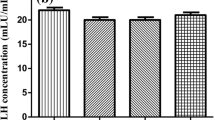
Abstract
Accumulating evidence has shown that inflammation is a key process in polycystic ovary syndrome (PCOS). Nucleotide-binding oligomerization domain-, leucine-rich repeat-, and pyrin domain-containing 3 (NLRP3) inflammasomes play an essential role in inflammation. We investigated the expression of NLRP3 inflammasome in PCOS and its underlying mechanisms. Human granulosa cells (GCs) were isolated from patients with PCOS and control women who underwent in vitro fertilization and embryo transfer. Ovarian specimens were collected from mice with polycystic ovarian changes induced by a high-fat diet and letrozole. RNA sequencing (RNA-Seq) was performed on a granulosa cell line (KGN) overexpressing NLRP3. Polymerase chain reaction (PCR) was performed to quantify the differentially expressed genes of interest. NLRP3 and caspase-1 expression was significantly higher in GCs from patients with PCOS than in GCs from the control group. Increased NLRP3 and caspase-1 expression was also detected by immunohistochemistry in the GCs of a mouse model of polycystic ovarian changes. The serum IL-18 concentration in PCOS-like mice was significantly higher than that in control mice. Following NLRP3 overexpression in KGN cells, the genes involved in N-glycan processing, steroidogenesis, oocyte maturation, autophagy, and apoptosis were upregulated. The RT-qPCR results revealed that the expression levels of GANAB, ALG-5, HSD3B2, ULK1, PTK2B, and Casp7 in KGN cells after NLRP3 overexpression were significantly higher than those in control cells, which was consistent with the RNA-Seq results. Taken together, the NLRP3 inflammasome-dependent pathway is involved in the pathogenesis of PCOS not only by mediating pyroptosis, but also by regulating glycan synthesis, sex hormone synthesis, autophagy, and apoptosis in GCs.





Similar content being viewed by others
Data availability
All data obtained or analyzed in this study are included in this article and are available from the corresponding authors.
Abbreviations
- NLRP3:
-
Nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing 3
- PCOS:
-
Polycystic ovary syndrome
- DEGs:
-
Differentially expressed genes
- GO:
-
Gene Ontology
- RNA-Seq:
-
RNA sequencing
- qRT PCR:
-
Quantitative RT-PCR
- KEGG:
-
Kyoto Encyclopedia of Genes and Genome
- GANAB:
-
Glucosidase II Alpha Subunit
- ALG5:
-
Asparagine-Linked Glycosylation 5
- HSD3B2:
-
Hydroxy-Delta-5-Steroid Dehydrogenase, 3 Beta- And Steroid Delta-Isomerase 2
- ULK1:
-
Unc-51 Like Autophagy Activating Kinase 1
- PTK2B:
-
Protein Tyrosine Kinase 2 Beta
- CASP7:
-
Caspase-7
References
Barrea L, Marzullo P, Muscogiuri G, Di Somma C, Scacchi M, Orio F, et al. Source and amount of carbohydrate in the diet and inflammation in women with polycystic ovary syndrome. Nutr Res Rev. 2018;31(2):291–301. https://doi.org/10.1017/S0954422418000136.
Lai Q, Xiang W, Li Q, Zhang H, Li Y, Zhu G, et al. Oxidative stress in granulosa cells contributes to poor oocyte quality and IVF-ET outcomes in women with polycystic ovary syndrome. Front Med. 2018;12(5):518–24. https://doi.org/10.1007/s11684-017-0575-y.
Gonzalez F, Considine RV, Abdelhadi OA, Acton AJ. Oxidative Stress in Response to Saturated Fat Ingestion Is Linked to Insulin Resistance and Hyperandrogenism in Polycystic Ovary Syndrome. J Clin Endocrinol Metab. 2019;104(11):5360–71. https://doi.org/10.1210/jc.2019-00987.
Mohammadi M. Oxidative Stress and Polycystic Ovary Syndrome: A Brief Review. Int J Prev Med. 2019;10:86. https://doi.org/10.4103/ijpvm.IJPVM_576_17.
