atlantic as inferred from height resolution stable isotope records off northeastern Brazil. Earth and Planetary Science Letters, 167, 105–117. https://doi.org/10.1016/S0012-821X(99)00025-4
Ballesteros, E., Torras, X., Pinedo, S., García, M., Mangialajo, L., & de Torres, M. (2007). A new methodology based on littoral community cartography dominated by macroalgae for the implementation of the European Water Framework Directive. Marine Pollution Bulletin, 55, 172–180. https://doi.org/10.1016/j.marpolbul.2006.08.038
Baptista Filho, L. S., Baptista Neto, J. A., Alves Martins, M. V., & Geraldes, M. C. (2019). Sources of pollutants in the Northern/Northeast area of Guanabara Bay (SE, Brazil) since the late nineteenth century using lead isotopes and metals concentrations. Journal of Sedimentary Environments, 4(3), 332–349. https://doi.org/10.12957/jheotb.2020.49099
Barcellos, C., Lacerda, L. D., & Ceradini, S. (1997). Sediment origin and budget in Sepetiba Bay (Brazil) - An approach based on multi-element analysis. Environmental Geology, 32, 203–209. https://doi.org/10.1007/s002540050208
Belart, P., Clemente, I., Raposo, D., Habib, R., Volino, E. K., Villar, A., Martins, M. V. A., Fontana, L. F., Lorini, M. L., Panigai, G., Frontalini, F., Figueiredo, M. S. L., Vasconcelos, S. C., & Laut, L. (2018). Living and dead Foraminifera as bioindicators in Saquarema Lagoon System, Brazil. Latin American Journal of Aquatic Research, 46(5), 1055–1072. https://doi.org/10.3856/vol46-issue5-fulltext-18
Belart, P., Habib, R., Raposo, D., Clemente, I., Alves Martins, M. V., Frontalini, F., Figueiredo, M. S. L., Lorini, M. L., & Laut, L. (2019). Seasonal dynamics of benthic foraminiferal biocoenosis in the Tropical Saquarema Lagoonal System Brazil. Estuaries and Coasts, 42(3), 822–841. https://doi.org/10.1007/s12237-018-00514-w
Bernasconi, E., & Cusminsky, G. (2015). Study of the distribution of Elphidium aff poeyanum (D’Orbigny) and Buccella peruviana (D’Orbigny) from the Colorado basin (South America): Holocene paleoenvironmental inferences. The Holocene, 25(5), 810–819. https://doi.org/10.1177/0959683615571424
Beurlen, G., & Hiltermann, H. (1983). As biocenoses de foraminíferos do mangue de Guaratiba, Rio de Janeiro. Brasil. Boletim Técnico Da PETROBRAS, 26(4), 259–267.
Boltovskoy, E., Giussani, G., Watanabe, S. & Wright, R. (1980). Atlas of benthic shelf foraminifera of the Southwest Atlantic. Dr. W. Junk b.v., Publishers, The Hague, 147 pp., pl. 36. E-book ISBN: 978–94–009–9188–0. https://doi.org/10.1016/S0025-3227(03)00044-6
Boltovskoy, E., & Totah, V. (1985). Diversity, similarity and dominance in benthic foraminiferal fauna along one transect of the Argentine shelf. Revue De Micropaléontologie, 28(1), 23–31.
Borges, H. V., & Nittrouer, C. A. (2016). Sediment accumulation in Sepetiba Bay (Brazil) during the Holocene: a reflex of the human influence. Journal of Sedimentary Environments, 1(1), 90–106. https://doi.org/10.12957/jse.2016.21868
Bouchet, V. M. P., Goberville, E., & Frontalini, F. (2018). Foraminifera to assess ecological quality status: The case of salmon fish farming. Ecological Indicators, 117, 106607. https://doi.org/10.1016/j.ecolind.2020.106607
Brönnimann, P., Moura, J.A., Dias-Brito, D. (1981c). Ecologia dos Foraminíferos e Microrganismos Associados da Área de Guaratiba/Sepetiba: Modelo Ambiental e sua Aplicação na Pesquisa de Hidrocarbonetos. Relatório 3549, PETROBRAS, 81p
Brönnimann, P., Whittaker, J. E. (1988). The Trochamminacea of the Discovery Reports. A Review of the Trochamminacea (Protozoa: Foraminiferida) described from South Atlantic and Antarctic Waters by Heron-Allen and Earland (1932) and Earland (1933, 1934, 1936). British Museum (Natural History), 152
Brönnimann, P., Moura, J. A., & Dias-Brito, D. (1981a). Estudos Ecológicos na Baía de Sepetiba, Rio de Janeiro, Brasil: Foraminíferos. Congresso Latino-Americano de Paleontologia (pp. 75–861). RS: Porto Alegre.
Brönnimann, P., Moura, J. A., & Dias-Brito, D. (1981b). Foraminíferos da Fácies Mangue da Planície de Maré de Guaratiba, Rio de Janeiro, Brasil. Congresso Latino-Americano de Paleontologia (pp. 91–861). RS: Porto Alegre.
Brönnimann, P., & Zaninetti, L. (1984). Agglutinated foraminifera, mainly Trochamminacea from the Baía de Sepetiba, near Rio de Janeiro. Brazil. Revue De Paléobiologie, 3(1), 63–115.
Carvalho, A. C. B., Moreira, V. A., Vicente, M. C., Bidone, E. D., Bernardes, M. C., & Sabadini-Santos, E. (2022). Sterol and PAHs fingerprint analysis of organic matter at Southeast Brazilian Bay. Marine Pollution Bulletin, 181, 113899. https://doi.org/10.1016/j.marpolbul.2022.113899
Castelo, W. F. L., Alves Martins, M. V., Ferreira, P. A. L., Figueira, R., Costa, C. F., Fonseca, L. B., et al. (2021b). Long-term eutrophication and contamination of the central area of Sepetiba Bay SW Brazil. Environmental Monitoring and Assessment, 193, 100. https://doi.org/10.1007/s10661-021-08861-1
Castelo, W. F. L., Alves Martins, M. V., Martínez-Colón, M., Guerra, J. V., Dadalto, T. P., Terroso, D., et al. (2021a). Disentangling natural vs. anthropogenic induced environmental variability during the Holocene: Marambaia Cove, SE sector of the Sepetiba Bay SE Brazil. Environmental Science Pollution Research, 28, 22612–22640. https://doi.org/10.1007/s11356-020-12179-9
Charrieau, L. M., Filipsson, H. L., Ljung, K., Chierici, M., Knudsen, K. L., & Kritzberg, E. (2018). The effects of multiple stressors on the distribution of coastal benthic foraminifera: A case study from the Skagerrak-Baltic Sea region. Marine Micropaleontology, 139, 42–56. https://doi.org/10.1016/j.marmicro.2017.11.004
Clarke, K. R., & Gorley, R. N. (2006). PRIMER v6: User Manual/Tutorial (Plymouth Routines in Multivariate Ecological Research). Plymouth: PRIMER-E.
