Abstract
The Annamites ecoregion harbors exceptional levels of species richness and endemism, but intensive snaring has decimated populations of terrestrial mammals. Ungulates, such as muntjacs, are susceptible to snaring, and in need of effective conservation action. At least three muntjacs occur sympatrically in the region: the Annamite dark muntjac species complex Muntiacus rooseveltorum/truongsonensis, the northern red muntjac Muntiacus vaginalis, and the large-antlered muntjac Muntiacus vuquangensis. We conducted a landscape-scale systematic camera-trapping survey in Nakai-Nam Theun National Park to gather information on the ecology and distribution of these muntjacs. We analyzed camera-trap records within an occupancy framework to evaluate responses to environmental and anthropogenic variables, and to predict distributions across the protected area. We found varying responses to the covariates, indicating complex drivers of occurrence, though all three muntjac had higher occupancies in more inaccessible areas. Mean (95%) PAO in the protected area was higher for large-antlered muntjac (0.33 [0.22–0.49]), followed by Annamite dark muntjac (0.28 [0.18–0.39]), and then northern red muntjac (0.27 [0.15–0.42]). Large-antlered muntjac and northern red muntjac were widespread, while dark muntjac was restricted to a single high elevation area. Overall, our results provide new insights into muntjac ecology, distribution, and population status, and we discuss how this information can be used to inform conservation efforts. Given the high occupancies that we found for the Critically Endangered large-antlered muntjac, we argue that Nakai-Nam Theun National Park may be vital for the long-term survival of the species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Globally, tropical rainforests account for high levels of mammal richness, including many range-restricted and endemic species (Ceballos and Ehrlich 2006), though populations have declined precipitously as a result of numerous anthropogenic pressures (Farris et al. 2017; Symes et al. 2018). In recent decades, unsustainable hunting has emerged as one of the main drivers of mammal declines (Harrison 2011; Benítez-López et al. 2017). Levels of unsustainable hunting have been particularly severe in Southeast Asia, where weak protected area governance (Graham et al. 2021), an expanding road network (Clements et al. 2014), and a thriving wildlife trade (Harrison et al. 2016; Gray et al. 2018) have contributed to the emptying of the region’s tropical forests. Several studies indicate that large-bodied mammals, including ungulates, are especially susceptible to overexploitation, due both to hunter preferences (Jerozolimski and Peres 2003; Chang et al. 2017) and life-history characteristics that make these species less resilient to high offtake levels (Cardillo et al. 2005). Overhunting has already contributed to the extinction of one Southeast Asian ungulate within the past 80 years (Duckworth et al. 2015). At the same time, other ungulates are currently threatened with extinction, such as the Critically Endangered saola Pseudoryx nghetinhensis (Timmins et al. 2020) and large-antlered muntjac Muntiacus vuquangensis (Timmins et al. 2016a). Therefore, urgent conservation efforts are needed to prevent continued local extirpations and extinctions of ungulates in the Southeast Asian biodiversity hotspot.
Within Southeast Asia, the Annamites ecoregion of Viet Nam and Laos harbours exceptionally high ungulate richness and endemism (Baltzer et al. 2001). Here, muntjac (deer of the genus Muntiacus native to South and Southeast Asia) diversity is particularly high: the large-antlered muntjac Muntiacus vuquangensis, northern red muntjac M. vaginalis, and Annamite dark muntjac species complex M. rooseveltorum/truongsonensis are all found within the ecoregion. The large-antlered muntjac is endemic to the Annamites (Timmins et al. 2016a), and it is possible that one or more species of the dark muntjac complex are also restricted to the area, though further studies are needed to confirm this (Timmins and Duckworth 2016a, b). Within the Annamites, as in other parts of Southeast Asia, muntjacs play an important role in ecosystem functioning, primarily through seed dispersal (Chen et al. 2001; McConkey et al. 2018). Thus, muntjacs are likely to be important to maintaining the long-term health of tropical forests in the region.
The Annamite forests also face extremely high levels of unsustainable hunting, primarily accomplished through the setting of indiscriminate wire snares (Gray et al. 2018). Widespread and intensive snaring has defaunated many forests in the region, resulting in population declines for all Annamite ungulates (Belecky and Gray 2020). The large-antlered muntjac is listed as Critically Endangered on The IUCN Red List of Threatened Species (Timmins et al. 2016a) and without immediate conservation efforts faces extinction in the near future. The northern red muntjac is listed as Least Concern, in part because there are stable populations across its widespread distribution, but populations in the Annamites are estimated to have declined in recent years (Timmins et al. 2016b).The Annamite dark muntjac species complex is currently listed as Data Deficient due to unresolved taxonomy and a lack of information on its status, though like all ungulates in the Annamites, its populations are probably declining (Timmins and Duckworth 2016a, b). To protect remaining populations of muntjacs in the Annamites, it is important to gather basic information on their ecology, distribution, and population status in priority areas. To date, few studies have focused on this species group, and those studies that have occurred have predominantly been limited in scope or not utilized systematically collected data (Timmins et al. 1998; Timmins and Evans 1996; Timmins and Duckworth 2013). Moreover, estimates of muntjac population declines in the Annamites are largely based on qualitative rather than quantitative assessments, and more rigorous data-driven approaches can provide additional information that can be used to inform conservation strategies. In recent decades, automatic camera-traps and new analytical techniques have revolutionized the ability to collect, examine, and interpret data on cryptic mammals living in tropical forests (Sollmann et al. 2013; Tobler et al. 2008). Moreover, because camera-traps can capture subtle morphological differences among closely related species (Coudrat and Nekaris 2013), the method is particularly well-suited for discriminating between taxonomically similar, sympatric species.
