Abstract
Aim
The study of islands biotas is fundamental to understand biodiversity patterns and process, both on evolutionary and ecological time scales. This study explores the influence of biogeographic and climatic factors on island species–area relationships (ISARs) in volcanic and continental islands, aiming to detect differences in slopes and intercepts between these island origins.
Methods
Data from 5049 vegetation plots on 58 Central Mediterranean Sea islands were collected from various sources. Islands were categorized as volcanic or continental based on their geological origin. area, isolation, maximum elevation, eccentricity, mean annual temperature and annual precipitation were calculated for each island. By using a moving window, we created groups of islands, and ISARs were fitted to each group using the Arrhenius power functions. Linear models and a permutation test were employed to examine how ISAR model parameters varied with the above-mentioned variables.
Results
While ISAR intercept values did not differ between island origins, volcanic islands showed higher ISAR slopes than continental islands. Whereas increasing island isolation and mean annual temperature increased ISAR intercepts on continental islands groups, it decreased on volcanic ones. Increasing annual precipitation decreased ISAR intercept on continental islands groups and increased on volcanic islands groups. Increasing island isolation, eccentricity and mean annual temperature increased the ISAR slope on volcanic islands groups while increasing annual precipitation decreased it. Increasing maximum elevation increased ISAR slope on continental islands groups.
Main conclusions
Our study provides evidence about the importance of island geological origin in determining the observed ISAR patterns. Biogeographic and climatic factors are pivotal in shaping species richness patterns on islands, exerting varying influences on both volcanic and continental islands.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Islands are a natural test bed for studying long-term evolutionary processes and formulating ecological theories. As a matter of fact, they constitute unique natural experiments where ecological and evolutionary processes are bounded by clear physical limits, constraining local biotic assemblages (Whittaker et al., 2017). Moreover, islands exhibit a large variability in terms of ecologically and evolutionary relevant features such as geological origin, geographic setting, biogeographic history and isolation through time. Such features have endowed biogeographers, evolutionary biologists and ecologists of a strong attraction and interest for island systems (Fernández-Palacios et al., 2015).
One of the best known patterns in island ecology and biogeography is the island species–area relationship (ISAR hereafter; Rosenzweig, 1995; Whittaker and Fernandez-Palacios 2007), which describes the nonlinear increase in species richness with increasing area. Since its formulation by Watson in 1835, it has been widely used to develop and support ecological and biogeographical investigations (Chiarucci et al., 2017; Lomolino, 2000; MacArthur & Wilson, 1967; Preston, 1962; Rosenzweig, 1995; Triantis et al., 2008, 2010). The most widely adopted model to describe the ISAR is the Arrhenius power function (\(S = c \cdot A^{2}\)), with easily understandable meaning and interpretable parameters. The interpretation of ISAR parameters and predictions of their patterns started with Preston (1962), who estimated a canonical value of 0.262 for the z parameter, often reported as slope. He predicted the slope value for true islands to be from 0.2 to 0.4 and that less isolated areas could result in values lower than 0.2. Subsequently, MacArthur and Wilson (1967) complemented Preston’s prediction and formulated the equilibrium theory of island biogeography (ETIB), according to which patterns of island species richness could be predicted based on island area and isolation.
Following the ETIB, larger islands are normally characterized by a greater resource base, reducing extinction rates and supporting more species (i.e., higher ISAR intercept) than smaller islands. Conversely, more isolated islands are harder to reach, leading to lower immigration rates and fewer species (i.e., lower ISAR intercept) than less isolated islands. As for ISAR slopes, MacArthur and Wilson (1967) highlighted that values may vary depending on the degree of isolation of smaller islands. When the ISAR is estimated for less isolated islands, the smaller islands of these sets are influenced by higher immigration rates and higher number of transient species that, in turn, inflate their species richness and may lead to flatter slopes. Conversely, in more isolated islands, the immigration rates approach zero, leading to overall lower species richness, hence a steeper slope (Patiño et al., 2014; Rosenzweig, 1995).
The previous studies analyzing ISAR parameters suggest that island isolation is one of the most important factors explaining species richness patterns and is tightly connected to island origin. Indeed, volcanic islands are isolated since their emergence and generally show steeper ISAR slopes and lower ISAR intercepts than continental islands (Kreft et al., 2007; Martin, 1981; Matthews et al., 2015; Patiño et al., 2014; Rosenzweig, 1995; Triantis et al., 2012). Although this is a widely recognized pattern previous work showed contradictory results (e.g., Chiarucci et al., 2021b; Connor & McCoy, 1979; Drakare et al., 2005), and empirical evidence on how ISAR parameters may vary as a function of other biogeographic and ecological factors (e.g., island altitude, island shape, temperature and precipitation) is still scarce. Furthermore, most of the existing literature focuses on animal species (e.g., Rahbek, 1997 and Kalmar & Currie, 2006, for birds; Fattorini et al., 2016, for land snails, isopods, centipedes, tenebrionids and reptiles; Ohyama et al., 2021, for ants and Franzén et al., 2012, for butterflies and moths), with fewer studies analyzing ISAR patterns for plants. In this context, it has been shown that the ISAR slopes are also influenced by climate, with more temperate islands exhibiting higher slopes than colder islands (Chown et al., 1998), while wetter islands showed lower slopes and higher intercepts than drier islands (Iliadou et al., 2014). Nevertheless, to our knowledge, other biogeographically relevant factors such as altitude and island shape were not accounted for when analyzing ISAR of plant species richness (but see Moraldi et al. 2020 for an analysis of nested SARs), nor was the interaction between such factors and island origin. Given the correlation between island area and elevation (Weigelt et al., 2013), islands with higher maximum elevation may exhibit higher ISAR intercepts and lower slopes. As for island shape, rounder islands may exhibit higher ISAR intercepts and lower slopes than more elongated islands, because of the increased core-to-edge ratio in the former. Overall, contrasted patterns between volcanic and continental ISAR as a function of environmental variables are to be expected.
