Abstract
Pesticide misuse and overuse severely pollute agricultural soils, water, and crop yields, harming people and animals. This situation raises serious concerns about environmental pollution on a global scale. As an eco-friendly material for soil remediation, biochar can efficiently immobilize pesticides in the soil. Several studies have focused on the feasibility of biochar in remediating polluted soil. However, its influences during the remediation of pesticide-polluted soils remain indistinct. The present review illuminates the positive and negative influences of biochar on the dissemination of pesticides, the underlying mechanisms, the regulating factors, and critical considerations in the ongoing development of biochar for pesticide use. It also delineates the positive and negative impacts of biochar on pesticides in the soil, evaluates potential pitfalls based on recent research, and offers suggestions for prospective biochar applications crucial for remediating contaminated soil. This review reveals that the fate and types of pesticides, along with the physicochemical properties of soil and biochar types, can significantly influence the remediation of pesticide-polluted soil using biochar. Biochar has the potential to enhance the abundance of certain bacteria and the colonization of arbuscular mycorrhizal fungi, both of which play crucial roles in soil remediation. Biochar can also modify soil moisture, microbial communities, and other factors that impact the rate of pesticide degradation while simultaneously reducing other types of arbuscular mycorrhizal fungi. This review underscores the importance of thoroughly understanding the properties of biochar before its application to polluted soils. This review can serve as a basis for subsequent studies on the biochar-mediated remediation of contaminated soils.
Graphical Abstract

Highlights
-
Biochar can significantly reduce the leaching and decomposition of pesticides in the soil.
-
The efficacy of biochar in the soil depends on its biological, physical, and chemical features.
-
The aging of biochar diminishes its adsorption capacity for pesticides in the soil environment.
-
Modification of biochar could enhance its capacity to remediate soil polluted by pesticides.
-
Biochar alters the structure and function of microbial communities that regulate pesticides.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Although the rise of pesticides use in modern agriculture enhances crop yields, uncontrolled use of pesticides associated with its geogenic sources poses a significant challenge to environmental and human health. Typically, pesticides could alter the physicochemical properties of agricultural soils (Diez et al. 2013; Safaei Khorram et al. 2016) and adversely affect microbiological and enzymatic activities (Arora et al. 2019; Wołejko et al. 2020; Borowik et al. 2023), which are essential for decomposing soil organic matter. On the other hand, pesticides can trigger and exacerbate health issues, including liver, colon, lung, and prostate cancers, in individuals exposed to them (Al-Ahmadi 2019; Cheng et al. 2022). Agricultural farm employees, including farmers, mixers, sprayers, formulators, transporters, and production workers, are considered high-risk groups due to repeated exposure to pesticides (Abdollahdokht et al. 2022; Tudi et al. 2022). However, the production and formulation entail substantial risks (Abdollahdokht et al. 2022). According to surveys, 1 in 5000 people working in agriculture gets poisoned yearly, and approximately 200,000 die due to severe exposure (Faber 2020). Therefore, remediating soil polluted with pesticides is highly urgent.
Typically, soil vapor extraction, washing, flushing, phytoremediation, chemical oxidation, and bioremediation are standard pesticide remediation methods. Nevertheless, some of these approaches are frequently impractical in the field due to their inherent deficiencies or the emergence of additional problems after implementation, such as fertility loss, soil erosion, high maintenance costs, and nutrient leaching (Zhang et al. 2020b; Kumar et al. 2022b). It is well known that some pesticides are manufactured to be persistent in the soil to ensure their effectiveness over time. This persistence makes them difficult to be broken down or removed from the soil. Other pesticides may penetrate deep into the soil, reaching levels that are hard to access with conventional remediation techniques. This type of contamination necessitates more intensive and costly methods for effective remediation. Due to this, studies on remediating soil polluted with pesticides are still being conducted to assess reliable and applicable methods (Mayakaduwa et al. 2017; Rajmohan et al. 2020). Therefore, an in-situ, eco-friendly, and cost-effective approach like bioremediation is being valued.