In this study, we conducted systematic landscape-scale camera-trapping in Nakai-Nam Theun National Park (NP), located in central Lao People’s Democratic Republic (hereafter, Laos), with the goal of understanding muntjac ecology and distribution, as well as to establish robust conservation baselines for these species. We hypothesized that muntjac occurrence would be influenced by both anthropogenic and environmental factors, as has been demonstrated in other Annamite landscapes (Tilker et al. 2020a). Because muntjac populations in the Annamites are believed to be negatively impacted by snaring pressure (Timmins and Duckworth 2016a, b; Timmins et al. 2016a, b), we predicted that occupancy for all three muntjacs would be negatively influenced by proxies for accessibility. Furthermore, we predicted that Annamite dark muntjac occupancy would increase with elevation, based on previous studies that suggest the species complex is more common in highland areas, and that large-antlered and red muntjac would have highest occupancies in lower- and mid-elevation forest areas (Long 2005; Timmins et al. 2016a, b; Timmins and Duckworth 2016a, b; Tilker et al. 2020a). We included other environmental covariates in our models that might affect muntjac occurrence, but given how little is known about the ecology of these muntjac, we did not make a priori predictions. To assess our predictions, we analyzed camera-trap data within an occupancy framework to account for imperfect detection, and used both environmental and anthropogenic covariates to better understand the factors that influence muntjac occurrence across the national park.
Our study is the first comprehensive and landscape-scale study on all three sympatric muntjacs that occur in the region, and one of the few studies to provide information that can be used for robust estimation of population trends and distributions over time. We discuss how our findings can be used to inform conservation strategies for the protection of Annamite muntjacs.
Materials and methods
Study area
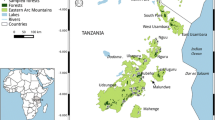
Nakai-Nam Theun NP (ca. 4000 km2) was established as a national protected area in 1993 in central-east Laos in the Annamite range (Fig. 1). The eastern boundary of the national park lies along the international border with Viet Nam. The national park contains diverse habitat types, including dry and wet evergreen, semi-evergreen, and coniferous forests. It encompases an elevational range from 500 to 2300 m above sea level (asl) and has annual precipitation from 1865 to 2620 mm (Coudrat and Nekaris 2013). The protected area is managed by the government institution “Nam Theun 2 Watershed Management and Protection Authority'', and receives technical support from various non-governmental organizations. Nakai-Nam Theun NP harbors a full suite of Annamite endemic mammals, including large-antlered muntjac, Annamite dark muntjac, Annamite striped rabbit Nesolagus timminsi, Owston’s civet Chrotogale owstoni, and possibly still holds saola. In addition, a number of globally threatened species have been recorded in the national park, though some, such as tiger Panthera tigris and leopard Panthera pardus, have been locally and nationally extirpated in recent decades (Rasphone et al. 2019; Coudrat et al. 2014; Coudrat 2021).
According to the local law, local communities are allowed to hunt certain species (including northern red muntjac and wild pig Sus scrofa) in allocated forest zones (referred to as “controlled use zones”), while in other zones, human activities are prohibited (referred to as “total protection zones”). Mammals taken from these activities can only be used for personal consumption; trading any wildlife catch is illegal (GoL 2007, 2019). Legal hunting can only take place during specific seasons with traditional hunting techniques; the use of hunting methods that may lead to rapid depletion of species populations (such as fence snares) is prohibited. Since 2017, the park management strategy has focused on three biodiversity priority zones that were designated based on their habitat diversity and occurrence of conservation-priority species (O’Kelly et al. 2018a, b): Thong Khacheng, Nam Chae, and Thong Khouang (Fig. 1). The three zones are ‘total protection zones’, where it is prohibited to conduct any human activities except for scientific research, and walking trails for ecotourism. The Thong Khacheng zone is an area with mainly primary evergreen forest of relatively high elevation (average of 1204 ± 150 m asl) and has experienced the lowest levels of snaring and other forms of hunting due to its remoteness. Nam Chae is of lower elevation (average of 827 ± 161 m asl) and has both primary and secondary forests of mixed broadleaf and coniferous forest patches. It underwent intensive logging in the mid-1990s to the early 2000s, during which time intensive hunting also took place, though current hunting mainly occurs from isolated snare traps (rather than intensive trade-driven snare fences) and gun hunting. Despite the prohibitions, villagers from inside and outside the national park also regularly collect non-timber forest products (for trade and/or local consumption) from Nam Chae. The Thong Khouang zone has an average elevation of 849 ± 220 m asl and is predominantly a primary evergreen forest. This zone has experienced the most intense trade-driven hunting through the use of fence snares.
Within these three zones, 16 ranger teams (composed mainly of six to seven hired and trained villagers from villages located inside the national park) patrol an average of 15 days/month. Reports indicate that much of the illegal hunting pressure originates from Vietnamese hunters crossing the border into the national park (O’Kelly et al. 2018a, b).
Data collection and preparation
Systematic camera-trap surveys (Table 1) were conducted between February 2018 and May 2019 across the three biodiversity management zones (Coudrat 2019). In total, 134 stations were established (50 in Thong Khacheng, 50 in Thong Khouang and 34 in Nam Chae zone) consisting of 256 individual camera-trap units at elevations ranging from 561 to 1717 m asl Cameras were set using a grid design, with stations spaced approximately 2.5 km apart, following Abrams et al. (2018). Each station consisted of two individual cameras spaced up to 20 m apart. For the Nam Chae and Thong Khacheng zones the cameras were set facing each other, and for Thong Khouang the cameras were set in different directions. Cameras were set at 20–40 cm above ground and operational for 24 h per day. Vegetation was cleared in front of each camera to ensure clear and consistent mammal detections. Three camera models were used: Bushnell Aggressor (two cameras), Covert Illuminator (100 cameras), and PantheraCam V6 (100 cameras).
