Abstract
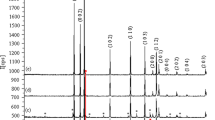
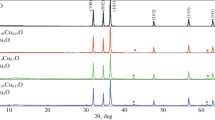
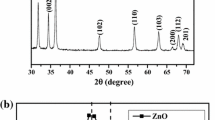
In this paper, the results of studying the formation conditions, crystal structure, thermal, spectral, and optical properties, as well as the electronic band structure of cobalt-doped zinc glycolate Zn1 − xCox(OCH2CH2O) (0˂x ≤ 0.2) are presented. Using X-ray powder diffraction data, it was shown that solid solutions are obtained by partial substitution of cobalt for zinc, while maintaining the crystal structure of Zn(OCH2CH2O). The vibrational spectra of Zn1 − xCox(OCH2CH2O) are identical to those of Zn(OCH2CH2O) and correlate completely with the results of structural analysis. As a result of heating in air at 600–900 °C, glycolate Zn1xCox(OCH2CH2O), where 0˂x ≤ 0.1, turns into oxide of the composition Zn1 − xCoxO with wurtzite structure, whose powders have a deep green color (Rinman’s green). The UV-Vis-NIR spectra of Zn1 − xCoxO contain bands typical of Co2+ ion transitions in the tetrahedral environment. When Zn1 − xCox(OCH2CH2O) is heated in helium atmosphere, composites (1-x)ZnO:xCo:nC are formed that include a phase with wurtzite structure, metallic cobalt, and elemental carbon. The electronic band structure, optical characteristics, and isosurfaces of wave functions of pure and cobalt-doped zinc glycolate and oxide were calculated. This allowed us to establish the reasons for the increase in the band gap width in glycolate compared to the oxide and its decrease during doping.


















Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
A. Kolodziejczak-Radzimska, T. Jesionowski, Zinc oxide-from synthesis to application: a review. Materials. 7, 2833–2881 (2014). https://doi.org/10.3390/ma7042833
H. Moradpoor, M. Safaei, H.R. Mozaffari, R. Sharifi, M.M. Imani, A. Golshahe, N. Bashardoust, An overview of recent progress in dental applications of zinc oxide nanoparticles. RSC Adv. 11, 21189 (2021). https://doi.org/10.1039/D0RA10789A
S. Deebansok, T. Amornsakchai, P. Sae-ear, P. Siriphannon, S.M. Smith, Sphere-like and flake-like ZnO immobilized on pineapple leaf fibers as easy-to-recover photocatalyst for the degradation of congo red. J Environ. Chem Eng. 9, 104746 (2021). https://doi.org/10.1016/j.jece.2020.104746
Y. Xiong, M. Fang, Q. Zhang, W. Liu, X. Liu, L. Ma, X. Xu, Reproducible and arbitrary patterning of transparent ZnO nanorod arrays for optic and biomedical device integration. J. Alloys Comp. 898, 163003 (2022). https://doi.org/10.1016/j.jallcom.2021.163003
D.K. Sharma, S. Shukla, K.K. Sharma, V. Kumar, A review on ZnO: Fundamental properties and applications. Mater. Today: Proc. 49, 3028–3035 (2022)
Y.Y. Kedruk, G.A. Baigarinova, L.V. Gritsenko, G. Cicero, K.A. Abdullin, Facile low-cost synthesis of highly photocatalitycally active zinc oxide powders. Front. Mater. 9, 869493 (2022). https://doi.org/10.3389/fmats.2022.869493
E.K. Droepenu, B.S. Wee, S.F. Chin, K.Y. Kok, M.F. Maligan, Zinc oxide nanoparticles synthesis methods and its effect on morphology: a review. Biointerface Res. Appl. Chem. 12, 4261–4292 (2022). https://doi.org/10.33263/BRIAC123.42614292
E. Alp, E.C. Araz, A.F. Buluc, Y. Güner, Y. Deger, H. Esgin, K.B. Dermenci, M.K. Kazmanli, S. Turan, A. Genc, Mesoporous nanocrystalline ZnO microspheres by ethylene glycol mediated thermal decomposition. Adv. Powder Technol. 29, 3455–3461 (2018). https://doi.org/10.1016/j.apt.2018.09.028
