Abstract
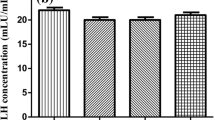
Male reproductive functions and bone health are both adversely affected by the high salt diet (HSD). Nevertheless, the underlying mechanism via which it alters the sperm function remains largely unknown. This study examines the mechanism by which HSD affects male fertility by impairing bone health. For investigating the same, male BALB/c mice were categorized into three groups—HSD group (fed with 4% NaCl), a low salt diet (LSD) group (fed with 0.4% NaCl), and a control group (fed with a normal diet) for 6 weeks and thereafter assessed for various sperm parameters, bone turnover markers, and testosterone levels. Furthermore, the quantitative assessment of testosterone biosynthesis enzymes was performed. Interestingly, we observed that mice fed with HSD showed significant alterations in sperm parameters—motility, count, and vitality, including morphological changes compared to both the LSD and the control groups. In addition, serum analysis showed an increase in bone resorption markers and a decrease in bone formation markers in the HSD group (p < 0.05). Further, HSD caused a decrease in the testosterone level and mRNA expression of testosterone biosynthesis enzymes. Importantly, a significant decrease in bone formation marker osteocalcin (OC) was observed to coincide with the dip in testosterone level in the HSD group. Given that OC plays a key role in maintaining male fertility, the above findings suggest that a decrease in OC levels may affect the testosterone biosynthesis pathway, reducing testosterone hormone secretion and thereby resulting in decreased spermatogenesis. The study for the first time delineates and bridges the mechanism of HSD-mediated bone loss (results in a deficiency of OC) with decreased testosterone biosynthesis and thus impaired male fertility.








Similar content being viewed by others
Data Availability
The data and material that support the findings of this study are available from the corresponding author upon reasonable request.
Code Availability
Not applicable.
References
Agarwal A, Mulgund A, Hamada A, Chyatte MR. A unique view on male infertility around the globe. Reproductive biology and endocrinology. Reprod Biol Endocrinol. 2015;13:1–9. https://doi.org/10.1186/s12958-015-0032-1.
Zegers-Hochschild F, Adamson GD, Dyer S, Racowsky C, De Mouzon J, Sokol R, et al. The international glossary on infertility and fertility care. Hum Reprod. 2017;32:1786–801. https://doi.org/10.1093/humrep/dex234.
Salas-Huetos A, James ER, Aston KI, Jenkins TG, Carrell DT. Diet and sperm quality: nutrients, foods and dietary patterns. Reprod Biol. 2019;19:219–24. https://doi.org/10.1016/j.repbio.2019.07.005.
Gray C, Long S, Green C, Gardiner SM, Craigon J, Gardner DS. Maternal fructose and/or salt intake and reproductive outcome in the rat: effects on growth, fertility, sex ratio, and birth order. Biol Reprod. 2013;89:51–1. https://doi.org/10.1095/biolreprod.113.109595.
Organization WH. Diet, nutrition, and the prevention of chronic diseases: report of a joint WHO/FAO expert consultation. World Health Organization; 2003. https://apps.who.int/iris/handle/10665/42665
Kaushik S, Kumar R, Kain P. Salt an essential nutrient: advances in understanding salt taste detection using drosophila as a model system. J Exp Neurosci. 2018;12:1179069518806894. https://doi.org/10.1177/1179069518806894.
Farquhar WB, Edwards DG, Jurkovitz CT, Weintraub WS. Dietary sodium and health: more than just blood pressure. J Am Coll Cardiol. 2015;65:1042–50. https://doi.org/10.1016/j.jacc.2014.12.039.
Cui Y, Sun K, Xiao Y, Li X, Mo S, Yuan Y, et al. High-salt diet accelerates bone loss accompanied by activation of ion channels related to kidney and bone tissue in ovariectomized rats. Ecotoxicol Environ Saf. 2022;244:114024. https://doi.org/10.1016/j.ecoenv.2022.114024.
Abdelnour SA, Abd El-Hack ME, Noreldin AE, Batiha GE, Beshbishy AM, Ohran H, et al. High salt diet affects the reproductive health in animals: an overview. Animals. 2020;10:590. https://doi.org/10.3390/ani10040590.
Fang Y, Zhong R, Sun X, Zhou D. High salt diet decreases reproductive performance in rams and down-regulates gene expression of some components of the renin-angiotensin system in the testis. Theriogenology. 2018;107:127–33. https://doi.org/10.1016/j.theriogenology.2017.11.005.