We collected information on both environmental and anthropogenic factors to assess potential influences on muntjac occurrence (Table 3). Elevation was derived from an SRTM 30 m digital elevation model. Previous studies indicate that elevation is a strong driver of mammal occurrence in the Annamites, including muntjacs (Tilker et al. 2020a). Terrain ruggedness was calculated in R v.4.0.2 (R Core Team 2020) using the raster package (Hijmans 2019) and a 3 × 3 neighbourhood size. Terrain ruggedness was included as a covariate as some ungulate species have been found to be associated with more rugged terrain (Estes et al. 2011), and the fact that more rugged areas may be harder for hunters to access. Distance to streams was calculated using a GIS layer obtained from the national park authority, and was used to assess if proximity to water sources influenced muntjac occurrence. We used the gdistance package (van Etten 2017) to calculate the Euclidean distance between stations and streams. The homogeneity covariate was derived from (Tuanmu and Jetz 2015) and based on the Enhanced Vegetation Index (EVI) at 1-km resolution. Homogeneity measures the similarity of EVI between pixels, and was used as a measure of spatial variability of habitat. Accessibility, defined as the time needed to reach a certain area by foot from the nearest access point, was used as a proxy for hunting pressure. Past studies have shown a correlation between hunting pressure and aspects of accessibility, such as road development (Espinosa et al. 2014) and distance to villages (Koerner et al. 2017). Here, we used villages, roads, and the border of the national park as access points to create a single accessibility raster, calculated from a 30-m DEM using Tobler’s hiking function (corrected for off-path travel) in the R package movecost (Alberti 2019). Finally, we measured canopy cover around each camera-trap station by converting five photos at each station to black and white binary images, representing foliage and sky, then calculating the percentage of each category (see Abrams et al. 2018 for details).
All continuous variables were scaled and standardized to have a mean value of 0 and a standard deviation of 1. No collinearity was observed among covariates using Pearson’s collinearity test with a threshold of 0.7 (Supplementary information Table S1).
Data analysis
All analyses were conducted in R v.4.0.2. We used the package camtrapR (Niedballa et al. 2016) for preparation of camera trapping data. Muntjac identification was conducted by co-authors experienced in the morphological characteristics of muntjacs in the Annamites. Initial identifications were done by one expert, then checked by the other two muntjac identification experts; any ambiguous or difficult to identify muntjac photos were discussed by the three co-authors working on muntjac identification, and then a final identification was given based on group consensus. We used several morphological characteristics for muntjac identification, including antler configuration for males, crown markings and coloration for females, and tail shape and length for both sexes (SI Fig. 1). At a minimum, muntjac photographs needed to clearly show the head and/or tail for identification. All muntjac photos were identified to species-level, with the exception of the dark muntjac complex, which was identified as M. rooseveltorum/truongsonensis. Muntjacs that could not be unambiguously identified (17% of total events) were not included in the analyses. We used a 1 h threshold to denote independent camera-trap events, following other recent camera-trapping studies from the region (Brozovic et al. 2018; Mohd-Azlan et al. 2018; Tilker et al. 2020a, b).
We analyzed the camera-trap data using single-species, single-season occupancy models (MacKenzie et al. 2002). Due to the large spacing between stations (2.5 km), muntjac detections were considered to be spatially independent. Although there is no available information on the home range size of the Annamite muntjacs, a previous study on Reeve’s muntjac M. reevsi showed an average home range size of 1.07 km2 (McCullough et al. 2000). To estimate occupancy, we first created detection histories for each of the three muntjacs for each camera-trap station, with a “1” indicating that the muntjac was detected during that occasion (at least one camera-trap photograph), and a “0” indicating that it was not detected (no camera-trap photograph). We used 5-day sampling occasions, resulting in a maximum of 25 occasions per station. Detection histories were created in camtrapR (Niedballa et al. 2016). We combined photographs from the two individual camera-traps at each station, following the protocol in Tilker et al. (2020a, b). We fit models in the unmarked package (Fiske and Chandler 2011). To avoid model overfitting, we tested individual covariates to assess variable importance before constructing the final models. We used covariates both on detection (p) and occupancy (Ψ). We included camera-trap effort (the number of active camera-trap nights per occasion for each station) as an a priori covariate on detection. Then, we tested biodiversity management zone and camera model as categorical covariates on detection. Including biodiversity management zones on detection also accounted for the different camera-trap setups among the three zones (ie. cameras facing each other in Nam Chae and Thong Khacheng zones vs. not facing each other in Thong Khouang zone; cf. Data collection and preparation).
We selected covariates on detection by including no covariates on occupancy and assessing model fit using AIC scores, using all covariate combinations. We followed a similar procedure for covariates on occupancy by testing each occupancy covariate while using the best detection covariates, then assessing covariate importance based on AIC ranking, p values and 95% CI. We considered any covariate on occupancy to be significant if it performed better than the null model while having p < 0.05 and 95% CI not overlapping zero. For elevation, canopy cover, terrain ruggedness, and homogeneity, we also explored nonlinear relationships, to test potential non-monotonous associations of occupancy with environmental conditions. Nonlinear relationships were defined as a combination of a linear and a quadratic term.
We used covariate responses from the final models to predict muntjac occupancy across Nakai-Nam Theun NP. Predictions were made at the 30 m spatial resolution, corresponding to the resolution for the elevation raster. We used the raster package to transform all other covariate layers to this spatial resolution. The canopy cover covariate was excluded from prediction maps, as this information was collected in situ, and, therefore, not available for predictions across the wider landscape. Finally, we estimated the percentage of area occupied (PAO) for each species based on the final models and covariate rasters at 300 m resolution. To account for model uncertainty in PAO estimation we created 10,000 random samples of each relevant model parameter (using their estimated means and standard errors as means and standard deviations of normal distributions), and for each random sample thus created, calculated occupancy probabilities and simulated binary occupancy at each cell (as a binomial random draw with the given occupancy probability). These binary occupancy predictions were then summarized across each biodiversity management zone to obtain mean PAO and 95% confidence intervals. Biodiversity management zones were delineated in QGIS (2020) via convex hulls around each set of camera-trap stations.