Liu Y, Liu H, Li Z, Fan H, Yan X, Liu X, et al. The Release of Peripheral Immune Inflammatory Cytokines Promote an Inflammatory Cascade in PCOS Patients via Altering the Follicular Microenvironment. Front Immunol. 2021;12:685724. https://doi.org/10.3389/fimmu.2021.685724.
Jung ES, Suh K, Han J, Kim H, Kang HS, Choi WS, et al. Amyloid-beta activates NLRP3 inflammasomes by affecting microglial immunometabolism through the Syk-AMPK pathway. Aging Cell. 2022;21(5):e13623. https://doi.org/10.1111/acel.13623.
Yao C, Veleva T, Scott L Jr, Cao S, Li L, Chen G, et al. Enhanced Cardiomyocyte NLRP3 Inflammasome Signaling Promotes Atrial Fibrillation. Circulation. 2018;138(20):2227–42. https://doi.org/10.1161/CIRCULATIONAHA.118.035202.
Williams BM, Cliff CL, Lee K, Squires PE, Hills CE. The Role of the NLRP3 Inflammasome in Mediating Glomerular and Tubular Injury in Diabetic Nephropathy. Front Physiol. 2022;13:907504. https://doi.org/10.3389/fphys.2022.907504.
Irandoost E, Najibi S, Talebbeigi S, Nassiri S. Focus on the role of NLRP3 inflammasome in the pathology of endometriosis: a review on molecular mechanisms and possible medical applications. Naunyn Schmiedebergs Arch Pharmacol. 2023;396(4):621–31. https://doi.org/10.1007/s00210-022-02365-6.
Fonseca BM, Pinto B, Costa L, Felgueira E, Rebelo I. Increased expression of NLRP3 inflammasome components in granulosa cells and follicular fluid interleukin(IL)-1beta and IL-18 levels in fresh IVF/ICSI cycles in women with endometriosis. J Assist Reprod Genet. 2023;40(1):191–9. https://doi.org/10.1007/s10815-022-02662-2.
Rogers LM, Serezani CH, Eastman AJ, Hasty AH, Englund-Ogge L, Jacobsson B, et al. Palmitate induces apoptotic cell death and inflammasome activation in human placental macrophages. Placenta. 2020;90:45–51. https://doi.org/10.1016/j.placenta.2019.12.009.
Zhu D, Zou H, Liu J, Wang J, Ma C, Yin J, et al. Inhibition of HMGB1 Ameliorates the Maternal-Fetal Interface Destruction in Unexplained Recurrent Spontaneous Abortion by Suppressing Pyroptosis Activation. Front Immunol. 2021;12:782792. https://doi.org/10.3389/fimmu.2021.782792.
Bergsbaken T, Fink SL, Cookson BT. Pyroptosis: host cell death and inflammation. Nat Rev Microbiol. 2009;7(2):99–109. https://doi.org/10.1038/nrmicro2070.
Schmidt J, Weijdegard B, Mikkelsen AL, Lindenberg S, Nilsson L, Brannstrom M. Differential expression of inflammation-related genes in the ovarian stroma and granulosa cells of PCOS women. Mol Hum Reprod. 2014;20(1):49–58. https://doi.org/10.1093/molehr/gat051.
Wang D, Weng Y, Zhang Y, Wang R, Wang T, Zhou J, et al. Exposure to hyperandrogen drives ovarian dysfunction and fibrosis by activating the NLRP3 inflammasome in mice. Sci Total Environ. 2020;745:141049. https://doi.org/10.1016/j.scitotenv.2020.141049.
Ryan GE, Malik S, Mellon PL. Antiandrogen Treatment Ameliorates Reproductive and Metabolic Phenotypes in the Letrozole-Induced Mouse Model of PCOS. Endocrinology. 2018;159(4):1734–47. https://doi.org/10.1210/en.2017-03218.
Herman R, Jensterle M, Janež A, Goričar K, Dolžan V. Genetic variability in antioxidative and inflammatory pathways modifies the risk for PCOS and influences metabolic profile of the syndrome. Metabolites. 2020;10(11):439 https://doi.org/10.3390/metabo10110439.
Lai H, Jia X, Yu Q, Zhang C, Qiao J, Guan Y, et al. High-fat diet induces significant metabolic disorders in a mouse model of polycystic ovary syndrome. Biol Reprod. 2014;91(5):127. https://doi.org/10.1095/biolreprod.114.120063.
