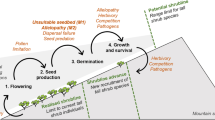
Abstract
Community assembly experiments in ecology provide a means to contrast abiotic and biotic influences that structure plant communities. Competition and facilitation are two common biotic predictors of success in plant communities. Abiotic limitations can include, but are not limited to, space, light, and water. Concurrent examination of spatial ecology of biotic and abiotic drivers with these interactions is critical to better understand plant invasion. Here, we test the relative importance of seed aggregation on the net outcome of interactions between a native desert annual Phacelia tanacetifolia and an invasive exotic species Bromus rubens. Our study proposes that seed aggregation can play a major role in determining whether an exotic species will co-exist or outcompete natives. We tested the ecological heteromyopia hypothesis that proposes interspecific competition is greater over shorter distances relative to intraspecific competition and that distance between species can thus lead to co-existence. We used competition trials with seeds and mimic plants via clumped and dispersed arrangements to test this hypothesis for one invasive and one native annual species. Monoculture and mixed seed trials were done under controlled greenhouse conditions for both species. To ensure findings that were not solely driven by physical interference or space constraints in pots, a plastic exotic mimic plant was used in a mixture treatment with natives to enable a contrast between biotic competition and space effects. Both P. tanacetifolia and B. rubens has significantly greater establishment and final abundances in monocultures with spatially clumped seeds. Dispersed seeds of B. rubens in monocultures had significantly lower establishment than the clumped sowing. Bromus rubens experienced a significant negative response to P. tanacetifolia in mixtures of clumped seeds. Additionally, the biomass of B. rubens was significantly higher in monocultures of clumped arrangement and mixtures of dispersed arrangement. The native P. tanacetifolia did not respond to the physical interference of the 3-D-printed plastic mimic brome invader. Light was also not a significant covariate in any of the competition trials. This pilot study suggests that seed aggregation is a potential key predictor of success for both the native and exotic species studied here in monocultures. Thus, the heteromyopia hypothesis was supported for these two dryland annual plant species because interspecific competition decreased as space and thus distance between individuals increased. This study highlights the importance of spatial structure of seeds in the seedbanks and seedlings in the emergent plant community in describing potential community assembly. Restoration efforts can consider seed sowing designs to decrease invasion by exotic species and increase native plant abundance by aggregating seeds at fine spatial scales.


Similar content being viewed by others
References
Abella, S. R., Craig, D. J., Chiquoine, L. P., Prengaman, K. A., Schmid, S. M., & Embrey, T. M. (2011). Relationships of native desert plants with red brome (Bromus rubens): Toward identifying invasion-reducing species. Invasive Plant Science and Management, 4(1), 115–124. https://doi.org/10.1614/ipsm-d-10-00013.1
Adler, F. R. (2010). The effects of intraspecific density dependence on species richness and species abundance distributions. Theoretical Ecology. https://doi.org/10.1007/s12080-010-0108-7
Aguiar, M. R., Soriano, A., & Sala, O. E. (1992). Competition and facilitation in the recruitment of seedlings in patagonian steppe. Functional Ecology, 6(1), 66. https://doi.org/10.2307/2389772
Amarasekare, P. (2002). Interference competition and species coexistence. In: Proceedings of the Royal Society of London. Series B: Biological Sciences, 269(1509), 2541–2550. https://doi.org/10.1098/rspb.2002.2181
Arredondo-Núñez, A., Badano, E., & Bustamante, R. (2009). How Beneficial are nurse plants? A meta-analysis of the effects of cushion plants on high-andean plant communities. Community Ecology, 10(1), 1–6. https://doi.org/10.1556/ComEc.10.2009.1.1
Bainbridge, D. A. (1995). A Beginner’s Guide to Desert Reclamation/Desert Restoration. University of California Libraries.