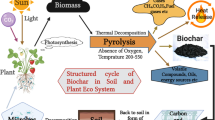
In bioremediation, biochar which is produced by the pyrolysis of organic materials, such as agricultural residues, wood, or other biomass, is a prodigious material that is used to remediate and rehabilitate pesticide-polluted soil environment. Biochar has garnered significant interest in recent years (Nie et al. 2018; de Souza et al. 2019) due to its porous structure and the availability of functional groups, including carboxyl, phenolic, and hydroxyl groups (Sakhiya et al. 2020; Twagirayezu et al. 2022). Numerous studies have demonstrated that biochar has the potential to immobilize organic and inorganic pollutants within the soil environment effectively (Sun et al. 2021; Qiu et al. 2022). For instance, the addition of biochar in soil near lakes or other watercourses could significantly reduce subsurface water pollution by lowering the concentration of pesticides that could seep into groundwater (Cheng et al. 2022; Rasool et al. 2022).
Typically, there is information on the impacts of biochar and its interactions with various pesticides in soil. For instance, the addition of biochar to the soil reduces pesticide leaching from the soil and absorption in the plants, thereby reducing the risk of water pollution and ensuring the safety and purity of agricultural products due to their excellent absorption capacity (Cheng et al. 2022, 2023). However, information about the interactions between soil microbial community structure and pesticides in the soil environment is limited. In addition, the influence of biochar on pesticides in the soil environment during remediation processes remains largely unclear. Therefore, understanding the intrinsic influencing factors and mechanism of biochar during the remediation of pesticide-polluted soil is highly desired in order to reduce the risks of environmental pollution. This could be immensely advantageous for biochar users seeking to regulate the fate of pesticides in the soil environment.
Therefore, this review aims to consolidate the positive and negative influence of biochar on pesticides. It also delineates the mechanisms regarding how biochar behaves on the properties of the soil polluted with pesticides. Moreover, it elucidates what needs to be done with biochar for future research on sustainably remediating pesticide-polluted soil.
2 Research methodology
The initial literature search was conducted in English, and relevant publications were either downloaded in open-access cases or requested in full text from the respective authors. The inclusion of papers was based on the following criteria: (1) accessibility of full text; (2) presence of information on biochar research; (3) documents offering sustainable applications of biochar for remediation; and (4) peer-reviewed scientific studies written in the English language. Conversely, publications failing to meet the following criteria were excluded from this review: (1) papers authored in languages other than English; (2) non reviewed preprint publications; (3) papers categorized as "grey literature," encompassing academic thesis, dissertations, technical reports, news articles, and summaries of scientific events; among others.
The critical review followed the PRISMA 2020 reporting guidelines outlined by Page et al. (2021). The review process followed a series of sequential steps, as illustrated in Fig. 1. Each stage of the search and screening process encompassed the inclusion, exclusion, and extraction of pertinent studies. The relevant search articles were exported and stored in CSV format. During the review process, 2345 articles were identified from Web of Science. Initially, 987 ineligible records were excluded from the initial reports. The 1358 remaining records underwent screening based on the title and abstract level, followed by a full-text assessment. After that, 674 articles were excluded because they did not cover biochar in areas such as remediation/degradation, health, adsorption/sorption, pesticides, waste and sludge. In total, 684 articles were obtained for in-depth analysis of their full texts. Of these, 296 were excluded because they were either preprints, reports, or opinion articles. Additionally, 203 articles were eliminated from the analysis due to their lack of focus on the review aim. Following the assessment of all exclusion and inclusion criteria, 185 articles were identified as pertinent to the objectives of our review. The authors scrutinized the records to ensure the reliability of the retrieved sources. The present study employed a systematic search strategy involving searching and screening. The search phase entailed the exploring publications directly linked to the investigation of the application of biochar in the remediation of pesticide-soil contamination through the Web of Science. The search involved meticulously collecting a raw database of target review themes using articles published from 01-01-2000 to 01-01-2024. In this context, the title, abstract, or keywords were used as the search criteria: biochar* and pesticides* and soil* or biochar* and wastes* or sludge*or water* with singular and plural. Scholarly articles, chapters from books, and brief communications were considered.