ISARs are also dependent on the sampling scheme used to collect species occurrences (Scheiner, 2003, 2011; Dengler, 2009) and by the adopted fitting approach. It is usually assumed that the figures used for fitting ISARs are based on lists of species which are complete for each area, but this is not always true due to undersampling or undetected rare species (Daru et al., 2018) or due to other factors that trigger different floristic richness across different authors (Bagella et al., 2020). The survey intensity, in terms of number of observed individuals or sampling units, determines the completeness of the list of species detected on an island, and several studies investigated the role of sampling design and scale on diversity estimates on islands (e.g., Chase et al., 2019a, 2019b; Gillespie et al., 2013; Sfenthourakis & Panitsa, 2012). Recently, Chiarucci et al. (2021a) showed that large vegetation databases represent a reliable source of information to estimate island plant species richness and fit ISARs, even if the resulting parameters are not identical to those obtained using floristic lists. Similarly, vegetation plot data have been used to identify island hotspots and coldspots, as well as information about species rarity (Testolin et al., 2023).
In this study, we focus on island species–area relationship curves (ISARs; type IV curves sensu Scheiner, 2003) applying the framework by Chiarucci et al. (2021a) and fitting the log–log Arrhenius’ power function. We analyzed the ISAR patterns based on plant occurrence data obtained by vegetation plots sampled in 27 islands of volcanic origin and 31 continental islands located in the Central Mediterranean Sea, with the aim to uncover biogeographical patterns related to island features, namely, island area, geographic isolation, elevation, island shape, precipitation, temperature and geological origin. Specifically, our research aims were: 1) to explore differences on ISAR slopes and intercepts between volcanic versus continental islands and 2) to detect ISAR slopes and intercepts changes along biogeographic and climatic gradients on volcanic versus continental islands.
Methods
Study area
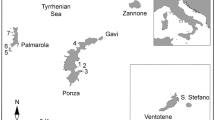
The Mediterranean Sea covers an approximate area of 2.51 × 106 km2; it is enclosed between the African and Eurasian continents and connected to the Atlantic Ocean across the strait of Gibraltar. A large number of islands and archipelagos with outstanding biodiversity and endemism are present in the basin, with the exact number of islands and islets ranging between 5000 (Vogiatzakis et al., 2016) and 10,000 (Médail, 2022; Sfenthourakis & Triantis, 2017), depending on definitions and area thresholds. The MEDIS geodatabase reported that the islands larger than 10,000 m2 in the Mediterranean basin are 2214 (Santi et al., 2024). Yet, only 157 of these islands are larger than 10 km2 (Médail, 2017). The basin experienced long-term anthropogenic changes started by ancient civilizations, triggering ecosystem transformation, biotic homogenization and biodiversity loss (Médail, 2017). From the conservation perspective, this area has been considered one of the most important global hotspots of biodiversity (Médail, 2022; Médail & Quézel, 1997; Myers et al., 2000). Our study system consists of a set of 58 islands located in the Central Mediterranean Sea, around peninsular Italy but not exclusively Italian (Fig. 1).
Map of the study area. The 58 islands included in the analyses are represented by progressive numbering. Islands represent 12 archipelagos and six solitary islands: Spezzino Archipelago (1 and 2), Tuscan Archipelago (3–10), Pontine Archipelago (11–16), Campanian Archipelago (17–19), Li Galli Islands (20 and 21), Isola di Cirella (22), Dino Island (23), Aeolian Archipelago (24–33), Ustica Island (34), Isola delle Femmine (35), Egadi Islands (36–38), Pantelleria Island (39), Pelagie Islands (40 and 42), Maltese Archipelago (43–46), Tremiti Islands (47–51), Venice Archipelago (52–54), Isola di Capo Passero (55) and Cyclops Archipelago (56–58). Detailed island features are given in Appendix S1.1
Data assembly
We obtained island species lists based on the plant occurrence data retrieved from vegetation plot data stored in the database AMS VegBank (Alessi et al., 2022) which contains published and unpublished vegetation surveys dataset (list of data sources in Appendix S2). The plots were sampled between 1950 and 2022, mostly for descriptive purposes, using opportunistically located units, the Braun–Blanquet cover scale and heterogeneous grain sizes (from less than 1 to 900 m2). Plot data have been recorded over a quite long time period, introducing some possible bias. However, this is the only possible way to obtain list of species for large areas lacking dedicated intense surveys, and it is normally adopted in biogeographical studies (e.g., Cao Pinna et al., 2021; Testolin et al., 2021). Taxa were standardized at species level, according to the Global Biodiversity Information Facility (GBIF) taxonomic backbone by means of the “taxadb” package (Norman et al., 2020).
Data analyses
Islands species richness estimation
For each island, we estimated the total number of vascular plant species using the Chao2 estimator on the basis of plot data. Such estimator derives from the Cauchy–Schwarz inequality, which has been applied to obtain a non-parametric estimation of species richness of an assemblage based on species incidence (detection/non-detection) data in multiple sampling units. The results are nearly unbiased when very rare/infrequent species have approximately the same detection probabilities (Chao & Colwell, 2017). As most of the other nonparametric estimators of species richness, Chao2 underestimates the total number of species in real communities and can be considered as a lower bound estimator (see, e.g., Xu et al., 2012; Chiarucci et al., 2018). The Chao2 estimator was calculated using the iNext package (version 3.0.0) in R (Hsieh et al., 2022).
