Abstract
This study aimed to investigate the effect of diagnosis and treatment of chronic endometritis (CE) on the outcome of assisted reproductive technology (ART) with or without repeated implantation failure (RIF). This retrospective analysis included patients who underwent pathological examination for diagnosis of CE at Yamagata University Hospital. The examination was performed for all patients planned for ART with or without RIF. Patients who were examined within 6 months of the first oocyte retrieval or embryo transfer were included. We counted the number of CD138-positive cells within the endometrial stroma in patients’ specimens and analyzed the patients’ clinical information. Clinical rates of pregnancy and implantation were determined. A total of 80 women met the inclusion criteria: 13 CE-negative patients (17.3%) and 67 CE-positive patients (83.7%). A significant decrease was noted in the CD138-positive cell count between the first biopsy and second biopsy after CE treatment (p < 0.001). In addition, no significant differences were noted in ongoing pregnancy rates between the CE-negative patients and those who underwent CE treatment. The CD138-positive cell counts at first biopsy tended to be lower in each pregnancy group than in the non-pregnancy group. For patients planned to undergo ART, examination for diagnosis of CE with or without RIF could be considered. Pathological CD138-positive cell counts were considered useful for CE diagnosis and treatment decision-making. The study findings suggest the efficacy of antimicrobial agents in CE treatment, contributing to improved pregnancy outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic endometritis (CE) is characterized by the persistence of bacteria or other antigens in the uterus after menstrual periods, eliciting an immunological response in the endometrium [1]. CE has been reported to be one of the causes of repeated implantation failure (RIF) in assisted reproductive technology (ART) [2], prompting widespread diagnostic and therapeutic efforts across various medical facilities.
Previous studies have reported that CE is diagnosed in approximately 14%–67.5% of RIF patients [3,4,5,6,7,8], and the efficacy of oral antibiotic treatment has been documented [5, 9, 10]. Furthermore, successful resolution of CE in RIF patients has been associated with improved fertility outcomes [11]. Despite these advancements, practical challenges persist with two major problems. First, the diagnosis of CE often occurs after multiple embryo transfer attempts for the following reason. Currently, examination for diagnosis of CE is indicated only for patients with RIF. RIF is typically diagnosed in ART settings when four or more good embryos and three or more transfers fail to result in pregnancy [2]. In other words, patients with CE are forced to undergo multiple embryo transfers before treatment of CE. There could be a substantial number of CE at the time of starting ART because CE has been reported in approximately 2.8%–56.8% of all infertile patients [12,13,14]. Therefore, we believe that early examination of CE, possibly conducted not for the patients with RIF but before the first embryo transfer like as screening test, may improve pregnancy outcomes by facilitating timely CE treatment. Second, methods of examination and diagnostic criteria for CE have not been established; therefore, indications for treatment vary across medical facilities. Currently, the main diagnostic method involves quantification of CD138-positive cells in endometrial tissue; however, common criteria with a universally accepted threshold for CE-positive cell count have not been defined [15]. It can be useful to create an index of diagnosis based on the count of CD138 cells, which is an objective assessment, rather than hysteroscopy, which relies on subjective assessment.
Therefore, this study aimed to examine the effect of the diagnosis and treatment of CE on the outcome of ART and report a retrospective review of our experience in examination and treatment of CE in ART patients, regardless of the diagnosis of RIF, at our institution.
Materials and Methods
This study was approved by the Ethics Committee of Yamagata University Faculty of Medicine (approval number: 202061).
Patients who underwent pathological examination for diagnosis of CE between January 2019 and December 2020 at Yamagata University Hospital were included in the study. During this period, if a patient undergoing ART requested for examination of CE, it was performed regardless of diagnosis of RIF. Therefore, the study cohort included both cases before and after the start of ART. To reduce the effect of timing of the examination, only patients who were examined within 6 months of the first oocyte retrieval or embryo transfer (ET) were included. Patient information including age, gravidity, parity, serum anti-mullerian hormone (AMH) level, antral follicle count (AFC), number of cycles of artificial insemination or ET performed prior to diagnosis of CE, and underlying diseases [uterine myoma, endometriosis, polycystic ovarian syndrome (PCOS), endometrial polyps] that may have contributed to infertility was extracted from their medical records. Patients with factors of infertility other than CE were excluded: those who were unable to obtain embryos due to poor ovarian reserve or fertilization failure, those who were unable to obtain an endometrial tissue biopsy, those who became pregnant by methods other than embryo transfer, and those who did not visit the hospital after pathological examination.