Results
We obtained data from 131 camera-trap stations totaling 20,183 trap nights. All three sympatric muntjacs were recorded (Table 2). Annamite dark muntjac was the most commonly photographed muntjac with 641 independent detections at 28 stations, followed by the northern red muntjac with 491 detections at 44 stations and the large-antlered muntjac with 272 detections at 63 stations. The Annamite dark muntjac was photographed only in the Thong Khacheng management zone. Northern red muntjac and the large-antlered muntjac were detected in all three management zones, though the majority of red muntjac detections were in Thong Khacheng.
Percentage of area occupied (PAO) varied considerably among muntjacs and biodiversity management zones. For Annamite dark muntjac, mean (95% CI) PAO was estimated to be 0.14 (0.06–0.29) in Nam Chae, 0.53 (0.35–0.67) in Thong Khacheng, 0.23 (0.13–0.36) in Thong Khouang and 0.28 (0.18–0.39) across all areas. For northern red muntjac, mean (95% CI) PAO was estimated to be 0.20 (0.09–0.36) in Nam Chae, 0.59 (0.40–0.74) in Thong Khacheng, 0.30 (0.22–0.41) in Thong Khouang and 0.27 (0.15–0.42) across all areas. Finally, for large-antlered muntjac, mean (95% CI) PAO was estimated to be 0.39 (0.27–0.52) in Nam Chae, 0.51 (0.42–0.60) in Thong Khacheng, 0.53 (0.44–0.62) in Thong Khouang and 0.33 (0.22–0.49) across all areas.
For all three muntjacs, the inclusion of biodiversity management zones on detection performed better than the null model (Table S2). Thus, both biodiversity management zone and camera-trap effort were included as detection covariates. For the Annamite dark muntjac and the northern red muntjac, the top detection model included camera effort and biodiversity management zones as detection covariates. For the large-antlered muntjac, the top model included camera effort and camera models as detection covariates. However, in the case of the large-antlered muntjac, the model that included camera effort and biodiversity management zones as covariates on detection had a ΔAIC ≤ 2 from the top model. Thus, we decided to include camera effort and biodiversity management zones as detection covariates for all three models, so that all three models include the same covariates on detection.
The most important covariates for occupancy varied with each muntjac (Tables 3, S3, S4, S6). Response curves for covariates used in the best models per species are shown in Fig. 2. For Annamite dark muntjac the best model included only elevation (positive relationship), and for large-antlered muntjac the best model included only accessibility. For northern red muntjac, occupancy was best explained by elevation (nonlinear positive relationship peaking at ~ 1250 m), accessibility (positive relationship), homogeneity (negative relationship), terrain ruggedness (positive relationship) and canopy cover (positive relationship). We ranked all possible combinations of these covariates (Table S5) and selected elevation, accessibility, homogeneity and canopy cover for the final model. Distance to streams was the only covariate not to strongly influence any of the three muntjacs.
Response curves obtained from single covariate models. Light gray shades indicate 95% confidence intervals. Rugs correspond to detections (top) and non-detections (bottom). Accessibility is defined as the time needed to reach a certain area by foot from the nearest access point. Canopy cover indicates the % of canopy cover at each station. Distance to streams is the Euclidean distance between stations and the nearest streams. Elevation is the station’s elevation above sea level (from SRTM 30 m DEM). Homogeneity is a measure for habitat heterogeneity. Terrain ruggedness index is the mean difference in elevation between a DEM cell and its 8 surrounding cells
Prediction maps for the three muntjacs are presented in Fig. 3. The distribution of the Annamite dark muntjac was restricted to higher elevation areas in the north-eastern part of Nakai–Nam Theun NP, as well as the highland areas in Thong Khouang. In comparison, the northern red muntjac and large-antlered muntjac both occurred more widely across the national park. Large-antlered muntjac occupancy was strongly influenced by accessibility, with the highest predicted occupancies in the Thong Khouang and Thong Khacheng areas, and lower predicted occupancy in Nam Chae. Northern red muntjac followed a similar pattern, but had higher predicted occupancy in the Nam Chae area than large-antlered muntjac.
Predicted probability of occurrence for the three muntjac species across the Nakai-Nam Theun National Park. Transparent colors indicate uncertain areas falling outside of the range of the covariates used. White dots with black outline represent camera-trap stations (left). Violin plots show occupancy probability per biodiversity priority zone and species (right). Mean and SD are shown inside each violin plot
Discussion
This study is the first, to our knowledge, to use quantitative methods to assess the ecology and population status of all three sympatric muntjacs in the Annamites. Our findings provide new insights into these species, and provide a basis for more informed conservation and management decisions in one of their key strongholds.
The factors influencing occupancy varied among the three muntjacs. The fact that Annamite dark muntjac was predominantly found at higher elevations confirms previous qualitative assessments that the species complex is more common in highland areas (Long 2005; Timmins and Duckworth 2016a, b). This finding is also consistent with results from a similar occupancy-based study in a forest area spanning Laos and Viet Nam (Tilker et al. 2020a). Timmins and Duckworth (2016a, b) suggest that the association of Annamite dark muntjac with higher elevations may be linked to the general tendency for highland areas to be characterized by more evergreen forest. Although we did not specifically test habitat associations in our study, such a conclusion seems reasonable for Nakai-Nam Theun NP, since the areas to the east near the border with Viet Nam are generally wetter than the lowland areas to the west (Timmins and Evans 1996). The reason for this association is unclear, but given that the pattern seems to hold for this species complex across multiple sites in the Annamites (Tilker et al. 2020a), it appears to be a consistent aspect of Annamite dark muntjac ecology.