Island features
For each island, we obtained data about surface area, geographical isolation, geological origin, maximum elevation, mean annual temperature, annual precipitation and shape eccentricity. The latter, calculated as the ratio between the longest and the shortest sides of the smallest rectangle enclosing the island, is a measure of island shape. Geographic isolation was calculated as the minimum distance to the mainland and maximum elevation as the elevation of the highest point of the island. Mean annual temperature and annual precipitation were derived from CHELSA 2.1 (Karger et al., 2017, 2018) as the mean of the values extracted within each island polygon. The geological origin was used to divide the total data (n = 58 islands) into volcanic (n = 27) and continental (n = 31) islands.
Variables with a skewed distribution were transformed as follows: Island area, species richness and maximum elevation were log10 transformed; geographic isolation and precipitation were root squared transformed; eccentricity was transformed to its inverse and mean annual temperature was not transformed. We then analyzed the differences between volcanic and continental islands as follows: For area, geographical isolation, eccentricity and annual precipitation, we employed two-sample t-tests, whereas for mean annual temperature and maximum elevation, we used two-sample Wilcoxon rank-sum tests due to the violation of normality assumptions.
Fitting island species–area relationship
We calculated ISARs separately for volcanic and continental islands by fitting the Arrhenius power function in an log–log transformed space:
where “S” is species richness, “A” island area and “c” and “z” are two fitted parameters.
Then, we investigated how the c (intercept) and z (slope) parameters of the fitted models vary as a function of the above-mentioned variables by performing an iterative analysis (moving window) to highlight the variation of ISAR parameters with increasing biogeographic and climatic gradients. For this purpose, we systematically analyzed two datasets, defined by the geological origin of the islands (continental, n = 31; and volcanic, n = 27). The moving window approach involved the arrangement of the islands in ascending order based on the target variable (e.g., area and island isolation). At each step, a group of nine islands was selected, representing those with the minimum values of the biogeographic or climatic variable under consideration at that specific point in the process. Subsequently, we calculated the mean of the target variable and fitted an ISAR model to this island group. In the following step, another set of nine islands is chosen. This selection excludes the first two islands from the previous iteration and introduces two new islands. We obtained 10 and 12 ISAR models fitted for volcanic and continental islands, respectively, as a function of each biogeographic and climatic variable. To our first aim, we performed a permutation test due the dependence of the sample. In this context, we evidenced whether slopes and intercept were significantly different between continental and volcanic islands. To our second aim, we fitted simple linear models to the resulting ISAR intercepts and slopes as a function of each biogeographic and climatic variable to evidence significant tendencies. All of our analyses were performed using R (R Core Team, 2022).
Results
Island features and plant species richness
The 58 islands included in the study have different geologic origins, 27 of them being volcanic and 31 being continental or land-bridge islands. Details about each island are given in Supporting Information Appendix S1.1. Cavallino-Treporti is the northernmost island (45.46° N latitude, 54 in Fig. 1) whereas Isola dei Conigli is the southernmost island (35.51° N latitude, 42 in Fig. 1). The island located nearest to the mainland is Monte Argentario (connected to the mainland by two strips of sand, so 0 km away; 9 in Fig. 1), whereas the farthest island from the continent is Linosa Island (162 and 173 km away from Tunisia and Sicily, respectively; 40 in Fig. 1). Malta is the largest island with 245.8 km2 (45 in Fig. 1), whereas Faraglione di Mezzo (56 in Fig. 1; Cyclops Archipelago) is the smallest with just 0.0004 km2. Despite a huge range of variation for all the considered biogeographic and climatic variables, the volcanic and continental islands did not show statistically significant differences for all the variables, except for mean annual temperature, which resulted slightly higher in volcanic than in continental islands (see Appendix S1.3 for details).
In the 5717 vegetation plots, we assembled in the 58 islands, we collected 81,093 occurrence records for a cumulative number of 1604 vascular plant species (see Appendix S1.1 and Appendix S1.2 in Supporting Information). The number of species recorded per island averaged 156 and ranged from a minimum of 10 species in Faraglione di Mezzo to a maximum of 524 species in Elba, while the estimated number of species per islands averaged 195 and ranged from 12 in Faraglione di Mezzo to a maximum of 603 species in Elba (Appendix S1.2).
ISAR parameters between volcanic and continental islands
Overall, ISAR intercepts did not show a statistically different value between volcanic and continental islands (p–value > 0.05; Fig. 2). Contrary, the slope of the ISAR was significantly higher for volcanic than for continental islands (p-values < 0.05; Fig. 2).
ISAR intercept and slope results based on the Arrhenius model in a log–log space \(\left( {\log \, S = \log \left( c \right) + z \, A} \right)\) calculated for continental (n = 31) and volcanic islands (n = 27). Different letters among island origins a, b show significant differences (p-value < 0.05)
Patterns of ISAR parameter variations on different island origins throughout biogeographic and climatic variables
We evidenced significant variation in ISAR intercepts between volcanic and continental islands along gradients of isolation, mean annual temperature and annual precipitation. Specifically, intercepts significantly decreased for volcanic islands and significantly increased for continental islands with increasing isolation (p-value = 0.008 and 0.004 for volcanic and continental model, respectively; Fig. 3a) and mean annual temperature (p-value < 0.001 and 0.004 for volcanic and continental model, respectively; Fig. 3c). On the other hand, intercepts significantly increased for volcanic islands and significantly decreased for continental islands with increasing annual precipitation (p-value < 0.021 and < 0.001 for volcanic and continental model, respectively; Fig. 3d). We did not detect significant intercept variation on volcanic and continental islands for eccentricity, maximum elevation and area variables.