Caldwell AS, Middleton LJ, Jimenez M, Desai R, McMahon AC, Allan CM, et al. Characterization of reproductive, metabolic, and endocrine features of polycystic ovary syndrome in female hyperandrogenic mouse models. Endocrinology. 2014;155(8):3146–59. https://doi.org/10.1210/en.2014-1196.
Kauffman AS, Thackray VG, Ryan GE, Tolson KP, Glidewell-Kenney CA, Semaan SJ, et al. A Novel Letrozole Model Recapitulates Both the Reproductive and Metabolic Phenotypes of Polycystic Ovary Syndrome in Female Mice. Biol Reprod. 2015;93(3):69. https://doi.org/10.1095/biolreprod.115.131631.
Zhao Y, Fu L, Li R, Wang LN, Yang Y, Liu NN, et al. Metabolic profiles characterizing different phenotypes of polycystic ovary syndrome: plasma metabolomics analysis. BMC Med. 2012;10:153. https://doi.org/10.1186/1741-7015-10-153.
Niu Z, Lin N, Gu R, Sun Y, Feng Y. Associations between insulin resistance, free fatty acids, and oocyte quality in polycystic ovary syndrome during in vitro fertilization. J Clin Endocrinol Metab. 2014;99(11):E2269–76. https://doi.org/10.1210/jc.2013-3942.
Mu YM, Yanase T, Nishi Y, Tanaka A, Saito M, Jin CH, et al. Saturated FFAs, palmitic acid and stearic acid, induce apoptosis in human granulosa cells. Endocrinology. 2001;142(8):3590–7. https://doi.org/10.1210/endo.142.8.8293.
Miller WL, Auchus RJ. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr Rev. 2011;32(1):81–151. https://doi.org/10.1210/er.2010-0013.
Nelson VL, Legro RS, Strauss JF 3rd, McAllister JM. Augmented androgen production is a stable steroidogenic phenotype of propagated theca cells from polycystic ovaries. Mol Endocrinol. 1999;13(6):946–57. https://doi.org/10.1210/mend.13.6.0311.
Hirsch A, Hahn D, Kempna P, Hofer G, Nuoffer JM, Mullis PE, et al. Metformin inhibits human androgen production by regulating steroidogenic enzymes HSD3B2 and CYP17A1 and complex I activity of the respiratory chain. Endocrinology. 2012;153(9):4354–66. https://doi.org/10.1210/en.2012-1145.
Viinikangas T, Khosrowabadi E, Kellokumpu S. N-Glycan Biosynthesis: Basic Principles and Factors Affecting Its Outcome. Exp Suppl. 2021;112:237–57. https://doi.org/10.1007/978-3-030-76912-3_7.
Freeze HH, Chong JX, Bamshad MJ, Ng BG. Solving glycosylation disorders: fundamental approaches reveal complicated pathways. Am J Hum Genet. 2014;94(2):161–75. https://doi.org/10.1016/j.ajhg.2013.10.024.
Ng BG, Freeze HH. Perspectives on Glycosylation and Its Congenital Disorders. Trends Genet. 2018;34(6):466–76. https://doi.org/10.1016/j.tig.2018.03.002.
Tharmalingam-Jaikaran T, Walsh SW, McGettigan PA, Potter O, Struwe WB, Evans AC, et al. N-glycan profiling of bovine follicular fluid at key dominant follicle developmental stages. Reproduction. 2014;148(6):569–80. https://doi.org/10.1530/REP-14-0035.
Spitzer D, Murach KF, Lottspeich F, Staudach A, Illmensee K. Different protein patterns derived from follicular fluid of mature and immature human follicles. Hum Reprod. 1996;11(4):798–807. https://doi.org/10.1093/oxfordjournals.humrep.a019257.
Meng XQ, Zheng KG, Yang Y, Jiang MX, Zhang YL, Sun QY, et al. Proline-rich tyrosine kinase2 is involved in F-actin organization during in vitro maturation of rat oocyte. Reproduction. 2006;132(6):859–67. https://doi.org/10.1530/rep.1.01212.
