Abstract
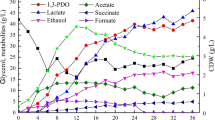
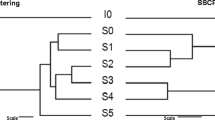
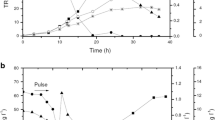
Glycerol is a by-product generated in the biodiesel industry and can be converted by biological processes into products such as hydrogen, alcohols and acids. This study aimed at comparing two sources of inoculum in dark fermentation using raw glycerol and at identifying the microorganisms present by high-performance sequencing. Granular sludge obtained from a vegetable oil industry (VOI) and anaerobic sludge from a wine industry (WI) were evaluated in batch reactors with raw glycerol. The highest hydrogen yield was obtained for VOI (1.12 mol H2 / mol glycerolconsumed). The WI was more efficient in producing soluble metabolites, with 6.2 g / L of 1,3 – propanediol, ethanol (2.5 g / L) and acids (122 mg / g glycerolconsumed). Both assays consumed more than 60% of the glycerol. A significant variation in microbial diversity was observed between the beginning and the end of the fermentation. The microbial community showed a predominance of Firmicutes and the Clostridiaceae and Peptostreptococcaceae families in different abundances among the inocula, which could lead to an increase in hydrogen production or a higher concentration of metabolites. Clostridium bifermentans was the only species isolated, showing a yield of H2 (0.135 mol H2 / mol glycerolconsumed) and the main metabolite produced was ethanol (670 mg / L). These results showed that two different kinds of seed sludge can lead to different metabolic profiles from the same substrate, highlighting the importance of choosing the inoculum and, consequently, that the knowledge regarding microorganisms can direct the process towards products of industrial interest and contribute to the generation of sustainable energy.
Graphical abstract





Similar content being viewed by others
Data availability
The data used to support the findings of this study is available upon request.
References
Almeida JRM, Fávaro LCL, Quirino BF (2012) Biodiesel biorefinery: opportunities and challenges for microbial production of fuels and chemicals from glycerol waste. Biotechnol Biofuels 5:1–16. https://doi.org/10.1186/1754-6834-5-48
Almena A, Bueno L, Díez M, Martín M (2018) Integrated biodiesel facilities: review of glycerol-based production of fuels and chemicals. Clean Technol Environ Policy 20:1639–1661. https://doi.org/10.1007/s10098-017-1424-z
ANP (2021) National Agency of Petroleum, Natural Gas and Biofuels. https://www.gov.br/anp/pt-br. Accessed 26 Jan 2021
APHA (2017) Standard methods for the examination of water and wastewater, 23rd edn. American Public Health Association, Washington
Badia-Fabregat M, Rago L, Baeza JA, Guisasola A (2019) Hydrogen production from crude glycerol in an alkaline microbial electrolysis cell. Int J Hydrogen Energy 44:17204–17213. https://doi.org/10.1016/j.ijhydene.2019.03.193
Bates ST, Berg-Lyons D, Caporaso JG et al (2011) Examining the global distribution of dominant archaeal populations in soil. ISME J 5:908–917. https://doi.org/10.1038/ismej.2010.171
Batlle-Vilanova P, Ganigué R, Ramió-Pujol S et al (2017) Microbial electrosynthesis of butyrate from carbon dioxide: production and extraction. Bioelectrochemistry 117:57–64. https://doi.org/10.1016/j.bioelechem.2017.06.004
Bergey DH, Whitman WB, De Paul V et al (2009) Bergey’s manual of systematic bacteriology, vol 3. Springer, New York