As represented in Fig. 2, the appropriately chosen keywords were grouped into six topical clusters. The most significant cluster, cluster 1 (red), represents biochar addition, amendment, agriculture, animal manure, bacterial community, additives, etc. Cluster 2 (green) contains absorption, agricultural residues, agricultural wastes, ash, biomass, biochar production, etc. Cluster 3 (blue) primarily focuses on activated biochar, activated carbons, adsorbents, adsorption behavior, adsorption capacity, adsorption mechanism, adsorption performance, adsorption removal, biochar, etc. Cluster 4 (yellow) includes soil, pesticides, atrazine, diuron, chlorpyrifos, accumulation, biodegradation, bioremediation, contaminants, contamination, degradation, desorption, etc. Cluster 5 (purple) contains activated sludge, bioconversion, biofertilizer, crop residue, food waste, fungi, kitchen waste, etc. Cluster 6 (baby blue) represents biocarbon, dairy manure, fly ash, livestock, waste recycling, etc.
Visualized network of 721 popular keywords of peer-reviewed publications from 01-01-2000 to 01-01-2024, with the minimum number of occurrences of a keyword is 6. The bibliographic data were retrieved from the “Web of Science Core Collection” with biochar* and pesticides* and soil* or biochar* and wastes* or sludge*or water* as keywords, with complete counting within keyword co-occurrence analysis performed using VOSviewer
3 Pesticides and biochar in the environment
3.1 Concerns about pesticides in the environment
Typically, there are chemical pesticides and biopesticides, of which biopesticides are further subdivided into plant-incorporated, biochemical, and microbial pesticides (Additional file 1: Fig. S1). Based on their mode of action, they may be categorized as destructive, repelling, or mitigating agents. Some of them are restricted, while others are unclassified. Restricted pesticides can harm people or the environment, while unclassified are all the remaining types. Based on pesticide origin, they can be generally sorted into natural pesticides, synthetic pesticides, and biopesticides. Natural pesticides are those extracted from their natural habitats, like plants or microorganisms (Table 1). Synthetic pesticides are created by modifying minerals or chemical substances and are considered the most widely used worldwide. Biopesticides are derived from living organisms, including bacteria, fungi, viruses, and certain plants. This category is often considered a subset of natural pesticides. As detailed in Table 2, pesticides are classified based on their target organisms.
As depicted in Fig. 3, although a considerable quantity of pesticides are sprayed into the environment, only a relatively small number of them reach the intended target. The remnants of pesticides are subsequently released into different parts of the ecosystem, increasing the concentration of potentially dangerous chemicals that affect the health of people and other life forms. A countrywide study conducted by the Environmental Protection Agency on 127 different pesticides revealed that 10 and 4% of community and rural household wells, respectively, exhibited detectable levels of at least one pesticide (Peshin 2014). Notably, pesticides are currently detected in various locations worldwide, including the soil, marine environments, drinking water, surface water, and groundwater. Pesticides have the potential to bring about severe health problems for people (Cao et al. 2023; Song et al. 2023). As shown in Fig. 4, pesticides might cause several adverse effects to the health of people due to several exposures.
It is well known that today and future generations will primarily rely on the soil, which provides food for them. However, excessive use of pesticides leads to the accumulation of toxic chemicals in the soil, affecting soil quality. Pesticides can change the properties of the soil, affecting its structure and texture (Karpouzas et al. 2016; Tripathi et al. 2020). Typically, changes in soil structure can impede water infiltration, root growth, and nutrient movement, creating an unfavorable environment for plants to uptake nutrients effectively. Pesticides can negatively impact the populations and activities of beneficial soil microorganisms (Arora et al. 2019; Satapute et al. 2019). These microorganisms are crucial in nutrient cycling and making nutrients available to plants (Cheng et al. 2022; Karimi et al. 2022). This shows that excessive pesticides may directly affect plant roots, inhibiting their ability to absorb water and essential nutrients. They may also interfere with the normal physiological processes of plants, leading to stunted growth or even the death of the plants.