Coates, S., Waugh, A., Anwar, A., & Robson, M. (2007). Efficacy of a multi-metric fish index as an analysis tool for the transitional fish component of the Water Framework Directive. Marine Pollution Bulletin, 55(1–6), 225–240. https://doi.org/10.1016/j.marpolbul.2006.08.029
Copeland, G., Monteiro, T., Couch, S., & Borthwick, A. (2003). Water quality in Sepetiba Bay, Brazil. Marine Environmental Research, 55, 385–408. https://doi.org/10.1016/S0141-1136(02)00289-1
Coppa, M. G., & Di Tuoro, A. (1995). Preliminary data on the Holocene Foraminifera of the Cilento continental shelf (Tyrrhenian Sea). Revista Española De Paleontología, 10(2), 161–174. https://doi.org/10.7203/sjp.24128
Corliss, B. H. (1991). Morphology and microhabitat preferences of benthic foraminifera from the northwest Atlantic Ocean. Marine Micropalaeontology, 17, 195–236. https://doi.org/10.1016/0377-8398(91)90014-W
Cunha, C. L. N., Rosman, P. C. C., Ferreira, A. P., & Monteiro, T. C. N. (2006). Hydrodynamics and water quality models applied to Sepetiba Bay. Continental Shelf Research, 26, 1940–1953. https://doi.org/10.1016/j.csr.2006.06.010
Cushman, J. A., & Bermúdez, P. J. (1946). A new genus, Cribropyrgo, and a new species of Rotalia. Contributions from the Cushman Laboratory for Foraminiferal Research, 22(119), 120.
Cushman, J. A., & Brönnimann, P. (1948). Additional new species of arenaceous Foraminifera from shallow waters of Trinidad. Contribution to Laboratory for Foraminiferal Research, 24(2), 37–42.
Damasceno, F. L., Alves Martins, M. V., Frontalini, F., et al. (2024). Assessment of the ecological quality status of the Sepetiba Bay (SE Brazil): When metabarcoding meets morphology on foraminifera. Marine Environmental Research, 195, 106340. https://doi.org/10.1016/j.marenvres.2024.106340
Damasio, B. V., Timoszczuk, C. T., Kim, B. S. M., Sousa, S. H. D. M. E., Bícego, M. C., Siegle, E., & Figueira, R. C. L. (2020). Impacts of hydrodynamics and pollutants on foraminiferal fauna distribution in the Santos Estuary (SE Brazil). Journal of Sedimentary Environments, 5, 61–86. https://doi.org/10.1007/s43217-020-00003-w
De, S., & Gupta, A. K. (2010). Deep-sea faunal provinces and their inferred environments in the Indian Ocean based on distribution of Recent benthic foraminifera. Palaeogeography, Palaeoclimatology, Palaeoecology, 291(3–4), 429–442. https://doi.org/10.1016/j.palaeo.2010.03.012
Debenay, J.-P., & Fernandez, J.-M. (2009). Benthic foraminifera records of complex anthropogenic environmental changes combined with geochemical data in a tropical bay of New Caledonia (SW Pacific). Marine Pollution Bulletin, 59, 311–322. https://doi.org/10.1016/j.marpolbul.2009.09.014
Debenay, J. P., Geslin, E., Eichler, B. B., Duleba, W., Sylvestre, F., & Eichler, P. (2001). Foraminiferal assemblages in a hypersaline lagoon, Araruama (RJ) Brazil. Journal of Foraminiferal Research, 31(2), 133–151. https://doi.org/10.2113/0310133
Denne, R. A., & Sen Gupta, B. K. (1991). Association of bathyal foraminifera with water masses in the northwestern Gulf of Mexico. Micropaleontology, 17, 173–193. https://doi.org/10.1016/0377-8398(91)90013-V
Dessandier, P.-A., Bonnin, J., Kim, J.-H., Bichon, S., Deflandre, B., Grémare, A., & Damsté, J. S. (2016). Impact of organic matter source and quality on living benthic foraminiferal distribution on a river-dominated continental margin: A study of the Portuguese margin. Journal of Geophysical Research, Biogeosciences, 121, 1689–1714. https://doi.org/10.1002/2015JG003231
Dessandier, P.-A., Bonnin, J., Kim, J.-H., Bichon, S., Grémare, A., Deflandre, B., De Stigter, H., & Malaizé, B. (2015). Lateral and vertical distributions of living benthic foraminifera off the Douro River (western Iberian margin): Impact of the organic matter quality. Marine Micropaleontology, 120, 31–45. https://doi.org/10.1016/j.marmicro.2015.09.002
Di Bella, L., Bergamin, L., & Frezza, V. (2012). Environmental changes by mean the foraminiferal assemblages in the Late Quaternary deposits of the Terracina basin (Central Tyrrhenian Sea, Italy). Journal of Mediterranean Earth Sciences. https://doi.org/10.3304/JMES.2012.002
Dias, B. B., Hart, M. B., Smart, C. W., & Hall-Spencer, J. M. (2010). Modern seawater acidification: The response of foraminifera to high-CO2 conditions in the Mediterranean Sea. Journal of Geological Society, 167, 843–846. https://doi.org/10.1144/0016-76492010-050
Dias-Brito, D., Moura, J., & Wurding, N., et al. (1988). Relationships between ecological models based on ostracods and foraminifers from Sepetiba Bay (Rio de Janeiro – Brazil). In T. Hanai (Ed.), Evolutionary biology of Ostracoda: Its fundamentals and applications Developments in Paleontology and Stratigraphy (pp. 467–485). Elsevier.
Díaz Morales, S. J., Guerra, J. V., Nunes, M. A. S., Souza, A. M., & Geraldes, M. C. (2019). Evaluation of the environmental state of the western sector of Sepetiba Bay (SE Brazil): trace metal contamination. Journal of Sedimentary Environments, 4, 174–188. https://doi.org/10.12957/jse.2019.43764
Dimiza, M. D., Triantaphyllou, M. V., Koukousioura, O., Hallock, P., Simboura, N., Karageorgis, A. P., & Papathanasiou, E. (2016). The Foram Stress Index: A new tool for environmental assessment of soft-bottom environments using benthic foraminifera. A case study from the Saronikos Gulf, Greece. Eastern Mediterranean. Ecological Indicators, 60, 611–621. https://doi.org/10.1016/j.ecolind.2015.07.030
Disaró, S. T. (2014). Caracterização da plataforma continental da Bacia de Campos (Brasil, SE) fundamentada em foraminíferos bentônicos recentes. PhD Thesis, Instituto de Geociências, Universidade Federal do Rio Grande do Sul.