A. Sinhamahapatra, D. Bhattacharjya, J.S. Yu, Green fabrication of 3-dimensional flower-shaped zinc glycerolate and ZnO microstructures for p-nitrophenol sensing. RSC Adv. 5, 37721 (2015). https://doi.org/10.1039/c5ra06286a
S.M. Mousavi, G. Behbudi, A. Gholami, S.A. Hashemi, Z.M. Nejad, S. Bahrani, W.H. Chiang, L.C. Wei, N. Omidifar, Shape-controlled synthesis of zinc nanostructures mediating macromolecules for biomedical applications. Biomaterials Res. 26, 4 (2022). https://doi.org/10.1186/s40824-022-00252-y
B. Patella, N. Moukri, G. Regalbuto, C. Cipollina, E. Pace, S. Di Vincenzo, G. Aiello, A. O’Riordan, Inguanta. Electrochemical synthesis of Zinc Oxide Nanostructures on flexible substrate and application as an Electrochemical Immunoglobulin-G Immunosensor. Materials. 15, 713 (2022). https://doi.org/10.3390/ma15030713
H. Ma, P.L. Williams, S.A. Diamond, Ecotoxicity of manufactured ZnO nanoparticles - a review. Environ. Pollut. 172, 76–85 (2013). https://doi.org/10.1016/j.envpol.2012.08.011
B. Poornaprakash, U. Chalapathi, K. Subramanyam, S.V.P. Vattikuti, S.H. Park, Wurtzite phase co-doped ZnO nanorods: morphological, structural, optical, magnetic, and enhanced photocatalytic characteristics. Ceram. Intern. 46, 2931–2939 (2020). https://doi.org/10.1016/j.ceramint.2019.09.289
X. Zhong, Y. Shen, S. Zhao, D. Wei, Y. Zhang, K. Wei, Hydrothermal growth of overlapping ZnO nanorod arrays on the porous substrate and their H2 gas sensing. Mater. Charact. 172, 110858 (2021). https://doi.org/10.1016/j.matchar.2020.110858
S. Doke, K. Sonawane, A. Banerjee, S. Mahamuni, Evidence of various stabilizing mechanisms in ferromagnetic Co doped ZnO nanocrystals. J. Alloys Comp. 726, 947–954 (2017). https://doi.org/10.1016/j.jallcom.2017.08.034
S.S. Ghosh, C. Choubey, A. Sil, Photocatalytic response of Fe, Co, Ni doped ZnO based diluted magnetic semiconductors for spintronics applications. Superlattices Microstruct. 125, 271–280 (2019). https://doi.org/10.1016/j.spmi.2018.10.028
M.A. Fagier, Plant-Mediated biosynthesis and photocatalysis activities of zinc oxide nanoparticles: a prospect towards dyes mineralization. J. Nanotechnol. 11, 1 (2021). https://doi.org/10.1155/2021/6629180
L.N. Wu, G.J. Zhang, S.T. Yang, J.X. Guo, S.Y. Wu, Theoretical examination of defect structures and spin hamiltonian parameters of manganese (II)- and cobalt (II)-doped ZnO nanowires. J. Phys. Chem. Solids. 165, 110657 (2022). https://doi.org/10.1016/j.jpcs.2022.110657
A.E. Oksuz, M. Yurddaskal, U. Kartal, T. Dikici, M. Erol, ZnO nanostructures for photocatalytic degradation of methylene blue: effect of different anodization parameters. J. Korean Ceram. Soc. 59, 859–868 (2022). https://doi.org/10.1007/s43207-022-00222-z
E. Ersöz, O.A. Yildirim, Green synthesis and characterization of Ag–doped ZnO nanofibers for photodegradation of MB, RhB and MO dye molecules. J. Korean Ceram. Soc. 59, 655–670 (2022). https://doi.org/10.1007/s43207-022-00202-3
S.S. Kumari, W. Nirmala, N. Chidhambaram, M. Prabu, V. Ganesh, I.S. Yahia, Tuning the physical properties of Sb–doped ZnO nanopowders toward elevated photosensing and photocatalytic activity. J. Korean Ceram. Soc. 60, 719–731 (2023). https://doi.org/10.1007/s43207-023-00298-1