Diagnosis of CE
We performed biopsy of endometrial tissue during the follicular phase within 6 months of the first oocyte retrieval or embryo transfer. First, hysteroscopic observation of intrauterine tissue was performed, and after removing sufficient saline solution from the uterine cavity, endometrial biopsy was conducted using endosuction® (HAKKO CO, Japan). The endometrium was pathologically fixed, and the specimen was stained with CD138. Pathological diagnosis of CE was performed through counting the number of CD138-positive cells within the endometrial stroma in the specimen. Immunostaining was performed, and the pathological specimens were evaluated by pathologists. The CD138 cell count was defined as the total number of positive cells in 10 high power fields (HPFs). A CE-positive specimen was defined as having at least one CD138-positive cell count in 10 HPFs, whereas a CE-negative specimen was defined as having a zero CD138-positive cell count in 10 HPFs. The successful therapy of CE was defined as zero CD138-positive cell count on the second biopsy after the antibiotic medication, whereas residual CE was defined as 1 or more CD138-positive cell count after antibiotic medication. Intrauterine findings using hysteroscopes were considered only for reference findings and were not considered as treatment criteria.
Treatment of CE
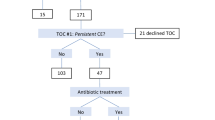
The treatment protocol for CE at our hospital between January 2019 and December 2020 is depicted in Fig. 1. For antimicrobial selection, we referred to the previous reports [5, 16] because 70–90% of plasma cells were reported to have been reduced. The first-line treatment comprised 100 mg of oral doxycycline, administered twice per day, for 14 days. The duration of dosing in the menstrual cycle were not specifically standardized. To determine the efficacy of CE treatment, the endometrial tissue was biopsied and immunostained again (second biopsy) in the proliferative phase immediately after the end of oral administration of the above-mentioned medication. The group that had residual CE after the first dose was treated with second-line drugs. The second-line treatment comprised oral metronidazole (250 mg, twice per day), ciprofloxacin hydrochloride (200 mg, twice per day), and antibiotic-resistant lactic acid bacteria (1 g, three times per day), administered for 14 days. After the second-line treatment was administered, the treatment of CE was terminated, and the plan was to perform ET in all patients.
Outcomes
Outcomes were defined as positive proof for pregnancy test (serum human chorionic gonadotropin: hCG levels rising above 20 mIU/mL around day 10 after embryo transfer) and ongoing pregnancy or live birth. An ongoing pregnancy was defined as a pregnancy that continued for more than 12 weeks.
Statistical Analysis
R software was used for statistical analysis. One-way analysis of variance (ANOVA), Fisher's exact probability, Mann-Whitney U, and Wilcoxon signed rank sum tests were used. A P-value < 0.05 was considered statistically significant.
Results
In this study, 110 patients underwent pathological examination of CE between January 2019 and December 2020; of these, 80 were included in the study. Overall, 30 patients were excluded as follows: 14 patients could not undergo endometrial biopsy (Timing of the test was not right, or there was insufficient specimen, etc.), 5 patients got pregnant without embryo transfer (natural pregnancy), and 11 patients could not undergo embryo transfer after the biopsy (Abandoned treatment, transferred to another hospital, etc.). At the first biopsy, there were 13 CE-negative patients (16.3%, defined as the “CE-negative group”) and 67 CE-positive patients (83.7%). At the second biopsy, wherein another biopsy was performed after the first-line treatment for the 67 CE-positive patients, CD138 cells disappeared in 21 patients (defined as “CE first-line group”), and the remaining 46 patients were subjected to second-line treatment (defined as “CE second-line group”). No significant differences were noted between these three groups, including history of pregnancy and infertility treatment (Table 1). A slightly higher number of patients with uterine myoma and endometriosis was observed in the CE-negative group; however, it was considered that the results would not be affected because of the small number of patients.