3.2 Concerns about biochar in the environment
As depicted in Fig. 5, the primary ingredients for biochar formation are sewage sludge, farming residues, animal manure, wood residues, and other waste, which are ubiquitous worldwide. Farming waste has traditionally been employed in a restricted number of uses, such as composting for manure generation and animal feed. However, there are currently possibilities of being utilized to produce biochar. For instance, around 3.1 million tons (Mt) of biochar in Indonesia can be produced from approximately 10.7 Mt of agricultural biomass residues (Susilawati et al. 2020; Awasthi et al. 2021). Specifically, the primary source of these biomass residues is rice husk, accounting for 6.8 Mt per year and producing biochar at a rate of up to 1.77 Mt per year. This constitutes approximately 56.48% of the country's biochar production capacity (Susilawati et al. 2020).
As a soil conditioner, biochar is gaining popularity because it improves soil aggregation, contaminant remediation, nutrient retention, carbon storage, promotion of soil microbes and mineralization of pesticides (Cheng et al. 2023). Manure like cow dung, agricultural waste such as sugarcane, weeds, maize, etc., as well as biosolids are only some of the biomass that can be used to produce biochar under oxygen-deprived and low-temperature pyrolysis (400–700 °C). Biochar and charcoal typically differ in their applications and purposes (Khiari et al. 2019). Biochar is primarily used for soil improvement and carbon sequestration, whereas charcoal is commonly used as a fuel source for cooking and heating. Biochar is produced from various materials such as wood, agricultural residues, chicken manure, or sludge at different pyrolysis temperatures. This results in distinct chemical and physical characteristics for the biochar produced. The physicochemical characteristics of biochar vary considerably due to the variety of feedstocks and pyrolysis procedures, exhibiting different specific surface areas, porosities, charges, and functional groups. These features affect pH value, conductivity (CEC), and surface adsorption capability. The standard size of the feedstock particle is employed to assess the biochar particle size. However, the biochar itself is often considerably smaller than the feedstock when the pyrolysis process causes shrinkage and attrition. The increased pyrolysis temperature enhances the tensile strength of the raw materials, resulting in biochar particles that are smaller in size (Albalasmeh et al. 2020; Ahmed et al. 2021; Wani et al. 2022). The functional groups on the ionic charges, surface, and porous structure of biochar aid in the detoxification (Alrashidi et al. 2020), physical adsorption (Zhang et al. 2020a), complexation, mobilization-immobilization (Hu et al. 2020), and co-precipitation (Deng et al. 2019) of metal pollutants and support phytoremediation potential of the hyperaccumulator. The physical and chemical characteristics of biochar produced from several feedstocks are displayed in Table 3.
4 Positive and negative influences of biochar on pesticides in the soil
Biochar is often added to the soil to improve sorption, speed up biodegradation, and decrease pesticide leakage (Cheng et al. 2022). The capacity of biochar to absorb pesticides depends on the biological, physical, and chemical features of the soil, influencing the movement of pesticides through the soil and their subsequent reach to watercourses. However, the impact of biochar on the adsorption of pesticides in the soil is variable and depends on the kind of pesticide, soil features, and biochar characteristics. The pores within biochar act as a microbial habitat, minimizing the probability of interactions between microbes and pesticides. This reduces the decomposition of pesticides in the soil.