S. Zhang, P. Yang, A. Zhang, R. Shi, Y. Zhu, Fabrication of hollow coupled-layered ZnO microstructures using zinc glycerolate precursors. Cryst. Eng. Comm. 15, 9090–9096 (2013). https://doi.org/10.1039/C3CE41218K
M.A. Melkozerova, V.N. Krasil’nikov, O.I. Gyrdasova, E.V. Zabolotskaya, E.V. Shalaeva, R.F. Samigullina, Nature of defects in nanocrystalline zinc oxide with particles of tubular morphology. Theoret. Exp. Chem. 48, 149–152 (2012). https://doi.org/10.1007/s11237-012-9253-y
V.N. Krasil’nikov, T.V. Dyachkova, A.P. Tyutyunnik, O.I. Gyrdasova, M.A. Melkozerova, I.V. Baklanova, Y.A. Perevozchikova, S.M. Emelyanova, H.W. Weber, V.V. Marchenkov, Magnetic and optical properties as well as EPR studies of polycrystalline ZnO synthesized from different precursors. Mater. Res. Bull. 97, 553–559 (2018). https://doi.org/10.1016/j.materresbull.2017.09.061
V.N. Krasil’nikov, A.P. Tyutyunnik, V.P. Zhukov, I.V. Baklanova, O.I. Gyrdasova, E.V. Chulkov, Zinc glycolate zn(OCH2CH2O): synthesis and structure, spectral and optical properties, electronic structure and chemical bonding. J. Alloys Comp. 924, 166320 (2022). https://doi.org/10.1016/j.jallcom.2022.166320
V.N. Krasil’nikov, O.I. Gyrdasova, L.Y. Buldakova, M.Y. Yanchenko, V.G. Bamburov, Synthesis and Photocatalytic Properties of highly dispersed Zinc Oxide Doped with Iron. Dokl. Chem. 437, 87–89 (2011). https://doi.org/10.1134/S0012500811040033
V.N. Krasil’nikov, O.I. Gyrdasova, L.Y. Buldakova, M.Y. Yanchenko, Synthesis and Photocatalytic Properties of Low Dimensional Cobalt Doped Zinc Oxide with different crystal shapes. Russ J. Inorg. Chem. 56, 145–151 (2011). https://doi.org/10.1134/S0036023611020136
M.A. Melkozerova, V.N. Krasil’nikov, O.I. Gyrdasova, E.V. Shalaeva, I.V. Baklanova, L.Y. Buldakova, M.Y. Yanchenko, Effect of doping with 3d elements (Co, Ni, Cu) on the intrinsic defect structure and Photocatalytic Properties of Nanostructured ZnO with tubular morphology of aggregates. Phys. Solid State. 55, 2459–2465 (2013). https://doi.org/10.1134/S106378341312024X
V.N. Krasilnikov, T.V. Dyachkova, Ð.I. Gyrdasova, Ð.P. Tyutyunnik, V.V. Marchenkov, H.W. Weber, Room-temperature ferromagnetism in polycrystalline Zn1 – xFexO (0 ≤ x ≤ 0.075) solid solutions synthesized by the precursor method. Mater. Chem. Phys. 162, 1–5 (2015). https://doi.org/10.1016/j.matchemphys.2015.05.025
O.I. Gyrdasova, V.N. Krasil’nikov, I.V. Baklanova, L.Y. Buldakova, M.Y. Yanchenko, Synthesis, structure, and Optical and Photocatalytic Properties of Quasi-One-Dimensional ZnO Doped with Со3O4 and Carbon, Bull. Russ Acad. Sci. : Phys. 80, 1298–1302 (2016). https://doi.org/10.3103/S1062873816110204
O.I. Gyrdasova, M.A. Melkozerova, I.V. Baklanova, L.Y. Buldakova, V.N. Krasil’nikov, M.Y. Yanchenko, N.S. Sycheva, V.G. Bamburov, Synthesis, structure, and Photocatalytic Properties of Zn1 – xCuxO:CuO composites with various morphologies of aggregates. Dokl. Chem. 474, 105–108 (2017). https://doi.org/10.1134/S0012500817050032
O.I. Gyrdasova, N.S. Sycheva, I.V. Baklanova, L.Y. Buldakova, M.Y. Yanchenko ,·K, V. Nefedova, Krasil’nikov, Synthesis, structure, optical, voltammetric and photocatalytic properties of manganese–activated ZnO. J. Mater. Sci. : Materials in Electronics. 30, 8820–8831 (2019). https://doi.org/10.1007/s10854-019-01207-1