ISAR intercepts resulted for continental (violet) and volcanic islands (blue) in a gradient of a island isolation, b eccentricity, c mean annual temperature, d annual precipitation, e maximum elevation and f island area. Blue circles and regression lines correspond to volcanic islands. Purple circles and regression lines correspond to continental islands. Bands represent the 95% confidence interval for volcanic and continental linear regressions. Error bars represent the variable standard errors
We detected significant contrasted variation of the slopes between volcanic and continental islands in the majority of the environmental gradients. While the slope on volcanic islands significantly increased with increasing isolation (p-value = 0.038; Fig. 4a), eccentricity (p-value = 0.002; Fig. 4b) and mean annual temperature (p-value = 0.032; Fig. 4c), and decreased with annual precipitation (p-value = 0.01; Fig. 4d), the slopes of continental islands significantly increased with increased maximum elevation (p-value = 0.001; Fig. 4e).
ISAR slopes resulted for continental (violet) and volcanic islands (blue) in a gradient of a island isolation, b eccentricity, c mean annual temperature, d annual precipitation and e maximum elevation and island area. Blue circles and regression lines correspond to volcanic islands. Purple circles and regression lines correspond to continental islands. Bands represent the 95% confidence interval for volcanic and continental linear regressions. Error bars represent the variable standard errors
Discussion
Several hypotheses have been tested seeking a deeper biogeographic meaning of the ISAR slopes and intercepts (Matthews et al., 2021; Triantis et al., 2012). By using 81,093 occurrence data on 1604 vascular plant species in 5717 vegetation plots from 58 Mediterranean islands, we confirmed the hypothesis that ISAR slope is higher in volcanic than continental islands (Kreft et al., 2007; Matthews et al., 2015; Patiño et al., 2014; Triantis et al., 2012), while the expected lower intercept in volcanic than continental islands was not supported by statistical evidence. We also confirmed a contrasted response of the ISAR slope and intercept values for several biogeographic and climatic gradients on volcanic versus continental islands. In this context, we showed that ISAR slopes were more influenced by biogeographic and climate variables on volcanic islands than on continental islands while ISAR intercepts were inversely influenced by island isolation, annual precipitation and mean annual temperature on both islands origins.
It has been described that steep SAR slopes could be generated by speciation-dominated systems, such as island systems (McArthur and Wilson 1967; Brown, 1971). While the dominance of speciation processes can be true in oceanic islands, situated in the middle of the oceans and very distant from the continents, this is hardly the case of volcanic islands in the Mediterranean basin, which are quite close to the coastline (range of distances 0.15–163.7 km) and have experienced periods with even lower isolation during the Quaternary sea level variations (see Whittaker and Fernández-Palacios et al. 2007). The smaller relevance of speciation processes in controlling species diversity and ISARs in Mediterranean volcanic islands is supported by the lower degree of endemism in these islands compared to real oceanic islands, such as Juan Fernández, Canaries, Hawaii or Galápagos (Caujapé-Castells et al., 2010; Vargas et al., 2014).
Differences in island isolation and geological origin have been described as the main factors that influence steeper ISAR slopes on oceanic than on continental islands (Kreft et al., 2007; Matthews et al., 2015; Patiño et al., 2014; Triantis et al., 2012). In the case of the Mediterranean volcanic islands, the geographic isolation is limited if compared with real oceanic islands, but the lack of pre-existing species pool at the moment of islands formation is certainly true, and while the speciation process can be limited because of the closeness of species pools and the easy immigration, the colonization process is likely lower in volcanic than in continental islands. In fact, volcanic islands are characterized by a lower immigration but also by lower colonization rates, since few species of those that arrive on volcanic islands survive and establish vital populations (MacArthur & Wilson, 1967). This also has the consequence of relaxing species competition, triggering adaptive radiations and species turnover (Patiño et al., 2014; Triantis et al., 2012). In this context, even in a frame of lower speciation in comparison with real oceanic islands, our results were in line with our expectations evidencing higher slopes for volcanic than for continental islands and increasing slopes with increasing isolation.
The ISAR intercepts, which can be interpreted as the number of species per area unit, is expected to be lower in volcanic than in continental islands, as well as with increasing isolation (MacArthur & Wilson, 1967). Yet, we did not evidence a significant difference between volcanic and continental islands. Moreover, the ISAR intercept increased with increasing isolation on continental islands. We hypothesized that these increases could be determined by the interplay of two processes: (1) area is positively correlated with island isolation allowing more species on continental islands with increasing isolation and/or (2) isolated islands host more endemic species formed from adaptive radiations or genetic drifts (Stuessy et al., 2006; Takayama et al., 2018). The first statement is supported by the significant positive correlation between the mean area and mean isolation of the continental islands grouped on each ISAR model (see Appendix S1.4 for further details). As for the second statement, it has been described that isolation and island area (also island elevation, positive and significantly correlated) influence speciation rates that combined with relaxed competition pressure could trigger adaptive radiations on islands (Givnish, 2010) generating, by consequence, higher ISAR intercepts. Nevertheless, this last claim requires a deeper analysis of our study area. In contrast, ISAR intercepts on volcanic islands were decreasing with increasing isolation, in line with our expectation and with the previous studies (Matthews et al., 2015; Triantis et al., 2012).
