Abstract
Life on Earth is complex and generally abounds in food webs with other living organisms in terms of an ecological community. Besides such complexity, and the fact that populations of most living organisms have never been studied in terms of their molecular ecology, it is best to tread carefully when describing a given species as a ‘generalist’, more especially in terms of dietary and habitat breadth. We very much doubt that population homogeneity ever exists—because populations are always undergoing molecular-genetic changes, sometimes rapid, in response to various ecological challenges (e.g. climate, intra- and interspecific competition). In any case, a population may already have begun to undergo cryptic speciation. Such entities can occupy different habitats or exhibit different dietary breadths as a result of various ecological interactions formed over different spatial scales. These scales include everything from local (including islands) to geographic. The fossil evidence reveals that specialisations have existed over vast swathes of time. Besides, as is well documented, evolution only occurs as a result of adaptations leading to specialisation, and ultimately, specialist entitles, i.e. species and lower levels of ecological-evolutionary divergence. Here, focusing on diet, we posit that the terms mono-, oligo-and polyphagous are more accurate in relation to the dietary breadth of animals, with omnivory adopted in the case of organisms with very different food items. Thus, we strongly urge that the dubious and unscientific term ‘generalism’ be dropped in favour of these more precise and scientifically accurate terms directly relating to levels of phagy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The terms ‘generalism’ and ‘specialism’ in animal species populations are usually equated with life history parameters like habitat preference, or more usually diet, and especially dietary breadth. (Fox & Morrow, 1981). Focusing on diet breadth, generalist animals are perceived as having a wide choice of food items. For example, the diet of the Holarctic brown bear, Ursus arctos L. (Mammalia: Carnivora: Ursidae), includes everything from moths, molluscs, roots, berries, honey, to large mammals such as fawns (Bojarska & Selva, 2012; Munro et al., 2006; Shipley et al., 2009). Thus this species may be described as omnivorous. In a stricter, more scientific sense, it can also be labelled as polyphagous (choosing many items to feed on) as opposed to oligophagous (choosing a few items to feed on) or monophagous at the other extreme, comprising a single preferred item of food, be that a plant, such as leaves, or single item of prey in the case of parasitic organisms or predators. Some scientists would claim that animals showing such a broad diet niche are ‘generalist’, a loose, fit-all category. But of course, unless we know what we are really dealing with in an evolutionary-ecological sense, such a term is likely to be used erroneously (e.g. Wray et al., 2022 in the case of bats). And such erroneous use is, in turn, likely to perpetrate and hence misconstrue the true state of affairs, and indeed the true nature of generalism, if such really exists. Does this matter? Besides the fact that it is important for biologists to use precise terminology when describing biological entities and their interactions, i.e. biological phenomena, the prospect of generalism occurring in nature has important ramifications for communities and ecosystem functioning (Richmond et al., 2005; Thébault et al., 2003). The topic is of course a highly contested one and the debate is ongoing, as the present contribution aims to reveal.
Insects are the largest and most diverse group of organisms on Earth (Gaston, 1991). In an earlier paper on dietary generalism versus specialism in nature (Loxdale et al., 2011), we argued that in insects, generalism was rare (Gaston, 1991), because of the nature of their community interactions (not only intraspecifically, and in the case of insects with asexual lifestyles or phases, intraclonally, but also interspecifically), and that most insects were dietary specialists to a greater or lesser extent. In a second paper (Loxdale & Harvey, 2016), we refined the concept, suggesting that insects could perhaps be seen as fitting within four broad categories of specialism and generalism. These four categories seemed at the time to encompass most of the major lifestyle examples we could find. Lastly, in a third paper (Loxdale et al., 2019), we moved away from the concept of generalism altogether, labelling it as a misnomer, and proposed that, in fact, all animal species, and by implication all living organisms, are broadly specialists and should be seen to be so. We concluded therefore that use of the term polyphagous (including omnivory where appropriate) was preferable, in a scientific sense, whilst acknowledging the reality of lower levels of polyphagy, i.e. oligophagy and ultimately, monophagy in relation to animals with a reduced dietary breadth.
Some would perhaps argue that in the end, discussions over terminology are frivolous and are merely semantics. However, we contest this by stating that the use of fundamentally incorrect scientific terminology is to the detriment of scientific thought and progress. This is especially true if this terminology is erroneously applied to describe observed phenomena (Fox & Morrow, 1981).
The uniqueness of the ecological niche
With animal populations, it seems clear that if a species population, or lower level of evolutionary-ecological divergence (e.g. subspecies, sibling and sister species, etho-species, race or biotype) moves into and fills a specific ecological niche, and is not displaced from that niche by a competing species population, then perhaps over the course of millions of years (as in the case of brown bears), that is the de facto specialism. What the animal species or subspecies then eats to maintain itself within that niche is a related, but not exclusive, question. This is because several ecological forces are at work here, which manifest themselves as ecological axes in a multidimensional hyperspace (e.g. food, climate, predators, parasites, etc.), as G. Evelyn Hutchinson (1903–91) realised and brilliantly espoused more than half a century ago (Colwell & Rangel, 2009; Hutchinson, 1959; Slobodkin, 1993).
We also noted (Loxdale & Harvey, 2016) that morphology-anatomy, and physiology-biochemistry, especially including chemical ecology (e.g. pheromones), are essential in defining the role of a living animal species within its own particular ecological niche. We further pointed out that these various influencing factors and elements cannot be divorced from one another. We would add that these factors interact with abiotic factors to determine a thermal niche that also influences the geographical distribution of a species. The niche concept applies to local forces, but this clearly has limitations at larger scales. Hence, it is surely naïve to talk of generalism, as if this blanket term can exist in spite of an array of underlying biological constraints, including ecological ones that confine an animal species, subspecies or population over a specific geographical area and within a defined niche (Fox & Morrow, 1981).
Over time, such an entity may disappear from its defined niche, either due to local extinction (the ultimate fate of all living species populations over time), or due to its displacement by a more vigorous and ’ecologically-superior’ competitor (which could in the case of asexuals be a superior clone, superior in terms of one or more life traits, including reproduction rate) (Amarasekare, 2003; Reitz & Trumble, 2002). The fossil record, stretching back hundreds of millions of years, is replete with many such examples of specialism, some broader than others, including of course co-evolved ones (Thompson, 1994). If an animal species or subspecific population moves to fill a new niche, it can only do so by an act of specialism and ecological specialisation,Footnote 1 brought about by allopatric, parapatric and sympatric mechanisms, sometimes, and perhaps often, involving large scale chromosomal re-arrangements, especially translocations (Avise, 1994; Coyne & Orr, 2004; Loxdale, 2010; Loxdale & Balog, 2018; Loxdale et al., 2020; White, 1978).