The main outcome of the study is shown in Table 2. We compared outcomes in the CE-negative, CE first-line, and CE second-line groups. No significant differences were noted in ongoing pregnancy/live birth rates between these three groups (CE-negative, CE first-line, and CE second-line groups: 46.1%, 47.6%, and 39.1%, respectively, p = 0.78); however, the CE second-line group (54.3%) showed significantly lower pregnancy rates than did the CE-negative group (84.6%, p = 0.01) or CE first-line group (76.1%, p = 0.02). The pregnancy rates were not significantly different between the CE-negative and CE first-line groups (p = 0.56).
A significant decrease was noted in the number of CD138-positive cells between the first and secondary biopsies (p < 0.001). Further, we evaluated whether the number of CD138-positive cells affected pregnancy outcome regardless of treatment. The number of CD138-positive cells at the first biopsy tended to be lower in the pregnancy groups than in the non-pregnancy group: 9 (4–15) cells /10 HPFs, 6 (4–9) cells /10 HPFs, p = 0.080 (Fig. 2A). Further, no significant difference was noted in the non-pregnancy group and ongoing pregnancy/live birth group: 6 (2.25–10.25) cells /10 HPFs, p = 0.073 (Fig. 2B). In addition, the number of CD138-positive cells at the second biopsy tended to be lower in the pregnancy group than in the non-pregnancy group: non-pregnancy and pregnancy group, 7 (3–8.5) cells /10 HPFs and 3 (1–6) cells /10 HPFs, respectively, p = 0.098. Meanwhile, the number of CD138-positive cells at the second biopsy was significantly lower in the ongoing pregnancy or live birth group than that in the non-pregnancy group (non-pregnancy, ongoing pregnancy or live birth: 7 (3–8.5) cells /10 HPFs, 2 (1–5) cells /10 HPFs, p = 0.020 (Fig. 3).
A: Comparison of CD138 positive cell counts between non-pregnancy and pregnancy groups at second biopsy performed to diagnose whether CE has been cured. B: Comparison of CD138 positive cell counts between non-pregnancy and ongoing pregnancy or live birth groups at second biopsy. CE: chronic endometritis
Discussion
This is the first study investigating the association between CE and ART outcomes regardless of the existence of RIF. Unexpectedly, we found that many patients undergoing ART before the diagnosis of RIF already had CE. For these patients, antibiotic treatment of CE certainly reduced the number of CD138-positive cells, serving as a possible predictor of subsequent fertility outcomes. These factors suggest that the diagnosis and treatment of CE may be considered not only for patients diagnosed with RIF but also for all patients planning to undergo ART. In addition, our study suggests the efficacy of evaluating CD138-positive cell count in the diagnosis and treatment of CE. We believe that the indications for the diagnosis and treatment of CE could be expanded beyond the currently existing ones.
CE is a chronic inflammation condition of the endometrium caused by various factors. Previous reports have included history of intrauterine device (IUD) insertion [17, 18], multiple pregnancies or abnormal uterine bleeding [17], and bacterial vaginosis, endometrial polyps, or endometriosis [19,20,21,22] as causes.
In this study, we evaluated whether background characteristics of patients, including history of intrauterine manipulation (artificial insemination or ET), affected the occurrence of CE; however, we could not find any specific correlation. Although significant differences were noted for myoma and endometriosis, these factors need not be necessarily considered, as the number of patients in this study were very small.