We expected higher slopes for round islands because they could contain greater habitat diversity than elongated islands (Blouin & Connor, 1985). In contrast with elongated islands, round ones could contain wider environmental gradients given by their diametric form and differences on sea, winds and sun exposure. We also expected this last pattern to be more pronounced on volcanic islands because they generally contained greater habitat diversity than continental islands (Whittaker & Fernández-Palacios, 2007). We rejected in part our hypothesis because only ISAR slopes on volcanic islands responded to the influence of eccentricity evidencing the opposite expected pattern, i.e., higher slopes with increasing eccentricity. However, this trend could be influenced by the isolation effect. As we showed, the mean eccentricities were positively correlated to the mean isolations resulting from the analyzed groups of islands on the sensitivity analysis (see Appendix S1.5 for further details). Other studies have described the challenges encountered when analyzing patterns of island shapes because these could be obscured by other variables that influence species richness such as isolation and habitat heterogeneity (Blouin & Connor, 1985). Despite species richness trends have been assessed by at least four different island shape measures finding contrasting patterns, the analysis has been carried out on mixed islands origin (Blouin & Connor, 1985), and these could have hidden the trends for strictly volcanic and/or continental islands (Cáceres-Polgrossi et al., 2023). Thus, our results suggest the need for further analysis, for example, on strictly volcanic islands for a deeper understanding of this trend.
According to Kreft et al. (2007), harsh climates may constrain the number of potential immigrant species that are able to tolerate the specific conditions and are thus able to successfully colonize an island. So, the intercepts could decrease, and the slopes increase when climatic conditions are unfavorable. Conversely, when climate is favorable, ISAR intercepts could increase, and the slopes decrease. In our study, we evidenced different dynamics of species richness between continental and volcanic islands along climate gradients. Following the hypothesis of Kreft et al. (2007), we could evidence that while vascular plant species richness on volcanic islands tends to decrease with increasing mean annual temperature and decreasing annual precipitation, we observed the opposite pattern on continental islands. Additionally, we attributed the absence of slope variation on continental islands to the fact that most plant species arrived when continental islands were connected to the mainland (Whittaker & Fernández-Palacios, 2007), so no climatic barriers had to be overcome. Although these interpretations need to be tested in depth and in a more global perspective, our study contributed to the existing knowledge gap on ISAR parameters variation as a function of climate variables.
Finally, we hypothesized that the general non-significant trends evidenced for the ISAR slopes on continental islands are suggesting a low influence of immigration and colonization dynamics in the majority of these islands (Brown, 1971). It would be interesting to investigate if continental islands often show difficulty in reflecting patterns on ISAR slopes. However, the significantly higher slopes with increasing island elevation on continental islands are also suggesting that islands with higher elevation presented some grade of speciation dynamics in their flora. For example, Illiadou et al. (2014) evidenced steeper slopes for Aegean islands that have higher elevations and a larger proportion of in situ speciation than Ionian Islands. In this context, our results suggest that some biogeographic variables could influence stronger ETIB dynamics on volcanic than on continental islands. At the same time, some biogeographic variables other than area and isolation (e.g., elevation, temperature and precipitation) could exclusively influence ETIB dynamics and that these could be easily evidenced by means of their ISAR even on systems with lower ETIB dynamics (e.g., continental islands of this study).
Conclusions
Using a large amount of vegetation plot data sampled on 58 Mediterranean islands, our study disentangled the contribution of different biogeographic and climatic variables on vascular plant species richness dynamics in continental and volcanic islands. Overall, ISARs reflected higher immigration and colonization dynamics for volcanic than for continental islands. Further research is needed to confirm and expand upon these findings in a broader context.
References
Alessi, N., Bruzzaniti, V., Buldrini, F., Centomo, E., Cervellini, M., Enea, M., Landi, S., Lelli, C., Montanari, I., Nascimbene, J., Pezzi, G., Virzí, G., Zannini, P., & Chiarucci, A. (2022). AMS-VegBank: a new database of vegetation plots for the Italian territory. Vegetation Classification and Survey, 3, 177–185. https://doi.org/10.3897/VCS.85083
Bagella, S., Becca, G., Bedini, G., Caria, M. C., Pisanu, S., & Rubani, M. (2020). Why so different? A case study about floras from a mediterranean island. Phytotaxa., 440, 129–158. https://doi.org/10.11646/phytotaxa.440.2.4
Blouin, M. S., & Connor, E. F. (1985). Is there a best shape for nature reserves? Biological Conservation, 32(3), 277–288. https://doi.org/10.1016/0006-3207(85)90114-4
Brown J (1971) Mammals on mountaintops. Nonequilibrium insular biogeography. The American naturalist, 105(945), 467–478. http://www.jstor.org/stable/2459514
Cáceres-Polgrossi, L., Di Rico, M., Parra, D., Seebens, H., & Galvin SD Boehmer, H. J. (2023). The relationship between naturalized alien and native plant species: insights from oceanic islands of the south-east Pacific over the last 200 years. Neobiota, 86, 21–432. https://doi.org/10.3897/neobiota.86.102661
Cao Pinna, L., Axmanová, I., Chytrý, M., Malavasi, M., Acosta, A. T. R., Giulio, S., Attorre, F., Bergmeier, E., Biurrun, I., Campos, J. A., Font, X., Küzmič, F., Landucci, F., Marcenò, C., Rodríguez-Rojo, M. P., & Carboni, M. (2021). The biogeography of alien plant invasions in the Mediterranean basin. Journal of Vegetation Science, 32, 1–13. https://doi.org/10.1111/jvs.12980
Caujapé-Castells, J., Tye, A., Crawford, D. J., Santos-Guerra, A., Sakai, A., Beaver, K., Lobin, W., Vincent Florens, F. B., Moura, M., & Jardim, R. (2010). Conservation of oceanic island floras: present and future global challenges. Perspectives Plant Ecology Evoluation and Systematics, 12(2), 107–129. https://doi.org/10.1016/j.ppees.2009.10.001
Chao, A., & Colwell, R. K. (2017). Thirty years of progeny from Chao’s inequality: Estimating and comparing richness with incidence data and incomplete sampling. SORT-Statistics Operations Research Transactions, 41(1), 3–54.