Morphology-anatomy and other life history aspects
When looking at the prospect of generalism as a natural phenomenon, what is often lost in such consideration is close inspection of the actual animal itself, its ecology and general biology. For example, the European pine marten, Martes martes L. (Mammalia: Carnivora: Mustelidae) may eat a wide range of food items, i.e. everything from small mammals, birds, insects, frogs, carrion, berries, birds’ eggs, nuts, and honey (Caryl et al., 2012; Posluszny et al., 2007). However, its anatomy and behaviour provide evidence of its adapted lifestyle. It has retractable claws (the only mustelid to do so), enabling it to run fast up and down tree trunks and branches, and hence it prefers an arboreal lifestyle, but with the ability to go across open ground when necessary. It is clear that it is a fierce, relentless predator, as shown by its lithe form, its skull with needle-sharp teeth, and its deft hunting ability and strategy; in other words, its ‘nature’. Its preferred food items, from a cost–benefit perspective, are small mammals and birds, which it hunts actively at dusk and at night, not to say that it is not opportunistic in eating other food items when it, by chance, finds them by sight or smell (e.g. birds’ eggs or fruit). Perhaps an even better example of a specialised mustelid predator is the North American fisher, Pekania (= Martes) pennanti (Erxleben), which is not only adept at killing snowshoe hares but also porcupines (Bowman et al., 2006).
If the species was long extinct, examination of its skeleton alone would be enough to conclude that this was a small mammal of the Order Carnivora, Family Mustelidae, probably with a fierce, active lifestyle. Other aspects of its biology and ecology would of course not be known, e.g. that it is territorial, what the colour of its pelt was, and whether or not it showed sexual dimorphism in term of pelt colour. But its basic lifestyle could be readily deduced. So why are most mustelids referred to as generalists? It is surely not a generalist, but a highly evolved small predator, to some large extent co-evolved with the European red squirrel, Sciurus vulgaris L. (Mammalia: Rodentia: Sciuridae), its preferred prey and in the British Isles, to the detriment to the historically-recently introduced grey squirrel of North America, Sciurus carolinensis Gmelin, (Mammalia: Rodentia: Sciuridae), which it has not co-evolved with over long stretches of time. As a consequence, in mainland Britain, it has increased its range from its stronghold in Scotland and England (after decades of persecution by humans, especially prior to legal protection in 1988; cf. MacPherson & Wright, 2021; Pullar, 2020), and is slowly extirpating local grey squirrel populations it encounters as it expands its range southwards (Sheehy et al., 2018; Twining et al., 2020; but see also Putman, 2000 who claims that voles are the main diet).
Effects of geographic range: birds as exemplars
Another important question is whether, even if a species population is generalist, it is universally so over a large spatial scale, i.e. across its whole geographic range? If it isn’t, then the epithet ‘generalist’ may be arbitrary, and thereby subjective, depending on where a particular population or subpopulation, including locally adapted forms, exist. For example, to start with the lowest level of specialism, monophagy, the European pied flycatcher, Ficedula hypoleuca (Pallas) (Aves: Passeriformes: Muscicapidae), a migrant to the British Isles in the spring and summer months, shows a clear westerly distribution according to the British Trust for Ornithology, (BTO) map for this species (Fig. 1). As the map reveals, it strangely appears absent from the island of Ireland, as many species of animal are, for whatever reason(s). The species is, according to Wikipedia (https://en.wikipedia.org/wiki/European_pied_flycatcher):-
“....mainly insectivorous, although its diet also includes other arthropods. This species commonly feeds on spiders, ants, bees and similar prey.”
But presumably the variation in prey animals of this specialist insectivore across the west and east of England and Scotland, if such exists, is not enough to account for the observed distribution. Other ecological forces must be at work here, presumably dominated by climate, which is comparatively milder and moister in the western side of the country, influenced by the Gulf Stream and predominantly westerly and southerly winds, than the eastern side, influenced by easterly and northerly winds coming off the North Sea and Scandinavia. So even if it is true that diet is theoretically broadly the same, other ecological constraints dominate the preferred habitat and ultimately breeding distribution of this bird (cf. also Burger et al., 2012; Nicolau et al., 2021).
Another bird species of interest in this respect is the Eurasian nuthatch, Sitta europaea L. (Aves: Passeriformes: Sittidae), which according to Wikipedia (https://en.wikipedia.org/wiki/Nuthatch):-
“....are omnivorous, eating mostly insects, nuts, and seeds. They forage for insects hidden in or under bark by climbing along tree trunks and branches, sometimes upside-down. They forage within their territories when breeding, but they may join mixed feeding flocks at other times.”
This bird thus appears to have a wider dietary breadth than the Pied Flycatcher, and may perhaps be described as oligophagous, but it also has a largely westerly distribution in England as shown on the BTO map, is largely absent from Scotland, and completely so from the island of Ireland (Fig. 2).The species is thought to be resident in the British Isles, with similar overlapping, but slightly different, species populations in mainland Europe, presumably the result of differentiation forces leading to local adaptation and hence evolution. The taxonomy of the species is apparently difficult with several species and subspecies across Europe and Asia, thereby producing a geographically-based species complex (cf. Nuthatch: Species boundaries; https://en.wikipedia.org/wiki/Nuthatch).
A last example that we present in this context of generalism and geography is the Eurasian Magpie, Pica pica L. (Aves: Passeriformes: Corvidae), which is both polyphagous and omnivorous (see below). This is a resident species throughout much of the British Isles, including the island of Ireland, and as shown on the broad distribution map, is found (or has been at some time recorded) over the entire length and breadth of these islands, with the exception of mid- and northern Scotland and the Orkney and Shetland Isles (Fig. 3a). However, when the Breeding Relative Abundance map is examined (Fig. 3b), a different pattern emerges. The species shows a far from uniform spatial distribution. Rather, the demography is centred in and around the major cities and conurbations, but interestingly, this trend is not so apparent in Northern Ireland, where the species appears abundant even away from the major cities such as Belfast.
With regard to their diet, according to the Royal Society for the Protection of Birds (https://www.rspb.org.uk/birds-and-wildlife/wildlife-guides/bird-a-z/magpie/what-do-magpies-eat/):-
“Their main diet [of the Magpie] in summer is grassland invertebrates, such as beetles, flies, caterpillars, spiders, worms and leatherjackets. In winter, they eat more plant material, such as wild fruits, berries and grains, with household scraps and food scavenged from bird tables or chicken runs, pet foods etc. They will eat carrion at all times and catch small mammals and birds. Occasionally, magpies prey on larger animals such as young rabbits. During the breeding season they will take eggs and young of other birds.”