Disaró, S.T., Aluizio, R., Ribas, E.R., Pupo, D.V., Tellez, I.R., Watanabe, S., Totah, V.I., & Koutsoukos, E.A.M. (2017). Foraminíferos bentônicos na plataforma continental da Bacia de Campos. In: Falcão, A.P.C., Lavrado, H.P. (ed.), Ambiente Bentônico: caracterização ambiental regional da Bacia de Campos, Atlântico Sudoeste. Rio de Janeiro: Habitats, vol. 3. p. 65–110.
Disaró, S.T., Totah, V.I., Watanabe, S., Ribas, E.R., & Pupo, D.V. (2022). Biodiversidade Marinha da Bacia de Potiguar: Foraminifera. Rio de Janeiro, Museu Nacional/UFRJ, 2022. 264 p. ISNN: 978–65–5729–015–6
Dong, S., Lei, Y., Li, T., & Jian, Z. (2020). Response of benthic foraminifera to pH changes: Community structure and morphological transformation studies from a microcosm experiment. Marine Micropaleontology, 156, 101819. https://doi.org/10.1016/j.marmicro.2019.101819
Donnici, S., Serandrei-Barbero, R., Bonardi, M., & Sperle, M. (2012). Benthic foraminifera as proxies of pollution: The case of Guanabara Bay (Brazil). Marine Pollution Bulletin, 64, 2015–2028. https://doi.org/10.1016/j.marpolbul.2012.06.024
Dourado, F., Cunha, J., Lima, A., Palermo, N. (2012). Os novos empreendimentos na Baía de Sepetiba e o passivo ambiental da CIA mercantil e Industrial Ingá. Baía de Sepetiba: estado da arte. Rio de Janeiro: Corbã, 253–262.
Dubois, A., Barras, C., Pavard, J. C., Donnay, A., Béatrix, M., & Bouchet, V. M. (2021). Distribution patterns of benthic foraminifera in fish farming areas (Corsica, France): Implications for the implementation of biotic indices in biomonitoring studies. Water, 13(20), 2821. https://doi.org/10.3390/w13202821
Duijnstee, I., Ernst, S., & Van der Zwaan, G. (2003). Effect of anoxia on the vertical migration of benthic foraminifera. Marine Ecology Progress Series, 246, 85–94. https://doi.org/10.3354/meps246085
Duleba, W., Gubitoso, S., Alves Martins, M. V., Teodoro, A. C., Pregnolato, L. A., & Prada, S. M. (2019). Evaluation of Contamination by Potentially Toxic Elements (PTE) of Sediments around the Petroleum Terminal Pipeline “Dutos e Terminais do Centro Sul (DTCS)”, SP. Brazil. Journal of Sedimentary Environments, 4(4), 387–402. https://doi.org/10.12957/jse.2019.46539
Duleba, W., Teodoro, A. C., Debenay, J. P., Alves Martins, M. V., Gubitoso, S., et al. (2018). Environmental impact of the largest petroleum terminal in SE Brazil: A multiproxy analysis based on sediment geochemistry and living benthic foraminifera. PLoS ONE, 13(2), e0191446. https://doi.org/10.1371/journal.pone.0191446
Eichler, P. P. B., McGann, M., Rodrigues, A. R., Mendonca, A., Amorim, A., Bonetti, C., Cordeiro de Farias, C., Sousa, S. H. M., Vital, H., & Gomes, M. P. (2018). The occurrence of the invasive foraminifera Trochammina hadai Uchio in Flamengo Inlet, Ubatuba, Sao Paulo State. Brazil. Micropaleontology, 64(5–6), 391–402. https://doi.org/10.47894/mpal.64.6.05
Ernst, S., Bours, R., Duijnstee, I., & van der Zwaan, B. (2005). Experimental effects of an organic matter pulse and oxygen depletion on a benthic foraminiferal shelf community. Journal of Foraminiferal Research, 35, 177–197. https://doi.org/10.2113/35.3.177
Ferreira, A. P., & Moreira, M. D. F. R. (2015). Metals pollution status in surface sediments along the Sepetiba Bay Watershed, Brazil. Journal of Integrated Coastal Zone Management, 18, 404. https://doi.org/10.4172/2473-3350.1000404
Filippos, L. S., Duleba, W., Hohenegger, J., Pregnolato, L. A., Bouchet, V. M. P., & Alves Martins, M. V. (2023). Domestic sewage outfall severely altered environmental conditions, foraminiferal communities, and ecological quality statuses in front of the nearshore beach of cigarras (SE Brazil). Water, 15, 405. https://doi.org/10.3390/w15030405
Fontanier, C., Mamo, B., Dubosq, N., Lamarque, B., Rigaud, S., Schmidt, S., Lebleu, P., Poirier, D., Cordier, M.-A., Grémare, A., & Deflandre, B. (2023). Seasonal variability of living benthic foraminifera from the West-Gironde mud patch (Bay of Biscay, NE Atlantic): Three contrasted periods under the stereomicroscope. Continental Shelf Research, 268(1), 105117. https://doi.org/10.1016/j.csr.2023.105117
Francescangeli, F., Du Chatelet, E. A., Billon, G., Trentesaux, A., & Bouchet, V. M. P. (2016). Palaeo-ecological quality status based on foraminifera of Boulogne-sur-Mer harbour (Pas-de-Calais, Northeastern France) over the last 200 years. Marine Environmental Research, 117, 32–43. https://doi.org/10.1016/j.marenvres.2016.04.002
Francescangeli, F., Portela, M., Du Chatelet, E. A., Billon, G., Andersen, T. J., Bouchet, V. M. P., & Trentesaux, A. (2018). Infilling of the Canche Estuary (eastern English Channel, France): Insight from benthic foraminifera and historical pictures. Marine Micropaleontology, 142, 1–12. https://doi.org/10.1016/j.marmicro.2018.05.003
Francescangeli, F., Quijada, M., Du Châtelet, E. A., Frontalini, F., Trentesaux, A., Billon, G., & Bouchet, V. M. P. (2020). Multidisciplinary study to monitor consequences of pollution on intertidal benthic ecosystems (Hauts de France, English Channel, France): Comparison with natural areas. Marine Environmental Research, 160, 105034. https://doi.org/10.1016/j.marenvres.2020.105034
Freret-Meurer, N. V., Andreata, J. V., Meurer, B. C., Manzano, F. V., Baptista, M. G. S., Teixeira, D. E., & Longo, M. M. (2010). Spatial distribution of metals in sediments of the Ribeira Bay, Angra dos Reis, Rio de Janeiro. Brazil. Marine Pollution Bulletin, 60(4), 627–629. https://doi.org/10.1016/j.marpolbul.2010.01.023
Frontalini, F., Cordier, T., Balassi, E., Armynot du Chatalet, E., Cermakova, K., Apothéloz-Perret-Gentil, L., Martins, M. V. A., Bucci, C., Scantamburlo, E., Treglia, M., Bonamin, V., & Pawlowski, J. (2020). Benthic foraminiferal metabarcoding and morphology-based assessment of the environmental impact of offshore gas platforms: Congruence and complementarity. Environment International, 144, 106049. https://doi.org/10.1016/j.envint.2020.106049
Gooday, A. (1988). A response by benthic Foraminifera to the deposition of phytodetritus in the deep sea. Nature, 332, 70–73. https://doi.org/10.1038/332070a0
GTZ (2021). Fundação Estadual de Engenharia do Meio Ambiente – (FEEMA)/ Deutsche Gesellschaft für Technische Zusammenarbeit (GTZ - Sociedade de Cooperação Técnica). Bacias Hidrográficas e Recursos Hídricos da Macroregião 2 – Bacia da Baía de Sepetiba. Rio de Janeiro. 178
Harikrishnan, S., & Senthil Nathan, D. (2023). Abundance and distribution of recent benthic foraminifera from the southwestern part of the Bay of Bengal. Geosystems and Geoenvironment, 2(4), 100209. https://doi.org/10.1016/j.geogeo.2023.100209
Hawkes, A. (2014). A Study of attached benthic foraminifera associated with the deep-sea coral Primnoa resedaeformis on the Scotian Margin. Ver 1 In OBIS Canada Digital Collections. Bedford Institute of Oceanography, Dartmouth, NS, Canada. Published by OBIS. http://ipt.vliz.be/obiscanada/ Accessed on Fev. 2024.
