Abstract
This paper will review a remarkable new approach to in vitro maturation “IVM” of oocytes from ovarian tissue, based on our results with in vitro oogenesis from somatic cells. As an aside benefit we also have derived a better understanding of ovarian longevity from ovary transplant. We have found that primordial follicle recruitment is triggered by tissue pressure gradients. Increased pressure holds the follicle in meiotic arrest and prevents recruitment. Therefore recruitment occurs first in the least dense inner tissue of the cortico-medullary junction. Many oocytes can be obtained from human ovarian tissue and mature to metaphase 2 in vitro with no need for ovarian stimulation. Ovarian stimulation may only be necessary for removing the oocyte from the ovary, but this can also be accomplished by simple dissection at the time of ovary tissue cryopreservation. By using surgical dissection of the removed ovary, rather than a needle stick, we can obtain many oocytes from very small follicles not visible with ultrasound. A clearer understanding of ovarian function has come from in vitro oogenesis experiments, and that explains why IVM has now become so simple and robust. Tissue pressure (and just a few “core genes” in the mouse) direct primordial follicle recruitment and development to mature oocyte, and therefore also control ovarian longevity. There are three distinct phases to oocyte development both in vitro and in vivo: in vitro differentiation “IVD” which is not gonadotropin sensitive (the longest phase), in vitro gonadotropin sensitivity “IVG” which is the phase of gonadotropin stimulation to prepare for meiotic competence, and IVM to metaphase II. On any given day 35% of GVs in ovarian tissue have already undergone “IVD” and “IVG” in vivo, and therefore are ready for IVM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Two objectives for improving preservation of fertility in cancer patients are IVM (in vitro maturation) of oocytes from ovarian tissue, and IVG (in vitro gametogenesis) of oocytes from somatic cells transformed into stem cells. Those two objectives are now clinically within reach, and both reinforce each other.
For IVM, studies in the last decade from Belgium and Denmark, more recently the USA, have shown that GV (germinal vesicle) oocytes retrieved from ovarian tissue dissection (not via ultrasound guided needle aspiration) can be readily cultured to M2 (metaphase 2) mature, functional oocytes [1,2,3,4]. Furthermore, these in vitro cultured M 2 oocytes have resulted in live births [3, 5,6,7].
For IVG, in mice, somatic cells have been transformed into functional M 2 oocytes leading to live births [8,9,10,11,12,13,14,15,16]. In humans normal PGCs (primordial germ cells) have been generated from skin biopsy in azoospermic men and in post menopausal women over 50 years of age [8,9,10,11,12,13,14,15,16,17,18,19,20]. It may be only a matter of time before these human PGCs can be fully transformed into functional oocytes. Skin cells have also been transformed into PGCs in endangered species in order to eventually resurrect them. The Northern white rhino is now extinct, but their skin cells have been transformed into beautiful IPS cells and PGCs [21].
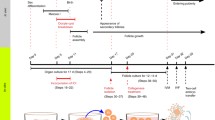
The success of in vitro gametogenesis in generating live offspring from somatic cell transformation has helped to elucidate the mechanism controlling primordial follicle arrest, recruitment, and ovarian longevity [12, 14, 22, 23]. In mice, using eight “core genes” and low pressure, primordial follicles can be unlocked and cultured to M 2 oocytes. Furthermore, in mice, fetal granulosa cells can also be produced from adult skin cells transformed into IPS cells (stem cells). Fetal, not adult, granulosa cells are necessary to convert PGCs into complete oocytes. These “manufactured” fetal granulosa cells can then be used to produce competent M 2 oocytes from already produced PGCL cells (PGCs) in mice [13]. Since PGCs have been robustly produced now in humans from skin biopsy cells, all that will be needed to produce competent M 2 oocytes in adult humans from skin biopsy is to find the species specific genes in the human (using Yoshino et al methods) that convert skin cells to fetal granulosa cells [13]. Even now, we can use this clearer knowledge of ovarian-oocyte physiology to simplify greatly the use of IVM for fertility preservation in humans from ovarian tissue (Fig. 1).
Comparison of in vivo oogenesis to in vitro oogenesis from stem cells [11]
In vitro Maturation Of Oocytes from Ovarian Tissue
It is clinically not yet possible to attempt the task of maturing human primordial follicles in vitro. However, it will be possible in the future utilizing the “eight core genes” approach [14]. But at present, it is simpler and indeed quite robust just to perform IVM on GV oocytes that have already been prepared “naturally” for maturation in vivo (by “IVD” and “IVG” phases). This simple approach arose out of our efforts in the mouse to transform skin cells to functional oocytes (in vitro gametogenesis), which has solved with clarity some of the mysteries of ovarian function. The problem in the past was doing too many steps that hindered rather than helped results. A clearer understanding of ovarian function and of in vitro gametogenesis explains the great simplicity of IVM.