So whilst the Magpie has a rather broad dietary breadth (much broader than the two aforementioned bird species), with different foods eaten due to seasonal abundance, and especially during the breeding season (early April)), nevertheless it is not found uniformly across the British Isles. Strangely, whilst it is a well-known carrion feeder, having thrived upon road kills in recent decades due to the rising level of traffic on British and Irish roads, the bird is clearly rarer or even largely absent from some areas with busy roads and thus an abundance of such road kills, including pheasants, rabbits, foxes, badgers and deer. Other ecological constraints are clearly at work here, possibly related to relative availably as well as security of nesting sites in human conurbations compared with more rural locations. The increasing number of small nesting birds now attracted to urban and suburban gardens in the spring (and here we emphasise this especially in terms of their eggs and chicks as potential prey items for the magpie) along with the greater availability of food put out for wild birds or discarded by humans generally are other factors worthy of consideration (cf. Hanmer, 2016). Either way, in this particular case, so-called generalism comes at a cost in terms of demography, which doesn’t appear to be related to climate per se.
Effects of geographic range: insects as exemplars
Insects are globally abundant, representing around 75% of the Earth’s animal species (Grimaldi & Engel, 2005) and fill innumerably diverse niches, especially in the tropics. Therefore, as entomologists, we feel obliged to provide a few examples of the geographic trends to which we are referring and basing our broad thesis upon. Because insects are such a huge taxon with so many species (approx. 1 million with doubtless many more to yet be discovered and described) (Gaston, 1991; Stork et al., 2015), many are poorly described scientifically. There is thus often very little or nothing known about their behaviour and ecology-molecular ecology. The strong possibility exists that many formally described species are in fact arrays of cryptic species, i.e. belonging to cryptic species complexes, or are introgressed/hybridised with other closely related species (Loxdale, 2018; Loxdale et al., 2016; Mallet, 2007). This of course will inflate the number of species that actually exist at any one time and place, possibly significantly.
The existence of cryptic species is bound to enter into any debate about the given dietary breadth of particular animal species, just because a large diet range (Fox & Morrow, 1981) may in fact be due to the presence of slightly differently adapted and evolved animals in slightly different niches. This might be so even though they appear superficially to be the same, i.e. genetically homogenous across a large geographic range. The aforementioned Nuthatch, Sitta eurpoaea sensu lato is one such organism, the diet of which may well differ significantly across its huge geographical range. This range stretches from Western Europe to China and North and South Korea, and also contains endemic species/subspecies, e.g. the Corsican nuthatch, Sitta whiteheadi Sharpe. (Bellamy et al., 1998).
Because of the problem of cryptic species and the potential introgression and hybridisation with related species (e.g. cf. Smith, 2014, in the case of Danaiid butterflies, Danaus chrysippus (L.) sensu lato (Lepidoptera: Nymphalidae: Danaus) species complexes in East Africa), it is not necessarily easy to compare the dietary breadth of various insect species in relation to geographical displacement, although a few examples do certainly exist. This is above and beyond morphological and genetic differentiation related to host plant adaptation, of which there are very many examples (Bernays, 1991; Forister et al., 2015; Rasmussen et al., 2020), notably in aphids (Insecta: Hemiptera: Aphididae): for example, the different chromosomal forms of the corn leaf aphid, Rhopalosiphum maidis Fitch on sorghum and maize, respectively (Blackman & Eastop, 2000; Brown & Blackman, 1988). Since maize is more commonly grown in North America than in Europe (although its acreage is yearly increasing; Kelly, 2019), there is likely to be ecological separation of the two karyotypic forms.
It is also known that other aphid species, notably the pea aphid, Acyrthosiphon pisum Harris, has sympatric host-adapted races on different species of Fabaceae related to divergence in both behaviour and genetics (Frantz et al, 2006). The aphid displays colour races, pink and green, which show host plant preference (Via, 1999; Via et al., 2000), a differential response to predator and parasitoid pressure (Bálint et al., 2018), whilst these forms seemingly show differences related to their facultative or secondary endosymbiotic bacterial communities (Leclair et al., 2021). Recent research has also shown that R. maidis infesting maize grown in chemically treated (pesticides and herbicides) and untreated fields have a different facultative endosymbiont profile (Csorba et al., 2022). Since these endosymbionts, both the primary one (Buchnera aphidicola) and the secondary ones, which live in special cells called mycetocytes (bacteriocytes) in the body cavity of the aphid host adjacent to the ovarioles, are involved in supplying nutrients to the host aphid (essential amino acids) and vitamins, as well as protection against primary parasitic wasps (Hymenoptera: Braconidae) (cf. Leclair et al., 2021 and references therein), these factors are very likely to lead to host adaptation and hence geographic host partitioning over different spatial scales.
There are several mechanisms that divide species into different populations. One such is the well-known and well-studied existence of hybrid zones which cause partitioning of species into effectively different cryptic populations. These divisions are sometimes based on karyotypic differences and, in turn, related to chromosomal fissions, fusions and translocations, the process occurring both allopatrically as well as sympatrically e.g. grasshoppers of the genus Vandiemenella (the viatica species group) (Kawakami et al., 2011). Other lines of species population demarcation exist and are more ambiguous. For example, as reviewed in Loxdale and Lushai (2001), in North America, populations of two noctuid moth species, the native hop vine borer, Hydraecia immanis (Guenée), and the introduced potato stem borer, H. micacea Esper are separated by host range differences found along the Great Lakes ecotone in Wisconsin and Michigan (Scriber et al., 1992). Accordingly,
“...the former species is more specialised in its diet and generally occurs south of the plant community transition zone, whereas the latter species is polyphagous and occurs in corn north of this zone. Of 19 enzyme loci screened, 6 showed fixed or nearly fixed allelic differences.” Hybridisation of the two Hydraecia species “may contribute to wider larval host plant use abilities, altered voltinism patterns, reduced developmental temperature thresholds of the larva and possibly changes in oviposition preferences” (cf. Scriber et al., 1992 and references therein).
Here migration and subsequent colonisation of either species into adjacent ecosystems is restricted, unless moths can adapt to the new ecological challenges presented.
Another famous example of such spatial distribution of cryptic populations of insects concerns dipterous flies of the North American tephritid genus Rhagoletis Loew., more especially R. pomonella (Walsh). The fly has been well studied by Jeffrey Feder and colleagues over many years because of the insect’s host switching from its native plant host hawthorn (Crataegus spp.) to apple (Malus spp.) in the last few centuries, probably since European colonisation of the North American continent and widespread establishment of domestic apple trees (Colleary, 2011). This host switch occurs sympatrically (Feder & Forbes, 2010; Feder et al., 1998) and is governed by attraction to host plant volatiles (Linn et al., 2003, 2004). There is known to be some degree of genetic divergence between the two host plant-adapted races on hawthorn and apple (Feder & Forbes, 2010; see below). Even, so, and most intriguingly, the transition of one host form to another is not absolute in terms of large spatial scales. Thus.