Hayward, B.W., Le Coze, F., Vachard, D., Gross, O. (2023). World Foraminifera Database. Accessed through: https://www.marinespecies.org/foraminifera/
Hayward, B. W., Grenfell, H. R., Nicholson, K., Parker, R., Wilmhurst, J., Horrocks, M., Swales, A., & Sabaa, A. T. (2004). Foraminiferal record of human impact on intertidal estuarine environments in New Zealand’s largest city. Marine Micropaleontology, 53, 37–66. https://doi.org/10.1016/j.marmicro.2004.03.001
Hayward, B. W., Grenfell, H. R., Reid, C. M., & Hayward, K. A. (1999). Recent New Zealand shallow-water benthic Foraminifera: Taxonomy, ecologic distribution, biogeography, and use in paleoenvironmental assessment. Institute of Geological and Nuclear Sciences Monograph., 21, 1–258.
Hayward, B. W., Holzmann, M., Pawlowski, J., Parker, J. H., Kaushik, T., Toyofuku, M. S., & Tsuchiya, M. (2021). Molecular and morphological taxonomy of living Ammonia and related taxa Foraminifera and their biogeography. Micropaleontology, 67, 109–313. https://doi.org/10.47894/mpal.67.2-3.01
Hermelin, J. O. R., & Shimmield, G. B. (1990). The importance of the oxygen minimum zone and sediment geochemistry in the distribution of Recent benthic foraminifera in the northwest Indian Ocean. Marine Geology, 91, 1–29.
Hess, S., Alve, E., Andersen, T. J., & Joranger, T. (2020). Defining ecological reference conditions in naturally stressed environments – how difficult is it? Marene Environmental Research, 156, 104885. https://doi.org/10.1016/j.marenvres.2020.104885
Hofker, J. (1971). The Foraminifera of Piscadera Bay, Curaçao. Studies on the Fauna of Curaçao and Other Caribbean Islands, 35(1), 1–62.
Holzmann, M., & Pawlowski, J. (2000). Taxonomic relationships in the genus Ammonia (Foraminifera) based on ribosomal DNA sequences. Journal of Micropalaeontology, 19, 85–95. https://doi.org/10.1144/jm.19.1.85
Ishiwada, Y. (1958). Studies on the brackish water. III. Recent Foraminifera from the brackish lake Haman-Ko. Reports, Geological Survey of Japan, 180, 1–19.
Jannink, N. T. (2001). Seasonality, biodiversity and microhabitats in benthic foraminiferal communities. Geologica Ultraiectina, 203, 192.
Javaux, E. J., & Scott, D. B. (2003). Illustration of Modern Benthic Foraminifera from Bermuda and Remarks on Distribution in Other Subtropical/tropical Areas. Palaeontologia Electronica, 6(4), 29.
Jesus, M. S. D. S., Frontalini, F., Bouchet, V. M. P., Yamashita, C., Sartoretto, J. R., Figueira, R. C. L., et al. (2020). Reconstruction of the palaeopaleo-ecological quality status in an anthropogenised estuary using benthic foraminifera: The Santos estuary (Sao Paulo state, SE Brazil). Marine Environmental Research, 162, 105121. https://doi.org/10.1016/j.marenvres.2020.105121
Jones, R. W. (1994). The Challenger Foraminifera (p. 150). London, Oxford University Press.
Jorissen, F. J., De Stigter, H. C., & Widmark, J. G. (1995). A conceptual model explaining benthic foraminiferal microhabitats. Marine Micropaleontology, 26, 3–15. https://doi.org/10.1016/0377-8398(95)00047-X
Kawagata, S. (2001). Tasman Frant shifts and associated paleoceanographic changes during the last 250,000 years: Foraminiferal evidence from the Lord Howe Rise. Marine Micropaleontology, 41, 167–191. https://doi.org/10.1016/S0377-8398(00)00058-X
Kjerfve, B., Dias, G. T. M., Filippo, A., & Geraldes, M. C. (2021). Oceanographic and environmental characteristics of a coupled coastal bay system: Baía de Ilha Grande-Baía de Sepetiba, Rio de Janeiro. Brazil. Regional Studies in Marine Sciences, 41, 101594. https://doi.org/10.1016/j.rsma.2020.101594
Koho, K. A., García, R., De Stigter, H. C., Epping, E., Koning, E., Kouwenhoven, T. J., & Van Der Zwaan, G. J. (2008). Sedimentary labile organic carbon and pore water redox control on species distribution of benthic foraminifera: A case study from Lisbon-Setúbal Canyon (southern Portugal). Progress in Oceanography, 79, 55–82. https://doi.org/10.1016/j.pocean.2008.07.004
Koho, K. A., Kouwenhoven, T. J., de Stigter, H. C., & van der Zwaan, G. J. (2007). Benthic foraminifera in the Nazaré Canyon, Portuguese continental margin: Sedimentary environments and disturbance. Marine Micropaleontology, 66, 27–51. https://doi.org/10.1016/j.marmicro.2007.07.005
Koho, K. A., LeKieffre, C., Nomaki, H., Salonen, I., Geslin, E., Mabilleau, G., Søgaard Jensen, L. H., & Reichart, G.-J. (2018). Changes in ultrastructural features of the foraminifera Ammonia spp. In response to anoxic conditions: Field and laboratory observations. Marine Micropaleontology, 138, 72–82. https://doi.org/10.1016/j.marmicro.2017.10.011
Kucera, M., & Kennett, J. P. (2000). Biochronology and evolutionary implications of Late Neogene California margin planktonic foraminiferal events. Marine Micropaleontology, 40, 67–81. https://doi.org/10.1016/S0377-8398(00)00029-3
Lacerda, L. D., Marins, R. V., Barcellos, C., & Molisani, M. M., et al. (2004). Sepetiba Bay A Case Study of the Environmental Geochemistry of Heavy Metals in a Subtropical Coastal Lagoon. In D. Lacerda (Ed.), Environmental Geochemistry in Tropical and Subtropical Environments (pp. 293–318). Berlin: SpringerVerlag.
