Abstract
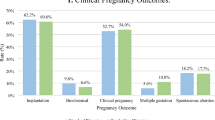
Oocyte cytoplasmic maturation is a crucial process during in vitro maturation (IVM), and finding an appropriate IVM medium that promotes oocyte competence is very critical in assisted reproductive technology (ART). The aim of this study was to investigate the effects of umbilical cord Wharton’s jelly (WJ-MSCs)-derived conditioned media on the maturation of immature oocytes and their developmental potential in humans after IVM, as well as apoptotic gene expression. A total of 392 germinal vesicle (GV) oocytes were collected from 207 women aged 25–35 years and divided into two IVM groups: (1) control group, which was cultured in CleavTM medium, and (2) experimental group, which was cultured in supernatants of umbilical cord Wharton’s jelly as a conditioned medium (CM). First, WJ-MSCs were isolated, and their purity was analyzed. The immunophenotypes of WJ-MSCs were analyzed by flow cytometry. The quantitative expression of BCL2, BAX, and BAG1 in matured oocytes and embryos was evaluated through quantitative real-time polymerase chain reaction (qRT-PCR). Our findings showed that WJ-MSCs have a high proliferating capacity. The purity of the isolated cells was further validated by immunophenotyping, which revealed that their surface antigen expression had phenotypical properties similar to WJ-MSCs. When compared to CD34 and CD45 surface markers, the enlarged cells were positive for CD90, CD105, and CD44. There were significant differences in cytoplasmic maturation of oocytes and embryo quality between the two groups. The mRNA expression levels of BCL-2, BAG1, and BAX in matured oocytes and embryos were also significantly different between the two groups. Therefore, WJ-MSCs medium indicated potential efficacy in terms of ameliorating oocyte maturation and in promoting the development and genes expression of BAX, BCL-2, and BAG1.



Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article.
Code Availability
Not applicable.
Change history
18 September 2023
A Correction to this paper has been published: https://doi.org/10.1007/s43032-023-01356-z
References
Parrella A, Irani M, Keating D, Chow S, Rosenwaks Z, Palermo GD. High proportion of immature oocytes in a cohort reduces fertilization, embryo development, pregnancy and live birth rates following ICSI. Reprod Biomed Online. 2019;39(4):580–7.
Chian R, Tan S. Maturational and developmental competence of cumulus-free immature human oocytes derived from stimulated and intracytoplasmic sperm injection cycles. Reprod Biomed Online. 2002;5(2):125–32.
Desai N, Alex A, AbdelHafez F, Calabro A, Goldfarb J, Fleischman A, et al. Three-dimensional in vitro follicle growth: overview of culture models, biomaterials, design parameters and future directions. Reprod Biol Endocrinol. 2010;8(1):1–12.
Miraki S, Mokarizadeh A, Banafshi O, Assadollahi V, Abdi M, Roshani D, et al. Embryonic stem cell conditioned medium supports in vitro maturation of mouse oocytes. Avicenna J Med Biotechnol. 2017;9(3):114–9.
Wang X, Catt S, Pangestu M, Temple-Smith P. Successful in vitro culture of pre-antral follicles derived from vitrified murine ovarian tissue: oocyte maturation, fertilization, and live births. Reproduction. 2011;141(2):183–91.
Chian R-C, Lim J-H, Tan S-L. State of the art in in-vitro oocyte maturation. Curr Opin Gynecol Obstet. 2004;16(3):211–9.
Picton H, Harris S, Muruvi W, Chambers E. The in vitro growth and maturation of follicles. Reproduction. 2008;136(6):703–15.
Akbari H, Eftekhar Vaghefi SH, Shahedi A, Habibzadeh V, Mirshekari TR, Ganjizadegan A, et al. Mesenchymal stem cell-conditioned medium modulates apoptotic and stress-related gene expression, ameliorates maturation and allows for the development of immature human oocytes after artificial activation. Genes. 2017;8(12):371–85.
Braga DPdAF, Zanetti BF, Setti AS, Iaconelli Jr A, Borges Jr E. Immature oocyte incidence: Contributing factors and effects on mature sibling oocytes in intracytoplasmic sperm injection cycles. JBRA Assist Reprod. 2020;24(1):70–6.