Ð.I. Gyrdasova, E.V. Shalaeva, V.N. Krasil’nikov, L.Y. Buldakova, I.V. Baklanova, M.A. Melkozerova, Ð.V. Kuznetsov, Yanchenko, Effect of Cu+ ions on the structure, morphology, optical and photocatalytic properties of nanostructured ZnO. Mater. Charact. 179, 111384 (2021). https://doi.org/10.1016/j.matchar.2021.111384
A. Mesaros, C.D. Ghitulica, M. Popa, R. Mereu, A. Popa, T.P. Jr. Gabor, A.I. Cadis, B.S. Vasile, Synthesis, structural and morphological characteristics, magnetic and optical properties of Co doped ZnO nanoparticles. Ceram. Intern. 40, 2835–2846 (2014). https://doi.org/10.1016/j.ceramint.2013.10.030
J.J. Beltran, C.A. Barrero, A. Punnoose, Combination of defects plus mixed Valence of Transition Metals: a strong strategy for ferromagnetic enhancement in ZnO Nanoparticles. J. Phys. Chem. C 120, 8969–8978 (2016). https://doi.org/10.1021/acs.jpcc.6b00743
K. Pandey, A.K. Shahi, J. Shah, R.K. Kotnala, R. Gopal, Giant ferromagnetism in Li doped ZnO nanoparticles at room temperature. J. Alloys Comp. 823, 153710 (2020). https://doi.org/10.1016/j.jallcom.2020.153710
J.M.P. Silva, N.F. Andrade Neto, M.C. Oliveira, R.A.P. Ribeiro, S.R. de Lazaro, Y.F. Gomes, C.A. Paskocimas, M.R.D. Bomio, F.V. Motta, Recent progress and approaches on the synthesis of Mn-doped zinc oxide nanoparticles: a theoretical and experimental investigation on the photocatalytic performance. New. J. Chem. 44, 8805 (2020). https://doi.org/10.1039/d0nj01530j
M. Carofiglio, S. Barui, V. Cauda, M. Laurenti, Doped Zinc Oxide Nanoparticles: synthesis, characterization and potential use in Nanomedicine. Appl. Sci. 10, 5194 (2020). https://doi.org/10.3390/app10155194
O. Muktaridha, M. Adlim, S. Suhendrayatna, I. Ismail, Progress of 3d metal-doped zinc oxide nanoparticles and the photocatalytic properties. Arab. J Chem. 14, 103175 (2021). https://doi.org/10.1016/j.arabjc.2021.103175
M.A. Wahba, S.M. Yakout, R. Khaled, Interface engineered efficient visible light photocatalytic activity of MWCNTs/Co doped ZnO nanocomposites: morphological, optical, electrical and magnetic properties. Opt. Mater. 115, 111039 (2021). https://doi.org/10.1016/j.optmat.2021.111039
Z. Yuan, J. Li, F. Meng, High response n-propanol sensor based on co-modified ZnO nanorods. J. Alloys Comp. 910, 164971 (2022). https://doi.org/10.1016/j.jallcom.2022.164971
D. Kumar, M. Kumar, S. Mohan, S. Mehata, Exploration of grown cobalt-doped zinc oxide nanoparticles and photodegradation of industrial dye. Mater. Res. Bull. 150, 111795 (2022). https://doi.org/10.1016/j.materesbull.2022.111795
P.T.L. Huong, N.V. Quang, M.T. Tran, D.Q. Trung, D.T.B. Hop,·T, T.H. Tam, N. Tu, V.D. Dao, Excellent visible light photocatalytic degradation and mechanism insight of Co2+–doped ZnO nanoparticles. Appl. Phys. A 128, 24 (2022). https://doi.org/10.1007/s00339-021-05140-1
A. Safeen, K. Safeen, M. Shafique, Y. Iqbal, N. Ahmed, M.A.R. Khan, G. Asghar, K. Althubeiti, S.A. Otaibi, G. Ali, W.H. Shah, R. Khan, The effect of Mn and Co dual-doping on the structural, optical, dielectric and magnetic properties of ZnO nanostructures. RSC Adv. 12, 11923 (2022). https://doi.org/10.1039/d2ra01798a
H.V.S. Pessoni, Magnetic properties in randomly diluted magnetic systems: co-doped ZnO polycrystalline ceramics. J. Alloys Comp. 923, 166264 (2022). https://doi.org/10.1016/j.jallcom.2022.166264. Jr.