As summarized in Table 4, biochar can positively and negatively impact the bio-physicochemical properties of the soil during remediation of pesticide-polluted soil. Biochar in the soil can change the microbial texture and activity. The impact of biochar on microbial abundance and population in the soil is studied using several techniques, including culture and plate counting (Jiang et al. 2017), respiration stimulated by the substrate (Kolb et al. 2009), total genomic DNA extracted (Igalavithana et al. 2019), fumigation extraction (Yuan et al. 2019), and extraction of phospholipid fatty acid (Ali et al. 2019). Qiu et al. (2019) and Ge et al. (2019) have also shown that the addition of biochar to soil makes microbial populations multiply faster. Biochar can affect microbial populations and abundance differently based on the kind of microorganism (Huang et al. 2020; Twagirayezu et al. 2022) and biochar properties. For instance, Shukla et al. (2017) found that the addition of biochar derived from eucalyptus wood in the soil at a rate of 0.6–6 t.ha−1 resulted in a 20–40% increase in the colonization of arbuscular mycorrhizal fungi in the root of Triticum aestivum. Conversely, the addition of biochar to the soil increased the tip number and growth rate of ectomycorrhizal infection in the roots of larch seedlings (Larix in Latin) by 19–157% (Shukla et al. 2017). In other studies, biochar reduced the relative proportion or quantity of soil microbes (Lehmann et al. 2011). Therefore, biochar in the soil can either boost or reduce the microbial population and abundance, altering pesticide decomposition in the soil environment.
Typically, inorganic carbon (SIC) and soil organic carbon (SOC) are the two main types of carbon in the soil (Dong et al. 2019). The behavior of organic and inorganic contaminants in soil is controlled by the SOC pool (Shahid et al. 2012). SOC plays a crucial role in influencing the fate and transport of pesticides in the soil environment (Dong et al. 2019). The concentration and composition of SOC can impact pesticide desorption (release from soil particles) and sorption (binding to soil particles) through various mechanisms. The increased SOC may enhance the sequestration of pesticides, reducing their availability for desorption and potential movement in the soil. Thus, SOC may significantly affect pesticide sorption and desorption in soil by forming various complexes with these pesticides. Abelmann et al. (2005) found that the aromaticity of SOC may affect the amount of pesticides that can be absorbed by the soil. However, the addition of biochar to the soil may not influence SOC in the soil, based on the study conducted by Cheng et al. (2016), where biochar produced at the lowest temperature enhanced the mineralization of SOC. Contrary to this, after a 60-day experiment, the mineralization of SOC was reduced relative to the control (without biochar). SIC or carbonate minerals, such as calcite and dolomite, can provide sorption sites for pesticides. The surface properties of these minerals and the specific interactions with pesticide molecules can influence sorption behavior. Biochar can alter the physical structure and properties of the soil matrix (Sun et al. 2021; Wang et al. 2022). This may impact the distribution of inorganic carbon and create microenvironments that influence pesticide sorption and desorption.
For instance, Zheng et al. (2019) demonstrated the adsorption of chlorpyrifos and chlorpyrifos-methyl onto biochar, as well as the influence of deashing and low molecular weight organic acid (LMWOA) aging as the temperature increased from 300 to 600 °C. The study results revealed that the adsorption capacity of biochar rose from 4.32 to 14.8 mg. g−1 and from 15.0 to 50.5 mg.g−1 for chlorpyrifos and chlorpyrifos-methyl, respectively. This is due to the fact that biochar produced at higher temperatures contains more aromatic units and pores suitable for entrapping a greater variety of sorbates. The deashing and LMWOA aging treatments enhanced the porosity of biochar and exposed more carbon surfaces, both of which are beneficial to sorption. Relative to the LMWOA aging treatment, the deashing treatment evinced enhanced adsorption. In addition, Sun et al. (2016) showed that the adsorption of sulfamethoxazole onto biochar increased by over fivefold as the concentration of low molecular weight organic acids (LMWOAs) rose from 0 to 100 mmol.L−1. This enhancement was primarily linked to the heightened microporosity of biochar after the treatment with LMWOAs (citric acid or malic acid).