Boboescu IZ, Ilie M, Gherman VD et al (2014) Revealing the factors influencing a fermentative biohydrogen production process using industrial wastewater as fermentation substrate. Biotechnol Biofuels 7:1–15. https://doi.org/10.1186/s13068-014-0139-1
Cabrol L, Marone A, Tapia-Venegas E et al (2017) Microbial ecology of fermentative hydrogen producing bioprocesses: useful insights for driving the ecosystem function. FEMS Microbiol Rev 41:158–181. https://doi.org/10.1093/femsre/fuw043
Canani RB, Di Costanzo M, Leone L (2012) The epigenetic effects of butyrate: potential therapeutic implications for clinical practice. Clin Epigenetics 4:4. https://doi.org/10.1186/1868-7083-4-4
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high- throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth0510-335
Castelló E, García y Santos C, Iglesias T et al (2009) Feasibility of biohydrogen production from cheese whey using a UASB reactor: links between microbial community and reactor performance. Int J Hydrogen Energy 34:5674–5682. https://doi.org/10.1016/j.ijhydene.2009.05.060
Castelló E, Braga L, Fuentes L, Etchebehere C (2018) Possible causes for the instability in the H2 production from cheese whey in a CSTR. Int J Hydrogen Energy 43:2654–2665. https://doi.org/10.1016/j.ijhydene.2017.12.104
Chao A (1984) Nonparametric estimation of the number of classes in a population. Scand J Stat 11:265–270
Chen Y, Yin Y, Wang J (2021) Comparison of fermentative hydrogen production from glycerol using immobilized and suspended mixed cultures. Int J Hydrogen Energy 46:8986–8994. https://doi.org/10.1016/j.ijhydene.2021.01.003
Cole JR, Wang Q, Fish JA et al (2014) Ribosomal database project: data and tools for high throughput rRNA analysis. Nucleic Acids Res 42:633–642. https://doi.org/10.1093/nar/gkt1244
Dahiya S, Venkata SM (2021) Selective enrichment of mixed consortia towards enhanced 1,3-Propanediol production from glycerol. Sustain Energy Technol Assess 47:101337. https://doi.org/10.1016/j.seta.2021.101337
de Souza BC, Bossardi FF, Furlan GR et al (2020) Validated high-performance liquid chromatographic (HPLC) method for the simultaneous quantification of 2,3-butanediol, glycerol, acetoin, ethanol, and phosphate in microbial cultivations. Anal Lett 54:2395–2410. https://doi.org/10.1080/00032719.2020.1869754
DeSantis TZ, Hugenholtz P, Larsen N et al (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72:5069–5072. https://doi.org/10.1128/AEM.03006-05
Dessì P, Porca E, Frunzo L et al (2018) Inoculum pretreatment differentially affects the active microbial community performing mesophilic and thermophilic dark fermentation of xylose. Int J Hydrogen Energy 43:9233–9245. https://doi.org/10.1016/j.ijhydene.2018.03.117
Dubé CD, Guiot SR (2015) Direct interspecies electron transfer in anaerobic digestion: a review. Adv Biochem Eng Biotechnol 151:101–115
Dutta S (2014) A review on production, storage of hydrogen and its utilization as an energy resource. J Ind Eng Chem 20:1148–1156. https://doi.org/10.1016/j.jiec.2013.07.037
Eder AS, Magrini FE, Spengler A et al (2020) Comparison of hydrogen and volatile fatty acid production by Bacillus cereus, Enterococcus faecalis and Enterobacter aerogenes singly, in co-cultures or in the bioaugmentation of microbial consortium from sugarcane vinasse. Environ Technol Innov 18:100638. https://doi.org/10.1016/j.eti.2020.100638
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461. https://doi.org/10.1093/bioinformatics/btq461
Edgar RC (2013) UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998. https://doi.org/10.1038/nmeth.2604