Bakacsy, L. (2019). Invasion impact is conditioned by initial vegetation states. Community Ecology, 20(1), 11–19. https://doi.org/10.1556/168.2019.20.1.2
Beatley, J. C. (1966). Ecological status of introduced brome grasses (Bromus Spp.) in desert vegetation of southern Nevada. Ecology, 47(4), 548–554. https://doi.org/10.2307/1933931
Bertness, M. D. (1989). Intraspecific competition and facilitation in a northern acorn barnacle population. Ecology, 70(1), 257–268. https://doi.org/10.2307/1938431
Boerner, R. E. J., & Harris, K. K. (1991). Effects of collembola (arthropoda) and relative germination date on competition between mycorrhizalPanicum virgatum (Poaceae) and non-mycorrhizalBrassica nigra (Brassicaceae). Plant and Soil, 136(1), 121–129. https://doi.org/10.1007/bf02465227
Bolker, B. M., Pacala, S. W., & Neuhauser, C. (2003). Spatial dynamics in model plant communities: What do we really know? The American Naturalist, 162(2), 135–148.
Calflora.” n.d. https://www.calflora.org/
Callaway, R. M., & Walker, L. R. (1997). Competition and facilitation: A synthetic approach to interactions in plant communities. Ecology, 78(7), 1958–1965.
Carothers, J. H., Jaksić, F. M., & Jaksic, F. M. (1984). Time as a niche difference: The role of interference competition. Oikos, 42(3), 403. https://doi.org/10.2307/3544413
Chen, S. S. C., & Thimann, K. V. (1966). Nature of seed dormancy in phacelia tanacetifolia. Science, 153(3743), 1537–1539. https://doi.org/10.1126/science.153.3743.1537
Colautti, R. I., Grigorovich, I. A., & MacIsaac, H. J. (2006). Propagule pressure: A null model for biological invasions. Biological Invasions, 8, 1023–1037.
Cousens, R. D., Wiegand, T., & Taghizadeh, M. S. (2008). Small-scale spatial structure within patterns of seed dispersal. Oecologia. https://doi.org/10.1007/s00442-008-1150-7
Craig, T. P., Itami, J. K., & Price, P. W. (1990). Intraspecific competition and facilitation by a shoot-galling sawfly. The Journal of Animal Ecology, 59(1), 147. https://doi.org/10.2307/5164
Crawley, M., & May, R. (1987). Population dynamics and plant community structure: Competition between annuals and perrenials. Journal of Theoretical Biology, 125(4), 475–489. https://doi.org/10.1016/s0022-5193(87)80215-1
De Luis, M., Raventos, J., Wiegand, T., & Gonzalez-Hidalgo, J. C. (2008). Temporal and spatial differentiation in seedling emergence may promote species coexistence in Mediterranean fire-prone ecosystems. Ecography, 31(5), 620–629.
Dimitri, L. A., & Longland, W. S. (2017). Distribution of Western Juniper Seeds Across an Ecotone andImplications for Dispersal. Western North American Naturalist, 77(2), 212–222. https://doi.org/10.3398/064.077.0209.
Dong, K., Hao, G., Yang, N., Zhang, J.-L., Ding, X.-F., Ren, H.-Q., & Gao, Y.-B. (2019). Community assembly mechanisms and succession processes significantly differ among treatments during the restoration of Stipa grandis – Leymus chinensis communities. Scientific Reports. https://doi.org/10.1038/s41598-019-52734-0
Filazzola, A., & Lortie, C. J. (2014). A systematic review and conceptual framework for the mechanistic pathways of nurse plants: A systematic review of nurse-plant mechanisms. Global Ecology and Biogeography, 23(12), 1335–1345. https://doi.org/10.1111/geb.12202
Gallardo-Cruz, J., Meave, J., Pérez-García, E., & Hernández-Stefanoni. (2010). Spatial structure of plant communities in a complex tropical landscape: Implications for β-diversity. Community Ecology, 11(2), 202–210. https://doi.org/10.1556/ComEc.11.2010.2.8
Garcia, D., & Houle, G. (2016). Fine-scale spatial patterns of recruitment in red oak (Quercus rubra): What matters most, abiotic or biotic factors? Ecoscience, 12(2), 223–235.
Gómez-Aparicio, L., Zamora, R., Gómez, J. M., Hódar, J. A., Castro, J., & Baraza, E. (2004). Applying plant facilitation to forest restoration: A meta-analysis of the use of shrubs as nurse plants. Ecological Applications, 14(4), 1128–1138. https://doi.org/10.1890/03-5084
Guo, Q., Rundel, P. W., & Goodall, D. W. (1998). Horizontal and vertical distribution of desert seed banks: Patterns, causes, and implications. Journal of Arid Environments, 38(3), 465–478. https://doi.org/10.1006/jare.1997.0353
Harper, J. L., Clatworthy, J. N., McNaughton, I. H., & Sagar, G. R. (1961). The evolution and ecology of closely related species living in the same area. Evolution, 15, 209–227.