Soil pH is essential for the fertility of the soil and the biogeochemistry of many inorganic and organic contaminants in soil. Soil pH can significantly change due to the addition of biochar (He et al. 2019). Earlier studies have shown that biochar pH values can be obtained < 4 and > 12 (Li et al. 2017). This variation in pH value relies on oxidation level (Cybulak et al. 2019), pyrolysis temperature, and feedstock kind (Xu et al. 2019; Kwak et al. 2019). Typically, the acid neutralization potential of biochar is contingent upon the feedstock. Any soil pH alteration may substantially affect pesticide desorption and sorption. Biochar effects on the pesticide sorption via changes in pH also depend on the kind of pesticide (Liu et al. 2018; Ogura et al. 2021). After addition of biochar to the soil, the sorption of some pesticides does not change according to the pH of the soil. Although the simazine content in the soil amended with biochar depends on the pyrolysis temperature, feedstock, and holding time (Cheng et al. 2018; Itoh et al. 2020; Pariyar et al. 2020) , several studies have confirmed that application of biochar to soil can regulate pesticide behaviors (Ali et al. 2019).
5 Mechanisms of biochar for influencing the fate of pesticides in the soil environment
The mechanisms by which biochar influences the fate of pesticides can be divided into two parts: the first part involves direct influence, where the adsorption and sorption of biochar depend on its various properties, leading to diverse influences. The second part encompasses indirect influence, wherein biochar regulates soil characteristics, microbial structure, and abundance, resulting in either positive or negative influences.
5.1 Direct influences of biochar on adsorption and sorption of pesticides
The strategic application of pesticides in cultivating high-value crops, like vegetables and fruits, is crucial for the agricultural industry. Nevertheless, inadequate management of pesticides not only diminishes their agricultural benefits but also poses serious hazards to the environment and human health (Yang et al. 2023). This has resulted in the banning certain pesticides, including dichlorodiphenyltrichloroethane (DDT), paraquat, endosulfan, aldrin, and dieldrin due to the acknowledge of their harmful effects on human health. Meanwhile, discussions persist regarding the potential ban of others, such as Glyphosate, alachlor, and atrazine due to concerns about their impact on human health. Therefore, there is an urgent need to create processes for mopping them up from contaminated sites (Cheng et al. 2022) to protect public health and the environment, and to enhance crop yield. For instance, soils containing excessive pesticides could adversely impact plant health and growth, ultimately reducing agricultural yield. Therefore, numerous studies are being conducted to address the usefulness of biochar in improving plant development and remediating polluted soil (Younis et al. 2016). Typically, the large surface area of biochar promotes an adsorptive activity. Several scanning electron microscopy studies have confirmed that biochar adsorbs contaminants on its surface area (Cederlund et al. 2016; Bai et al. 2023).
The influence of biochar on the desorption, adsorption, leaching, and degradation of pesticides in the soil environment has been revealed by several studies (Cederlund et al. 2017). Wang et al. (2015) showed that wheat straw biochar generated at 750 °C in the aqueous phase displayed a high affinity for immobilizing pesticides like chlorpyrifos. It well evinced that the sorption of organic molecules onto biochar changes based on the characteristics of biochar (Pignatello et al. 2017). The pyrolysis temperature is a critically important characteristic (Zhao et al. 2013). The aging process and the co-existence of organic acids with varying molecular weights in a rhizosphere environment may impact the interactions between contaminants and biochar. The increasing amounts of two low molecular weight organic acids (malic and citric acid) ranging from 0 to 100 mmol. L−1 increased the sorption of ionizable sulfamethoxazole onto straw biochar (Sun et al. 2016).