Etchebehere C, Castelló E, Wenzel J et al (2016) Microbial communities from 20 different hydrogen-producing reactors studied by 454 pyrosequencing. Appl Microbiol Biotechnol 100:3371–3384. https://doi.org/10.1007/s00253-016-7325-y
Faber MO, Ferreira-Leitão VS (2016) Optimization of biohydrogen yield produced by bacterial consortia using residual glycerin from biodiesel production. Bioresour Technol 219:365–370. https://doi.org/10.1016/j.biortech.2016.07.141
Faust K, Raes J (2012) Microbial interactions: from networks to models. Nat Rev Microbiol 10:538–550. https://doi.org/10.1038/nrmicro2832
Fountoulakis MS, Manios T (2009) Enhanced methane and hydrogen production from municipal solid waste and agro-industrial by-products co-digested with crude glycerol. Bioresour Technol 100:3043–3047. https://doi.org/10.1016/j.biortech.2009.01.016
Fuentes L, Braga L, Castelló E, Etchebehere C (2018) Work scheme to isolate the different micro-organisms found in hydrogen-producing reactors: a study of effectiveness by pyrosequencing analysis. J Appl Microbiol 125:96–110. https://doi.org/10.1111/jam.13763
Gan Y, Qiu Q, Liu P et al (2012) Syntrophic oxidation of propionate in rice field soil at 15 and 30 C under methanogenic conditions. Appl Environ Microbiol 78:4923–4932. https://doi.org/10.1128/AEM.00688-12
Hammer Ø, Harper DAT, Ryan PD (2001) PAST–Palaeontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9
Iltchenco J, Almeida LG, Beal LL et al (2020) Microbial consortia composition on the production of methane from sugarcane vinasse. Biomass Convers Biorefinery 10:299–309. https://doi.org/10.1007/s13399-019-00426-0
Kamalaskar LB, Dhakephalkar PK, Meher KK, Ranade DR (2010) High biohydrogen yielding Clostridium sp. DMHC-10 isolated from sludge of distillery waste treatment plant. Int J Hydrogen Energy 35:10639–10644. https://doi.org/10.1016/j.ijhydene.2010.05.020
Katarzyna L, Kamila M, Agnieszka KO-S et al (2014) Selection and characterization of Clostridium bifermentans strains from natural environment capable of producing 1,3-propanediol under microaerophilic conditions. African J Microbiol Res 8:1187–1197. https://doi.org/10.5897/ajmr213.6516
Kato S, Hashimoto K, Watanabe K (2012) Methanogenesis facilitated by electric syntrophy via (semi)conductive iron-oxide minerals. Environ Microbiol 14:1646–1654. https://doi.org/10.1111/j.1462-2920.2011.02611.x
Lazaro CZ, Perna V, Etchebehere C, Varesche MBA (2014) Sugarcane vinasse as substrate for fermentative hydrogen production: the effects of temperature and substrate concentration. Int J Hydrogen Energy 39:6407–6418. https://doi.org/10.1016/j.ijhydene.2014.02.058
Magrini FE, Almeida GM, Soares DM et al (2020) Effect of different heat treatments of inoculum on the production of hydrogen and volatile fatty acids by dark fermentation of sugarcane vinasse. Biomass Conv Bioref. https://doi.org/10.1007/s13399-020-00687-0
Magrini FE, Almeida GM, Soares DM et al (2021) Variation of the prokaryotic and eukaryotic communities after distinct methods of thermal pretreatment of the inoculum in hydrogen-production reactors from sugarcane vinasse. Curr Microbiol 78:2682–2694. https://doi.org/10.1007/s00284-021-02527-4
Mahata C, Dhar S, Ray S, Das D (2021) Effect of thermal pretreated organic wastes on the dark fermentative hydrogen production using mixed microbial consortia. Fuel 284: https://doi.org/10.1016/j.fuel.2020.119062
Mangayil R, Karp M, Santala V (2012) Bioconversion of crude glycerol from biodiesel production to hydrogen. Int J Hydrogen Energy 37:12198–12204. https://doi.org/10.1016/j.ijhydene.2012.06.010