“.... populations of R. pomonella showed geographic differentiation. Populations from the eastern USA, the native range of the fly, were highly polymorphic and showed homogeneity within and among populations. In contrast, populations from the western USA where it has colonised recently (c. 1980), generally lacked genetic variation (McPheron, 1990), although at the remaining polymorphic loci, showed spatial heterogeneity suggestive of founder events. Other data by Feder et al. (1990) (6 enzyme loci) also showed that flies (R. pomonella) not only differed at loci between hosts (apple or hawthorn) but that allele frequencies were influenced by latitude. This produced a north-south cline in the eastern USA and Canada superimposed on inter-host patterns. It appears that adult flies do not migrate between host species, but rather, tend to infest the same host that they colonised as larvae.” (Reviewed in Loxdale & Lushai, 2001).
Hence, the distribution of the host-adapted forms is not homogenous over its geographical range in North America; rather it shows clear cryptic differentiation into host related, genetically-separated sub-populations.
Such divergence, including the establishment of clinal populations, is well known in other species, including north–south clines in aphids (Llewellyn et al., 2003; Simon et al., 1999). In the case of aphids, this latitudinal change is related to life cycle polymorphisms, e.g. the bird cherry-oat aphid, Rhopalosiphum padi (L.) (Martinez et al., 1997).
Another good example of a lack of genetic homogeneity within a geographic population of one apparently ‘good’ species, i.e. in terms of genetic identity, concerns the Fall Armyworm moth, Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae). This moth is a major pest of cash crops in eastern and central North America and South America. In the mid-1980s, the moth was shown to display host plant associated genetic variation (Pashley, 1986; Pashley et al., 1985). Two main strains were identified using molecular electrophoretic diagnostic markers (esterase allozyme and protein) such that a corn strain feeding principally on maize, sorghum, and cotton could be discriminated from a rice strain attacking rice and other grasses. Later, DNA-based markers were developed to distinguish the strains. The discovery of up to 16% hybridisation in the field suggested that these host-adapted forms might be incipient rather than fully differentiated species (Groot et al., 2010). According to Groot et al., despite the high level of hybridisation between the strains where they co-occur, even so the two strains are found throughout North and South America and that.
“Apparently, there is an array of reproductive isolating barriers active throughout the Western Hemisphere that prevents these strains from merging into one panmictic population. Apart from habitat isolation, two behavioural isolation mechanisms may contribute to reproductive isolation in S. frugiperda: differences in the female pheromone composition.... and differential timing of reproductive activity at night.” Furthermore, as they argue: “No intrinsic hybrid unviability has been found for the two host strains (unpubl. res.), but extrinsic ecological or behavioural sterility may affect overall reproductive isolation.” (cf. Groot et al., 2010 for further details and references therein).
In an earlier paper, Groot et al. (2009), examining moths taken from 5 geographical areas in eastern North America and Mexico,
“.....found significant variation in the ratios of sex pheromone blend components as well as in male response, not only between geographic regions but also within a region between consecutive years.” They also observed temporal variation “of a similar magnitude as the geographic variation.”
They concluded that the geographic variation.
“...seems to at least partly be controlled by genetic factors, and to be correlated with the quality of the local chemical environment. However, the pattern of temporal variation within populations suggests that optimization of the [sex] pheromonal signal also may be driven by within-generation physiological adjustments by the moths in response to their experience of the local chemical environment.”
So a complex ecological scenario is clearly at work here, involving both innate genetic and physiological factors in concert with prevailing environmental ones, including climate and soil chemistry. Whatever the exact factors governing the clear geographic separation of the two strains, the population of S. frugiperda sensu lato cannot be treated as a single, genetically and physiologically homogenous entity. This, in turn, means that the overall diet breadth of the moth cannot be taken as representing a meaningful sample of its true dietary range, since two distinct host-adapted, sometimes sympatric, populations are in evidence.
Discussion
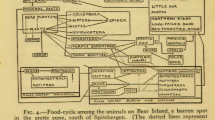
This brief and far from extensive array of examples as aforementioned shows clearly that some, and possibly many, animal species are not homogenous genetically over their geographical range. Rather, they present a heterogeneous, often cryptic population genetic structuring, including spatial patterning, sometimes as latitudinal clines in gene frequency, including life style and lifecycle forms. This means, especially in relation to the topic of generalism, that this inaccurate and misleading term should be abandoned in favour of levels of phagy, ranging from mono, oligo through to polyphagy, the latter including where appropriate, the broad ancillary term omnivorous. With aphids showing clinal changes in lifecycle reproductive forms, the species in question often also shows host preference changes, alternating from a spring and summer herbaceous host or hosts (e.g. Brassicales or Poaceae), to a primary overwintering host, (e.g. Rosaceae), on which cold hardy overwintering eggs are laid in the autumn following mating of the sexual forms on this latter host, i.e. wingless sexual females (oviparae) and males, these usually winged, but not exclusively so, depending on species or race (cf. Blackman, 2010; Blackman & Eastop, 2000; Dixon, 1998; Moran, 1992; Peccoud & Simon, 2010; Peccoud et al., 2009a, 2009b). To emphasise this point further, the peach-potato aphid, Myzus persicae (Sulzer) (Fig. 4), which is apparently highly polyphagous on plants from 40 families, mostly its herbaceous spring and summer secondary plant hosts (Blackman & Eastop, 2000) (but again may in fact be an array of cryptic, host-adapted species or subspecific entities; Loxdale & Balog, 2018), is virtually monophagous on its primary host, principally peach, Prunus persica (L.) Batsch (Blackman, 2010; Blackman & Eastop, 2000; Tatchell et al., 1983). Hence, these various species cannot be descried as generalist but rather, as is our current view (Loxdale & Harvey, 2019), are universally specialist to some degree, often highly so, and often co-evolved with their host (plant or animal in the case of parasites/parasitoids) or prey, whatever this may be (Thompson, 1994).
Peach-potato aphid, Myzus persicae; apterous (wingless) individuals with nymphs (© R.D. Dransfield & R. Brightwell, influentialpoints.com, reproduced with permission). The species is reportedly highly polyphagous on a large number of plant species, notably secondary herbaceous spring and summer hosts, but virtually monophagous on its primary overwintering host, Peach, Prunus persica, on which the sexual forms mate and the females (ovipara) lay overwintering eggs. Clearly, this is not a so-called generalist aphid, host alternating as it does between polyphagous and monophagous phases of its life cycle, i.e. asexual (on the secondary host/s) and sexual (on the primary woody host). Instead, it is highly specialised in terms of its life style (including morphology and behaviour) and lifecycle, which in turn impacts on its diet breadth. Aphids generally do significantly better in terms of reproductive success on the natal plants on which they were reared (e.g. Via, 1999, but see also McLean et al., 2009 and references therein)
The fact that many species populations are subject to hybridisation/introgression events (e.g. Mallet, 2007; Smith, 2014; Zolotareva et al., 2021) and that many, if not most, species have never been examined using high resolution molecular markers in terms of their population genetic fidelity and identity (which is/are anyway undergoing adaptive/evolutionary changes of one kind and another, perhaps rapidly, as found with many insect species, especially pests; Loxdale, 2010), only adds to the doubts and confusion surrounding what may be claimed to be a single, homogenous species population, consuming one group or groups of food stuffs over its entire geographic range. This is especially so in considerations of spatial scale, ranging from micro- (e.g. plant level), to local (including island populations; Williamson, 1981), to large geographic areas, hundreds of kilometres in extent.