Northern red muntjac occupancy peaked at approximately 1250 m asl., but declined at elevations above this. Such a finding is broadly consistent with previous assessments that in Laos the northern red muntjac may be uncommon above 1500 m asl (Timmins and Duckworth 2016a). Northern red muntjac also showed higher occupancies in more remote areas. As with Annamite dark muntjac, this finding may be explained by lower occupancies in areas that are more accessible to hunters. Surprisingly, we found that red muntjac occupancy increased with canopy cover (as measured around the camera trap stations), but decreased with habitat homogeneity (as measured at a 1-km resolution). One possible explanation for this finding is that red muntjac is associated with denser forest at a microhabitat scale, but more heterogeneous habitat at larger spatial scales. Liwei et al. (2004) found higher levels of site use in areas with higher canopy cover, and suggested that this might be due to antipredator behavior. The association with more heterogeneous habitat follows previous studies that show that red muntjac can tolerate a range of habitat types (Liwei et al. 2004; Gray and Phan 2011).
Large-antlered muntjac occupancy was strongly influenced by accessibility, and was the only one of the three muntjacs to be affected exclusively by anthropogenic factors. We interpret this finding to indicate, as with the other two muntjacs, that large-antlered muntjac populations may be depressed by hunting pressure in less remote areas. From a conservation perspective, such a finding is more worrisome for large-antlered muntjac than the other two Annamite muntjacs, given its globally threatened status. Although elevation was not included as a covariate in our final models, the documentation of large-antlered muntjac at higher elevations in Nakai-Nam Theun NP was surprising given previous speculation that the species predominantly occurs below 1000 m asl due to ecological habitat associations (Timmins et al. 2016a). In contrast, median elevation for our large-antlered muntjac records was 932 m asl in a range of 589–1717 m asl. Our highest large-antlered muntjac record was 1717 m asl, with 29/63 stations with detections above 1000 m a.s.l, and 4/63 stations with detections above 1500 m asl. These findings suggest that large-antlered muntjac may be more widespread within Nakai-Nam Theun NP than was previously believed.
Our findings also highlight the importance of considering how even closely related taxa may respond differently to anthropogenic and environmental factors. Several previous studies have grouped taxonomically similar species into one group for analytical purposes, often because of difficulties in unambiguously identifying focal species through camera-trap photos (Bernard et al. 2014; Brodie et al. 2015; Sollmann et al. 2017). While this may be unavoidable in some situations, we recommend that scientists carefully consider the possible implications of grouping different species with potentially different responses to ecological and anthropogenic factors, and interpret the results with appropriate caution.
Percentage of Area Occupied (PAO), occupancy estimates, and predictive maps all provide information that can be used to inform conservation decisions in Nakai-Nam Theun NP. Assessing population changes over time is an integral part of adaptive management. Our occupancy estimates provide a robust conservation baseline for each muntjac and management zone, which can in turn be used to assess population trends and, ultimately, the effectiveness of conservation interventions. The Thong Khacheng area had the highest predicted richness for all three muntjacs, likely because it is more remote than the other two areas, and because it has the high elevation areas that are suitable for the dark muntjac complex. We recommend that this area should be a priority for in situ conservation efforts. Indeed, this zone holds several other threatened and endemic terrestrial mammal species—including Owston’s civet Chrotogale owstoni (Endangered), sun bear Helarctos malayanus (Vulnerable), Asiatic black bear Ursus thibetanus (Endangered), Annamite striped rabbit Nesolagus timminsi (Endangered), pangolin Manis spp. (Critically Endangered), as well as large populations of arboreal white-cheeked gibbon Nomascus spp. (Critically Endangered) and red-shanked douc Pygathrix nemaeus (Critically Endangered) (Coudrat 2019, 2020)—providing further support for the idea that, among the three management zones, it is the least impacted by hunting pressure. However, from a more general muntjac conservation perspective, it is imperative to protect all large-antlered muntjac populations. Because large-antlered muntjac was distributed across all management zones, it is important that all three areas are afforded the strictest level of protection. In Nakai-Nam Theun NP, this primarily means maintaining consistent patrolling efforts to prevent poaching in these core areas. We suggest that, in addition to patrolling, conservation stakeholders should pursue a multi-faceted approach to proactively reduce poaching, which could include basic village outreach education and outreach (COTE 2018) to more involved crime prevention strategies (Viollaz et al. 2021). There is no question that implementing these activities represents a challenging task; despite the fact that Nakai-Nam Theun NP is the most funded protected area in Laos, it still has the nascent protected area management capacities typical of most protected areas in Laos (Coudrat 2021, in press).
Our findings provide further evidence that Nakai-Nam Theun NP is a global stronghold for large-antlered muntjac. The fact that large-antlered muntjac is still widespread across much of the protected area stands in sharp contrast to the situation across most of the species’ range, especially in Viet Nam, where both current and past hunting pressure has been more intense (Timmins et al. 2016a). We recorded 272 independent detections of large-antlered muntjac in 48% of camera-trapping stations in Nakai-Nam Theun NP. For comparison, camera-trapping using the same systematic design from 2014 to 2020 (611 camera-trap stations with 76.662 camera-trap nights) across 10 forest areas in Viet Nam and two areas in Laos, all of which are within the historical distribution of the species, has recorded only 23 large-antlered muntjac independent detections in 4 (0.65%) stations (Tilker et al. 2020a; Leibniz-IZW, WWF Viet Nam, WWF Laos unpublished information). While it is possible that some of these areas would have had naturally low densities of large-antlered muntjac due to habitat associations (Timmins et al. 2016a), there is little question that, given the exceptionally high levels of snaring pressure across the Annamites (Gray et al. 2018), the primary reason for such low encounter rates is that the species has been extirpated from large parts of its former range. These findings further indicate that the global population of large-antlered muntjac is small and highly threatened. That other hunting-sensitive mammal species appear to be in decline in Nakai-Nam Theun NP (Coudrat et al. 2014) presents a worrisome picture for large-antlered muntjac populations in the protected area. Despite the high survey effort of our study, numerous species which historically occurred in Nakai-Nam Theun NP—several of which were camera-trapped between 2007 and 2010, such as mainland clouded leopard Neofelis nebulosa, Asiatic golden cat Catopuma temminckii, and dhole Cuon alpinus (Coudrat et al. 2014)—were not recorded during this study. Securing large-antlered muntjac, as well as other Annamite endemic species that are still present in Nakai-Nam Theun NP, is critical for the conservation of these species in the Annamite mountains.