Mockaitis G, Bruant G, Guiot SR et al (2020) Acidic and thermal pre-treatments for anaerobic digestion inoculum to improve hydrogen and volatile fatty acid production using xylose as the substrate. Renew Energy 145:1388–1398. https://doi.org/10.1016/j.renene.2019.06.134
Öner EB, Akyol Ç, Bozan M et al (2018) Bioaugmentation with Clostridium thermocellum to enhance the anaerobic biodegradation of lignocellulosic agricultural residues. Bioresour Technol 249:620–625. https://doi.org/10.1016/j.biortech.2017.10.040
Pachapur VL, Kutty P, Brar SK, Ramirez AA (2016a) Enrichment of secondary wastewater sludge for production of hydrogen from crude glycerol and comparative evaluation of mono-, co- and mixed-culture systems. Int J Mol Sci 17:1–14. https://doi.org/10.3390/ijms17010092
Pachapur VL, Sarma SJ, Brar SK et al (2016b) Surfactant mediated enhanced glycerol uptake and hydrogen production from biodiesel waste using co-culture of Enterobacter aerogenes and Clostridium butyricum. Renew Energy 95:542–551. https://doi.org/10.1016/j.renene.2016.03.097
Paiders M, Nikolajeva V, Makarenkova G et al (2021) Changes in freshwater sediment microbial populations during fermentation of crude glycerol. Electron J Biotechnol 49:34–41. https://doi.org/10.1016/j.ejbt.2020.10.007
Palomo-Briones R, Montoya-Rosales JJ, Razo-Flores E (2021) Advances towards the understanding of microbial communities in dark fermentation of enzymatic hydrolysates: diversity, structure and hydrogen production performance. Int J Hydrogen Energy 46:27459–27472. https://doi.org/10.1016/j.ijhydene.2021.06.016
Pathak K, Reddy KM, Bakhshi NN, Dalai AK (2010) Catalytic conversion of glycerol to value added liquid products. Appl Catal A Gen 372:224–238. https://doi.org/10.1016/j.apcata.2009.10.036
Poleto L, Souza P, Magrini FE et al (2016) Selection and identification of microorganisms present in the treatment of wastewater and activated sludge to produce biohydrogen from glycerol. Int J Hydrogen Energy 41:4374–4381. https://doi.org/10.1016/j.ijhydene.2015.06.051
Rabelo CABS, Soares LA, Sakamoto IK et al (2018) Optimization of hydrogen and organic acids productions with autochthonous and allochthonous bacteria from sugarcane bagasse in batch reactors. J Environ Manage 223:952–963. https://doi.org/10.1016/j.jenvman.2018.07.015
Ratti RP, Botta LS, Sakamoto IK, Varesche MBA (2013) Microbial diversity of hydrogen-producing bacteria in batch reactors fed with cellulose using leachate as inoculum. Int J Hydrogen Energy 38:9707–9717. https://doi.org/10.1016/j.ijhydene.2013.05.089
Ratti RP, Delforno TP, Okada DY, Varesche MBA (2015) Bacterial communities in thermophilic H2-producing reactors investigated using 16S rRNA 454 pyrosequencing. Microbiol Res 173:10–17. https://doi.org/10.1016/j.micres.2015.01.010
Rodrigues CV, Santana KO, Nespeca MG et al (2016) Crude glycerol by transesterification process from used cooking oils: characterization and potentialities on hydrogen bioproduction. Int J Hydrogen Energy 41:14641–14651. https://doi.org/10.1016/j.ijhydene.2016.06.209
Rodrigues CV, Nespeca MG, Sakamoto IK et al (2019) Bioconversion of crude glycerol from waste cooking oils into hydrogen by sub-tropical mixed and pure cultures. Int J Hydrogen Energy 4:144–154. https://doi.org/10.1016/j.ijhydene.2018.02.174
Rodrigues CV, Santana KO, Nespeca MG et al (2020) Energy valorization of crude glycerol and sanitary sewage in hydrogen generation by biological processes. Int J Hydrogen Energy 45:11943–11953. https://doi.org/10.1016/j.ijhydene.2020.02.168