Lacerda, L. D., Marins, R. V., Paraquetti, H. H. M., Mounier, S., Benaim, J., & Fevrier, D. (2001). Mercury distribution and reactivity in waters of a subtropical coastal lagoon, Sepetiba Bay, SE Brazil. Journal of the Brazilian Chemical Society, 12, 93–98. https://doi.org/10.1590/S0103-50532001000100013
Langlet, D., Baal, C., Geslin, E., Metzger, E., Zuschin, M., Riedel, B., Risgaard-Petersen, N., Stachowitsch, M., & Jorissen, F. (2014). Foraminiferal species responses to in situ, experimentally induced anoxia in the Adriatic Sea. Biogeosciences, 11, 1775–1797. https://doi.org/10.5194/bg-11-1775-2014
Laut, L., Belart, P., Carelli, T., Martins, M. V. A., & Laut, V. (2022). Insights into the Ecology of Foraminifera from the Most Hypersaline Lagoon in Brazil: Vermelha Lagoon. Estuaries and Coasts, 45(8), 2632–2649. https://doi.org/10.1007/s12237-022-01073-x
Laut, L. M., Koutsoukos, E. A. M., & Rodrigues, M. A. C. (2006). Review of mangrove foraminifera from the Guaratiba tidal plain, Rio de Janeiro, SE Brazil, collected in the early 70’s. Anuário Do Instituto De Geociências UFRJ, 29(1), 427–442.
Laut, L., Matta, G., Camara, C., Belart, P., Clemente, I., Ballalai, J., Volino, E., & Couto, E. C. G. (2021). Living and dead foraminifera assemblages as environmental indicators in the Almada River Estuary, Ilhéus, northeastern Brazil. Journal of South American Earth Sciences, 105, 102883. https://doi.org/10.1016/j.jsames.2020.102883
Laut, L. L. M., & Rodrigues, M. A. C. (2011). Foraminíferos do manguezal de Guaratiba, Rio de Janeiro: Revisão taxonômica e aplicação de análises multivariadas. Paleontologia: Cenários da Vida. Editora Interciência, 3, 231–240.
Laut, L. L. M., Silva, F. S., Martins, V., Rodrigues, M. A. C., Mendonça, J. O., Clemente, I. M. M. M., Laut, V. M., & Mentzigen, L. G. (2012). Foraminíferos do Complexo Sepetiba/Guaratiba. In M. A. C. Rodrigues, S. D. Pereira, & S. B. Santos (Eds.), Baía de Sepetiba: Estado da Arte (pp. 115–150). Corbã.
Levy, A., Mathieu, R., Poignant, A., Rosset-Moulinier, M., Ubaldo, M. L., & Lebreiro, S. (1995). Foraminiferes actuels de la marge continentale portuguaise-inventaire et distribution. Memórias Do Instituto Geológico e Mineiro, Lisboa, 32, 116.
Lintner, M., Lintner, B., Wanek, W., Keul, N., Von der Kammer, F., Hofmann, T., & Heinz, P. (2021). Effects of heavy elements (Pb, Cu, Zn) on algal food uptake by Elphidium excavatum (Foraminifera). Heliyon, 7, 11. https://doi.org/10.1016/j.heliyon.2021.e08427
Loeblich, A. R., & Tappan, H. (1987). Foraminiferal genera and their classification (p. 970). Van Nostrand Reinhold.
Loeblich, A. R., & Tappan, H. N. (1994). Foraminifera of the Sahul Shelf and Timor Sea. Cushman Foundation for Foraminiferal Research, Special Publication, 31, 661.
Lutze, G. F., & Coulbourn, W. T. (1984). Recent benthic foraminifera from the continental margin off Northwest Africa: Community structure and distribution. Marine Micropalaeontology, 8, 361–401. https://doi.org/10.1016/0377-8398(84)90002-1
Mackensen, A., Grobe, H., Kuhn, G., & Fütterer, D. K. (1990). Benthic foraminiferal assemblages from the Eastern Weddel Sea between 68º and 73º S: Distribution, ecology and fossilization potential. Marine Micropalaeontology, 16, 241–283. https://doi.org/10.1016/0377-8398(90)90006-8
Magalhães, V. F., Carvalho, C. E. V., & Pfeiffer, W. C. (2001). Arsenic Contamination and Dispersion in the Engenho Inlet, Sepetiba Bay, SE, Brazil. Water, Air, and Soil Pollution, 129, 83–90. https://doi.org/10.1023/A:1010381902874
Magno, M. C., Bergamin, L., Finoia, M. G., Pierfranceschi, G., Venti, F., & Romano, E. (2012). Correlation between textural characteristics of marine sediments and benthic foraminifera in highly anthropogenically-altered coastal areas. Marine Geology, 315, 143–161. https://doi.org/10.1016/j.margeo.2012.04.002
Marine Strategy Framework Directive (2008/56/CE). Directive 2008/56/EC of the European Parliament and of the Council of 17 June 2008 establishing a framework for community action in the field of marine environmental policy (Marine Strategy Framework Directive) (Text with EEA relevance). https://www.eea.europa.eu/policy-documents/2008-56-ec, accessed in September 2023
Martins, M. V. A., Quintino, V., Tentúgal, R. M., Frontalini, F., Miranda, P., Laut, L. L. M., Martins, R., & Rodrigues, A. M. (2015b). Characterization of bottom hydrodynamic conditions on the central western Portuguese continental shelf based on benthic foraminifera and sedimentary parameters. Marine Environmental Research, 109, 52–68. https://doi.org/10.1016/j.marenvres.2015.06.006
Martins, M. V., Silva, F., Laut, L. L., Frontalini, F., Clemente, I. M., Miranda, P., Figueira, R., Sousa, S. H., & Dias, J. M. A. (2015a). Response of benthic foraminifera to organic matter quantity and quality and bioavailable concentrations of metals in Aveiro Lagoon (Portugal). PLoS ONE, 10, e0118077. https://doi.org/10.1371/journal.pone.0118077
Martins, M. V. A., Soares-Gomes, A., Yamashita, C., Pinto, A. F. S., Frontalini, F., Sequeira, C., Laut, L. L. M., Belart, P., Zaaboub, N., Miranda, P., Sousa, S. H. M., Figueira, F., Pena, A. L., Terroso, D. L., & Rocha, F. (2016). Similarity between the dead and living foraminiferal assemblages of the NE sector of Guanabara Bay (Brazil). Journal of Sedimentary Environments, 1(4), 411–431. https://doi.org/10.12957/jse.2016.26875
Martins, V., Ferreira da Silva, E., Sequeira, C., Rocha, F., & Duarte, A. C. (2010). Evaluation of the ecological effects of heavy metals on the assemblages of benthic foraminifera of the canals of Aveiro (Portugal). Estuarine, Coastal and Shelf Science, 87, 293–304. https://doi.org/10.1016/j.ecss.2010.01.011