From a community ecological perspective, where the interaction of animals and plants with one another is often complex and to date not well explored (as is true in many species, especially insects, and especially those in the tropics, the centre of biodiversity and interacting webs of species), then caution should be exercised in labelling such animals ‘generalist’. Finally, it is worth repeating the mantra, as my wife Nicola argues, “If generalism existed, evolution would effectively stop, since it is the process of specialism that governs evolutionary adaptations and population divergence, by whatever means (allopatric, sympatric, parapatric)”. (Nicola von Mende-Loxdale, cited in Loxdale, 2021). The fossil record alone (e.g. Penney & Jepson, 2014 in the case of insects) shows that indeed specialism was—and still very much is—the prevailing dietary-ecological state, and has been as far as we can ascertain for hundreds of millions of years, probably since life first appeared on our planet.
Conclusions
Because natural populations are rarely homogeneously structured, either as a result of climate, or genetically, due to various mutational forces such as chromosomal changes, and also hybridisation and introgression, and clinal changes in gene frequencies, natural populations can never be unequivocally described as generalist. Rather, such populations may be heterogeneously structured, involving cryptic population changes, even up to the level of species.Footnote 2 Even without the occurrence of cryptic speciation events, the anatomy-morphology of the animal in question often gives vital clues as to its diet, especially beaks in birds and jaws and teeth in mammals, and general skeleton in both groups. In light of these aspects and concerns, in the absence of direct genetic testing using high resolution molecular markers, it is better to verge on the side of caution, and label such organisms with regard to their apparent level of phagy only.
We have here presented several examples from two speciose groups of animals, avian and insect, where population genetic homogeneity or demographic homogeneity at large spatial scales is clearly absent, and from this standpoint, are hence loath to use the term ‘generalist’ when describing such species populations or subspecific ones. In the end, this whole debate essentially resolves around the fact that nature is often much more complex than we may at first assume it to be; that because of mutation, adaptation and evolution, a population is not necessarily just one thing, but may comprise several things or indeed even many things, i.e. different cryptic populations existing below the subspecific level. These can be formed by various selective processes (positive directional selection or genetic drift) and due to various genetic mechanisms and forces, either allopatrically, parapatrically or sympatrically. This is the nature of nature, with dynamic ecological-genetic changes an ever present reality or likelihood.
Notes
A good example of how weird such novel adaptations can be is surely the Aardwolf, Proteles cristata Sparrman (Carnivora: Feliformia: Hyaenidae: Protelinae), a highly specialised insectivorous hyena native to East and South Africa that feeds mainly on ants and termites using its long sticky tongue, and which seemingly evolved from typical hyena stock some 10 million years ago (Koepfli et al., 2006).
A ‘classic’ example of this concerns the East African bush babies or galagos (Mammalia: Primates: Strepsirrhini: Lorisoidea: Galagidae) where, following examination of vocalisation patterns, hand prints, and other morphological characters, the hitherto anomalous behaviour and spatial distribution of the animals (vertical within the forest canopy) could finally be understood, i.e. it was found that the assumed homogeneous species populations studied actually comprised an array of cryptic species (cf. Anderson, 1998a, 1998b, Bearder et al., 1995 and Ceballos & Ehrlich, 2009 for further details).
References
Amarasekare, P. (2003). Competitive coexistence in spatially structured environments: A synthesis. Ecology Letters, 6, 1109–1122. https://doi.org/10.1046/j.1461-0248.2003.00530.x
Anderson, M. (1998a). Comparative morphology and speciation in galagos. Folia Primatologica, 69(supplement), 325–331. https://doi.org/10.1159/000052721
Anderson, M. (1998b). The use of hand morphology in the taxonomy of galagos. Primate Eye, 63, 28. (Abstract)
Avise, J. C. (1994). Molecular markers, natural history and evolution. Chapman & Hall.
Bálint, J., Benedek, K., Loxdale, H. D., Kovács, E., Ábrahám, B., & Balog, A. (2018). How host plants and predators influence pea aphid (Acyrthosiphon pisum Harris) populations in a complex habitat. North-West Journal of Zoology, 14(149–158), e171103.
Balmer, D. (2013). Bird atlas 2007–11.https://www.bto.org/our-science/projects/birdatlsa
Bearder, S. K., Honess, P. E., & Ambrose, L. (1995). Species diversity among galagos with special reference to mate recognition. In L. Alterman, M. K. Izard, & G. A. Doyle (Eds.), Creatures of the dark: The nocturnal prosimians (pp. 331–352). New York: Plenum Press.
Bellamy, P. E., Brown, N. J., Enoksson, B., Firbank, L. G., Fuller, R. J., Hinsley, S. A., & Schotman, A. G. M. (1998). The influences of habitat, landscape structure and climate on local distribution patterns of the nuthatch (Sitta europaea L.). Oecologia, 115, 127–136. https://doi.org/10.1007/s004420050499
Bernays, E. A. (1991). Evolution of insect morphology in relation to plants. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences, 333, 257–264. https://doi.org/10.1098/rstb.1991.0075
Blackman, R. L., & Eastop, V. F. (2000). Aphids on the World’s Crops: An identification and information guide (2nd ed., p. 476). Chichester: John Wiley & Sons Ltd.