Santos SC, Rosa PRF, Sakamoto IK et al (2014) Organic loading rate impact on biohydrogen production and microbial communities at anaerobic fluidized thermophilic bed reactors treating sugarcane stillage. Bioresour Technol 159:55–63. https://doi.org/10.1016/j.biortech.2014.02.051
Sanz JL, Köchling T (2007) Molecular biology techniques used in wastewater treatment: an overview. Process Biochem 42:119–133. https://doi.org/10.1016/j.procbio.2006.10.003
Sarma SJ, Brar SK, Sydney EB et al (2012) Microbial hydrogen production by bioconversion of crude glycerol: a review. Int J Hydrogen Energy 37:6473–6490. https://doi.org/10.1016/j.ijhydene.2012.01.050
Schmieder R, Edwards R (2011) Quality control and preprocessing of metagenomic datasets. Bioinformatics 27:863–864. https://doi.org/10.1093/bioinformatics/btr026
Schwengber CA, Alves HJ, Schaffner RA et al (2016) Overview of glycerol reforming for hydrogen production. Renew Sustain Energy Rev 58:259–266. https://doi.org/10.1016/j.rser.2015.12.279
Seifert K, Waligorska M, Wojtowski M, Laniecki M (2009) Hydrogen generation from glycerol in batch fermentation process. Int J Hydrogen Energy 34:3671–3678. https://doi.org/10.1016/j.ijhydene.2009.02.045
Selembo PA, Perez JM, Lloyd WA, Logan BE (2009) High hydrogen production from glycerol or glucose by electrohydrogenesis using microbial electrolysis cells. Int J Hydrogen Energy 34:5373–5381. https://doi.org/10.1016/j.ijhydene.2009.05.002
Shah AT, Favaro L, Alibardi L et al (2016) Bacillus sp. strains to produce bio-hydrogen from the organic fraction of municipal solid waste. Appl Energy 176:116–124. https://doi.org/10.1016/j.apenergy.2016.05.054
Si B, Liu Z, Zhang Y et al (2015) Effect of reaction mode on biohydrogen production and its microbial diversity. Int J Hydrogen Energy 40:3191–3200. https://doi.org/10.1016/j.ijhydene.2015.01.030
Silva FMS, Oliveira LB, Mahler CF, Bassin JP (2017) Hydrogen production through anaerobic co-digestion of food waste and crude glycerol at mesophilic conditions. Int J Hydrogen Energy 42:22720–22729. https://doi.org/10.1016/j.ijhydene.2017.07.159
Sittijunda S, Reungsang A (2017) Fermentation of hydrogen, 1,3-propanediol and ethanol from glycerol as affected by organic loading rate using up-flow anaerobic sludge blanket (UASB) reactor. Int J Hydrogen Energy 42:27558–27569. https://doi.org/10.1016/j.ijhydene.2017.05.149
Sivagurunathan P, Sen B, Lin CY (2014) Batch fermentative hydrogen production by enriched mixed culture: combination strategy and their microbial composition. J Biosci Bioeng 117:222–228. https://doi.org/10.1016/j.jbiosc.2013.07.015
Sun L, Huang A, Gu W et al (2015) Hydrogen production by Enterobacter cloacae isolated from sugar refinery sludge. Int J Hydrogen Energy 40:1402–1407. https://doi.org/10.1016/j.ijhydene.2014.11.121
Sun YQ, Shen JT, Yan L et al (2018) Advances in bioconversion of glycerol to 1,3-propanediol: Prospects and challenges. Process Biochem 71:134–146. https://doi.org/10.1016/j.procbio.2018.05.009
Tapia-Venegas E, Cabrol L, Brandhoff B et al (2015) Adaptation of acidogenic sludge to increasing glycerol concentrations for biohydrogen production. Appl Microbiol Biotechnol 99:8295–8308. https://doi.org/10.1007/s00253-015-6832-6
Toledo-Alarcón J, Cabrol L, Jeison D et al (2020) Impact of the microbial inoculum source on pre-treatment efficiency for fermentative H2 production from glycerol. Int J Hydrogen Energy 45:1597–1607. https://doi.org/10.1016/j.ijhydene.2019.11.113