Li H, Jia G-H, Lu X-L, Zhang G, Tian K-Y, Li J-T, et al. In vitro maturation of oocytes is not a risk factor for adult metabolic syndrome of mouse offspring. Eur J Obstet Gynecol Reprod Biol. 2014;174:96–9.
Dong L, Hao H, Liu J, Ti D, Tong C, Hou Q, et al. A conditioned medium of umbilical cord mesenchymal stem cells overexpressing Wnt7a promotes wound repair and regeneration of hair follicles in mice. Stem Cells Int. 2017;2017:1–13.
Zhang X, Zhang L, Li Y, Yin Z, Feng Y, Ji Y. Human umbilical cord mesenchymal stem cells (hUCMSCs) promotes the recovery of ovarian function in a rat model of premature ovarian failure (POF). Gynecol Endocrinol. 2021;37(4):353–7.
Virant-Klun I, Knez K, Tomazevic T, Skutella T. Gene expression profiling of human oocytes developed and matured in vivo or in vitro. BioMed Res Int. 2013;2013:1–20.
Ma W, Zhang D, Hou Y, Li Y-H, Sun Q-Y, Sun X-F, et al. Reduced expression of MAD2, BCL2, and MAP kinase activity in pig oocytes after in vitro aging are associated with defects in sister chromatid segregation during meiosis II and embryo fragmentation after activation. Biol Reprod. 2005;72(2):373–83.
Asgari HR, Akbari M, Abbasi M, Ai J, Korouji M, Aliakbari F, et al. Human Wharton’s jelly-derived mesenchymal stem cells express oocyte developmental genes during co-culture with placental cells. Iran J Basic Med Sci. 2015;18(1):22–9.
Ranjbaran H, Abediankenari S, Mohammadi M, Jafari N, Khalilian A, Rahmani Z, et al. Wharton's jelly derived-mesenchymal stem cells: Isolation and characterization. Acta Medica Iranica. 2018;56(1):28–33.
Ling B, Feng D, Zhou Y, Gao T, Wei H, Tian Z. Effect of conditioned medium of mesenchymal stem cells on the in vitro maturation and subsequent development of mouse oocyte. Braz J Med Biol Res. 2008;41:978–85.
Sumapraja K, Hestiantoro A, Liem IK, Boediono A, Jacoeb TZ. Effect of conditioned medium of umbilical cord-derived mesenchymal stem cells as a culture medium for human granulosa cells: An experimental study. Int J Reprod BioMed. 2021;19(12):1037.
Shen J, Cao D, Sun J-L. Ability of human umbilical cord mesenchymal stem cells to repair chemotherapy-induced premature ovarian failure. World J Stem Cells. 2020;12(4):277–87.
Varaa N, Azandeh S, Khodabandeh Z, Gharravi AM. Wharton’s jelly mesenchymal stem cell: Various protocols for isolation and differentiation of hepatocyte-like cells; narrative review. Iranian J Med Sci. 2019;44(6):437–48.
Parekkadan B, Van Poll D, Suganuma K, Carter EA, Berthiaume F, Tilles AW, et al. Mesenchymal stem cell-derived molecules reverse fulminant hepatic failure. PloS One. 2007;2(9):e941.
Nasr-Esfahani M, Salehi M, Razavi S, Mardani M, Bahramian H, Steger K, et al. Effect of protamine-2 deficiency on ICSI outcome. Reprod Biomed Online. 2004;9(6):652–8.
Peters VM, Spray DC, Mendez-Otero R. Effect of mesenchymal stem cells and mouse embryonic fibroblasts on the development of preimplantation mouse embryos. In Vitro Cell Dev Biol Anim. 2016;52(4):497–506.
Rios GL, Buschiazzo J, Mucci NC, Kaiser GG, Cesari A, Alberio R. Combined epidermal growth factor and hyaluronic acid supplementation of in vitro maturation medium and its impact on bovine oocyte proteome and competence. Theriogenology. 2015;83(5):874–80.