Blackman, R.L (2010). Aphids - Aphidinae (Macrosiphini). Handbooks for the Identification of British Insects (Volume 2, Part 7). Royal Entomological Society, Field Studies Council, Shrewsbury
Bojarska, K., & Selva, N. (2012). Spatial patterns in brown bear Ursus arctos diet: The role of geographical and environmental factors. Mammal Review, 42, 120–143. https://doi.org/10.1111/j.1365-2907.2011.00192.x
Bowman, J., Donovan, D., & Rosatte, R. C. (2006). Numerical response of fishers to synchronous prey dynamics. Journal of Mammalogy, 87, 480–484. https://doi.org/10.1644/05-MAMM-A-202R2.1
Brown, P. A., & Blackman, R. L. (1988). Karyotype variation in the corn leaf aphid, Rhopalosiphum maidis (Fitch), species complex (Hemiptera: Aphididae) in relation to host-plant and morphology. Bulletin of Entomological Research, 78, 351–363. https://doi.org/10.1017/S0007485300013110
Burger, C., Belskii, E., Eeva, T., Laaksonen, T., Mägi, M., Mänd, R., Qvarnström, A., Slagsvold, T., Veen, T., Visser, M. E., Wiebe, K. L., Wiley, C., Wright, J., & Both, C. (2012). Climate change, breeding date and nestling diet: how temperature differentially affects seasonal changes in pied flycatcher diet depending on habitat variation. Journal of Animal Ecology, 81, 926–936. https://doi.org/10.1111/j.1365-2656.2012.01968.x
Caryl, F. M., Raynor, R., Quine, C. P., & Park, K. J. (2012). The seasonal diet of British pine marten determined from genetically identified scats. Journal of Zoology, 288, 252–259. https://doi.org/10.1111/j.1469-7998.2012.00951.x
Ceballos, G., & Ehrlich, P. R. (2009). Discoveries of new mammal species and their implications for conservation and ecosystem services. Proceedings of the National Academy of Sciences of the United States of America. 106, 3841–3846. https://doi.org/10.1073/pnas.0812419106
Colleary, E. (2011). Apples in America: a very brief history. The American Table. http://www.americantable.org/2011/10/apples-in-america/
Colwell, R. K., & Rangel, T. F. (2009). Hutchinson’s duality: The once and future niche. Proceedings of the National Academy of Sciences of the United States of America, 106, 19651–19658. https://doi.org/10.1073/pnas.0901650106
Coyne, J. A., & Orr, H. A. (2004). Speciation (p. 545). Sunderland: Sinauer Associates Inc.
Csorba, A. B., Fora, C. G., Bálint, J., Felföldi, T., Szabó, A., Máthé, I., Loxdale, H. D., Kentelky, E., Nyárádi, I.-I., & Balog, A. (2022). Endosymbiotic bacterial diversity of Corn Leaf Aphid, Rhopalosiphum maidis Fitch (Hemiptera: Aphididae) associated with maize management systems. Microorganisms, 10(939), 12. https://doi.org/10.3390/microorganisms10050939
Dixon, A. F. G. (1998). Aphid ecology (2nd ed., p. 300). Chapman & Hall.
Feder, J. L., Berlocher, S. H., & Opp, S. B. (1998). Sympatric host race formation and speciation in Rhagoletis (Diptera: Tephritidae): A tale of two species for Charles D. In S. Mopper & S. Y. Strauss (Eds.), Genetic structure and local adaptation in natural insect populations (pp. 408–441). Chapman & Hall.
Feder, J. L., Chilcote, C. A., & Bush, G. L. (1990). The geographic pattern of genetic differentiation between host associated populations of Rhagoletis pomonella (Diptera: Tephritidae) in the eastern United States and Canada. Evolution, 44, 570–594. https://doi.org/10.1111/j.1558-5646.1990.tb05939.x
Feder, J. L., & Forbes, A. (2010). Sequential speciation and the diversity of parasitic insects. Ecological Entomology, 35(Suppl. 1), 67–76. https://doi.org/10.1111/j.1365-2311.2009.01144.x
Forister, M. L., Novotny, V., Panorska, A. K., Baje, L., Basset, Y., Butterill, P. T., Cizek, L., Coley, P. D., Dem, F., Diniz, I. R., & Drozd, P. (2015). The global distribution of diet breadth in insect herbivores. Proceedings of the National Academy of Sciences of the United States of America, 112, 442–447. https://doi.org/10.1073/pnas.142304211
Fox, L. R., & Morrow, P. A. (1981). Specialization: species property or local phenomenon? Science, 211, 887–891. https://doi.org/10.1126/science.211.4485.887
Frantz, A., Plantegenest, M., Mieuzet, L., & Simon, J. -C. (2006). Ecological specialization correlates with genotypic differentiation in sympatric host-populations of the pea aphid. Journal of Evolutionary Biology, 19, 392–401. https://doi.org/10.1111/j.1420-9101.2005.01025.x
Gaston, K. J. (1991). The magnitude of global insect species richness. Conservation Biology, 5(3), 283–296. https://doi.org/10.1111/j.1523-1739.1991.tb00140.x
Grimaldi, D., & Engel, M. S. (2005). Evolution of the insects (p. 772). Cambridge University Press.
Groot, A. T., Inglis, O., Bowdridge, S., Santangelo, R. G., Blanco, C., López, J. D., Vargas, A. T., Gould, F., & Schal, C. (2009). Geographic and temporal variation in moth chemical communication. Evolution, 63, 1987–2003. https://doi.org/10.1111/j.1558-5646.2009.00702.x
Groot, A. T., Marr, M., Heckel, D. G., & Schöfl, G. (2010). The roles and interactions of reproductive isolation mechanisms in fall armyworm (Lepidoptera: Noctuidae) host strains. Ecological Entomology, 35(Suppl. 1), 105–118. https://doi.org/10.1111/j.1365-2311.2009.01138.x
Hanmer, H. (2016). Nest predation in urban gardens: Does feeding birds increase local nest predation? British Ornithologists’ Union Blog, https://bou.org.uk/blog-hanmer-garden-nest-predation/
Hutchinson, G. E. (1959). Homage to Santa Rosalia or why are there so many kinds of animals? The American Naturalist, 93, 145–159.
Kawakami, T., Butlin, R. K., & Cooper, S. J. B. (2011). Chromosomal speciation revisited: modes of diversification in Australian morabine grasshoppers (Vandiemenella, viatica species group). Insects, 2, 49–61. https://doi.org/10.3390/insects2010049
Kelly, P. (2019). The EU cereals sector: Main features, challenges and prospects. https://www.europarl.europa.eu/RegData/etudes/BRIE/2019/640143/EPRS_BRI(2019)640143_EN.pdf
Koepfli, K.-P., Jenks, S. M., Eizirik, E., Zahirpoura, T., Van Valkenburgh, B., & Wayne, R. K. (2006). Molecular systematics of the Hyaenidae: Relationships of a relictual lineage resolved by a molecular supermatrix. Molecular Phylogenetics and Evolution, 38, 603–620. https://doi.org/10.1016/j.ympev.2005.10.017
Leclair, M., Buchard, C., Mahéo, F., Simon, J.-C., & Outreman, Y. (2021). A link between communities of protective endosymbionts and parasitoids of the pea aphid revealed in unmanipulated agricultural systems. Frontiers in Ecology & Evolution, 9(618331), 14. https://doi.org/10.3389/fevo.2021.618331
Linn, C. E., Darnbroski, H. R., Feder, J. L., Berlocher, S. H., Nojima, S., & Roelofs, W. L. (2004). Postzygotic isolating factor in sympatric speciation in Rhagoletis flies: Reduced response of hybrids to parental host-fruit odors. Proceedings of the National Academy of Sciences of the United States of America, 101, 17753–17758. https://doi.org/10.1073/pnas.040825510
Linn, C., Feder, J. L., Nojima, S., Dambroski, H. R., Berlocher, S. H., & Roelofs, W. (2003). Fruit odor discrimination and sympatric host race formation in Rhagoletis. Proceedings of the National Academy of Sciences of the United States of America, 100, 11490–11493. https://doi.org/10.1073/pnas.163504910
Llewellyn, K. S., Loxdale, H. D., Harrington, R., Brookes, C. P., Clark, S. J., & Sunnucks, P. (2003). Migration and genetic structure of the grain aphid (Sitobion avenae) in Britain related to climatic adaptation and clonal fluctuation revealed using microsatellites. Molecular Ecology, 12, 21–34. https://doi.org/10.1046/j.1365-294X.2003.01703.x
Loxdale, H. D., & Lushai, G. (2001). Use of genetic diversity in movement studies of flying insects. In I. P. Woiwod, D. R. Reynolds, & C. D. Thomas (Eds.), Insect movement: mechanisms and consequences. Royal entomological society 20th international symposium volume (pp. 361–386). Wallingford: CABI.