Availability of data and materials
Considering the sensitive information on endangered species in the present study, as well as the illegal wildlife trade, no data will be publicly archived. Instead, all supporting data is accessible from authors upon request.
Code availability
Not applicable.
Change history
19 October 2022
Funding information was updated.
References
Abrams JF, Axtner J, Bhagwat T, Mohamed A, Nguyen A, Niedballa J, Wilting A (2018) Studying terrestrial mammals in tropical rainforests. a user guide for camera-trapping and environmental DNA. Leibniz-IZW, Berlin
Alberti G (2019) movecost: an R package for calculating accumulated slope-dependent anisotropic cost-surfaces and least-cost paths. SoftwareX 10:100331
Baltzer M, Nguyen D, Shore R (2001) Towards a vision for biodiversity conservation in the forests of the Lower Mekong ecoregion complex: summary of the biological assessment for the ecoregion biodiversity conservation program in the forests of the Lower Mekong ecoregion complex. WWF Indochina
Belecky M, Gray TNE (2020) Silence of the Snares: Southeast Asia’s Snaring Crisis. WWF International, Gland
Benítez-López A, Alkemade R, Schipper AM, Ingram DJ, Verweij PA, Eikelboom JAJ, Huijbregts MAJ (2017) The impact of hunting on tropical mammal and bird populations. Science 356(6334):180–183
Bernard H, Baking EL, Giordano AJ, Wearn OR, Ahmad AH (2014) Terrestrial mammal species richness and composition in three small forest patches within an oil palm landscape in Sabah, Malaysian Borneo. Mammal Study 39(3):141–154
Brodie JF, Giordano AJ, Ambu L (2015) Differential responses of large mammals to logging and edge effects. Mamm Biol 80(1):7–13
Brozovic R, Abrams JF, Mohamed A, Wong ST, Niedballa J, Bhagwat T, Sollmann R, Mannan S, Kissing J, Wilting A (2018) Effects of forest degradation on the moonrat Echinosorex gymnura in Sabah, Malaysian Borneo. Mamm Biol 93(1):135–143
Cardillo M, Mace GM, Jones KE, Bielby J, Bininda-Emonds OR, Sechrest W, Orme CDL, Purvis A (2005) Multiple causes of high extinction risk in large mammal species. Science 309(5738):1239–1241
Ceballos G, Ehrlich PR (2006) Global mammal distributions, biodiversity hotspots and conservation. PNAS 103(51):19374–19379. https://www.pnas.org/content/103/51/19374
Chang CH, Barnes ML, Frye M, Zhang M, Quan RC, Reisman L, Levin SA, Wilcove DS (2017) The pleasure of pursuit: recreational hunters in rural Southwest China exhibit low exit rates in response to declining catch. Ecol Soc J Integr Sci Resil Sustain 22(1):43. https://doi.org/10.5751/ES-09072-220143
Chen J, Deng XB, Bai ZL, Yang Q, Chen GQ, Liu Y, Liu ZQ (2001) Fruit characteristics and Muntiacus muntijak vaginalis (Muntjac) visits to individual plants of Choerospondias axillaris. Biotropica 33(4):718–722
Clements GR, Lynam AJ, Gaveau D, Yap WL, Lhota S, Goosem M, Laurance S, Laurance WF (2014) Where and how are roads endangering mammals in Southeast Asia’s forests? PLoS One 9(12):e115376
COTE (2018) Draft Community Outreach and Conservation Awareness Strategy for Nakai Nam Theun National Protected Area 2018–2022. Prepared by the Consortium of Technical Experts (COTE) for the Nam Theun 2 Watershed Management and Protection Authority (WMPA)
Coudrat CNZ (2019) Camera-trap surveys in Nakai—Nam Theun National Park for wildlife population monitoring—Report for surveys conducted in 2018–2019. Association Anoulak, Lao PDR.
Coudrat CNZ (2020) Camera-trap surveys in Nakai—Nam Theun National Park for wildlife population monitoring—Report for surveys conducted in 2020. Association Anoulak, Lao PDR.
Coudrat CNZ (2021) The Annamite mountains—a biodiverse ecosystem at risk: a case study in Nakai-Nam Theun National Park, Lao P.D.R. Imperiled: The Encyclopedia of Conservation. Elsevier, Oxford (In press)
Coudrat CNZ and Nekaris KAI (2013) Modelling niche differentiation of co-existing, elusive and morphologically similar species: A case study of four macaque species in Nakai-Nam Theun National Protected Area, Laos. Animals 3, pp 45–62. https://www.mdpi.com/2076-2615/3/1/45
Coudrat CNZ, Nanthavong C, Sayavong S, Johnson A, Johnston JB, Robichaud WG (2014) Non-Panthera cats in Nakai-Nam Theun National Protected Area, Lao PDR. Cat News Spec 8:45–52
Duckworth JW, Robichaud W, Timmins R (2015) Rucervus schomburgki, The IUCN Red List of Threatened Species 2015: e.T4288A79818502. https://doi.org/10.2305/IUCN.UK.2015-3.RLTS.T4288A79818502.en
Espinosa S, Branch LC, Cueva R (2014) Road development and the geography of hunting by an Amazonian indigenous group: consequences for wildlife conservation. PLoS One 9(12):e114916
Estes LD, Mwangi AG, Reillo PR, Shugart HH (2011) Predictive distribution modeling with enhanced remote sensing and multiple validation techniques to support mountain bongo antelope recovery. Anim Conserv 14(5):521–532
Farris ZJ, Gerber BD, Valenta K, Rafaliarison R, Razafimahaimodison JC, Larney E, Rajaonarivelo T, Randriana Z, Wright PC, Chapman CA (2017) Threats to a rainforest carnivore community: a multi-year assessment of occupancy and co-occurrence in Madagascar. Biol Cons 210:116–124
Fiske I, Chandler R (2011) Unmarked: an R package for fitting hierarchical models of wildlife occurrence and abundance. J Stat Softw 43(10):1–23
Government of the Lao PDR (GoL) (2007) Wildlife and Aquatic Law No 07/NA. National Assembly of the Lao People’s Democratic Republic, Vientiane, Lao P.D.R.