It is much easier to obtain many GVs from dissection then needle aspiration. That is one key to the robustness of IVM. Do not interfere with natural meiotic competence by ovarian stimulation which only is useful for follicle enlargement. At the time of oophorectomy for ovarian tissue cryopreservation, when the ovarian cortex has been dissected from the medulla (Fig. 2) and divided into slices for cryopreservation, the “spent” medium in which the dissection took place should be examined for many free, loose cumulus complexes, which usually contain immature germinal vesicle oocytes [4]. These cumulus complexes are then placed in culture with widely varying concentrations of FSH and human chorionic gonadotrophin (HCG) or LH. The cumulus is stripped between 24 and 44 h later (Fig. 3a–d). The reason that a variety of media and gonadotrophin concentrations were employed in our center is based on our previously published data from in-vitro gametogenesis in mice [8,9,10,11,12, 14]. We wished to see whether IVM could proceed in a variety of various media because the oocytes have already been able to develop meiotic competence from in vivo “IVD” and IVG” (Fig. 1). IVD is the phase of primordial follicle development (3 weeks in mouse; 4.5 months in human) that is inert to gonadotropin. IVG in this model does not mean in vitro gametogenesis. It means in vitro gonadotropin sensitivity.
a Day 0 Compact appearance of cumulus complexes (CCs) at time of dissection [4]. b Day 1 Spreading of cumulus complexes after 24 h in culture with HCG or LH [4]. c Day 1 Metaphase II oocytes after 24 h in culture with HCG or LH [4]. d Three normal M II oocytes, one M I oocyte, and one degenerated oocyte, resulting from cultured ovarian tissue GVs, on day 2 [4]
IVD is the 4-month (in the human) stage of development from primordial follicle to secondary follicle, and IVG is the 11-day stage of sensitivity to gonadotropins that makes the GVs meiotically competent. No prior hormonal stimulation is administered. Any oocytes that advance in culture to mature MII stage from GV (usually about 35%) are then vitrified in standard fashion [2, 3, 5, 6, 24, 25]. The reason why all centers doing IVM get this average of 35% maturation is explainable by understanding IVD and IVG. We noted no difference in results whether the culture media was Quinn’s Cleavage, Sage IVM, or Medicult IVM media. There was also no difference in results whether HCG concentration was either extreme of 10 mIU/ml or 1000 mIU/ml (Tables 1, 2). Poor results were only obtained if the cumulus was stripped immediately, or if the patient had undergone previous chemotherapy, or if she was pre-pubertal. An intact cumulus is essential, but a variety of ordinary medium was used because of our previous results with in-vitro gametogenesis demonstrating the stages of IVD (in vitro development), IVG, (in vitro gonadotropin sensitivity), and IVM (LH or HCG induced). We postulated that many (35%) of these germinal vesicles would already have completed the full phases of IVD and IVG in vivo in the ovary. We thus suspected and demonstrated that no special IVM media would be needed and that almost any concentration of HCG would suffice [4] (Tables 1, 2).
Note that with needle aspiration, one is not able to obtain the small follicles that are right at the cortico-medullary junction (Fig. 4a, b). Yet these are likely to be the best follicles for IVM because of the location of FSH in the granulosa cells [26,27,28]. The FSH concentration is important for sensitizing LH receptors for IVM. The FSH concentration is highest in the mural granulosa cells of the follicle, and not in the corona in the granulosa cells by a ratio of 100:1. A high concentration of FSH in mural cells is only found in the smaller CCs (cumulus complexes) closer to the outer rim of the cortex and these smaller CCs nonetheless have large 100 microns GVs that are ready for IVM. So not only is surgical dissection more likely to successfully retrieve GVs but it is also more likely to retrieve the LH-sensitive mural granulosa cells.