Chase, J. M., McGill, B. J., Thompson, P. L., Antão, L. H., Bates, A. E., Blowes, S. A., et al. (2019b). Species richness change across spatial scales. Oikos, 128, 1079–1091. https://doi.org/10.1111/oik.05968
Chase, J. M., Gooriah, L., May, F., Ryberg, W. A., Schuler, M. S., Craven, D., et al. (2019a). A framework for disentangling ecological mechanisms underlying the island species-area relationship. Frontiers of Biogeography, 11, e40844. https://doi.org/10.21425/F5FBG40844
Chiarucci, A., Di Biase, R. M., Fattorini, L., Marcheselli, M., & Pisani, C. (2018). Joining the incompatible: exploiting purposive lists for the sample-based estimation of species richness. The Annals of Applied Statistics, 12(3), 1679–1699. https://doi.org/10.1214/17-AOAS1126
Chiarucci, A., Guarino, R., Pasta, S., Rosa, A. L., Cascio, P. L., Médail, F., Pavon, D., Fernández-Palacios, J. M., & Zannini, P. (2021b). Species–area relationship and small-island effect of vascular plant diversity in a young volcanic archipelago. Journal of Biogeography, 48(11), 2919–2931. https://doi.org/10.1111/jbi.14253
Chiarucci, A., Fattorini, S., Foggi, B., Landi, S., Lazzaro, L., Podani, J., & Simberloff, D. (2017). Plant recording across two centuries reveals dramatic changes in species diversity of a Mediterranean archipelago. Science and Reports, 7(1), 5415. https://doi.org/10.1038/s41598-017-05114-5
Chiarucci, A., Buldrini, F., Cervellini, M., Guarino, R., Caccianiga, M., & Foggi, B. (2021a). Habitat type and island identity as drivers of community assembly in an archipelago. Journal of Vegetation Science, 32(1), e12953. https://doi.org/10.1111/jvs.12953
Chown, S. L., Gremmen, N. J. M., & Gaston, K. J. (1998). Ecological biogeography of southern ocean islands: species-area relationships, human impacts, and conservation. The American Naturalist, 152(4), 562–575. https://doi.org/10.1086/286190
Connor, E. F., & McCoy, E. D. (1979). The statistics and biology of the species-area relationship. The American Naturalist, 113(6), 791–833.
Daru, B. H., Park, D. S., Primack, R. B., Willis, C. G., Barrington, D. S., & Whitfeld, T. J. S. (2018). Widespread sampling biases in herbaria revealed from large-scale digitization. New Phytologist, 217, 939–955. https://doi.org/10.1111/nph.14855
Dengler, J. (2009). Which function describes the species-area relationship best? A review and empirical evaluation. Journal of Biogeography, 36, 728–744. https://doi.org/10.1111/j.1365-2699.2008.02038.x
Drakare, S., Lennon, J. J., & Hillebrand, H. (2005). The imprint of the geographical, evolutionary and ecological context on species-area relationships. Ecology Letters, 9, 215–227. https://doi.org/10.1111/j.1461-0248.2005.00848.x
Fattorini, S., Borges, P. A. V., Dapporto, L., & Strona, G. (2016). What can the parameters of the species-area relationship (SAR) tell us? Insights from Mediterranean islands. Journal of Biogeography, 44, 1018–1028. https://doi.org/10.1111/jbi.12874
Fernández-Palacios JM, Kueffer C, Drake D (2015) A new golden era in island biogeography. Frontiers Biogeography, 7(1), https://doi.org/10.21425/F5FBG24493
Franzén, M., Schweiger, O., & Betzholtz, P.-E. (2012). Species-area relationships are controlled by species traits. PLoS ONE, 7(5), e37359. https://doi.org/10.1371/journal.pone.0037359
Gillespie, T. W., Keppel, G., Pau, S., Price, J. P., O’Neill, J. T., & K. (2013). Scaling species richness and endemism of tropical dry forests on oceanic islands. Diversity and Distributions, 19, 896–906. https://doi.org/10.1111/ddi.12036
Givnish, T. J. (2010). Ecology of plant speciation. Taxon, 59, 1326–1366. https://doi.org/10.1002/tax.595003
Hsieh TC, Ma KH, Chao A (2022) iNEXT: iNterpolation and EXTrapolation for species diversity. R package, version 3.0.0 http://chao.stat.nthu.edu.tw/wordpress/software-download/.
Iliadou, E., Kallimanis, A. S., Dimopoulos, P., & Panitsa, M. (2014). Comparing the two Greek archipelagos plant species diversity and endemism patterns highlight the importance of isolation and precipitation as biodiversity drivers. Journal of Biological Research-Thessaloniki, 21(1), 16. https://doi.org/10.1186/2241-5793-21-16
Kalmar, A., & Currie, D. J. (2006). A global model of island biogeography. Global Ecology and Biogeography, 15(1), 72–81.