Martins, V. A., Frontalini, F., Tramonte, K. M., Figueira, R. C. L., Miranda, P., Sequeira, C., Fernández-Fernández, S., Dias, J. A., Yamashita, C., Laut, L. M., Sobrinho, F., Rodrigues, M. A., Bernardes, C., Nagai, R., Sousa, S. H. M., Mahiques, M., Rubio, B., Bernabeu, A., Rey, D., & Rocha, F. (2013). Assessment of the health quality of Ria de Aveiro (Portugal): Heavy metals and benthic foraminifera. Marine Pollution Bulletin, 70, 18–33. https://doi.org/10.1016/j.marpolbul.2013.02.003
Mendes, I., Rosa, F., Dias, J. A., Schönfeld, J., Ferreira, Ó., & Pinheiro, J. (2010). Inner shelf paleoenvironmental evolution as a function of land–ocean interactions in the vicinity of the Guadiana River, SW Iberia. Quaternary International, 221, 58–67. https://doi.org/10.1016/j.quaint.2009.10.037
Molisani, M. M., Kjerfve, B., Silva, A. P., & Lacerda, L. D. (2006). Water discharger and sediment load to Sepetiba Bay from and anthropogenically-altered drainage basin, SE Brazil. Journal of Hydrology, 331, 425–433. https://doi.org/10.1016/j.jhydrol.2006.05.038
Molisani, M. M., Marins, R. V., Machado, W., Paraquetti, H. H. M., Bidone, E. D., & Lacerda, L. D. (2004). Environmental changes in Sepetiba Bay, SE Brazil. Regional Environmental Change, 4, 17–27. https://doi.org/10.1007/s10113-003-0060-9
Moura, J. A., Dias-Brito, D., & Bronniman, P. (1982). Modelo ambiental de laguna costeira clastica- Baía de Sepetiba. Atlas do Simpósio do Quaternário do Brasil: RJ.
Murray, J. W. (1991). Ecology and Paleoecology of Benthic Foraminifera (p. 397). Longman.
Murray, J. W. (2006). Ecology and Applications of Benthic Foraminifera. Cambridge University Press. https://doi.org/10.1017/CBO9780511535529
Murray, J. W. (2013). Living benthic foraminifera: Biogeographical distributions and the significance of rare morphospecies. Journal of Micropaleontology, 32(1), 1–58. https://doi.org/10.1144/jmpaleo2012-010
CONAMA Nº 454/2012 - Estabelece as diretrizes gerais e os procedimentos referenciais para o gerenciamento do material a ser dragado em águas sob jurisdição nacional. - Data da legislação: 01/11/2012 - Publicação DOU, de 08/11/2012, Seção 1, pág. 66. Status: Revoga as Resoluções nº 344 de 2004 e nº 421 de 2010. http://www2.mma.gov.br/port/conama/Legiabre.cfm?codlegi=693
Nunes, M., Alves Martins, M. V., Frontalini, F., Bouchet, V. M. P., Francescangeli, F., Hohenegger, J., Figueira, R., Senez-Mello, T. M., Castelo, W. F. L., Damasceno, F. L., Laut, L., Duleba, W., Melloe Sousa, S. H., Antonioli, L., & Geraldes, M. C. (2023). Inferring the ecological quality status based on living benthic foraminiferal indices in transitional areas of the Guanabara Bay (SE Brazil). Environmental Pollution. https://doi.org/10.1016/j.envpol.2023.121003
Oliveira-Silva, P. (2003). Biofaciologia, relações ecológicas e paleoecológicas na baía de Sepetiba, Rio de Janeiro, com base na associação de foraminíferos bentônicos recentes. Master Thesis, Universidade do Estado do Rio de Janeiro, 136p
Paraquetti, H. H. M., Ayres, G. A., Almeida, M. D., Molisani, M. M., & Lacerda, L. D. (2004). Mercury distribution, speciation and flux in the Sepetiba Bay tributaries, SE Brazil. Water Research, 38, 1439–1448. https://doi.org/10.1016/j.watres.2003.11.039
Potratz, Gl., Geraldes, M. C., Bizzi, S., Nogueira, L., & Alves Martins, M. V. (2019). Using lead isotopes and potentially toxic elements to trace pollutant sources in the northern region of Guanabara Bay, southeastern Brazil. Marine Pollution Bulletin, 144, 216–223. https://doi.org/10.1016/j.marpolbul.2019.04.057
Pregnolato, L. A., de Almeida Viana, R., Passos, C. C., Misailidis, M. L., & Duleba, W. (2018). Ammonia-Elphidium index as a proxy for marine pollution assessment. Northeast Brazil. Journal of Sedimentary Environments, 3(3), 176–186. https://doi.org/10.12957/jse.2018.38001
Quaresma, V. S., Aguiar, V. M. C., Bastos, A. C., Oliveira, K. S., Vieira, F. V., Sá, F., & Baptista Neto, J. A. (2021). The impact of trace metals in marine sediments after a tailing dam failure: The Fundão dam case (Brazil). Environmental Earth Sciences, 80, 571. https://doi.org/10.1007/s12665-021-09817-x
Raposo, D., Clemente, I., Figueiredo, M., Vilar, A., Lorini, M. L., Frontalini, F., Martins, M. V. A., Belart, P., Fontana, L., Habib, R., & Laut, L. (2018). Benthic foraminiferal and organic matter compounds as proxies of environmental quality in a tropical coastal lagoon: The Itaipu lagoon (Brazil). Marine Pollution Bulletin, 129, 114–125. https://doi.org/10.1016/j.marpolbul.2018.02.018
Raposo, D., Frontalini, F., Clemente, I., et al. (2022). Benthic foraminiferal response to trace elements in a tropical mesotidal Brazilian estuary. Estuaries and Coasts, 45, 2610–2631. https://doi.org/10.1007/s12237-022-01095-5
Raposo, D., Laut, V., Clemente, I., Martins, V., Frontalini, F., Silva, F., Lorini, M. L., Fortes, R., & Laut, L. (2016). Recent benthic foraminifera from the Itaipu Lagoon, Rio de Janeiro (southeastern Brazil). Check List, 12(5), 1959–1959. https://doi.org/10.15560/12.5.1959
Ribeiro, A. P., Figueiredo, A. M., dos Santos, J. O., Dantas, E., Cotrim, M. E., Figueira, R. C., et al. (2013). Combined SEM/AVS and attenuation of concentration models for the assessment of bioavailability and mobility of metals in sediments of Sepetiba Bay (SE Brazil). Marine Pollution Bulletin, 15, 55–63. https://doi.org/10.1016/j.marpolbul.2012.12.023
Rincón, C. H. L., Bolívar, G., Neira, R., & Peña, E. J. (2008). Utilización de la macrofauna bentónica como indicador de calidad ambiental en la desembocadura del Río Anchicaya, Pacífico colombiano. Ingeniería De Recursos Naturales y Del Ambiente, 7, 94–101.