Adekunbi DA, Ogunsola OA, Oyelowo OT, Aluko EO, Popoola AA, Akinboboye OO. Consumption of high sucrose and/or high salt diet alters sperm function in male Sprague–Dawley rats. Egypt J Basic Appl Sci. 2016;3:194–201. https://doi.org/10.1016/j.ejbas.2016.03.003.
Wube T, Haim A, Fares F. Effect of increased dietary salinity on the reproductive status and energy intake of xeric and mesic populations of the spiny mouse. Acomys Physiol Behav. 2009;96(1):122–7. https://doi.org/10.1016/j.physbeh.2008.09.006.
Iranloye BO, Oludare GO, Morakinyo AO, Esume NA, Ekeh LC. Reproductive parameters and oxidative stress status of male rats fed with low and high salt diet. J Hum Reprod Sci. 2013;6(4):267–72. https://doi.org/10.4103/0974-1208.126308.
Lee EY, Cho KH. High intake of sodium chloride exacerbates degradation of human lipoproteins and impairment of reproductive capacity via testicular toxicity in zebrafish. Atherosclerosis. 2016;252:e116. https://doi.org/10.1016/j.atherosclerosis.2016.07.623.
Efrat M, Stein A, Pinkas H, Unger R, Birk R. Dietary patterns are positively associated with semen quality. Fertil Steril. 2018;109:809–16. https://doi.org/10.1016/j.fertnstert.2018.01.010.
Cutillas-Tolín A, Adoamnei E, Navarrete-Muñoz EM, Vioque J, Moñino-García M, Jørgensen N, et al. Adherence to diet quality indices in relation to semen quality and reproductive hormones in young men. Hum Reprod. 2019;34:1866–75. https://doi.org/10.1093/humrep/dez157.
Danielewicz A, Morze J, Przybyłowicz M, Przybyłowicz KE. Association of the dietary approaches to stop hypertension, physical activity, and their combination with semen quality: a cross-sectional study. Nutrients. 2019;12:39. https://doi.org/10.3390/nu12010039.
Liu C-Y, Chou Y-C, Chao JC-J, Hsu C-Y, Cha T-L, Tsao C-W. The association between dietary patterns and semen quality in a general Asian population of 7282 males. PloS One. 2015;10:e0134224. https://doi.org/10.1371/journal.pone.0134224.
Dar HY, Singh A, Shukla P, Anupam R, Mondal RK, Mishra PK, et al. High dietary salt intake correlates with modulated Th17-Treg cell balance resulting in enhanced bone loss and impaired bone-microarchitecture in male mice. Sci Rep. 2018;8:1–13. https://doi.org/10.1038/s41598-018-20896-y.
Wu L, Luthringer BJC, Feyerabend F, et al. Increased levels of sodium chloride directly increase osteoclastic differentiation and resorption in mice and men. Osteoporos Int. 2017;28:3215–28. https://doi.org/10.1007/s00198-017-4163-4.
Frassetto LA, Morris RC Jr, Sellmeyer DE, Sebastian A. Adverse effects of sodium chloride on bone in the aging human population resulting from habitual consumption of typical American diets. J Nutr. 2008;138:419S–22S. https://doi.org/10.1093/jn/138.2.419S.
Remer T. High salt intake: detrimental not only for blood pressure, but also for bone health? Endocrine. 2015;49:580–2. https://doi.org/10.1007/s12020-015-0626-6.
Tiyasatkulkovit W, Aksornthong S, Adulyaritthikul P, Upanan P, Wongdee K, Aeimlapa R, et al. Excessive salt consumption causes systemic calcium mishandling and worsens microarchitecture and strength of long bones in rats. Sci Rep. 2021;11:1–15. https://doi.org/10.1038/s41598-021-81413-2.
Oury F. A crosstalk between bone and gonads. Ann NY Acad Sci. 2012;1260:1–7. https://doi.org/10.1111/j.1749-6632.2011.06360.x.
Oury F, Sumara G, Sumara O, Ferron M, Chang H, Smith CE, et al. Endocrine regulation of male fertility by the skeleton. Cell. 2011;144:796–809. https://doi.org/10.1016/j.cell.2011.02.
Karsenty G, Oury F. Regulation of male fertility by the bone-derived hormone osteocalcin. Mol Cell Endocrinol. 2014;382:521–6. https://doi.org/10.1016/j.cell.2011.02.004.
