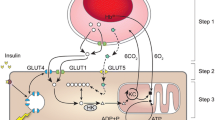
Abstract
A tightly regulated glucose homeostasis is essential for nectar-feeding bats, to manage and avoid hyperglycemia. The role of insulin and the contribution of glycogen reserves during rest periods and the extent of its involvement in blood glucose homeostasis during exercise have not been described yet and was the main objective of this study. Bats (Anoura caudifer) were assigned to the following groups: (1) Water and Resting (WR): received water (0.25 mL) and rested for zero, 45 or 90 min; (2) Glucose and Resting (GR): fed with a glucose solution (5.4 g/Kbw) and rested for 45 or 90 min; (3) Glucose and Exercise (GE): fed with a glucose solution (5.4 g/Kbw) and were induced to fly for a total of 45 or 90 min. Oxidative stress and metabolic markers were analyzed in all groups. We also describe morphometric and stereological parameters in pancreas from WR bats. Blood glucose concentrations were increased 45 min after glucose intake in resting bats compared to bats forced to fly and also compared to resting bats which received no glucose. Blood glucose levels after 90 min were the same in all bats, returning to baseline levels in GR bats. Plasma insulin concentrations and liver and muscle glycogen content did not show any differences among groups. Our results provide evidence that insulin secretion seems to play a role on glucose regulation when bats are at rest and that the increased nitric oxide production in the kidneys found in GE90 may contribute to renal protection during exercise.






Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Ayala-Berdon J, Rodríguez-Peña N, Villaseñor-Orduna M, Stoner KE, Kelm DH, Schondube JE (2011) Foraging behavior adjustments related to changes in nectar sugar concentration in phyllostomid bats. Comp Biochem Physiol Part A Mol Integr Physiol 160:143–148. https://doi.org/10.1016/j.cbpa.2011.05.030
Barros MA, Rui AM, Fabian ME (2013) Seasonal variation in the diet of the bat Anoura caudifer (Phyllostomidae: Glossophaginae) at the southern limit of its geographic range. Acta Chiropterologica 15(1):77–84
Bock T, Pakkenberg B, Buschard K (2005) Genetic background determines the size and structure of the endocrine pancreas. Diabetes 54:133–137. https://doi.org/10.2337/diabetes.54.1.133
Bonner-Weir S, Deery D, Leahy JL, Weir GC (1989) Compensatory growth of pancreatic p-cells in adult rats after short-term glucose infusion. Diabetes 3:49–53. https://doi.org/10.2337/diab.38.1.49
Boulé NG, Weisnagel SJ, Lakka TA, Tremblay A, Bergman RN, Rankinen T, Leon AS, Skinner JS, Wilmore JH, Rao DC, Bouchard C (2005) Effects of exercise training on glucose homeostasis. Diabetes Care 28:108–114. https://doi.org/10.2337/diacare.28.1.108
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310. https://doi.org/10.1016/S0076-6879(78)52032-6
Cerf ME (2013) Beta cell dysfunction and insulin resistance. Front Endocrinol 4:1–12. https://doi.org/10.3389/fendo.2013.00037
Costantini D, Lindecke O, Pētersons G, Voigt CC (2019) Migratory flight imposes oxidative stress in bats. Current Zoology 65(2):147–153
Díaz MM, Solari S, Aguirre LF, Aguiar LMS, Barquez RM (2016) Clave de identificación de los murciélagos de Sudamerica. Primeira Edição. Programa de Conservación de los Murciélagos de Argentina. Tucumán