Maturation of Cumulus Complexes Recovered in the Media After Cortico-medullary Dissection
The number of CCs recovered can vary widely, between 3 and 80 or more cumulus complexes, depending on the age of the women and whether there had been any prior chemotherapy. For IVM as we have reported, a total of 34 females underwent oophorectomy for ovary tissue freezing and IVM [4]. In 14 of those patients there was prior chemotherapy or they were pre-pubertal and there were no CCs found. In the remaining 20, IVM was performed. [4] (Tables 1, 2). The rate of maturation to an MII oocyte (Fig. 3a–d) varied from a low of 19% to a high of 56% (average 35%) Once exception is where the cumulus was dissected. The average values from other centers publishing on IVM has also varied between 30 and 39% [2, 3, 8, 9, 11, 12, 29,30,31]. Maturation of germinal vesicle to MII oocytes is detected between 24 and 48 h of exposure to the HCG-containing media. Interestingly, Previous research indicated that the number of mature oocytes we obtain from IVM is similar to what would be obtained from ovarian stimulation [2]. A variety of media and gonadotropin concentrations were intentionally used in light of the mechanisms of in vitro oogenesis, in order to see if this new understanding of ovarian function could allow for a simplification of IVM [4, 8,9,10,11,12, 14].
As these results (and those reported by other groups) with IVM demonstrate, one might consider with cancer patients the option to dispense with ovarian stimulation, and go right to oophorectomy, with no delay in cancer treatment. There might be no need for stimulation to obtain many mature oocytes [2, 3, 5, 6, 25]. Mature oocytes may simply be directly obtained (as an extra benefit) from the excised ovarian tissue before cryopreservation of the tissue. Other groups have preceded our current efforts at IVM from excised ovarian tissue and achieved pregnancy with healthy infants [1, 3, 5,6,7, 25].
Mechanism In vivo and In vitro of Oocyte Maturation
What is most fascinating about this very simple IVM is why does it work? It might at first seem puzzling why IVM from ovarian tissue suddenly seems so easy at many different centers, when it has been so difficult in the past. There are several potential reasons for this. First, we were not trying to mature primordial follicles, although that should become possible in the future with the “8 core genes” approach, which works well in mice [14]. However, culturing germinal vesicle oocytes “GVs” that have already become meiotically competent by in vivo “IVD” and in vivo “IVG” should not be expected to be difficult. For example, in a young woman on average every single day about 30 GVs will leave the inert IVD phase, and will become sensitive to gonadotropin. About 120 GVs will have been exposed to gonadotropin for more than 7, and less than 12 days IN VIVO. Of course, one must obtain many CCs containing GVs for this to be practical, and it is far easier to obtain many germinal vesicle oocytes with cortical dissection rather than with a needle. Figure 4a and b demonstrate clearly why dissection of the cortico-medullary junction is so much more effective than trying to obtain these CCs from tiny follicles than with a needle. The results of in vitro gametogenesis and IVM also reveal the limited role of the ovulatory cycle and ovarian stimulation in oocyte development other than to remove the oocyte from the ovary [9, 10, 29], 2013; [2, 11, 30, 31].
Intrinsic tissue pressure (along with eight “core genes”) has been shown with in-vitro gametogenesis to be the initiating mechanism at work to control primordial follicle recruitment and development to antral follicle status [2, 3, 5, 6, 11, 12, 23, 25, 32, 33]. The in vivo cortical tissue pressure gradient is a major regulator of primordial follicle recruitment and ovarian longevity [22, 34, 35] (Fig. 5a–c). This pressure theory is supported clinically by the changes in AMH and FSH observed and previously reported and discussed in fresh and frozen ovary tissue transplants [22, 32, 36] (Fig. 6). As the FSH comes down to normal levels 4–5 months after the transplant, at the same moment AMH begins to rise from zero up to very high levels, and then 9 months later comes down again to low levels, indicating an over-recruitment of primordial follicles because of tissue pressure reduction at the time of transplant. Then despite low AMH, the graft continues to function for many years because as ovarian reserve decreases, follicle recruitment also decreases.
Relation of recovery of FSH to AMH and Over Recruitment. There is a 4-–5-month period after ovary tissue transplant where FSH remains high and AMH is undetectable. Then when the FSH starts coming down, the AMH rises indicating that primordia follicles have finally differentiated into gonadotropin sensitivity, releasing estrogen, inhibiting FSH, and secreting AMH. However, the AMH then goes up very high for 4–6 months indicating over-recruitment from primordial follicle 4.5 months earlier. Because of subsequent depletants, the AMH then goes down. It stays down for many years because of the well-known phenomenon that as the ovarian reserve goes down, the rate of primordial follicle recruitment also goes down. So despite low AMH, these grafts (only ½ of an ovary) last for 8–10 years
Primordial follicle arrest in the highly compact ovarian cortex is thought to be a possible key to saving the oocyte from disappearing after the fetal initiation of meiosis and prevents its continuation all the way through meiosis and subsequent apoptosis [8,9,10,11,12]. It may also be a key to the gradual recruitment every month of a limited number of oocytes in the adult to develop over 4 months into gonadotrophin-sensitive antral and graafian follicles, which spares the resting oocytes from sudden total depletion [23] (Fig. 5a–c). The IVD stage in mice of 3 weeks from primordial follicle to gonadotropin sensitive secondary follicle is all controlled by decreasing pressure gradient and just eight “core genes.” In humans this stage is much longer (4 months). In culture systems for in vitro gametogenesis (as opposed to in vivo), oocytes developed from IPS cells will differentiate quickly through IVD. However, if these cultures are performed in a high pressure incubator, the nuclei start rotating and development is arrested. Then when placed into a normal low pressure environment, the nuclei stop rotating, and the oocytes resume developing [12] (Fig. 7a–c).