Harper, J. L., Williams, J. T., & Sagar, G. R. (1965). The behaviour of seeds in soil: 1. The heterogeneity of soil surfaces and its role in determining the establishment of plants from Seed. Journal of Ecology, 53(2), 273–286.
Hart, S. P., & Marshall, D. J. (2009). Spatial arrangement affects population dynamics and competition independent of community composition. Ecology, 90(6), 1485–1491. https://doi.org/10.1890/08-1813.1
Hoef, V., Jay, M., & Boveng, P. L. (2007). Quasi-poisson vs. negative binomial regression: How should we model overdispersed count data? Ecology, 88(11), 2766–2772. https://doi.org/10.1890/07-0043.1
Kraft, N. J. B., Adler, P. B., Godoy, O., James, E. C., Fuller, S., & Levine, J. M. (2014). Community assembly, coexistence and the environmental filtering metaphor. Community Phylogenetics and Ecosystem Functioning, 29(5), 592–599.
Li, M., Du, Z., Pan, H., Yan, C., Xiao, W., & Lei, J. (2012). Effects of neighboring woody plants on target trees with emphasis on effects of understorey shrubs on overstorey physiology in forest communities: A mini-review. Community Ecology, 13(1), 117–128. https://doi.org/10.1556/ComEc.13.2012.1.14
“LI-250A.” (2021). LI-COR. 2021. https://www.licor.com/env/products/light/light_meter
Lortie, C. J., Brooker, R. W., Choler, P., Kikvidze, Z., Michalet, R., Pugnaire, F. I., & Callaway, R. M. (2004). Rethinking plant community theory. Oikos, 107(2), 433–438. https://doi.org/10.1111/j.0030-1299.2004.13250.x
Lortie, C. J., Ellis, E., Novoplansky, A., & Turkington, R. (2005). Implications of spatial pattern and local density on community-level interactions. Oikos, 109(3), 495–502.
Lortie, C. J., & Turkington, R. (2002a). The effect of initial seed density on the structure of a desert annual plant community. Journal of Ecology, 90, 435–445.
Lortie, C. J., & Turkington, R. (2002b). The facilitative effects by seeds and seedlings on emergence from the seed bank of a desert annual plant community. Ecoscience, 9(1), 106–111.
Lovich, J. E. (1999). Anthropogenic Degradation of the Southern California Desert Ecosystem and Prospectsfor Natural Recovery and Restoration. Environmental Management, 24(3), 309–326. https://doi.org/10.1007/s002679900235.
Lucero, J. E., Noble, T., Haas, S., Westphal, M., Butterfield, H. S., & Lortie, C. J. (2019). The dark side of facilitation: Native shrubs facilitate exotic annuals more strongly than native annuals. NeoBiota, 44, 75–93. https://doi.org/10.3897/neobiota.44.33771
Mccallum, K. P., Lowe, A. J., Breed, M. F., & Paton, D. C. (2018). Spatially designed revegetation-why the spatial arrangement of plants should be as important to revegetation as they are to natural systems. Restoration Ecology, 26(3), 446–455. https://doi.org/10.1111/rec.12690
McIntire, E. J. B., & Fajardo, A. (2014). Facilitation as a ubiquitous driver of biodiversity. New Phytologist, 201(2), 403–416. https://doi.org/10.1111/nph.12478
Moore, J. L., Mouquet, N., Lawton, J. H., & Loreau, M. (2003). Coexistence, saturation and invasion in simulated plant assemblages. Oikos, 94(2), 303–314.
Murrell, D. J., & Law, R. (2003). Heteromyopia and the spatial coexistence of similar competitors. Ecology Letters, 6, 48–59. https://doi.org/10.1046/j.1461-0248.2003.00397.x
Nelder, J. A., & Wedderburn, R. W. M. (1972). Generalized linear models. Journal of the Royal Statistical Society. Series A (general), 135(3), 370. https://doi.org/10.2307/2344614
Pearson, D. E., Ortega, Y. K., & Sears, S. J. (2012). Darwin’s naturalization hypothesis up-close: Intermountain grassland invaders differ morphologically and phenologically from native community dominants. Biological Invasions, 14, 901–913.