Karger, D., Conrad, O., Böhner, J., Kawohl, T., Kreft, H., Soria-Auza, R. W., et al. (2017). Climatologies at high resolution for the earth’s land surface areas. Scientific Data, 4, 170122. https://doi.org/10.1038/sdata.2017.122
Karger DN, Conrad O, Böhner J, Kawohl T, Kreft H, Soria-Auza RW et al (2018) Data from: climatologies at high resolution for the earth’s land surface areas. EnviDat, https://doi.org/10.16904/envidat.159.
Kreft, H., Jetz, W., Mutke, J., Kier, G., & Barthlott, W. (2007). Global diversity of island floras from a macroecological perspective. Ecology Letters, 11, 116–127.
Lomolino, M. V. (2000). Ecology’s most general, yet protean pattern: the species–area relationship. Journal of Biogeography, 27, 17–26. https://doi.org/10.1111/j.1461-0248.2007.01129.x
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton, NJ: princeton university press. https://www.jstor.org/stable/j.ctt19cc1t2
Martin, T. E. (1981). Species-area slopes and coefficients: a caution on their interpretation. The American Naturalist, 118(6), 823–837.
Matthews, T. J., Guilhaumon, F., Triantis, K. A., Borregaard, M. K., & Whittaker, R. J. (2015). On the form of species-area relationships in habitat islands and true islands. Global Ecology and Biogeography, 25, 847–858. https://doi.org/10.1111/geb.12269
Matthews TJ, Rigal F, Proios K, Triantis KA, Whittaker RJ (2021) Explaining variation in island species–area relationship (ISAR) model parameters between different archipelago types: expanding a global model of ISARs. The species–area relationship, 51–77.
Médail, F. (2017). The specific vulnerability of plant biodiversity and vegetation on Mediterranean islands in the face of global change. Regional Environmental Change, 17, 1775–1790. https://doi.org/10.1007/s10113-017-1123-7
Médail, F. (2022). Plant biogeography and vegetation patterns of the mediterranean islands. Botanical Review, 88, 63–129. https://doi.org/10.1007/s12229-021-09245-3
Médail, F., & Quézel, P. (1997). Hot-spots analysis for conservation of plant biodiversity in the Mediterranean basin. Annals of Missouri Botanical Garden, 84, 112–127. https://doi.org/10.2307/2399957
Moradi, H., Fattorini, S., & Oldeland, J. (2020). Influence of elevation on the species–area relationship. Journal of Biogeography, 47(9), 2029–2041. https://doi.org/10.1111/jbi.13851
Myers, N., Mittermeier, R., Mittermeier, C., Mittermeier, C. G., da Fonseca, G. A. B., & Kent, J. (2000). Biodiversity hotspots for conservation priorities. Nature, 403, 853–858. https://doi.org/10.1038/35002501
Norman, K. E. A., Chamberlain, S., & Boettiger, C. (2020). taxadb: a high-performance local taxonomic database interface. Methods in Ecology and Evolution, 11, 1153–1159. https://doi.org/10.1111/2041-210X.13440
Ohyama, L., Holt, R., Matthews, T. J., & Lucky, A. (2021). The species-area relationship in ant ecology. Journal of Biogeography, 48(8), 1824–1841. https://doi.org/10.1111/jbi.14149
Patiño, J., Weigelt, P., Guilhaumon, F., Kreft, H., Triantis, K. A., Naranjo-Cigala, A., et al. (2014). Differences in species-area relationships among the major lineages of land plants: a macroecological perspective. Global Ecology and Biogeography, 23, 1275–1283. https://doi.org/10.1111/geb.12230
Preston, F. W. (1962). The canonical distribution of commonness and rarity: Part I. Ecology, 43, 185–215. https://doi.org/10.2307/1933371
R Core Team (2022) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. https://www.R-project.org/.
Rahbek, C. (1997). The relationship among area, elevation, and regional species richness in neotropical birds. The American Naturalist, 149(5), 875–902.
Rosenzweig, M. L. (1995). Species diversity in space and time. New York: Cambridge University Press.