Perhaps the most striking scientific postulate about ovarian function resulting from this series is the high rate of maturation of germinal vesicle oocytes from ovary tissue to MII in less than 2 days, with no ovarian stimulation. The success of IVM did not correlate with the specifics of the media used or the concentration of gonadotrophin. The success of IVM appears to be intrinsic to the cumulus and they have already achieved meiotic competence by in vivo exposure to the “core genes” (IVD), and to in vivo FSH (IVG) [14].
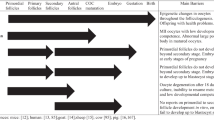
On average the ovarian cortex of a young woman contains about 200,000 oocytes. Every month, about 1000 are recruited from “resting” follicles in the cortex, and they require 4–5 months (IVD) thereafter to become sensitive to gonadotrophins and enter the ovulatory cycle. In-vitro gametogenesis studies in mice have termed this phase “IVD,” i.e., in vitro differentiation to germinal vesicle stage. This phase, also called “PPT” (primordial to primary follicle transition) represents the non-gonadotropin-sensitive growth from either recruited oocytes or pluripotent stem cell to germinal vesicle oocytes (which are now sensitive to gonadotrophin). IVD requires 3 weeks in mice, but closer to 4 months in humans [8, 10, 22, 32, 34, 37]. IVD in vivo is a constantly occurring process, as also is IVG. There is a continuous exposure to FSH of IVG-ready oocytes in the intact in vivo ovary. The IVG process usually takes about 8–11 days in both mice, and presumably in humans. Oocytes that have completed IVG but then are not immediately exposed to HCG in vitro, or to the ovulatory LH surge in vivo, become post-mature and gradually degenerate. It is the population of oocytes retrieved from the dissected ovarian cortex that have just recently completed IVG (35%) that mature readily with exposure to HCG in a variety of concentrations in ordinary culture media.
It should be noted from Fig. 8, the most important figure in this paper, that on any given day there are about 35% of oocytes that have undergone at least 7–8 days of IVG, but also less than 12 days where degeneration occurs. There is thus a “sweet spot” between 7 days and 11 days of gonadotropin exposure where GVs can mature in vitro and not degenerate (Fig. 8). Less than seven days means not enough exposure to in vivo gonadotropin to become sensitive to HCG or LH. More than 11 days exposure of in vivo means they are post-mature and they will degenerate. That is why on any given day 35% of GVs will mature to M2 if exposed to HCG. If exposure to FSH after completing IVD is less than 7-8 days the GV will not mature in HCG. If exposure to FSH has been greater than 12 days, the GV will just degenerate.
Recruitment from Primordial Follicle to GV Oocyte
The phase referred to as “IVG,” i.e., gonadotrophin (FSH)-induced meiotic competence of germinal vesicle oocytes, usually requires 7–11 days, remarkably similar to ovarian stimulation in human IVF. The unstimulated ovary has already been exposed to FSH in vivo. Many (average 35%)of the germinal vesicle oocytes (in cumulus complexes) recovered from the ovarian dissection have already gone through the “IVD” and “IVG” phases in vivo, and therefore are meiotically competent, having already had adequate exposure to endogenous FSH. Therefore just 1–2 days of exposure to LH or HCG is all that is needed for these specific germinal vesicle oocytes to develop to mature MII oocytes. What has previously not been understood is how the primordial follicles are recruited and nourished to go through IVD and become gonadotropin sensitive GVs.
The eight “core genes” in mice are all that are needed to convert stem cells to oocytes (Fig. 9). However, the oocytes that develop directly from stem cells are not fully competent. Full competence requires culture with fetal granulosa cells, which can also be produced from stem cells using a more complex culture system [13]. However, if these eight core genes are used to recruit from the primordial follicles (that have already been exposed to granulosa cells when the ovary was just fetal), then the resulting M2 oocytes are fully functional and can result in normal offspring.