Generally, diagnosis and treatment of CE should be performed on patients with RIF; however, we consider that all patients who are planning to undergo ART should be indicated. Currently, CE has been reported in approximately 2.8%–56.8% of infertile patients [12,13,14], approximately 14%–67.5% of RIF patients [3,4,5,6,7,8], and approximately 9.3%–67.6% of patients with repeated miscarriages [23, 24]. This variation among reports might be attributed to the different examination methods and diagnostic criteria. In this study, we defined CE positivity as “CD138-positive cells ≥ 1/10 HPFs,” with reference to a previous study [25], and the positivity rate was as high as 83.7% for patients planning to undergo ART with or without RIF. Although a relatively strict definition, such as the one mentioned above, may have influenced this high rate, it was suggested that a significant proportion of patients planning ART could already have CE. Meanwhile, the number of CD138-positive cells was significantly decreased when antimicrobial agents were administered to patients with CE in this study. A high number of CD138-positive cells in the endometrium is thought to have a negative effect on the pregnancy and ongoing pregnancy rates in ART [26,27,28]. Hence, decreasing CD138 positive cells with antimicrobial therapy for such CE patients may be beneficial for pregnancy. In practice, no significant differences were noted in the ongoing pregnancy rates between the CE-negative group and the groups after CE treatment. It is possible that CE treatment reduced the number of CD138-positive cells, resulting in improved and comparable pregnancy outcomes in the CE-positive patients. However, the low pregnancy rate in the CE second-line group suggests that the treatment effect may not be sufficient in severe cases; therefore, early diagnosis of CE and first-line treatment may be beneficial before it becomes severe. In the ongoing pregnancy or live birth group, CD138-positive cell counts were significantly lower at the second biopsy (Fig. 3), suggesting that the reduction in the number of CD138 cells caused by first-line antibiotic treatment could be important. Taken together, it may be useful to consider patients with diagnosis of CE and antibiotic treatment before the planned ART stage.
Our results also suggest the efficacy of evaluating the number of CD138-positive cells in the endometrium in the diagnosis of CE. To the best of our knowledge, no study has reported on pathological evaluation before and after antimicrobial therapy. Currently, CE is diagnosed through hysteroscopy, pathological examination, bacteriological testing, or a combination of two of these methods, with many medical facilities generally using both hysteroscopy and pathological examinations, given their simplicity [8, 16, 21]. During hysteroscopy, “strawberry” aspect, focal hyperemia, hemorrhagic spots, diffuse micropolyps, and stromal edema have been reported as findings that predict CE [29]. Using hysteroscopy, the diagnosis can be made immediately upon examination. However, even in cases where hysteroscopy shows no abnormal findings, pathological examinations often reveal CE. A review by Fani et al. [8] stated that hysteroscopy was not suitable as a first choice for CE diagnosis because of the high risk of bias. Similarly, in our study, the findings of hysteroscopy were often viewed differently by each physician. The positive predictive value of CE through hysteroscopy to pathological examination was 0.886 (95% confidence interval 0.754–0.962), whereas the negative predictive value was only 0.222 (95% confidence interval 0.101–0.392) (data not shown). Therefore, it was considered that hysteroscopy should be in the category of adjunctive diagnosis, as it could not provide reproducible results. Meanwhile, CD138 immunostaining specific for plasma cells has been commonly used for pathological diagnosis of CE. A higher number of CD138-positive cells was associated with reduced subsequent fertility outcomes, and the number of CD138-positive cells was considered to be able to assess the disease status of CE [13, 14, 23]. Although pathological examination took longer than hysteroscopy, it had the advantage of greater objectivity and less bias [8]. Our study also suggested that the number of CD138-positive cells could be a predictor of pregnancy outcome through evaluating CD138-positive cells before and after treatment.