Shahedi A, Hosseini A, Khalili MA, Norouzian M, Salehi M, Piriaei A, et al. The effect of vitrification on ultrastructure of human in vitro matured germinal vesicle oocytes. Eur J Obstet Gynecol Reprod Biol. 2013;167(1):69–75.
Trounson A, Wood C, Kausche A. In vitro maturation and the fertilization and developmental competence of oocytes recovered from untreated polycystic ovarian patients. Fertil Steril. 1994;62(2):353–62.
Magli MC, Gianaroli L, Ferraretti AP, Lappi M, Ruberti A, Farfalli V. Embryo morphology and development are dependent on the chromosomal complement. Fertil Steril. 2007;87(3):534–41.
de Fried EP, Ross P, Zang G, Divita A, Cunniff K, Denaday F, et al. Human parthenogenetic blastocysts derived from noninseminated cryopreserved human oocytes. Fertil Steril. 2008;89(4):943–7.
Ferreira EM, Vireque AA, Adona PR, Ferriani RA, Navarro PA. Prematuration of bovine oocytes with butyrolactone I reversibly arrests meiosis without increasing meiotic abnormalities after in vitro maturation. Eur J Obstet Gynecol Reprod Biol. 2009;145(1):76–80.
Yang MY, Rajamahendran R. Expression of Bcl-2 and Bax proteins in relation to quality of bovine oocytes and embryos produced in vitro. Anim Reprod Sci. 2002;70(3–4):159–69.
Metcalfe AD, Hunter HR, Bloor DJ, Lieberman BA, Picton HM, Leese HJ, et al. Expression of 11 members of the BCL-2 family of apoptosis regulatory molecules during human preimplantation embryo development and fragmentation. Mol Reprod Dev. 2004;68(1):35–50.
Piltan A, Totonchi M, Rezazadeh M, Gourabi H, Karimian L, Baghaban Eslaminejad M, et al. Quantitative expression of BAG1, BAX and BCL-2 genes in human embryos with different fragmentation grades derived from ART. Yakhteh Med J. 2010;12:257–66.
Melka M, Rings F, Hölker M, Tholen E, Havlicek V, Besenfelder U, et al. Expression of apoptosis regulatory genes and incidence of apoptosis in different morphological quality groups of in vitro-produced bovine pre-implantation embryos. Reprod Domest Anim. 2010;45(5):915–21.
Zhang G-M, Gu C-H, Zhang Y-L, Sun H-Y, Qian W-P, Zhou Z-R, et al. Age-associated changes in gene expression of goat oocytes. Theriogenology. 2013;80(4):328–36.
Devreker F, Englert Y. In vitro development and metabolism of the human embryo up to the blastocyst stage. Eur J Obstet Gynecol Reprod Biol. 2000;92(1):51–6.
Spanos S, Rice S, Karagiannis P, Taylor D, Becker D, Winston R, et al. Caspase activity and expression of cell death genes during development of human preimplantation embryos. Reproduction. 2002;124(3):353–63.
Leon J, Acuña-Castroviejo D, Escames G, Tan DX, Reiter RJ. Melatonin mitigates mitochondrial malfunction. J Pineal Res. 2005;38(1):1–9.
Acknowledgements
We thank the members of the Department of Biology, Arak University. This research was supported by Arak University. Also, we appreciate the members of Department of the Stem Cells of the Academic Center for Education, Culture, and Research (ACER), Qom.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Monireh Mahmoodi, Ebrahim Cheraghi, and Alireza Riahi. The first draft of the manuscript was written by Alireza Riahi, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
This study was approved by the research ethics committee (the University of Qom Ethical Committee [IR.QOM.REC.1400.006]).
Consent to Participate
Written, informed consent was obtained from the patients.
Consent for Publication
No identifying details of the participants are published in this manuscript; hence, consent for publication is not obtained.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mahmoodi, M., Cheraghi, E. & Riahi, A. The Effect of Wharton’s Jelly-Derived Conditioned Medium on the In Vitro Maturation of Immature Oocytes, Embryo Development, and Genes Expression Involved in Apoptosis. Reprod. Sci. 31, 190–198 (2024). https://doi.org/10.1007/s43032-023-01345-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01345-2