Without the dense pressure of the ovarian cortex in the fetus, which induces the formation of primordial follicles, fetal oocytes would continue in meiosis and be completely depleted by birth [8,9,10, 38,39,40]; Hikabe [22, 32, 41,42,43]. Nagamatsu and colleagues demonstrated that dense cortical tissue pressure caused oocyte nuclei to rotate, and holds the primordial follicles in arrest [12]. As they encounter less tissue pressure internally, the rotation stops, and the primordial follicles are recruited [12] (Fig. 7a, b). These eight “core genes” can also recruit stem cells or IPS (induced pluripotent stem cell) cells to transform all the way to MII oocyte-like cells that are not competent but recruitment from primordial follicles (from ovarian tissue) results in normal, competent oocytes. [14, 44,45,46]; Winkler-Crepaz [32, 47, 48]. Therefore, with IVM it should become possible to obtain normal MII oocytes from ovarian tissue. However, for competent oocytes to develop directly from IPS cells in vitro, will require co-cultures with fetal (not adult) granulosa cells. That will require transforming IPS cells in a separate culture system using the Yoshino approach into species-specific fetal granulosa cells [13].
Conclusions
The only way to be certain of the functional competence any of these MII oocytes is if live births can be obtained. Several reports have already shown this to be the case with IVM from ovary tissue [1,2,3, 5, 6, 25]. What is a new and perhaps shocking suggestion is the relative ease with which this can be achieved using ordinary media. This would be expected from our group’s previously published studies of in-vitro gametogenesis from stem cells in mice. It is easy to collect many germinal vesicle oocytes from the tiny follicles at the cortico-medullary junction when you have the ovary in hand instead of using a needle, and their intrinsic 30–40% meiotic competence is universal. So why do we even need ovarian stimulation, or even the normal ovulatory cycle? The normal ovulation cycle is not needed for meiotic competence. Ovarian stimulation is only required to allow oocytes to exit the ovary. To apply this simple and robust IVM to women undergoing IVF with ultrasound and needle will require a special technique to aspirate the smallest follicles of 2mm to 6mm [49,50,51]. It is easy to obtain CCs from these follicles using dissection. It will be more difficult (but possible) using ultrasound guided needle aspiration.
Simple cortical tissue pressure (associated with primordial follicle nuclear rotation) has been found to be a key regulator of primordial follicle arrest, recruitment and ovarian longevity in humans, similar to mice [12]. However, eight “core genes” (in mice) and exit of intranuclear FOX3 are also necessary to allow the primordial follicles to escape arrest and develop to meiotically competent germinal vesicle oocytes. The ability of ovarian tissue germinal vesicle oocytes to undergo normal IVM indicates that the normal ovulatory cycle and ovarian stimulation is not necessary for oocyte maturation. Eventually, we can project that all that will be needed to make competent MII oocytes in our patients of any age is a skin biopsy as a source for IPS cells. We already can make competent human PGCs from IPS cells. Now we have to make fetal granulosa cells from IPS cells.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
De Vos M, Grynberg M, Ho TM, Yuan Y, Albertini DF, Gilchrist RB. Perspectives on the development and future of oocyte IVM in clinical practice. J Assist Reprod Genet. 2021;38(6):1265–80.
Nikiforov D, Junping C, Cadenas J, Shukla V, Blanshard R, Pors SE, Kristensen SG, Macklon KT, Colmorn L, Ernst E, Bay-Bjørn AM, Ghezelayagh Z, Wakimoto Y, Grøndahl ML, Hoffmann E, Andersen CY. Improving the maturation rate of human oocytes collected ex vivo during the cryopreservation of ovarian tissue. J Assist Reprod Genet. 2020 Apr;37(4):891–904.
Segers I, Mateizel I, Van Moer E, Smitz J, Tournaye H, Verheyen G, De Vos M. In vitro maturation (IVM) of oocytes recovered from ovariectomy specimens in the laboratory: a promising "ex vivo" method of oocyte cryopreservation resulting in the first report of an ongoing pregnancy in Europe. J Assist Reprod Genet. 2015;32(8):1221–31.
Silber SJ, Goldsmith S, Castleman L, Hurlbut K, Fan Y, Melnick J, Hayashi K. In-vitro maturation and transplantation of cryopreserved ovary tissue: understanding ovarian longevity. Reprod Biomed Online. 2022;44(3):504–14.