Meng XQ, Cui B, Cheng D, Lyu H, Jiang LG, Zheng KG, et al. Activated proline-rich tyrosine kinase 2 regulates meiotic spindle assembly in the mouse oocyte. J Cell Biochem. 2018;119(1):736–47. https://doi.org/10.1002/jcb.26237.
Cong Y, Wu H, Bian X, Xie Q, Lyu Q, Cui J, et al. Ptk2b deletion improves mice folliculogenesis and fecundity via inhibiting follicle loss mediated by Erk pathway. J Cell Physiol. 2021;236(2):1043–53. https://doi.org/10.1002/jcp.29914.
Zheng Y, Ma L, Liu N, Tang X, Guo S, Zhang B, et al. Autophagy and apoptosis of porcine ovarian granulosa cells during follicular development. Animals (Basel). 2019;9(12):1111 https://doi.org/10.3390/ani9121111.
Bhardwaj JK, Paliwal A, Saraf P, Sachdeva SN. Role of autophagy in follicular development and maintenance of primordial follicular pool in the ovary. J Cell Physiol. 2022;237(2):1157–70. https://doi.org/10.1002/jcp.30613.
Choi JY, Jo MW, Lee EY, Yoon BK, Choi DS. The role of autophagy in follicular development and atresia in rat granulosa cells. Fertil Steril. 2010;93(8):2532–7. https://doi.org/10.1016/j.fertnstert.2009.11.021.
Li X, Qi J, Zhu Q, He Y, Wang Y, Lu Y, et al. The role of androgen in autophagy of granulosa cells from PCOS. Gynecol Endocrinol. 2019;35(8):669–72. https://doi.org/10.1080/09513590.2018.1540567.
Biasizzo MN, Kopitar-Jerala N. Interplay Between NLRP3 Inflammasome and Autophagy. Front Immunol. 2020;11:591803. https://doi.org/10.3389/fimmu.2020.591803.
Sreerangaraja Urs DB, Wu WH, Komrskova K, Postlerova P, Lin YF, Tzeng CR, et al. Mitochondrial function in modulating human granulosa cell steroidogenesis and female fertility. Int J Mol Sci. 2020;21(10):3592. https://doi.org/10.3390/ijms21103592.
Salehi E, Aflatoonian R, Moeini A, Yamini N, Asadi E, Khosravizadeh Z, et al. Apoptotic biomarkers in cumulus cells in relation to embryo quality in polycystic ovary syndrome. Arch Gynecol Obstet. 2017;296(6):1219–27. https://doi.org/10.1007/s00404-017-4523-5.
Yu YY, Sun CX, Liu YK, Li Y, Wang L, Zhang W. Promoter methylation of CYP19A1 gene in Chinese polycystic ovary syndrome patients. Gynecol Obstet Invest. 2013;76(4):209–13. https://doi.org/10.1159/000355314.
Panghiyangani R, Soeharso P, Andrijono DA, Suryandari B, Wiweko M. Kurniati, et al. CYP19A1 Gene Expression in Patients with Polycystic Ovarian Syndrome. J Hum Reprod Sci. 2020;13(2):100–3. https://doi.org/10.4103/jhrs.JHRS_142_18.
Acknowledgements
The authors are grateful to Prof. Hongli Yan and the medical staff of Shanghai Changhai Hospital for helping with the case samples collection and information.
Funding
This work was supported by the Programs Foundation of Xinhua Hospital, School of Medicine, Shanghai Jiaotong University, under Grant (No. YJ-12, 2016–2018, Bo Wang). Shanghai Key Laboratory of Embryo Original Diseases (Shelab201902, Bo Wang, Guolian Ding).
Author information
Authors and Affiliations
Contributions
WB, DGL, and HHF contributed to study design, data acquisition and interpretation, and manuscript drafting. WB, SMF, YCJ, PH, SHQ, BL, and XD collected samples and contributed to data analysis and interpretation. DGL, YCJ, DYT, and SJZ critically revised the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of the Xinhua Hospital, School of Medicine, Shanghai Jiaotong University.
Consent for participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, B., Shi, M., Yu, C. et al. NLRP3 Inflammasome-dependent Pathway is Involved in the Pathogenesis of Polycystic Ovary Syndrome. Reprod. Sci. 31, 1017–1027 (2024). https://doi.org/10.1007/s43032-023-01348-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01348-z