The standard treatment for CE comprises administration of antimicrobial agents, but no general regimen has been established to date. Kitaya et al. reported that 92.3% of patients with CE improved upon treatment with doxycycline (200 mg/day for 14 days), while the remaining patients received ciprofloxacin (400 mg/day for 14 days) and metronidazole (500 mg/day for 14 days), and reported an overall response rate of 99.1% [16]. We referred to this as the treatment protocol at our facility. In other studies, a combination of doxycycline, ciprofloxacin, and metronidazole [5] or ofloxacin and metronidazole [9] has been reported to be effective for the treatment of CE. Cicinelli et al. treated CE with a systematic antibiotic regimen according to the microbial profile of the endometrium [10]. As described above, various studies have reported on antimicrobial therapy for CE; however, previous reports have only determined whether CE was cured and have not discussed the reduction in the number of CD138-positive cells after antibiotic treatment. In this study, we found for the first time that the number of CD138-positive cells was significantly reduced after antimicrobial therapy, which can be useful as an indicator for evaluating the efficacy of antimicrobial treatment.
This study has several limitations. We could not evaluate the number of CD138-positive cells after second-line antimicrobial treatment pathologically. Therefore, the CE of some patients in the treatment group may not yet be cured. In such cases, further treatment of CE may need to be considered. Second, because this study was a retrospective study, and all patients who had CE were treated, direct comparisons between untreated and treated cases were not possible. Further studies with prospective designs are needed to evaluate the accurate effect of CE treatment. However, the usefulness of the pathological examination of CE and the therapeutic effect were at least demonstrated through our study.
CE was found to be more common than that expected in patients planning for ART. The findings suggest that antimicrobial therapy in these patients may have benefited pregnancy outcomes. Further, the diagnosis and treatment of CE should be considered not only for patients with RIF but also for all patients planning to undergo ART like as screening test. In addition, the evaluation of CD138-positive cell count was found to be objective as well as useful as an indicator of treatment. Therefore, pathological examination could be the preferred assessment for diagnosis of CE. Furthermore, many CE-positive patients are said to have an abnormal pattern in endometrial receptivity analysis (ERA) [30]. Taken together, early detection of CE is expected to be a novel key investigation in future ART treatment strategies. Further research is needed on these relationships, including the availability and efficacy of treatment of CE and endometrial receptivity. We plan to conduct these additional studies of early CE treatment and other adjunctive therapies.
In conclusion, for all patients planning to undergo ART, examination for diagnosis of CE should be considered, regardless of existence of RIF, and pathological CD138-positive cell counts could be useful as a method for diagnosis of CE and treatment decision-making. In addition, antimicrobial agents could be useful in the treatment of CE, contributing to better pregnancy outcomes.
Data Availability
The data are available from the corresponding author, I.T. upon reasonable request.
Code Availability
Not applicable.
References
Greenwood SM, Moran JJ. Chronic endometritis: morphologic and clinical observations. Obstet Gynecol. 1981;58:176–84.
Coughlan C, Ledger W, Wang Q, Liu F, Demirol A, Gurgan T, et al. Recurrent implantation failure: definition and management. Reprod Biomed Online. 2014;28:14–38. https://doi.org/10.1016/j.rbmo.2013.08.011.
Liu Y, Chen X, Huang J, Wang CC, Yu MY, Laird S, et al. Comparison of the prevalence of chronic endometritis as determined by means of different diagnostic methods in women with and without reproductive failure. Fertil Steril. 2018;109:832–9. https://doi.org/10.1016/j.fertnstert.2018.01.022.
Bouet PE, El Hachem H, Monceau E, Gariépy G, Kadoch IJ, Sylvestre C. Chronic endometritis in women with recurrent pregnancy loss and recurrent implantation failure: prevalence and role of office hysteroscopy and immunohistochemistry in diagnosis. Fertil Steril. 2016;105:106–10. https://doi.org/10.1016/j.fertnstert.2015.09.025.
Johnston-MacAnanny EB, Hartnett J, Engmann LL, Nulsen JC, Sanders MM, Benadiva CA. Chronic endometritis is a frequent finding in women with recurrent implantation failure after in vitro fertilization. Fertil Steril. 2010;93:437–41. https://doi.org/10.1016/j.fertnstert.2008.12.131.
Song D, Feng X, Zhang Q, Xia E, Xiao Y, Xie W, et al. Prevalence and confounders of chronic endometritis in premenopausal women with abnormal bleeding or reproductive failure. Reprod Biomed Online. 2018;36:78–83. https://doi.org/10.1016/j.rbmo.2017.09.008.