Prasath EB, Chan ML, Wong WH, Lim CJ, Tharmalingam MD, Hendricks M, Loh SF, Chia YN. First pregnancy and live birth resulting from cryopreserved embryos obtained from in vitro matured oocytes after oophorectomy in an ovarian cancer patient. Hum Reprod. 2014;29(2):276–8.
Uzelac PS, Delaney AA, Christensen GL, Bohler HC, Nakajima ST. Live birth following in vitro maturation of oocytes retrieved from extracorporeal ovarian tissue aspiration and embryo cryopreservation for 5 years. Fertil Steril. 2015;104(5):1258–60.
Vesztergom D, Segers I, Mostinckx L, Blockeel C, De Vos M. Live births after in vitro maturation of oocytes in women who had suffered adnexal torsion and unilateral oophorectomy following conventional ovarian stimulation. J Assist Reprod Genet. 2021;38(6):1323–9.
Hayashi K, Ogushi S, Kurimoto K, Shimamoto S, Ohta H, Saitou M. Offspring from oocytes derived from in vitro primordial germ cell-like cells in mice. Science. 2012;338:971–5.
Hayashi K, Ohta H, Kurimoto K, Aramaki S, Saitou M. Reconstitution of the mouse germ cell specification pathway in culture by pluripotent stem cells. Cell. 2011;146:519–32.
Hayashi K, Saitou M. Generation of eggs from mouse embryonic stem cells and induced pluripotent stem cells. Nat Protoc. 2013;8:1513–24.
Hikabe O, Hamazaki N, Nagamatsu G, Obata Y, Hirao Y, Hamada N, Shimamoto S, Imamura T, Nakashima K, Saitou M, Hayashi K. Reconstitution in vitro of the entire cycle of the mouse female germ line. Nature. 2016;539:299–303.
Nagamatsu G, Shimamoto S, Hamazaki N, Nishimura Y, Hayashi K. Mechanical stress accompanied with nuclear rotation is involved in the dormant state of mouse oocytes. Science. Advances. 2019;5(6):[eaav9960].
Yoshino T, Suzuki T, Nagamatsu G, Yabukami H, Ikegaya M, Kishima M, Kita H, Imamura T, Nakashima K, Nishinakamura R, Tachibana M, Inoue M, Shima Y, Morohashi KI, Hayashi K. Generation of ovarian follicles from mouse pluripotent stem cells. Science. 2021;373(6552):eabe0237.
Hamazaki N, Kyogoku H, Araki H, Miura F, Horikawa C, Hamada N, Shimamoto S, Hikabe O, Nakashima K, Kitajima TS, Ito T, Leitch HG, Hayashi K. Reconstitution of the oocyte transcriptional network with transcription factors. Nature. 2021;589(7841):264–9.
Hayashi K, Galli C, Diecke S, Hildebrandt TB. Artificially produced gametes in mice, humans and other species. Reprod Fertil Dev. 2021;33(2):91–101.
Pandolfi EC, Hsu FM, Duhon M, Zheng Y, Goldsmith S, Fu J, Silber SJ, Clark AT. In vitro germ cell induction from fertile and infertile monozygotic twin research participants. Cell Rep Med. 2022;3(10):100782.
Pandolfi EC, Hunt TJ, Goldsmith S, Hurlbut K, Silber SJ, Clark AT. Generation of three human induced pluripotent stem cell sublines (UCLAi004-A, UCLAi004-B, and UCLAi004-C) for reproductive science research. Stem Cell Res. 2021a;54:102446.
Pandolfi EC, Hunt TJ, Goldsmith S, Hurlbut K, Silber SJ, Clark AT. Generation of three human induced pluripotent stem cell sublines (UCLAi005-A, UCLAi005-B and UCLAi005-C) for reproductive science research. Stem Cell Res. 2021b;54:102409. https://doi.org/10.1016/j.scr.2021.102409.
Pandolfi EC, Sosa E, Hunt TJ, Goldsmith S, Hurlbut K, Silber SJ, Clark AT. Generation of six human induced pluripotent stem cell sublines (MZT01E, MZT01F, MZT01N and MZT02D, MZT02G and MZT02H) for reproductive science research. Stem Cell Res. 2021c;51:102204.
Pandolfi EC, Rojas EJ, Sosa E, Gell JJ, Hunt TJ, Goldsmith S, Fan Y, Silber SJ, Clark AT. Generation of three human induced pluripotent stem cell sublines (MZT04D, MZT04J, MZT04C) for reproductive science research. Stem Cell Res. 2019;40:101576.