Santi, F., Testolin, R., Zannini, P., Di Musciano, M., Micci, V., Ricci, L., Guarino, R., Bacchetta, G., Fernández-Palacios, J.M., Fois, M. & Kougioumoutzis, K (2024) MEDIS—A comprehensive spatial database on Mediterranean islands for biogeographical and evolutionary research. Abstract Global Ecology and Biogeography. https://doi.org/10.1111/geb.13855
Scheiner, S. M. (2003). Six types of species-area curves. Global Ecology and Biogeography, 12, 441–447. https://doi.org/10.1046/j.1466-822X.2003.00061.x
Scheiner, S. M., Chiarucci, A., Fox, G. A., Helmus, M. R., McGlinn, D. J., & Willig, M. R. (2011). The underpinnings of the relationship of species richness with space and time. Ecological Monographs, 81, 195–213. https://doi.org/10.1890/10-1426.1
Sfenthourakis, S., & Triantis, K. A. (2017). The Aegean archipelago: a natural laboratory of evolution, ecology and civilisations. J Biol Res Thessalon, 24(1), 1–13. https://doi.org/10.1186/s40709-017-0061-3
Sfenthourakis, S., & Panitsa, M. (2012). From plots to islands: species diversity at different scales. Journal of Biogeography, 39, 750–759. https://doi.org/10.1111/j.1365-2699.2011.02639.x
Stuessy, T. F., Jakubowsky, G., Salguero Gómez, R., Pfosser, M., Schlüter, P. M., Fer, T., Sun, B. Y., & Kato, H. (2006). Anagenetic evolution in island plants. Journal of Biogeography, 33, 1259–1265. https://doi.org/10.1111/j.1365-2699.2006.01504.x
Takayama, K., Crawford, D. J., López-Sepúlveda, P., Greimler, J., & Stuessy, T. F. (2018). Factors driving adaptive radiation in plants of oceanic islands: a case study from the Juan Fernández Archipelago. Journal of Plant Research, 131(3), 469–485. https://doi.org/10.1007/s10265-018-1023-z
Testolin, R., Attorre, F., Bruzzaniti, V., Guarino, R., Jiménez-Alfaro, B., Lussu, M., Martellos, S., Di Musciano, M., Pasta, S., Sabatini, F. M., Santi, F., Zannini, P., & Chiarucci, A. (2023). Plant species richness hotspots and related drivers across spatial scales in small Mediterranean islands. Journal of Systematics and Evolution. https://doi.org/10.1111/jse.13034
Testolin, R., Attorre, F., Borchardt, P., Brand, R. F., Bruelheide, H., Chytrý, M., De Sanctis, M., Dolezal, J., Finckh, M., Haider, S., Hemp, A., Jandt, U., Kessler, M., Korolyuk, A. Y., Lenoir, J., Makunina, N., Malanson, G. P., Montesinos-Tubée, D. B., Noroozi, J., & Jiménez-Alfaro, B. (2021). Global patterns and drivers of alpine plant species richness. Global Ecology and Biogeography, 30(6), 1218–1231. https://doi.org/10.1111/geb.13297
Triantis, K. A., Nogués-Bravo, D., Hortal, J., Borges, A. V. P., Adsersen, H., & Fernández-Palacios, J. M. (2008). Measurements of area and the (island) species–area relationship: new directions for an old pattern. Oikos, 117, 1555–1559. https://doi.org/10.1111/j.0030-1299.2008.16808.x
Triantis, K. A., Borges, P. A. V., Ladle, R. J., Hortal, J., Cardoso, P., & Gaspar, C. (2010). Extinction debt on oceanic islands. Ecography, 33, 285–294. https://doi.org/10.1111/j.1600-0587.2010.06203.x
Triantis, K. A., Guilhaumon, F., & Whittaker, R. J. (2012). The island species–area relationship: biology and statistics. Journal of Biogeography, 39, 215–231. https://doi.org/10.1111/j.1365-2699.2011.02652.x
Vargas, R., Smith-Ramírez, C., González, C., & Fernández, M. (2014). Reserva de la Biosfera Archipiélago Juan Fernández: endemismo para conservar. In A. Moreira-Muñoz & A. Borsdorf (Eds.), Reservas de la Biosfera de Chile: Laboratorios para la sustentabilidad (pp. 126–145). Santiago: Serie GEOlibros.
Vogiatzakis, I. N., Mannion, A. M., & Sarris, D. (2016). Mediterranean island biodiversity and climate change: the last 10,000 years and the future. Biodiversity and Conservation, 25(13), 2597–2627. https://doi.org/10.1007/s10531-016-1204-9
Weigelt, P., Jetz, W., & Kreft, H. (2013). Bioclimatic and physical characterization of the world’s islands. Proceed Natl Acad Sci, 110(38), 15307–15312. https://doi.org/10.1073/pnas.1306309110
Whittaker, R. J., Fernández-Palacios, J. M., Matthews, T. J., Borregaard, M. K., & Triantis, K. A. (2017). Island biogeography: taking the long view of nature’s laboratories. Science, 357, eaam8326. https://doi.org/10.1126/science.aam8326
Whittaker, R. J., & Fernández-Palacios, J. M. (2007). Island biogeography: Ecology, evolution, and conservation. Oxford University Press.
Xu, H., Liu, S., Li, Y., Zang, R., & He, F. (2012). Assessing non-parametric and area-based methods for estimating regional species richness. Journal of Vegetation Science, 23, 1006–1012. https://doi.org/10.1111/j.1654-1103.2012.01423.x
Acknowledgements
We would like to thank Marcelino J. Del Arco Aguilar for his kindness and expertise in the field of phytosociology. His invaluable insights and mentorship have greatly enriched our understanding. We would like to express our gratitude to the Carolina Foundation for providing opportunities for students from Central and South America to study in Spain. AC and RT were funded by the European Union—NextGenerationEU, under the National Recovery and Resilience Plan (NRRP), Project title “National Biodiversity Future Center -NBFC” (project code CN_00000033) CUP J33C22001190001. Finally, we thank to the ERASMUS program for facilitating and promoting European cooperation and knowledge exchange.
Funding
Open access funding provided by Alma Mater Studiorum - Università di Bologna within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
L.C.-P. and A.C. conceived the ideas; L.C.-P., A.C., P.Z., V.B., F.B. and M.C. collected the data; L.C.-P. and R.T. analyzed the data and L.C.-P. and A.C. led the writing, with major contributions by J.M.F.-P. and R.T. All authors checked, improved and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. The authors have no financial or proprietary interests in any material discussed in this article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cáceres-Polgrossi, L., Buldrini, F., Bruzzaniti, V. et al. Climatic and biogeographic factors show contrasted effects on continental and volcanic ISARs. COMMUNITY ECOLOGY (2024). https://doi.org/10.1007/s42974-024-00195-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42974-024-00195-5