F.C. Romeiro, N.S. Castro, L. Scolfaro, P.D. Borges, R.C. Lima, Theoretical and experimental study of effects of Co2+ doping on structural and electronic properties of ZnO. J. Phys. Chem. Solids. 162, 110501 (2022). https://doi.org/10.1016/j.jpcs.2021.110501
Y. Shen, J. Yang, Y. Zhang, T. Zeng, Q. Wan, Fabrication of a branch–like Ni–Doped ZnO/Graphene Nanoplatelet Composite for enhanced Electrochemical determination of 17β–Estradiol and Acetaminophen. J. Electron. Mater. 51, 5310–5321 (2022). https://doi.org/10.1007/s11664-022-09763-9
H.V.S. Pessoni, P. Banerjeea, A. Franco Jr., Colossal dielectric permittivity in co-doped ZnO ceramics prepared by a pressure-less sintering method, cite this. Phys. Chem. Chem. Phys. 20, 28712 (2018). https://doi.org/10.1039/c8cp04215b
M.A. Mahmood, R. Khan, S.A. Otaibi, K. Althubeiti, S.S. Abdullaev, N. Rahman, M. Sohail, S. Iqbal, The Effect of Transition Metals Co-Doped ZnO Nanotubes based-diluted magnetic Semiconductor for Spintronic Applications. Crystals. 13, 984 (2023). https://doi.org/10.3390/cryst13070984
Q. Yue, L. Yan, J.Y. Zhang, E.Q. Gao, Novel Functionalized Metal-Organic Framework based on unique hexagonal prismatic clusters. Inorg. Chem. 49, 8647–8649 (2010). https://doi.org/10.1021/ic100558x
L.E. Kreno, K. Leong, O.K. Farha, M. Allendorf, R.P. Van Duyne, J.T. Hupp, Metal-Organic Framew. Mater. as Chem. Sens. Chem Rev. 112, 1105–1125 (2012). https://doi.org/10.1021/cr200324t
K.V. Lawler, Z. Hulveyz, P.M. Forster, Nanoporous metal formates for krypton/xenon separation. Chem. Commun. 49, 10959 (2013). https://doi.org/10.1039/c3cc44374d
H. Li, L. Li, R.-B. Lin, W. Zhou, Z. Zhang, S. Xiang, B. Chen, Porous metal-organic frameworks for gas storage and separation: Status and challenges. Energy Chem. 1, 100006 (2019). https://doi.org/10.1016/j.enchem.2019.100006
B. Wu, Y.T.A. Wong, B.E.G. Lucier, P.D. Boyle, Y. Huang, Exploring host-guest interactions in the α–Zn3(HCOO)6 Metal-Organic Framework. ACS Omega. 4, 4000–4011 (2019). https://doi.org/10.1021/acsomega.8b03623
R.B. Lin, S. Xiang, W. Zhou, B. Chen, Microporous Metal-Organic Framework materials for gas separation. Chem. 6, 337–363 (2020). https://doi.org/10.1016/j.chempr.2019.10.012
H. Zhang, Y. Fan, R. Krishna, X. Feng, L. Wang, F. Luo, Robust metal-organic framework with multiple traps for trace Xe/Kr separation. Sci. Bull. 66, 1073–1079 (2021). https://doi.org/10.1016/j.scib.2020.12.031
R.D. Shannon, Revised effective ionic Radii and systematic studies of interatomie distances in Halides and Chaleogenides. Acta Cryst. A32, 751–767 (1976). https://doi.org/10.1107/S0567739476001551
N. Chakroune, G. Viau, S. Ammar, N. Jouini, P. Gredin, M.J. Vaulaya, F. Fievet, Synthesis, characterization and magnetic properties of disk-shaped particles of a cobalt alkoxide: CoII(C2H4O2). New. J. Chem. 29, 355–361 (2005). https://doi.org/10.1039/B411117F
B.H. Toby, EXPGUI, a graphical user interface for GSAS. J. Appl. Crystallogr. 34, 210–2013 (2001). https://doi.org/10.1107/S0021889801002242
A.C. Larson, R.B. Von Dreele, General Structure Analysis System (GSAS) (Los Alamos National Laboratory Report LAUR 86–748, Los Alamos, New Mexico, 2004)