Dieterich S, Bieligk U, Beulich K, Hasenfuss G, Prestle J (2000) Gene expression of antioxidative enzymes in the human heart: increased expression of catalase in the end-stage failing heart. Circulation 101:33–39. https://doi.org/10.1161/01.CIR.101.1.33
Etgen GJ, Memon AR, Thompson GA, Ivy JL (1993) Insulin- and contraction-stimulated translocation of GTP-binding proteins and GLUT4 protein in skeletal muscle. J Biol Chem 268:20164–20169. https://doi.org/10.1016/S0021-9258(20)80708-X
Freitas MB, Queiroz JF, Gomes CID, Collares-Buzato CB, Barbosa HC, Boschero AC, Gonçalves CA, Pinheiro EC (2013) Reduced insulin secretion and glucose intolerance are involved in the fasting susceptibility of common vampire bats. Gen Comp Endocrinol 183:1–6. https://doi.org/10.1016/j.ygcen.2012.11.023
Garg A, Grundy SM, Koffler M (1992) Effect of high carbohydrate intake on hyperglycemia, islet function, and plasma lipoproteins in NIDDM. Diabetes Care 15:1572–1580. https://doi.org/10.2337/diacare.15.11.1572
Gonzalez-Terrazas TP, Medellin RA, Knörnschild M, Tschapka M (2012) Morphological specialization influences nectar extraction efficiency of sympatric nectar-feeding bats. J Exp Biol 215(22):3989–3996
Griess P (1879) Bemerkungen zu der Abhandlung der HH. Weselsky und Benedikt “Ueber einige Azoverbindungen.” Chem Ber 12:426–428
Gundersen G, Bendtsen TF, Korbo L, Marcussen N, Moller A, Nyengaard JR, Pakkenberg B, Sorensen FB, Vesterby A (1988) Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. APMIS 96:379–394. https://doi.org/10.1111/j.1699-0463.1988.tb05320.x
Gutiérrez-Guerrero YT, Ibarra-Laclette E, Martínez del Río C, Barrera-Redondo J, Rebollar EA, Ortega J et al (2020) Genomic consequences of dietary diversification and parallel evolution due to nectarivory in leaf-nosed bats. GigaScience 9(6):giaa059
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases: the first enzymatic step in mercapturic and formation. J Biol Chem 249:7130–7140
Helversen OV, Reyer HU (1984) Nectar intake and energy expenditure in a flower visiting bat. Oecologia 63:178–184
Hernandez A, Martinez C (1992) Intestinal disaccharides in five species of phyllostomoid bats. Comp Biochem Physiol B Biochem Mol Biol 103:105–111
Ito D, Cao P, Kakihana T, Sato E, Suda C, Muroya Y, Ogawa Y, Hu G, Ishii T, Ito O, Kohzuki M, Kiyomoto H (2015) Chronic running exercise alleviates early progression of nephropathy with upregulation of nitric oxide synthases and suppression of glycation in Zucker diabetic rats. PLoS ONE 10:1–21. https://doi.org/10.1371/journal.pone.0138037
Jensen TE, Richter EA (2012) Regulation of glucose and glycogen metabolism during and after exercise. J Physiol (lond) 590:1069–1076. https://doi.org/10.1113/jphysiol.2011.224972
Jensen TE, Sylow L, Rose AJ, Madsen AB, Angin Y, Maarbjerg SJ, Richter EA (2014) Contraction-stimulated glucose transport in muscle is controlled by AMPK and mechanical stress but not sarcoplasmatic reticulum Ca2+ release. Mol Metab 3(7):742–753
Kawahito S, Kitahata H, Oshita S (2009) Problems associated with glucose toxicity: role of hyperglycemia-induced oxidative stress. World J Gastroenterol 15:4137–4142. https://doi.org/10.3748/wjg.15.4137
Keegan DJ (1977) Aspects of the assimilation of sugars by Rousettus aegyptiacus. Comp Biochem Physiol 58a:349–352
Kellogg DL, McCammon KM, Hinchee-Rodriguez KS, Adamo ML, Roman LJ (2017) Neuronal nitric oxide synthase mediates insulin- and oxidative stress-induced glucose uptake in skeletal muscle myotubes. Free Radic Biol Med 110:261–269. https://doi.org/10.1016/j.freeradbiomed.2017.06.018