Government of the Lao PDR (GoL) (2019) Forestry Law No 64/NA. National Assembly of the Lao People’s Democratic Republic, Vientiane, Lao P.D.R
Graham V, Geldmann J, Adams VM, Grech A, Deinet S, Chang HC (2021) Management resourcing and government transparency are key drivers of biodiversity outcomes in Southeast Asian protected areas. Biol Conserv 253:108875
Gray TN, Phan C (2011) Habitat preferences and activity patterns of the larger mammal community in Phnom Prich Wildlife Sanctuary, Cambodia. Raffles Bull Zool 59(2):311–318
Gray TNE, Hughes AC, Laurance WF, Long B, Lynam AJ, O’Kelly H, Ripple WJ, Seng T, Scotson L, Wilkinson NM (2018) The wildlife snaring crisis: an insidious and pervasive threat to biodiversity in Southeast Asia. Biodivers Conserv 27(4):1031–1037
Harrison RD (2011) Emptying the forest: hunting and the extirpation of wildlife from tropical nature reserves. BioScience 61(11):919–924. https://academic.oup.com/bioscience/article/61/11/919/223755
Harrison RD, Sreekar R, Brodie JF, Brook S, Luskin M, O’Kelly H, Rao M, Scheffers B, Velho N (2016) Impacts of hunting on tropical forests in Southeast Asia. Conserv Biol 30(5):972–981
Hijmans RJ (2019) raster: Geographic data analysis and modeling. R package version 2.8-19
Jerozolimski A, Peres CA (2003) Bringing home the biggest bacon: a cross-site analysis of the structure of hunter-kill profiles in Neotropical forests. Biol Conserv 111:415–425
Koerner SE, Poulsen JR, Blanchard EJ, Okouyi J, Clark CJ (2017) Vertebrate community composition and diversity declines along a defaunation gradient radiating from rural villages in Gabon. J Appl Ecol 54(3):805–814
Liwei T, Zhensheng L, Yan-Ling S, Zhigao Z (2004) Forage and bed sites characteristics of Indian muntjac (Muntiacus muntjak) in Hainan Island, China. Ecol Res 19(6):675–681
Long B (2005) Identification of priority areas for integrated conservation management in Quang Nam province, Vietnam. (Doctoral dissertation). University of Kent, UK
MacKenzie DI, Nichols JD, Lachman GB, Droege S, Andrew Royle J, Langtimm CA (2002) Estimating site occupancy rates when detection probabilities are less than one. Ecology 83(8):2248–2255
McConkey KR, Nathalang A, Brockelman WY, Saralamba C, Santon J, Matmoon U, Somnuk R, Srinoppawan K (2018) Different megafauna vary in their seed dispersal effectiveness of the megafaunal fruit Platymitra macrocarpa (Annonaceae). PLoS One 13(7):e0198960
McCullough DR, Pei KC, Wang Y (2000) Home range, activity patterns, and habitat relations of Reeves' muntjacs in Taiwan. J Wildl Manag 64(2):430–441
Mohd-Azlan J, Nurul-Asna H, Jailan TS, Tuen AA, Engkamat L, Abdillah DN, Zainudin R, Brodie JF (2018) Camera trapping of terrestrial animals in Tanjung Datu National Park, Sarawak, Borneo. Raffles Bull Zool 66:587–594
Niedballa J, Sollmann R, Courtiol A, Wilting A (2016) camtrapR: an R package for efficient camera trap data management. Methods Ecol Evol 7(12):1457–1462
O’Kelly H, Bousa A, Coudrat CNZ, Khiewvongphachan X, Vongkhamheng C (2018a) Law Enforcement Strategy for Nakai-Nam Theun National Protected Area. Prepared by the Consortium of Technical Experts (COTE) for the Nam Theun 2 Watershed Management and Protection Authority (WMPA)
O’Kelly H, Coudrat CNZ and Vongkhamheng C (2018b) Biological Monitoring Strategy for Nakai-Nam Theun National Protected Area. Prepared by the Consortium of Technical Experts (COTE) for the Nam Theun 2 Watershed Management and Protection Authority (WMPA)
QGIS Development Team (2020) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. https://wwwr-project.org/
Rasphone A, Kéry M, Kamler JF, Macdonald DW (2019) Documenting the demise of tiger and leopard, and the status of other carnivores and prey, in Lao PDR’s most prized protected area: Nam et-Phou louey. Glob Ecol Conserv. https://doi.org/10.1016/j.gecco.2019.e00766
Sollmann R, Mohamed A, Kelly MJ (2013) Camera trapping for the study and conservation of tropical carnivores. Raffles Bull Zool 28:21–42
Sollmann R, Mohamed A, Niedballa J, Bender J, Ambu L, Lagan P, Mannan S, Ong RC, Langner A, Gardner B, Wilting A (2017) Quantifying mammal biodiversity co-benefits in certified tropical forests. Divers Distrib 23(3):317–328
Symes WS, Edwards DP, Miettinen J, Rheindt FE, Carrasco LR (2018) Combined impacts of deforestation and wildlife trade on tropical biodiversity are severely underestimated. Nat Commun 9(1):1–9
Tilker A, Abrams JF, Nguyen A, Hörig L, Axtner J, Louvrier J, Rawson BM, Nguyen HAQ, Guegan F, Nguyen TV, Le M (2020a) Identifying conservation priorities in a defaunated tropical biodiversity hotspot. Divers Distrib 26(4):426–440
Tilker A, Nguyen A, Abrams JF, Bhagwat T, Le M, Van Nguyen T, Nguyen AT, Niedballa J, Sollmann R, Wilting A (2020b) A little-known endemic caught in the South-east Asian extinction crisis: the Annamite striped rabbit Nesolagus timminsi. Oryx 54(2):178–187
Timmins RJ, Duckworth JW (2013) Distribution and habitat of Assamese macaque Macaca assamensis in Lao PDR, including its use of low-altitude karsts. Primate Conserv 26(1):103–114