Phacelia Tanacetifolia. (2021). In The Jepson Herbarium. University of California, Berkley. https://ucjeps.berkeley.edu/eflora/eflora_display.php?tid=37579
Pielou, E. C. (1960). A single mechanism to account for regular, random and aggregated populations. The Journal of Ecology, 48(3), 575. https://doi.org/10.2307/2257334
R Core Team. (2021). R (version 3.6.1).
Rice, K. J., & Dyer, A. R. (2001). Seed aging, delayed germination and reduced competitive ability in Bromus tectorum. Plant Ecology, 155(2), 237–243. https://doi.org/10.1023/a:1013257407909
Richardson, P. J., MacDougall, A. S., & Larson, D. W. (2012). Fine-scale spatial heterogeneity and incoming seed diversity additively determine plant establishment. Journal of Ecology, 100(4), 939–949.
Salo, L. F. (2004). Population dynamics of red brome (Bromus madritensis subsp. rubens): times for concern, opportunities for management. Journal of Arid Environments, 57(3), 291–296. https://doi.org/10.1016/s0140-1963(03)00110-1
Salo, L. F. (2005). Red brome (Bromus rubens subsp. madritensis) in North America: Possible modes for early introductions, subsequent spread. Biological Invasions, 7(2), 165–180. https://doi.org/10.1007/s10530-004-8979-4
Serrato-Valenti, G., Mariotti, M. G., Cornara, L., & Corallo, A. (1998). A histological and structural study of phacelia tanacetifolia endosperm in developing, mature, and germinating seed. International Journal of Plant Sciences, 159(5), 753–761. https://doi.org/10.1086/297594
Stohlgren, T. J., Binkley, D., Chong, G. W., Kalkhan, M. A., Schell, L. D., Bull, K. A., et al. (1999). Exotic plant species invade hot spots ofnative plant diversity. Ecological Monographs, 69(1), 25–46. https://doi.org/10.1890/0012-9615(1999)069[0025:EPSIHS]2.0.CO;2.
Thébault, A., Stoll, P., & Buttler, A. (2012). Complex interactions between spatial pattern of resident species and invasiveness of newly arriving species affect invasibility. Oecologia, 170(4), 1133–1142. https://doi.org/10.1007/s00442-012-2376-y
Turnbull, L., Coomes, D., Purves, D., & Reesi, M. (2007). How spatial structure alters population and community dynamics in a natural plant community. Journal of Ecology, 95(1), 79–89.
USDA Plant Database. n.d. https://plants.sc.egov.usda.gov/home/.
Vahl, W. K., Meer, J. V. D., Weissing, F. J., Dullemen, D. V., & Piersma, T. (2005). The mechanisms of interference competition: Two experiments on foraging waders. Behavioral Ecology, 16(5), 845–855. https://doi.org/10.1093/beheco/ari073
Velázquez, J., Garrahan, J. P., & Eichhorn, M. P. (2014). Spatial complementarity and the coexistence of species. PLoS ONE. https://doi.org/10.1371/journal.pone.0114979
Wassmuth, B. E., Stoll, P., Tscharntke, T., & Thies, C. (2009). Spatial aggregation facilitates coexistence and diversity of wild plant species in field margins. Perspectives in Plant Ecology, Evolution and Systematics, 11(2), 127–135. https://doi.org/10.1016/j.ppees.2009.02.001
Acknowledgements
This study was possible thanks to the National Sciences and Engineering Research Council of Canada (NSERC) grant awarded to CJL.
Author information
Authors and Affiliations
Contributions
CC and CJL designed the experiment; CC performed the experiments and wrote the manuscript; CJL analyzed the data; NG co-wrote and thoroughly edited the manuscript; MO and JB contributed critically.
Corresponding author
Appendix
Appendix

Rights and permissions
About this article
Cite this article
Ghazian, N., Braun, J., Owen, M. et al. Seed aggregation tips the scale in plant competition. COMMUNITY ECOLOGY 22, 403–412 (2021). https://doi.org/10.1007/s42974-021-00064-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42974-021-00064-5