Rato L, Alves MG, Dias TR, Lopes G, Cavaco JE, Socorro S, Oliveira PF. High-energy diets may induce a pre-diabetic state altering testicular glycolytic metabolic profile and male reproductive parameters. Andrology. 2013;1(3):495–504.
World Health Organization. WHO laboratory manual for the examination and processing of human semen. World Health Organization; 2021.
Jaâ M. A simple and practical method for rat epididymal sperm count (Rattus norvegicus). Biol Med Nat Prod Chem. 2015;4(1):1–3.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods. 2001;25(4):402–8. https://doi.org/10.1006/meth.2001.1262.
Coskun G, Sencar L, Tuli A, Saker D, Alparslan M, Polat S. Effects of osteocalcin on synthesis of testosterone and INSL3 during adult Leydig cell differentiation. Int J Endocrinol. 2019;2019:1–18. https://doi.org/10.1155/2019/1041760.
Tsai-Morris C-H, Sheng Y, Gutti R, Li J, Pickel J, Dufau ML. Gonadotropin-regulated testicular RNA helicase (GRTH/DDX25) gene: cell-specific expression and transcriptional regulation by androgen in transgenic mouse testis. J Cell Biochem. 2010;109(6):1142–7. https://doi.org/10.1002/jcb.22493.32.
Sheng Y, Tsai-Morris C-H, Gutti R, Maeda Y, Dufau ML. Gonadotropin-regulated testicular RNA helicase (GRTH/Ddx25) is a transport protein involved in gene-specific mRNA export and protein translation during spermatogenesis. J Biol Chem. 2006;281:35048–56. https://doi.org/10.1074/jbc.M605086200.
Liu H-C, Zhu D, Wang C, Guan H, Li S, Hu C, et al. Effects of etomidate on the steroidogenesis of rat immature Leydig cells. PloS One. 2015;10:e0139311. https://doi.org/10.1371/journal.pone.0139311.
Nwangwa JN, Udefa AL, Amama EA, Inah IO, Ibrahim HJ, Iheduru SC, et al. Cyperus esculentus L.(tigernut) mitigates high salt diet-associated testicular toxicity in Wistar rats by targeting testicular steroidogenesis, oxidative stress and inflammation. Andrologia. 2020;52(11):e13780. https://doi.org/10.1111/and.13780.
Domazetovic V, Marcucci G, Iantomasi T, Brandi ML, Vincenzini MT. Oxidative stress in bone remodeling: role of antioxidants. Clin Cases Miner Bone Metab. 2017;14:209. https://doi.org/10.11138/ccmbm/2017.14.1.209.
Dai P, Mao Y, Sun X, Li X, Muhammad I, Gu W, et al. Attenuation of oxidative stress-induced osteoblast apoptosis by curcumin is associated with preservation of mitochondrial functions and increased Akt-GSK3β signaling. Cell Physiol Biochem. 2017;41(2):661–77. https://doi.org/10.1159/000457945.
Acknowledgements
The authors wish to thank Dr. Sushil Saini at National Dairy Research Institute for assistance in RNA isolation and qPCR, Dr. Kriti for ELISA analysis, and Miss Kajal Sihag for sperm microscopy.
Funding
This study was supported by the AIIMS intramural project A-619 sanctioned to Dr. Shrabani Saugandhika.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics Approval
The study protocol was reviewed and approved by the Animal Ethics Committee of All Institute of Medical Sciences, New Delhi, India (Approval number- 64/1AEC-1/2018).
Consent to Participate
All authors had final approval of the submitted versions.
Consent for Publication
The authors and institution agreed with this publication.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Materials
Fig 1 Correlation analysis of Testosterone with OC and other bone turnover markers: (Pearson correlation) a) OC, Testosterone correlation; *p < 0.05, r2=0.972 b) OPG, Testosterone correlation; *p < 0.05, r2=0.97 c) CTX-II, Testosterone correlation; *p < 0.05, r2=0.904, d) RANKL, Testosterone correlation; *p < 0.05, r2=0.905, e) ALP, Testosterone correlation; *p < 0.05, r2=0.974. (DOCX 90 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saugandhika, S., Sapra, L., Kumari, K. et al. High Salt Diet Impairs Male Fertility in Mice via Modulating the Skeletal Homeostasis. Reprod. Sci. 30, 3339–3352 (2023). https://doi.org/10.1007/s43032-023-01278-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01278-w