Tersoglio AE, Salatino DR, Reinchisi G, Gonzalez A, Tersoglio S, Marlia C. Repeated implantation failure in oocyte donation. What to do to improve the endometrial receptivity? JBRA Assist Reprod. 2015;19:44–52. https://doi.org/10.5935/1518-0557.20150012.
Yang R, Du X, Wang Y, Song X, Yang Y, Qiao J. The hysteroscopy and histological diagnosis and treatment value of chronic endometritis in recurrent implantation failure patients. Arch Gynecol Obstet. 2014;289:1363–9. https://doi.org/10.1007/s00404-013-3131-2.
McQueen DB, Bernardi LA, Stephenson MD. Chronic endometritis in women with recurrent early pregnancy loss and/or fetal demise. Fertil Steril. 2014;101:1026–30. https://doi.org/10.1016/j.fertnstert.2013.12.031.
Cicinelli E, Matteo M, Tinelli R, Lepera A, Alfonso R, Indraccolo U, et al. Prevalence of chronic Endometritis in repeated unexplained implantation failure and the IVF success rate after antibiotic therapy. Hum Reprod. 2015;30:323–30. https://doi.org/10.1093/humrep/deu292.
Vitagliano A, Saccardi C, Noventa M, Di Spiezio SA, Saccone G, Cicinelli E, et al. Effects of chronic endometritis therapy on in vitro fertilization outcome in women with repeated implantation failure: A systematic review and metaanalysis. Fertil Steril. 2018;110:103-112.e1. https://doi.org/10.1016/j.fertnstert.2018.03.017.
Kasius JC, Fatemi HM, Bourgain C, Sie-Go DM, Eijkemans RJ, Fauser BC, et al. The impact of chronic endometritis on reproductive outcome. Fertil Steril. 2011;96:1451–6. https://doi.org/10.1016/j.fertnstert.2011.09.039.
Cicinelli E, Resta L, Nicoletti R, Tartagni M, Marinaccio M, Bulletti C, et al. Detection of chronic endometritis at fluid hysteroscopy. J Minim Invasive Gynecol. 2005;12:514–8. https://doi.org/10.1016/j.jmig.2005.07.394.
Cicinelli E, Matteo M, Trojano G, Mitola PC, Tinelli R, Vitagliano A, et al. Chronic endometritis in patients with unexplained infertility: Prevalence and effects of antibiotic treatment on spontaneous conception. Am J Reprod Immunol. 2018;79:e12782. https://doi.org/10.1111/aji.12782.
Kimura F, Takebayashi A, Ishida M, Nakamura A, Kitazawa J, Morimune A, et al. Review: chronic endometritis and its effect on reproduction. J Obstet Gynaecol Res. 2019;45:951–60. https://doi.org/10.1111/jog.13937.
Kitaya K, Matsubayashi H, Takaya Y, Nishiyama R, Yamaguchi K, Takeuchi T, et al. Live birth rate following oral antibiotic treatment for chronic endometritis in infertile women with repeated implantation failure. Am J Reprod Immunol. 2017;78:12719. https://doi.org/10.1111/aji.12719.
Kitaya K, Yasuo T. Immunohistochemistrical and clinicopathological characterization of chronic endometritis. Am J Reprod Immunol. 2011;66:410–5. https://doi.org/10.1111/j.1600-0897.2011.01051.x.
Moyer DL, Mishell DR Jr, Bell J. Reactions of human endometrium to the intrauterine device. Correlation of the endometrial histology with the bacterial environment of the uterus following short-term insertion of the IUD. Am J Obstet Gynecol. 1970;106:799–809. https://doi.org/10.1016/0002-9378(70)90470-9.
Beruchashvili M, Gogiashvili L, Datunashvili E, Topuria Z, Tsagareli Z. Morphological peculiarities of endometrium in chronic endometritis associated with bacterial vaginosis. Georgian Med News. 2010;181:59–63.