Timmins RJ, Duckworth JW (2016a) Muntiacus rooseveltorum, The IUCN Red List of Threatened Species 2016a: e.T13928A2. https://doi.org/10.2305/IUCN.UK.2016a-1.RLTS.T13928A22160435.en. Accessed 5 Oct 2020
Timmins RJ and Duckworth JW (2016b) Muntiacus truongsonensis, The IUCN Red List of threatened species 2016b: e.T44704A2. https://doi.org/10.2305/IUCN.UK.2016b-1.RLTS.T44704A22154056.en. Accessed 5 Oct 2020
Timmins RJ, Evans TD (1996) Wildlife and Habitat Survey of the Nakai-Nam Theun National Biodiversity Conservation Area. Wildlife Conservation Society, Vientiane, p 59
Timmins RJ, Evans TD, Khounboline K, Sisomphone C (1998) Status and conservation of the giant muntjac Megamuntiacus vuquangensis, and notes on other muntjac species in Laos. Oryx 32(1):59–67
Timmins RJ, Duckworth JW, Robichaud W, Long B, Gray TNE, Tilker A (2016a) Muntiacus vuquangensis. The IUCN Red List of Threatened Species 2016a: e.T44703A22153828. https://doi.org/10.2305/IUCN.UK.2016a.2.RLTS.T44703A22153828.en. Downloaded on 13 January 2021
Timmins RJ, Steinmetz R, Samba Kumar N, Anwarul IM, Sagar BH (2016b) Muntiacus vaginalis. The IUCN Red List of Threatened Species 2016b: e.T136551A22165292. https://doi.org/10.2305/IUCN.UK.2016b.1.RLTS.T136551A22165292.en. Downloaded on 13 January 2021
Timmins RJ, Hedges S and Robichaud W (2020) Pseudoryx nghetinhensis (amended version of 2016 assessment) The IUCN Red List of Threatened Species 2020: e.T18597A166485696. https://doi.org/10.2305/IUCN.UK.2020.1.RLTS.T18597A166485696.en
Tobler MW, Carrillo-Percastegui SE, Pitman RL, Mares R, Powell G (2008) An evaluation of camera traps for inventorying large-and medium-sized terrestrial rainforest mammals. Anim Conserv 11(3):169–178
Tuanmu M, Jetz W (2015) A global, remote sensing-based characterization of terrestrial habitat heterogeneity for biodiversity and ecosystem modelling. Glob Ecol Biogeogr 24(11):1329–1339
van Etten J (2017) R package gdistance: distances and routes on geographical grids. J Stat Softw 76(13). https://www.jstatsoft.org/article/view/v076i13
Viollaz J, Long B, Trung CT, Kempinski J, Rawson BM, Quang HX, Hiền NN, Liên NTB, Dũng CT, Huyền HT, McWhirter R, Gore ML (2021) Using crime script analysis to understand wildlife poaching in Vietnam. Ambio 50(7):1378–1393
Acknowledgements
This project was conducted in collaboration with the Nam Theun 2 Watershed Management and Protection Authority (NT2 WMPA) governed under the Ministry of Agriculture and Forestry of the Lao P.D.R. As part of a Memorandum of Understanding between Association Anoulak and WMPA, Association Anoulak provides technical advice to the national park to implement a long-term systematic wildlife monitoring program. We are specifically grateful to the director of the national park Mr. Savanh Chanthakhoumman for endorsing our work. The team leaders involved in the field implementation in 2018–2019 were Mr. Chanthalaphone, Mr. Khammay, Mr. Khanthaly, Mr. Souliyo, Mr. Vilakhone, Mr. Ounchai, Mr. Soulixai, Mr. Chanla, Mr. Thongla, Mr. Chamsy, Mr. Phouxay. Villagers from numerous villages inside the national park participated in the field work as porters or field assistants; we are grateful for their dedicated work and assistance throughout the project. The project was funded by Association Anoulak (via grants received from the National Geographic Society grant WW-040C-17, The Mohamed bin Zayed Species Conservation Fund project number 172516260, and Beauval Nature) and by the national park authority. We would like to thank Anirban Mukhopadhyay and Jan Axtner for providing technical assistance in the data management process. We thank Wai-Ming Wong for assisting in the procurement of the Panthera cameras used as part of this project.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was funded by Association Anoulak (via grants received from the National Geographic Society Grant WW-040C-17, The Mohamed bin Zayed Species Conservation Fund project number 172516260, and Beauval Nature) and by the national park authority. The publication of this article was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – project number 491292795.
Author information
Authors and Affiliations
Contributions
Formal analysis: IA, CNZC. Visualization: IA, JFA. Writing—original draft: IA, AT. Writing—review and editing: JFA, CNZC, AN, AW, AT, JN. Conceptualization: CNZC, AW, AT. Funding acquisition: CNZC. Investigation: CNZC, CN, AN. Methodology: CNZC, AW. Project administration: CNZC, AW. Resources: CNZC, AW. Supervision: CNZC, AW.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Αll authors have provided their consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling editor: Sabine Begall.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alexiou, I., Abrams, J.F., Coudrat, C.N.Z. et al. Camera-trapping reveals new insights in the ecology of three sympatric muntjacs in an overhunted biodiversity hotspot. Mamm Biol 102, 489–500 (2022). https://doi.org/10.1007/s42991-022-00248-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-022-00248-0