Loxdale, H. D. (2010). Rapid genetic changes in natural insect populations. Ecological Entomology, 35, 155–164. https://doi.org/10.1111/j.1365-2311.2009.01141.x
Loxdale, H. D., & Harvey, J. A. (2016). The ‘generalism’ debate: misinterpreting the term in the empirical literature focusing on dietary breadth in insects. Biological Journal of the Linnean Society, 119, 265–282. https://doi.org/10.1111/bij.12816
Loxdale, H. D., & Balog, A. (2018). Aphid specialism as an example of ecological-evolutionary divergence. Biological Reviews, 93, 642–657. https://doi.org/10.1111/brv.12361
Loxdale, H. D., Balog, A., & Harvey, J. A. (2019). Generalism in nature - the great misnomer: Aphids and wasp parasitoids as examples. Insects, 10, 314. https://doi.org/10.3390/insects10100314
Loxdale, H. D. (2018). Aspects, including pitfalls, of temporal sampling of flying insects, with special reference to aphids. Insects, 9(153), 21. https://doi.org/10.3390/insects9040153
Loxdale, H. D. (2021). Hanging onto your crocodile and hippo gods; conservatism in science, including entomology. Antenna (Journal of the Royal Entomological Society), 45(2), 78–80.
Loxdale, H. D., Balog, A., & Biron, D. G. (2020). Aphids in focus: Unravelling their complex ecology and evolution using genetic and molecular approaches. Biological Journal of the Linnean Society, 129, 507–531. https://doi.org/10.1093/biolinnean/blz194
Loxdale, H. D., Davis, B. J., & Davis, R. A. (2016). Known knowns and unknowns in biology. Biological Journal of the Linnean Society, 117, 386–398. https://doi.org/10.1111/bij.12646
Loxdale, H. D., Lushai, G., & Harvey, J. A. (2011). The evolutionary improbability of ‘generalism’ in nature, with special reference to insects. Biological Journal of the Linnean Society, 103, 1–18. https://doi.org/10.1111/j.1095-8312.2011.01627.x
MacPherson, J. & Wright, P. (2021). Long-term strategic recovery plan for pine martens in Britain. Vincent Wildlife Trust, pp. 72, https://www.vwt.org.uk/wp-content/uploads/2021/07/Pine-Marten-Recovery-Plan-VWT-10June2021.pdf
Mallet, J. (2007). Hybrid speciation. Nature, 446, 279–283. https://doi.org/10.1038/nature05706
Martinez-Torres, D., Moya, A., Hebert, P. D. N., & Simon, J. -C. (1997). Geographic distribution and seasonal variation of mitochondrial DNA haplotypes in the aphid Rhopalosiphum padi (Hemiptera: Aphididae). Bulletin of Entomological Research, 87, 161–167. https://doi.org/10.1017/S0007485300027309
McLean, A. H. C., Ferrari, J., & Godfray, H. C. J. (2009). Effects of the maternal and pre-adult host plant on adult performance and preference in the pea aphid, Acyrthosiphon pisum. Ecological Entomology, 34, 330–338. https://doi.org/10.1111/j.1365-2311.2008.01081.x
McPheron, B. A. (1990). Genetic structure of apple maggot fly (Diptera: Tephritidae) populations. Annals of the Entomological Society of America, 83, 568–577. https://doi.org/10.1093/aesa/83.3.568
Moran, N. A. (1992). The evolution of life cycles in aphids. Annual Review of Entomology, 37, 321–348. https://doi.org/10.1146/annurev.en.37.010192.001541
Munro, R. H. M., Nielsen, S. E., Price, M. H., Stenhouse, G. B., & Boyce, M. S. (2006). Seasonal and diel patterns of grizzly bear diet and activity in west-central Alberta. Journal of Mammalogy, 87, 1112–1121. https://doi.org/10.1644/05-MAMM-A-410R3.1
Nicolau, P. G., Burgess, M. D., Marques, T. A., Baillie, S. R., Moran, N. J., Leech, D. I., & Johnston, A. (2021). Latitudinal variation in arrival and breeding phenology of the pied flycatcher Ficedula hypoleuca using large-scale citizen science data. Journal of Avian Biology. https://doi.org/10.1111/jav.02646
Pashley, D. P. (1986). Host-associated genetic differentiation in Fall Armyworm (Lepidoptera: Noctuidae): a sibling species complex? Annals of the Entomological Society of America, 79, 898–904. https://doi.org/10.1093/aesa/79.6.898
Pashley, D. P., Johnson, S. J., & Sparks, A. N. (1985). Genetic population structure of migratory moths: The fall armyworm (Lepidoptera: Noctuidae). Annals of the Entomological Society of America, 78, 756–762. https://doi.org/10.1093/aesa/78.6.756
Peccoud, J., Ollivier, A., Plantegenest, M., & Simon, J.-C. (2009a). A continuum of genetic divergence from sympatric host races to species in the pea aphid complex. Proceedings of the National Academy of Sciences of the United States of America, 106, 7495–7500. https://doi.org/10.1073/pnas.081111710
Peccoud, J., Simon, J.-C., McLaughlin, H. J., & Moran, N. A. (2009b). Post-Pleistocene radiation of the pea aphid complex revealed by rapidly evolving endosymbionts. Proceedings of the National Academy of Sciences of the United States of America, 106, 16315–16320. https://doi.org/10.1073/pnas.0905129106
Peccoud, J., & Simon, J.-C. (2010). The pea aphid complex as a model of ecological speciation. Ecological Entomology, 35, 119–130. https://doi.org/10.1111/j.1365-2311.2009.01147.x
Penney, D., & Jepson, J. E. (2014). Fossil insects: An introduction to palaeoentomology (p. 222). Rochdale: Siri Scientific Press.