Hayashi M, Zywitza V, Naitou Y, Hamazaki N, Goeritz F, Hermes R, Holtze S, Lazzari G, Galli C, Stejskal J, Diecke S, Hildebrandt TB, Hayashi K. Robust induction of primordial germ cells of white rhinoceros on the brink of extinction. Sci Adv. 2022;8(49):eabp9683.
Silber S, Pineda J, Lenahan K, DeRosa M, Melnick J. Fresh and cryopreserved ovary transplantation and resting follicle recruitment. Reprod BioMed Online. 2015;30:643–50.
Woodruff TK, Shea LD. A new hypothesis regarding ovarian follicle development: ovarian rigidity as a regulator of selection and health. J Assist Reprod Genet. 2011;28:3–6.
Kuwayama M, Vajta G, Kato O, Leibo SP. Highly efficient vitrification method for cryopreservation of human oocytes. Reprod Biomed Online. 2005;11(3):300–8.
Segers I, Bardhi E, Mateizel I, Van Moer E, Schots R, Verheyen G, Tournaye H, De Vos M. Live births following fertility preservation using in-vitro maturation of ovarian tissue oocytes. Hum Reprod. 2020;35(9):2026–36.
Cadenas J, Pors SE, Kumar A, Kalra B, Kristensen SG, Andersen CY, Mamsen LS. Concentrations of oocyte secreted GDF9 and BMP15 decrease with MII transition during human IVM. Reprod Biol Endocrinol. 2022;20(1):126.
McLaughlin M, Albertini DF, Wallace WHB, Anderson RA, Telfer EE. Metaphase II oocytes from human unilaminar follicles grown in a multi-step culture system. Mol Hum Reprod. 2018;24(3):135–42.
Telfer EE. FERTILITY PRESERVATION: Progress and prospects for developing human immature oocytes in vitro. Reproduction. 2019;158(5):F45–54.
Andersen CY, Rosendahl M, Byskov AG, Loft A, Ottosen C, Dueholm M, Schmidt KL, Andersen AN, Ernst E. Two successful pregnancies following autotransplantation of frozen/thawed ovarian tissue. Hum Reprod. 2008;23(10):2266–72.
Rosendahl M, Schmidt KT, Ernst E, Rasmussen PE, Loft A, Byskov AG, Andersen AN, Andersen CY. Cryopreservation of ovarian tissue for a decade in Denmark: a view of the technique. Reprod BioMed Online. 2011;22(2):162–71.
Schmidt KT, Rosendahl M, Ernst E, Loft A, Nyboe Andersen A, Dueholm M, Ottosen C, Yding AC. Autotransplantation of cryopreserved ovarian tissue in 12 women with chemotherapy-induced premature ovarian failure: The Danish experience. Fertil Steril. 2011;95:695–701.
Ayuandari S, et al. Follicular growth after xenotransplantation of cryopreserved/thawed human ovarian tissue in SCID mice: dynamics and molecular aspects. J Assist Reprod Genet. 2016;33:1585–93. https://doi.org/10.1007/s10815-016-0769-2.
Xiao S, Zhang J, Romero M, et al. In vitro follicle growth supports human oocyte meiotic maturation. Sci Rep. 2015;5:17323.
Silber S. Unifying theory of adult resting follicle recruitment and fetal oocyte arrest. Reprod BioMed Online. 2015;31:472–5.
Silber S. Ovarian tissue cryopreservation and transplantation: scientific implications. J Assist Reprod Genet. 2016;33(12):1595–603.
Silber SJ, DeRosa M, Goldsmith S, Fan Y, Castleman L, Melnick J. Cryopreservation and transplantation of ovarian tissue: results from one center in the USA. J Assist Reprod Genet. 2018;35(12):2205–13.
Donnez J, Dolmans MM, Demylle D, Jadoul P, Pirard C, Squifflet J, Martinez-Madrid B, Van Langendonckt A. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004;364:1405–10.
Donnez J, Dolmans MM, Pellicer A, Diaz-Garcia C, Sanchez Serrano M, Schmidt KT, Ernst E, Luyckx V, Andersen CY. Restoration of ovarian activity and pregnancy after transplantation of cryopreserved ovarian tissue: a review of 60 cases of reimplantation. Fertil Steril. 2013;99:1503–13.
Donnez J, Dolmans MM. Fertility preservation in women. N Engl J Med. 2017;377:1657–65.