Kelm DH, Simon R, Kuhlow D, Voigt CC, Ristow M (2011) High activity enables life on a high-sugar diet: blood glucose regulation in nectar-feeding bats. Proc R Soc B 278:3490–3496. https://doi.org/10.1098/rspb.2011.0465
Király MA, Campbell J, Park E, Bates HE, Yue JTY, Rao V, Matthews SG, Bikopoulos G, Rozakis-Adcock M, Giacca A, Vranic M, Riddell MC (2009) Exercise maintains euglycemia in association with decreased activation of c-Jun NH 2 -terminal kinase and serine phosphorylation of IRS-1 in the liver of ZDF rats. Am J Physiol Endocrinol Metab 298:E671–E682. https://doi.org/10.1152/ajpendo.90575.2008
Lemieux I, Pascot A, Couillard C, Lamarche B, Tchernof A, Alméras N, Bergeron J, Gaudet D, Tremblay G, Prud’homme D, Nadeau A, Després JP (2000) Hypertriglyceridemic Waist: A marker of the atherogenic metabolic triad (Hyperinsulinemia; Hyperapolipoprotein B; Small, Dense LDL) in men? Circulation 102:179–184
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement whit the folin phenol reagent. J Biol Chem 193:265–275
Machado-Santos C, Aquino JCF, Mikalauka JS, Abidu-Figueiredo M, Mendes RMM, Sales A (2013) What difference exists in the pancreas of mammals with sanguivorous diet? A morphological, stereological and immunohistochemical study of the pancreatic islets of the hematophagous bat Diphylla ecaudata. Regul Pept 183:62–68. https://doi.org/10.1016/j.regpep.2013.03.015
Machado-Santos C, Aquino JCF, Rocha PA, Abidu-Figueiredo M, Brito-Gitirana L, Sales A (2017) Influence of feeding habits in the endocrine pancreas of insectivore bat Pteronotus personatus and nectarivore bat Anoura geoffroyi: a comparative stereological and immunohistochemical study. Tissue Cell 49:1–7. https://doi.org/10.1016/j.tice.2017.01.001
Mandarim-De-Lacerda CA (2003) Stereological tools in biomedical research. An Acad Bras Ciênc 75:469–486. https://doi.org/10.1590/S0001-37652003000400006
McConell GK, Rattigan S, Lee-Young RS, Wadley GD, Merry TL (2012) Skeletal muscle nitric oxide signaling and exercise: a focus on glucose metabolism. Am J Physiol Endocrinol Metab 303:E301–E307. https://doi.org/10.1152/ajpendo.00667.2011
Munshi-South J, Wilkinson GS (2010) Bats and birds: exceptional longevity despite high metabolic rates. Ageing Res Rev 9:12–19
Musi N, Fujii N, Hirshman MF, Ekberg I, Fröberg S, Ljungqvist O, Thorell A, Goodyear LJ (2001) AMP-activated protein kinase (AMPK) is activated in muscle of subjects with type 2 diabetes during exercise. Diabetes 50:921–927. https://doi.org/10.2337/diabetes.50.5.921
Oprea M, Aguliar L, Wilson DE (2009) Anoura caudifer (Chiroptera: Phyllostomidae). Mamm Species 844:1–8
Peng X, He X, Liu Q, Sun Y, Liu H, Zhang Q, Liang J, Peng Z, Liu Z, Zhang L (2017) Flight is the key to postprandial blood glucose balance in the fruit bats Eonycteris spelaea and Cynopterus sphinx. Ecol Evol 47:8804–8811. https://doi.org/10.1002/ece3.3416
Pereira Freitas RM, Oliveira JM, Justinico Castro DL, Matias Sarandy M, Gonçalves RV, Freitas MB (2019) The antioxidant status of three Neotropical bat species with different feeding habits. Acta Chiropterologica 21(2):395–402
Polakof S, Mommsen TP, Soengas JL (2011) Glucosensing and glucose homeostasis: from fish to mammals. Comp Biochem Physiol B Biochem Mol Biol 160:123–149. https://doi.org/10.1016/j.cbpb.2011.07.006