Varrone C, Rosa S, Fiocchetti F et al (2013) Enrichment of activated sludge for enhanced hydrogen production from crude glycerol. Int J Hydrogen Energy 38:1319–1331. https://doi.org/10.1016/j.ijhydene.2012.11.069
Vartoukian SR, Palmer RM, Wade WG (2010) Strategies for culture of “unculturable” bacteria. FEMS Microbiol Lett 309:1–7. https://doi.org/10.1111/j.1574-6968.2010.02000.x
Wadjeam P, Reungsang A, Imai T, Plangklang P (2019) Co-digestion of cassava starch wastewater with buffalo dung for bio-hydrogen production. Int J Hydrogen Energy 44:14694–14706. https://doi.org/10.1016/j.ijhydene.2019.04.138
Wang CC, Chang CW, Chu CP et al (2003) Producing hydrogen from wastewater sludge by Clostridium bifermentans. J Biotechnol 102:83–92. https://doi.org/10.1016/S0168-1656(03)00007-5
Wenzel J, Fuentes L, Cabezas A, Etchebehere C (2017) Microbial fuel cell coupled to biohydrogen reactor: a feasible technology to increase energy yield from cheese whey. Bioprocess Biosyst Eng 40:807–819. https://doi.org/10.1007/s00449-017-1746-6
Wong YM, Wu TY, Ling TC et al (2018) Evaluating new bio-hydrogen producers: clostridium perfringens strain JJC, Clostridium bifermentans strain WYM and Clostridium sp. strain Ade.TY. J Biosci Bioeng 125:590–598. https://doi.org/10.1016/j.jbiosc.2017.12.012
Yang G, Yin Y, Wang J (2019) Microbial community diversity during fermentative hydrogen production inoculating various pretreated cultures. Int J Hydrogen Energy 44:13147–13156. https://doi.org/10.1016/j.ijhydene.2019.03.216
Yin Y, Chen Y, Wang J (2021) Co-fermentation of sewage sludge and algae and Fe2+ addition for enhancing hydrogen production. Int J Hydrogen Energy 46:8950–8960. https://doi.org/10.1016/j.ijhydene.2021.01.009
Zahedi S, Solera R, García-Morales JL, Sales D (2016) Effect of the addition of glycerol on hydrogen production from industrial municipal solid waste. Fuel 180:343–347. https://doi.org/10.1016/j.fuel.2016.04.063
Zappi A, Hernandez R, Holmes WE (2021) A review of hydrogen production from anaerobic digestion. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-020-03117-w
Zhou JJ, Shen JT, Jiang LL et al (2017) Selection and characterization of an anaerobic microbial consortium with high adaptation to crude glycerol for 1,3-propanediol production. Appl Microbiol Biotechnol 101:5985–5996. https://doi.org/10.1007/s00253-017-8311-8
Acknowledgements
We thank High-Performance Computing Lab—LAD/PUCRS for allowing access to run the high-throughput sequence analyses.
Author information
Authors and Affiliations
Contributions
AS designed the study, analyzed the data and wrote the paper; LGA and DA carried out the experiments and analyzed the data; FEV analyzed the data and wrote the paper; LM, LGB and AG performed bioinformatics analyses and modified the manuscript; SP wrote the paper and supervised the research.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
The authors declare to have participated in the study and authorize its publication without personal financial interests.
Consent for publication
All authors approved the manuscript and agree to the submission to the Brazilian Journal of Chemical Engineering.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Paesi, S., Schiavenin, A., Almeida, L.G. et al. Comparison of two different kinds of seed sludge and characterization of microorganisms producing hydrogen and soluble metabolites from raw glycerol. Braz. J. Chem. Eng. 39, 387–402 (2022). https://doi.org/10.1007/s43153-021-00212-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-021-00212-4