Peipert JF, Montagno AB, Cooper AS, Sung CJ. Bacterial vaginosis as a risk factor for upper genital tract infection. Am J Obstet Gynecol. 1997;177:1184–7. https://doi.org/10.1016/s0002-9378(97)70038-3.
Takebayashi A, Kimura F, Kishi Y, Ishida M, Takahashi A, Yamanaka A, et al. The association between endometriosis and chronic endometritis. PLoS ONE. 2014;9:e88354. https://doi.org/10.1371/journal.pone.0088354.
Carvalho FM, Aguiar FN, Tomioka R, de Oliveira RM, Frantz N, Ueno J. Functional endometrial polyps in infertile asymptomatic patients: A possible evolution of vascular changes secondary to endometritis. Eur J Obstet Gynecol Reprod Biol. 2013;170:152–6. https://doi.org/10.1016/j.ejogrb.2013.05.012.
Zolghadri J, Momtahan M, Aminian K, Ghaffarpasand F, Tavana Z. The value of hysteroscopy in diagnosis of chronic endometritis in patients with unexplained recurrent spontaneous abortion. Eur J Obstet Gynecol Reprod Biol. 2011;155:217–20. https://doi.org/10.1016/j.ejogrb.2010.12.010.
Kitaya K. Prevalence of chronic endometritis in recurrent miscarriages. Fertil Steril. 2011;95:1156–8. https://doi.org/10.1016/j.fertnstert.2010.09.061.
Kitaya K, Yasuo T. Aberrant expression of selectin E, CXCL1, and CXCL13 in chronic endometritis. Mod Pathol. 2010;23:1136–46. https://doi.org/10.1038/modpathol.2010.98.
Fan X, Li X, Li Y, Liao J, Chen H, Li Y, et al. Endometrial CD138 count appears to be a negative prognostic indicator for patients xho have experienced previous embryo transfer failure. Fertil Steril. 2019;112:1103–11. https://doi.org/10.1016/j.fertnstert.2019.08.006.
Li Y, Xu S, Yu S, Huang C, Lin S, Chen W, et al. Diagnosis of chronic endometritis: How many CD138+ cells/HPF in endometrial stroma affect pregnancy outcome of infertile women? Am J Reprod Immunol. 2021;85:e13369. https://doi.org/10.1111/aji.13369.
Rimmer MP, Fishwick K, Henderson I, Chinn D, Al Wattar BH, Quenby S. Quantifying CD138+ cells in the endometrium to assess chronic endometritis in women at risk of recurrent pregnancy loss: A prospective cohort study and rapid review. J Obstet Gynaecol Res. 2021;47:689–97. https://doi.org/10.1111/jog.14585.
Cicinelli E, Vitagliano A, Kumar A, Lasmar RB, Bettocchi S, Haimovich S, et al. Unified diagnostic criteria for chronic endometritis at fluid hysteroscopy: Proposal and reliability evaluation through an international randomized-controlled observer study. Fertil Steril. 2019;112:162-173.e2. https://doi.org/10.1016/j.fertnstert.2019.03.004.
Kroda K, Horikawa T, Moriyama A, Nakao K. Juen H er al, Impact of chronic endometritis on endometrial receptivity analysis results and pregnancy outcomes. Immun Inflamm Dis. 2020;8:650–8. https://doi.org/10.1002/iid3.354.
Author information
Authors and Affiliations
Contributions
KS designed the study, performed statistical analyses, and drafted the manuscript. IT participated in coordinating the study design and revised the manuscript. KS, IT, HK, FN, NN, KT, JM, and KM participated in data collection. SN revised and approved the final version of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The study was approved by our institution’s IRB (IRB No. 202061).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sakai, K., Takehara, I., Kaneko, H. et al. Impact of Diagnosis and Treatment of Chronic Endometritis on Outcomes Before Starting Assisted Reproductive Technology: A Retrospective Study. Reprod. Sci. (2024). https://doi.org/10.1007/s43032-024-01633-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43032-024-01633-5