G. Williamson, W. Hall, X-ray line broadening from filed aluminium and wolfram. Acta Metall. 1, 22–31 (1953). https://doi.org/10.1016/0001-6160(53)90006-6
G. Kresse, M. Marsman, J. Furthmüller. Vienna ab-initio Simulationpackage. VASP the Guide (Universität Wien, Wien, 2018), p. 233
S.C. Abrahams, J.L. Bernstein, Remeasurement of the structure of Hexagonal ZnO. Acta Crystallogr. B 25, 1233–1236 (1969). https://doi.org/10.1107/S0567740869003876
M. Mamak, P.Y. Zavalij, M.S. Whittingham, Layered structure of Lithium Ethylene Glycolate, Li(OCH2CH2OH). Acta Crystallogr. Sect. C 54, 937–939 (1998). https://doi.org/10.1107/S0108270198002121/da1011sup2.hkl
R. Nesper, H.G. von Schnering, Zinn(II)-Ethylenglykolat, SnC2H4O2, ein polymerer Chelatkomplex mit 2-fach funktionellem Sauerstoff. Z. Naturforsch B: Chem. Sci. 37, 1144–1145 (1982). https://doi.org/10.1515/znb-1982-0910
J. Teichert, M. Ruck, Influence of Common Anions on the Coordination of Metal Cations in Polyalcohols. Eur. J. Inorg. Chem. 2019, 2267–2276 (2019). https://doi.org/10.1002/ejic.201801540
S.D. Birajdar, V.R. Bhagwat, A.B. Shinde, K.M. Jadhav, Effect of Co2+ ions on structural, morphological and optical properties of ZnO nanoparticles synthesized by sol-gel auto combustion method. Mater. Sci. Semicond. Proc. 41, 441–449 (2016). https://doi.org/10.1016/j.mssp.2015.10.002
K.N. Hutchings, M. Wilson, P.A. Larsen, R.A. Cutler, Kinetic and thermodynamic considerations for oxygen absorption/desorption using cobalt oxide. Solid State Ionics. 177, 45–51 (2006). https://doi.org/10.1016/j.ssi.2005.10.005
C.W. Tang, C.B. Wang, S.H. Chien, Characterization of cobalt oxides studied by FT-IR, Raman, TPR and TG-MS. Thermochim. Acta. 473, 68–73 (2008). https://doi.org/10.1016/j.tca.2008.04.015
P. Martin-Ramos, M. Susano, F.P.S.C. Gil, P.S. Pereira da Silva, J. Martin-Gil, M.R. Silva, Facile synthesis of three Kobolds: introducing students to the structure of pigments and their characterization. J. Chem. Educ. 95, 1340–1344 (2018). https://doi.org/10.1021/acs.jchemed.7b00402
M. Scepanovic, M. Grujic-Brojcin, K. Vojisavljevic, S. Bernik, T. Sreckovic, Raman study of structural disorder in ZnO nanopowders. J. Raman Spectroscopy. 41, 914–921 (2010). https://doi.org/10.1002/jrs.2546
B. Hadzic, N. Romcevic, M. Romcevic, I. Kuryliszyn-Kudelska, W. Dobrowolski, R. Wrobel, U. Narkiewicz, D. Sibera, Raman study of surface optical phonons in ZnO(mn) nanoparticles. J. Alloys Comp. 585, 214–219 (2014). https://doi.org/10.1016/j.jallcom.2013.09.132
R.L. de Sousa, A.F Jr. Silva, Raman spectroscopy study of structural disorder degree of ZnO ceramics. Mater. Sci. Semicond. Proc. 119(2020)
M. Fang, Z.W. Liu, Structure and properties variations in Zn1 – xCoxO nanorods prepared by microwave-assisted hydrothermal method. Mater. Sci. Semicond. Proc. 57, 233–238 (2017). https://doi.org/10.1016/j.mssp.2016.10.041
S.B. Yahia, L. Znaidi, A. Kanaev, J.P. Petitet, Raman study of oriented ZnO thin films deposited by sol-gel method. Spectrochim. Acta Part A 71, 1234–1238 (2008). https://doi.org/10.1016/j.saa.2008.03.032