Posłuszny, M., Pilot, M., Goszczyński, J., & Gralak, B. (2007). Diet of sympatric pine marten (Martes martes) and stone marten (Martes foina) identified by genotyping of DNA from faeces. Annales Zoologici Fennicii, 44, 269–284.
Pullar, P. (2020). The return of the Taghan. https://www.walkhighlands.co.uk/news/the-return-of-the-taghan/
Putman, R. J. (2000). Diet of pine martens Martes martes L. in west Scotland. Journal of Natural History, 34, 793–797. https://doi.org/10.1080/002229300299426
Rasmussen, C., Engel, M. S., & Vereecken, N. J. (2020). A primer of host-plant specialization in bees. Emerging Topics in Life Sciences, 4, 7–17. https://doi.org/10.1042/ETLS20190118
Reitz, S. R., & Trumble, J. T. (2002). Competitive displacement among insects and arachnids. Annual Review of Entomology, 47, 435–465. https://doi.org/10.1146/annurev.ento.47.091201.145227
Richmond, C. E., Breitburg, D. L., & Rose, K. A. (2005). The role of environmental generalist species in ecosystem function. Ecological Modelling, 188, 279–295. https://doi.org/10.1016/j.ecolmodel.2005.03.002
Scriber, J. M., Bossart, J. L., & Snider, D. (1992). Diagnostic alleles from electrophoresis distinguish two noctuid pest species, Hydraecia immanis and H. micacea (Lepidoptera: Noctuidae). The Great Lakes Entomologist, 25, 91–98.
Sheehy, E., Sutherland, C., O’Reilly, C., & Lambin, X. (2018). The enemy of my enemy is my friend: Native pine marten recovery reverses the decline of the red squirrel by suppressing grey squirrel populations. Proceedings of the Royal Society B: Biological Sciences, 285, 2017–2603. https://doi.org/10.1098/rspb.2017.2603
Shipley, L. A., Forbey, J. S., & Moore, B. D. (2009). Revisiting the dietary niche: When is a mammalian herbivore a specialist? Integrative and Comparative Biology, 49, 274–290. https://doi.org/10.1093/icb/icp051
Simon, J.-C., Baumann, S., Sunnucks, P., Hebert, P. D. N., Pierre, J.-S., Le Gallic, J.-F., & Dedryver, C.-A. (1999). Reproductive mode and population genetic structure of the cereal aphid Sitobion avenae studied using phenotypic and microsatellite markers. Molecular Ecology, 8, 531–545. https://doi.org/10.1046/j.1365-294x.1999.00583.x
Slobodkin, L. B. (1993). An appreciation: George Evelyn Hutchinson. Journal of Animal Ecology, 62, 390–394. https://doi.org/10.2307/5370
Smith, D. A. S. (2014). African Queens and their kin: A Darwinian Odyssey (p. 810). Taunton: Brambleby Books Ltd.
Stork, N. E., McBroom, J., Gely, C., & Hamilton, A. J. (2015). New approaches narrow global species estimates for beetles, insects, and terrestrial arthropods. Proceedings of the National Academy of Sciences of the United States of America, 112, 7519–7523. https://doi.org/10.1073/pnas.15024081
Tatchell, G. M., Parker, S. J., & Woiwod, I. P. (1983). Synoptic monitoring of migrant insect pests in Great Britain and western Europe IV. Host plants and their distribution for pest aphids in Great Britain. Annual Report of Rothamsted Experimental Station, 2, 45–159.
Thébault, E., & Loreau, M. (2003). Food-web constraints on biodiversity–ecosystem functioning relationships. Proceedings of the National Academy of Sciences of the United States of America, 100, 14949–14954. https://doi.org/10.1073/pnas.2434847100
Thompson, J. N. (1994). The coevolutionary process (p. 376). University of Chicago Press.
Twining, J. P., Montgomery, W. I., & Tosh, D. G. (2020). The dynamics of pine marten predation on red and grey squirrels. Mammalian Biology, 100, 285–293. https://doi.org/10.1007/s42991-020-00031-z
Via, S. (1999). Reproductive isolation between sympatric races of pea aphids. I. Gene flow restriction and habitat choice. Evolution, 53, 1446–1457. https://doi.org/10.1111/j.1558-5646.1999.tb05409.x
Via, S., Bouck, A. C., & Skillman, S. (2000). Reproductive isolation between divergent races of pea aphids on two hosts. II. Selection against migrants and hybrids in the parental environments. Evolution, 54, 1626–1637. https://doi.org/10.1111/j.0014-3820.2000.tb00707.x
White, M. J. D. (1978). Modes of speciation (p. 455). San Francisco: W.H. Freeman & Company.
Williamson, M. (1981). Island populations (p. 200). Oxford University Press.
Wray, A. K., Gratton, C., Jusino, M. A., Wang, J. J., Kochanski, J. M., Palmer, J. M., Banik, M. T., Lindner, D. L., & Peery, M. Z. (2022). Disease-related population declines in bats demonstrate non-exchangeability in generalist predators. Ecology and Evolution, 12(e8978), 13. https://doi.org/10.1002/ece3.8978
Zolotareva, K. I., Belokon, M. M., Belokon, Y. S., Rutovskaya, M. V., Hlyap, L. A., Starykov, V. P., Politov, D. V., Lebedev, V. S., & Bannikova, A. A. (2021). Genetic diversity and structure of the hedgehogs Erinaceus europaeus and Erinaceus roumanicus: Evidence for ongoing hybridization in Eastern Europe. Biological Journal of the Linnean Society, 132, 174–195. https://doi.org/10.1093/biolinnean/blaa135
Acknowledgements
We thank Dr Andrew Davis for kindly inviting us to write this article, my (HDL’s) wife Nicola von Mende-Loxdale, PhD for her critical comments on the first draft of the manuscript, and Mike Toms, Head of Communications at the BTO, for kindly allowing us to reproduce the bird UK distribution maps (from Bird Atlas 2007–11 (Balmer et al., 2013), which is a joint project between BTO, BirdWatch Ireland and the Scottish Ornithologists' Club), as shown in Figs. 1, 2, 3a, b, and Bob Dransfield & Bob Brightwell for kindly allowing us to use their excellent colour photo of Myzus persicae.
Funding
No funding bodies were involved during the writing and preparation of the manuscript, which we both contributed to.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Loxdale, H.D., Harvey, J.A. Generalism in nature: a community ecology perspective. COMMUNITY ECOLOGY 24, 113–125 (2023). https://doi.org/10.1007/s42974-022-00130-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42974-022-00130-6