Rodrigues, A.R., Díaz, T.L., & Pellizari, V.H. (2014). Living Foraminifera in a Brazilian Subtropical Coastal Environment (Flamengo Inlet, Ubatuba, São Paulo State–Brazil). In: Kitazato, H., M. Bernhard, J. (eds) Approaches to Study Living Foraminifera. Environmental Science and Engineering. Springer, Tokyo, pp. 195-227
Rodrigues, S. K., Machado, W., Guerra, J. V., Geraldes, M., Morales, S., & Vinzón, S. B. (2020). Changes in Cd and Zn distribution in sediments after closure of an electroplanting industry, Sepetiba Bay. Brazil. Marine Pollution Bulletin, 161, 111758. https://doi.org/10.1016/j.marpolbul.2020.111758
Rostami, A. M., Frontalini, F., Armynot du Châtelet, E., Francescangeli, F., Alves Martins, M. V., et al. (2023). Understanding the distributions of benthic foraminifera in the Adriatic Sea with Gradient Forest and Structural Equation Models. Applied Sciences, 13(2), 794. https://doi.org/10.3390/app13020794
Rudorff, N. M., Bonetti, C. H. C., & Bonetti Filho, J. (2012). Suspended shellfish culture impacts on the benthic layer: A case study in Brazilian subtropical waters. Brazilian Journal of Oceanography, 60(2), 219–232. https://doi.org/10.1590/S1679-87592012000200012
Santos, A. L. F., Ponte, L., Peixoto, R. S., Rosman, P. A., & Rosman, P. C. C. (2018). Projeto Baías do Brasil, Baías de Ilha Grande e Sepetiba – RJ. COPPE UFRJ: SisBaHiA - Sistema Base de Hidrodinâmica Ambiental.
Schmiedl, G., Bovée, F., Buscail, R., Charrière, B., Hemleben, C., Medernach, L., & Picon, P. (2000). Trophic control of benthic foraminiferal abundance and microhabitat in the bathyal Gulf of Lions, western Mediterranean Sea. Marine Micropaleontology, 40, 167–188. https://doi.org/10.1016/S0377-8398(00)00038-4
Schonfeld, J., Alve, E., Geslin, E., Jorissen, F., Korsun, S., Spezzaferri, S., Abramovich, S., Almogi-Labin, A., Du Chatelet, E. A., Barras, C., Bergamin, L., Bicchi, E., Bouchet, V., Cearreta, A., Di Bella, L., Dijkstra, N., Disaro, S. T., Ferraro, L., Frontalini, F., … members of the Fobimo Group. (2012). The FOBIMO (FOraminiferal Bio-MOnitoring) initiative – towards a formalised protocol for benthic foraminiferal monitoring studies. Marine Micropaleontology, 94–95, 1–13. https://doi.org/10.1016/j.marmicro.2012.06.001
Schratzberger, M., & Ingels, J. (2018). Meiofauna matters: The roles of meiofauna in benthic ecosystems. Journal of Experimental Marine Biology and Ecology, 502, 12–25. https://doi.org/10.1016/j.jembe.2017.01.007
Sen Gupta, B. K., & Machain-Castillo, M. L. (1993). Benthic foraminifera in oxygen-poor habitats. Marine Micropaleontology, 20(3–4), 183–201. https://doi.org/10.1016/0377-8398(93)90032-S
Signorini, S. R. (1980). A study of the circulation in Bay of Ilha Grande and Bay of Sepetiba: Part I. a survey of the circulation based on experimental field data. Boletim Do Instituto Oceanográfico, 29, 41–55. https://doi.org/10.1590/S0373-55241980000100004
Silva, L. C., Alves Martins, M. V., Castelo, W. F. L., Saibro, M. B., Rangel, D., Pereira, E., Bergamaschi, S., Sousa, S. H. M., Varela, J., Laut, L., Frontalini, F., Chaves, H., Reis, A. T., Aguilera, O., Zaaboub, N., Cheriyan, E., & Geraldes, M. C. (2022a). Trace metals enrichment and Potential Ecological Risk in sediments of the Sepetiba Bay (Rio de Janeiro, SE Brazil). Marine Pollution Bulletin, 177(2022), 113485. https://doi.org/10.1016/j.marpolbul.2022.113485
Silva, L. C., Alves Martins, M. V., Figueira, R., Frontalini, F., Pereira, E., Senez-Mello, T. M., Castelo, W. F. L., Saibro, M. B., Francescangeli, F., Melloe Sousa, S. H., Bergamaschi, S., Antonioli, L., Bouchet, V. M. P., Terroso, D., & Rocha, F. (2022b). Unraveling the Anthropocene paleoenvironmental conditions combining sediment and foraminiferal data: proof-of-concept in the Sepetiba Bay (SE, Brazil). Frontiers in Ecology and Evolution, 10, 852439. https://doi.org/10.3389/fevo.2022.852439
Sliter, W. V. (1965). Laboratory experiments on the life cycle and ecologic controls of Rosalina globularis d’Orbigny. The Journal of Protozoology, 12(2), 210–215. https://doi.org/10.1111/j.1550-7408.1965.tb01838.x
Snyder, S. W. (1990). Relationship between benthic foraminiferal assemblages and Neogene phosphatic sediments, North Carolina coastal plain and continental shelf. In W. C. Burnett & S. R. Riggs (Eds.), Phosphate Deposits of the World (pp. 444–464). Cambridge Univ.
Sousa, S.H., Yamashita, C., Nagai, R.H., Martins, M.V.A., Ito, C., Vicente, T., Taniguchi, N., Burone, L., Fukumoto, M., Aluizio, R., Koutsoukos, E.A.M. (2017). Foraminíferos bentônicos no talude continental, Platô de São Paulo e cânions da Bacia de Campos. In: Falcão, A.P.C., Lavrado, H.P., editoras. Ambiente Bentônico: caracterização ambiental regional da Bacia de Campos, Atlântico Sudoeste. Rio de Janeiro: Elsevier. Habitats, 3: 111–144.