Greve T, Clasen-Linde E, Andersen MT, Andersen MK, Sorensen SD, Rosendahl M, Ralfkiaer E, Andersen CY. Cryopreserved ovarian cortex from patients with leukemia n complete remission contains no apparent viable malignant cells. Blood. 2012;120:4311–6.
Jensen AK, Kristensen SG, Macklon KT, Jeppesen JV, Fedder J, Ernst E, Andersen CY. Outcomes of transplantations of cryopreserved ovarian tissue to 41 women in Denmark. Hum Reprod. 2015;30:2838–45.
Nesbit ME Jr, Robison LL, Ortega JA, Sather HN, Donaldson M, Hammond D. Testicular relapse in childhood acute lymphoblastic leukemia: association with pretreatment patient characteristics and treatment. A report for Childrens Cancer Study Group. Cancer. 1980;45:2009–16.
Ortega JJ, Javier G, Toran N. Testicular relapses in childhood acute lymphoid leukaemia (author’s transl). Sangre. 1981;26:168–79.
Lind T, Holte J, Olofsson JI, Hadziosmanovic N, Gudmundsson J, Nedstrand E, Lood M, Berglund L, Rodriguez-Wallberg K. Reduced live-birth rates after IVF/ICSI in women with previous unilateral oophorectomy: results of a multicenter cohort study. Hum Reprod. 2018;33(2):238–47.
Thomas-Teinturier C, El Fayech C, Oberlin O, et al. Age at menopause and its influencing factors in a cohort of survivors of childhood cancer: earlier but rarely premature. Hum Reprod. 2013;28:488–95. https://doi.org/10.1093/humrep/des391.
Wallace WH, Kelsey TW. Human ovarian reserve from conception to the menopause. PLoS One. 2010;5:e8772. https://doi.org/10.1371/journal.pone.0008772.
Yasui T, Hayashi K, Mizunuma H, Kubota T, Aso T, Matsumura Y, Lee JS, Suzuki S. Factors associated with premature ovarian failure, early menopause and earlier onset of menopause in Japanese women. Maturitas. 2012;72:249–55. https://doi.org/10.1016/j.maturitas.2012.04.002.
Zhai A, Axt J, Hamilton EC, Koehler E, Lovvorn HN. 3rd Assessing gonadal function after childhood ovarian surgery. J Pediatr Surg. 2012;47:1272–9. https://doi.org/10.1016/j.jpedsurg.2012.03.038.
Gilchrist R, Ho T, De Vos M, Sanchez F, Romero S, Ledger WL, Anckaert E, Vuong L, Smitz, J. A fresh start for IVM: capacitating the oocyte for development using pre-IVM, Human Reproduction Update. 2023; dmad023. https://doi.org/10.1093/humupd/dmad023
Vuong LN, Le AH, Ho VNA, Pham TD, Sanchez F, Romero S, De Vos M, Ho TM, Gilchrist RB, Smitz J. Live births after oocyte in vitro maturation with a prematuration step in women with polycystic ovary syndrome. J Assist Reprod Genet. 2020;37(2):347–57.
Vuong LN, Ho VNA, Ho TM, Dang VQ, Phung TH, Giang NH, Le AH, Pham TD, Wang R, Smitz J, Gilchrist RB, Norman RJ, Mol BW. In-vitro maturation of oocytes versus conventional IVF in women with infertility and a high antral follicle count: a randomized non-inferiority controlled trial. Hum Reprod. 2020;35(11):2537–47.
Author information
Authors and Affiliations
Contributions
Writing and preparing manuscript: Sherman Silber and Sierra Goldsmith. Clinical and scientific review: Leilani Castleman. Overall scientific review and ultimate advice: Katsuhiko Hayashi
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
Local Hospital IRB St. Luke’s Hospital (irbnet.org) ID: 1803756-2, ID: 852330-12. Informed consent was obtained prior to performing any procedures including permission for publication on all data.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
The authors affirm that human research participants provided informed consent for publication of any of the images used.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Capsule
A new understanding of in vitro maturation “IVM” and ovarian longevity derives from experiments with in vitro oogenesis. Many mature oocytes can now be obtained from human ovary tissue IVM with no need for ovarian stimulation, using just simple media. Tissue pressure and just a few “core genes” control ovarian longevity.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Silber, S.J., Goldsmith, S., Castleman, L. et al. In Vitro Maturation, In Vitro Oogenesis, and Ovarian Longevity. Reprod. Sci. 31, 1234–1245 (2024). https://doi.org/10.1007/s43032-023-01427-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01427-1