Protzek AOP, Rafacho A, Viscelli BA, Bosqueiro JR, Cappelli AP, Paula FMM, Boschero AC, Pinheiro EC (2010) Insulin and glucose sensitivity, insulin secretion and β-cell distribution in endocrine pancreas of the fruit bat Artibeus lituratus. Comp Biochem Physiol Part A Mol Integr Physiol 157:142–148. https://doi.org/10.1016/j.cbpa.2010.05.016
Röder PV, Wu B, Liu W, Han W (2016) Pancreatic regulation of glucose homeostasis. Exp Mol Med 48:219
Rodriguez-Peña N, Price ER, Caviedes-Vidal E, Flores-Ortiz CM, Karasov WH (2016) Intestinal paracellular absorption is necessary to support the sugar oxidation cascade in nectarivorous bats. J Exp Biol 219:779–782. https://doi.org/10.1242/jeb.133462
Rose AJ, Richter EA (2005) Skeletal muscle glucose uptake during exercise: how is it regulated? Physiology 20:260–270
Simmons NB (2005) Order chiroptera. Mammal Species World Taxon Geogr Ref 1:312–529
Sjögren B, Nordenskjöld T, Holmgren H, Möllerström J (1938) Beitrag zur Kenntnis der Leberrhythmik. Pflugers Arch Gesamte Physiol Menschen Tiere 240:427–448
Suarez RK, Welch KC (2017) Sugar metabolism in hummingbirds and nectar bats. Nutrients 9:1–16. https://doi.org/10.3390/nu9070743
Suarez RK, Herrera LGM, Welch KC (2011) The sugar oxidation cascade: aerial refueling in hummingbirds and nectar bats. J Exp Biol 214:172–178. https://doi.org/10.1242/jeb.047936
Thomas SP (1975) Metabolism during flight in two species of bats, Phyllostomus hastatus and Pteropus gouldii. J Exp Biol 63(1):273–293
Voigt CC, Speakman JR (2007) Nectar-feeding bats fuel their high metabolism directly with exogenous carbohydrates. Funct Ecol 21:913–921. https://doi.org/10.1111/j.1365-2435.2007.01321.x
Wachtman LM, Kramer JA, Miller AD, Hachey AM, Curran EH, Mansfield KG (2011) Differential contribution of dietary fat and monosaccharide to metabolic syndrome in the common marmoset (Callithrix jacchus). Obesity 19:1145–1156. https://doi.org/10.1038/oby.2010.303
Welch KC Jr, Herrera MLG (2008) Dietary sugar as a direct fuel for flight in the nectarivorous bat Glossophaga soricina. J Exp Biol 211:310–316. https://doi.org/10.1242/jeb.012252
Yacoe ME, Cummings JW, Myers P, Creighton GK (1982) Muscle enzyme profile, diet, and flight in South American bats. Am J Physiol Regul Integr Comp Physiol 242(3):R189–R194
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethics approval
All procedures involving animals were approved by the University Animal Care and Use Committee (88/2016) and all captures were approved by the Biodiversity Authorization and Information System (SISBIO, Brazil) (56105-1).
Consent for participate
Not applicable.
Consent for publication
All the authors of this manuscript accepted that the paper is submitted for publication in the Mammalian biology journal, and report that this paper has not been published or accepted for publication in another journal, and it is not under consideration at another journal.
Additional information
Handling editor: Yoshiyuki Henning.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Castro, D.L.J., Freitas, R.M.P., Silva, S.B. et al. Insulin and glucose regulation at rest and during flight in a Neotropical nectar-feeding bat. Mamm Biol 101, 987–996 (2021). https://doi.org/10.1007/s42991-021-00146-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-021-00146-x