V. Gandhi, R. Ganesan, H.H.A. Syedahamed, M. Thaiyan, Effect of Cobalt Doping on Structural, Optical, and magnetic Properties of ZnO Nanoparticles synthesized by Coprecipitation Method. J. Phys. Chem. C 118, 9715–9725 (2014). https://doi.org/10.1021/jp411848t
K.S. Al-Namshah, M. Shkir, F.A. Ibrahim, M.S. Hamdy, Auto combustion synthesis and characterization of Co doped ZnO nanoparticles with boosted photocatalytic performance. Phys. B: Condens. Matter. 625, 413459 (2022). https://doi.org/10.1016/j.physb.2021.413459
R. Bhargava, P.K. Sharma, R.K. Dutta, S. Kumar, A.C. Pandey, N. Kumar, Influence of co-doping on the thermal, structural, and optical properties of sol-gel derived ZnO nanoparticles. Mater. Chem. Phys. 120, 393–398 (2010). https://doi.org/10.1016/j.matchemphys.2009.11.024
B. Hadzic, N. Romcevic, M. Romcevic, I. Kuryliszyn-Kudelska, W. Dobrowolski, J. Trajic, D. Timotijevic, U. Narkiewicz, D. Sibera, Surface optical phonons in ZnO(Co) nanoparticles: Raman study. J. Alloys Comp. 540, 49–56 (2012). https://doi.org/10.1016/j.jallcom.2012.06.076
V. Ney, S. Ye, T. Kammermeier, A. Ney, H. Zhou, J. Fallert, H. Kalt, F.Y. Lo, A. Melnikov, A.D. Wieck, Structural, magnetic, and optical properties of Co- and Gd-implanted ZnO (0001) substrates. J. Appl. Phys. 104, 083904 (2008). https://doi.org/10.1063/1.3000452
O.F. Kolomys, V.V. Strelchuk, S.V. Rarata, R. Hayn, A. Savoyant, F. Giovannelli, F. Delorme, V. Tkach, Optical and structural properties of individual co-doped ZnO microwires. Superlattices Microstruct. 118, 7–15 (2018). https://doi.org/10.1016/j.spmi.2018.04.005
P.R. Chithira, T.T. John, Correlation among oxygen vacancy and doping concentration in controlling the properties of cobalt doped ZnO nanoparticles. J. Magn. Magn. Mater. 496, 165928 (2020). https://doi.org/10.1016/j.jmmm.2019.165928
J.S. Kumar, K. Pavani, A.M. Babu, N.K. Giri, S.B. Rai, L.R. Moorthy, Fluorescence characteristics of Dy3+ ions in calcium fluoroborate glasses. J. Luminescence. 130, 1916–1923 (2010). https://doi.org/10.1016/j.jlumin.2010.05.006
A.B. Djurisic, Y.H. Leung, K.H. Tam, L. Ding, W.K. Ge, H.Y. Chen, S. Gwo, Green, yellow and orange defect emission from ZnO nanostructures: influence of excitation wavelength. Appl. Phys. Lett. 88, 103107 (2006). https://doi.org/10.1063/1.2182096
A.B. Djurisic, Y.H. Leung, K.H. Tam, Y.F. Hsu, L. Ding, W.K. Ge, Y.C. Zhong, K.S. Wong, W.K. Chan, H.L. Tam, Defect emissions in ZnO nanostructures. Nanotechnology. 18, 095702 (2007). https://doi.org/10.1088/0957-4484/18/9/095702
H. Ji, C. Cai, S. Zhou, W. Liu, Structure, photoluminescence, and magnetic properties of co-doped ZnO nanoparticles. J. Mater. Sci. : Materials in Electronics. 29, 12917–12926 (2018). https://doi.org/10.1007/s10854-018-9411-7.S
J. Kammoun, El ghoul, Structural and optical investigation of co-doped ZnO nanoparticles for nanooptoelectronic devices. J. Mater. Sci. : Materials in Electronics. 32, 7215–7225 (2021). https://doi.org/10.1007/s10854-021-05430-7
N. Pushpa, M.K. Kokila, Effect of Cobalt doping on structural, thermo and photoluminescent properties of ZnO nanopowders. J. Luminescence. 190, 100–107 (2017). https://doi.org/10.1016/j.jlumin.2017.05.032