Souza, A. M., Rocha, D. S., Guerra, J. V., Cunha, B. A., Martins, M. V. A., & Geraldes, M. C. (2021). Metal concentrations in marine sediments of the Rio de Janeiro Coast (Brazil): A proposal to establish new acceptable levels of contamination. Marine Pollution Bulletin, 165, 112113. https://doi.org/10.1016/j.marpolbul.2021.112113
Subba Rao, M., Vedantam, D., & D., & Nageswara Rao, J. (1979). Distribution and ecology of benthonic foraminifera in the sediments of the Visakhapatnam shelf, east coast of India. Palaeogeography, Palaeoclimatology, Palaeoecology, 27, 349–369. https://doi.org/10.1016/0031-0182(79)90107-X
Suguio, K., Vieira, E. M., Barcelo, J. H., & Silva, M. S. (1979). Interpretação ecológica dos foraminíferos de sedimentos modernos da Baía de Sepetiba e adjacências Rio de Janeiro. Revista Brasileira De Geociências, 9(4), 233–247.
Teodoro, A. C., Duleba, W., Gubitoso, S., Prada, S. M., Lamparelli, C. C., & Bevilacqua, J. E. (2010). Analysis of foraminifera assemblages and sediment geochemical properties to characterise the environment near Araçá and Saco da Capela domestic sewage submarine outfalls of São Sebastião Channel, São Paulo State. Brazil. Marine Pollution Bulletin, 60(4), 536–553. https://doi.org/10.1016/j.marpolbul.2009.11.011
Tinoco, I. M. (1965). Contribuição à Sedimentologia e Microfauna da Baía de Sepetiba (Estado do Rio de Janeiro). Instituto Oceanográfico, Universidade Federal De Pernambuco, 7(8), 123–135.
Uchimura, H., Nishi, H., Takashima, R., Kuroyanagi, A., Yamamoto, Y., & Kutterolf, S. (2017). Distribution of Recent Benthic Foraminifera off Western Costa Rica in the Eastern Equatorial Pacific Ocean. Paleontological Research, 21(4), 380–396. https://doi.org/10.2517/2017PR003
Uchio, T. (1960). Ecology of living benthonic foraminifera from the San Diego, California, area Cushman Foundation for Foraminiferal Research. Cushman Special Publications, 5, 71.
Yamashita, C., Omachi, C., Santarosa, A. C. A., et al. (2020). Living benthic foraminifera of Santos continental shelf, southeastern Brazilian continental margin (SW Atlantic): Chlorophyll-a and particulate organic matter approach. Journal of Sedimentary Environments, 5, 17–34. https://doi.org/10.1007/s43217-019-00001-7
Yassini, I., & Jones, B. G. (1995). Foraminiferida and ostracoda from estuarine and shelf environments on the southeastern coast of Australia. Wollong, N (p. 484). S. W. The University os Wollongong Press.
Zaghloul, A., Saber, M., Gadow, S., et al. (2020). Biological indicators for pollution detection in terrestrial and aquatic ecosystems. Bulletin of the National Research Centre, 44, 127. https://doi.org/10.1186/s42269-020-00385-x
Zaninetti, L. (1979). L’étude des Foraminifère des Mangrove Actuelles: Réflexion sur les Objectifs et sur l’état des Connaissances. Archieves Des Science, 32, 151–161.
Zaninetti, L., Brönnimann, P., Beurlen, G., & Moura, J. A. (1976). La mangrove de Guaratiba et la Baie de Sepetiba, Etat de Rio de Janeiro, Bresil: Foraminiferes et ecólogie. Note Préliminaire, 11, 39–44.
Zaninetti, L., Brönnimann, P., Beurlen, G., & Moura, J. A. (1977). La mangrove de Guaratiba et la Baie de Sepetiba, Etat de Rio de Janeiro, Bresil: Foraminiferes et ecólogie. Archives Des Sciences, 30(2), 161–178.
Acknowledgements
This paper is a contribution to the project “Anthropized Coastal Systems: Innovative Proxies for Application to Environmental Biomonitoring and Reconstitution of Quaternary Paleoenvironments” financed by the Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro—FAPERJ (Edital Projetos Temáticos no Estado do Rio de Janeiro—2021, process # E-26/211.278/2021). Maryane Soares is grateful for the Scientific Initiation scholarship to UERJ (DECARH; PIBIC 2022). Wellen Castelo thanks the Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro, FAPERJ, for the post-doctoral fellowship (#E-26/205.702/2022). Fabrício Damasceno, thanks for the PhD fellowship to FAPERJ (process #E-26/204.093/2022). Murilo is grateful for the PhD fellowship to the National Council for Scientific and Technological Development, CNPq (Process 141863/2023-4). Virgínia Martins is grateful for the financial support received from FAPERJ (# E-26/211.278/2021, #202.927/2019, #E-26/200.333/2023) and the CnPQ processes # 443662/2018-5 # 302676/2019-8. The authors would like to thank the Fundação para a Ciência e a Tecnologia—FCT, Portugal (GeoBioTec funding UID/GEO/04035/2019; UIDB/04035/2020) for financial support.
Funding
Open access funding provided by FCT|FCCN (b-on). Conselho Nacional de Desenvolvimento Científico e Tecnológico of Brazil, CnPQ – processes # 443662/2018–5, # 302676/2019–8 and, # 141863/2023–4. Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro—FAPERJ (Edital Projetos Temáticos no Estado do Rio de Janeiro—2021, process # E-26/211.278/2021 and research grants: processes # E-26/202.927/2019, # E-26/200.333/2023, #E-26/205.702/2022, #E-26/204.093/2022. Fundação para a Ciência e a Tecnologia—FCT, Portugal (GeoBioTec funding UID/GEO/04035/2019; UIDB/04035/2020).
Author information
Authors and Affiliations
Contributions
M.F.S.—Data analysis, Writing—Original Draft. M.V.A.M.—Conceptualization, Methodology, Data acquisition, Species identification, Species description, Data Curation, Formal data analysis, Investigation, Writing and Editing, Supervision, Financial Acquisition. W.F.L.C.—Data analysis, Investigation, Supervision, Writing—Original Draft. M.B.S.—Data analysis, Investigation, Writing—Original Draft. A.B.R.O. -Data analysis, Investigation Writing—Original Draft. F.L.D.—Mapping, Writing—Original Draft. D.S.L.L.– Investigation. L.L.– Investigation. C.V. – Investigation. C.S.—SEM photos. F.R. – Investigation, Financial Acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
The authors followed the ethical standards.
Consent to participate
Authors have consent to participate.
Consent to publish
Authors have consent to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
43217_2024_173_MOESM3_ESM.png
Supplementary file3 Figure S1: Sectorization of Sepetiba Bay by Brönnimann et al. (1981a), adapted from Laut et al. (2012) (PNG 1990 KB)
43217_2024_173_MOESM4_ESM.png
Supplementary file4 Figure S2: Zones in Sepetiba Bay based on foraminifera, according to Oliveiria-Silva (2003), adapted from Laut et al. (2012) (PNG 2186 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Soares, M.F., Alves Martins, M.V., Castelo, W.F.L. et al. Species richness of living foraminifera in Sepetiba Bay (SE Brazil): a species checklist. J. Sediment. Environ. (2024). https://doi.org/10.1007/s43217-024-00173-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43217-024-00173-x