J. Kazmi, P.C. Ooi, S.R.A. Raza, B.T. Goh, S.S.A. Karim, M.H. Samat, M.K. Lee, M.F.M.R. Wee, M.F.M. Taibd, M.A. Mohamed, Appealing stable room-temperature ferromagnetism by well-aligned 1D co-doped zinc oxide nanowires. J. Alloys Comp. 872, 159741 (2021). https://doi.org/10.1016/j.jallcom.2021.159741
P. Koidl, Optical absorption of Co2+ in ZnO. Phys. Rev. B 15, 2493–2499 (1977). https://doi.org/10.1103/PhysRevB.15.2493
L. Reinert, M. Zeiger, S. Suarez, V. Presser, F. Mucklich, Dispersion analysis of carbon nanotubes, carbon onions, and nanodiamonds for their application as reinforcement phase in nickel metal matrix composites. RSC Adv. 5, 95149–95159 (2015). https://doi.org/10.1039/C5RA14310A
Y.L. Fu, J.L. Ren, Z.W. Xu, S.W. Ng, Cobalt(II) formate hydroxide. Acta Cryst. E61, m2395–m2396 (2005). https://doi.org/10.1107/S1600536805033817
H. Yue, Y. Zhao, X. Ma, J. Gong, Ethylene glycol: properties, synthesis, and applications. Chem. Soc. Rev. 41, 4218–4244 (2012). https://doi.org/10.1039/c2cs15359a
P. Sulcova, M. Trojan, New green pigments: ZnO-CoO. Dyes Pigm. 4, 83–86 (1998). https://doi.org/10.1016/S0143-7208(98)00036-9
N. Zhou, Y. Zhang, S. Nian, W. Li, J. Li, W. Cao, Z. Wu, Synthesis and characterization of Zn1 – xCoxO green pigments with low content cobalt oxide. J. Alloys Comp. 711, 406–413 (2017). https://doi.org/10.1016/j.jallcom.2017.04.015
I. Mjejri, S. Mornet, M. Gaudon, From nano-structured polycrystalline spheres with Zn1 – xCoxO composition to core-shell Zn1 – xCoxO@SiO2 as green pigments. J. Alloys Comp. 777, 1204–1210 (2019). https://doi.org/10.1016/j.jallcom.2018.10.333
N. Zhou, S. Sha, Y. Zhang, S. Li, S. Xu, J. Luan, Coprecipitation synthesis of a green co-doped wurtzite structure high near-infrared reflective pigments using ammonia as precipitant. J. Alloys Comp. 820, 153183 (2020). https://doi.org/10.1016/j.jallcom.2019.153183
Acknowledgements
The X-ray study was carried out at the Multiple-Access Center for X-ray Structure Analysis at the Institute of Solid State Chemistry, UB RAS. The UV-Vis spectra were recorded using the equipment of the Multiple-Access Center for Spectroscopy and Analysis of Organic Compounds at the Postovsky Institute of Organic Synthesis, UB RAS. This work was carried out in accordance with the scientific and research plans and as defined in the state assignment for the Institute of Solid State Chemistry, UB RAS (grant No. AAAA-A19–119031890025-9). The electronic structure calculations were performed with the URAN cluster in the Institute of Mathematics and Mechanics, UB RAS. E.V.C. acknowledges support from Saint Petersburg State University (grant No. ID 94031444).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Krasil’nikov, V.N., Tyutyunnik, A.P., Baklanova, I.V. et al. Cobalt-doped zinc glycolate as a precursor for the production of Zn1 − xCoxO oxide with nanostructured octahedral particles: synthesis, crystal structure, thermal, spectral, and optical properties. J. Korean Ceram. Soc. 60, 990–1009 (2023). https://doi.org/10.1007/s43207-023-00323-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43207-023-00323-3