Nonetheless, mechanisms on how biochar influences the adsorption of hydrophobic pesticides (such as chlorpyrifos and chlorpyrifos-methyl) are well displayed in Fig. 6. Biochar in the soil contains hydrophobic sites, pores, and several minerals, sustaining the soil properties and microorganisms, etc. On the other hand, pesticides in the soil are characterized by solubility, mobility volatility, degradation, bioaccumulation and environmental fate, etc. Soil can interact with soil components and other substances applied to the soil, particularly pesticides, leading to positive and negative impacts on the soil environment. The different types of biochar (unmodified, modified, and aged) act differently in the soil depending on pyrolysis temperature, feedstock, etc. Ultimately, biochar characteristics can improve the adsorption capacity of pesticides due to the enhancement of microporosity. Adsorption disparity may be attributed to distinct chemical and physicochemical characteristics and the kind of the pollutants along with the characteristics of biochar. For instance, when citric acid is not present, Ca2+ can fill biochar sorption sites via Ca2+–π interaction (Frontera et al. 2011) and surface complexation or electrostatic interaction between oxygen and Ca2+-containing functional groups such as −OH and COO− (Uchimiya 2014). Ultimately, the mechanism of pesticide adsorption by biochar in the soil is principally due to ligand exchange, followed by average pore diameter and the biochar surface.
Sorption marks the initial phase in the soil following the addition of pesticides (Safaei Khorram et al. 2016; Cheng et al. 2022). Higher sorption rates of pesticides are achieved by a combination of factors, including large porous structures, surface area, and high OC concentration in biochar or feedstock (Zheng et al. 2010; Ogura et al. 2021). For instance, there was an increase in the Freundlich sorption coefficient (Kf) of atrazine when biochar produced from the chicken litter was added to the soil at a dosage of 10 t ha−1 (Tatarková et al. 2013). According to the study of Tatarková et al. (2013), the sorption of 4-chloro-2-methylphenoxyacetic acid in the soil with biochar (1.0% M/M) was 82 and 2.53 times higher than that of the soil without biochar, respectively. Previous research has revealed that the addition of wheat biochar (1%) to sandy soil increased the Kf value of atrazine by 5 while adding it to clay soil increased it by 4.3 times (Loganathan et al. 2009).
The addition of rice straw biochar at concentrations of 0.1% and 0.5% resulted in a 1.5-fold and threefold increase in both the sorption capacity and the sorption coefficient (Kf values) of the soil (Xu et al. 2008). The temperature of the pyrolysis procedures alters the properties of the biochar, which in turn changes the interaction between biochar and herbicide retention. Sawdust biochar paralyzed at 300 °C (5% M/M) evinced a high sorption capacity for acetochlor and atrazine in the sand soil (Spokas et al. 2009). This was due to high OC content (69%) and specific surface area (1.6 m2 g−1) of biochar. According to the findings of the glyphosate study (Hall et al. 2018), higher values of pesticide sorption were obtained in proportion to an increased pyrolysis temperature, with the highest retention revealed for biochar generated at 900 °C. The influences of biochar on the sorption of pesticides are shown in Table 5. Ultimately, biochar amendments to the soil stabilize pesticides through sorption, reducing their bioaccumulation, ecotoxicity, biomagnification, and ecotoxicity (Egamberdieva et al. 2021). Generally, the significant consequences of pesticides in the soil environment may be mitigated by biochar, which also depends on several features listed below.
5.1.1 pH of biochar
The pH of biochar can influence the adsorption of ionic pesticides onto its surface. Biochar surface characteristics, including pH, are crucial in determining its ability to adsorb different substances, including pesticides. Ionic pesticides can be positively or negatively charged depending on their chemical structure and the pH of the surrounding environment. If the surface of biochar is positively charged, it may attract negatively charged pesticides, and vice versa. Higher biochar pH values in soil enhance the solubility of alkali minerals, whereby the alkali catalytic processes are required for the hydrolysis of carbamates and organophosphate pesticides (Zhang et al. 2013).
The feedstock and pyrolysis temperature used during biochar production influence the surface area, porosity, and pH of biochar. The surface functionality of biochar is considerably associated with pH owing to the variety of feedstock qualities, which may vary from mineral-poor woody feedstocks to mineral-rich manures or crop residues like rice hulls (Zhou et al. 2019). An elevated pH level mainly accompanies a high mineral ash ratio in biochar. The pH of any given feedstock rises as the temperature rises, and the pH of biochar can either increase or fall based on the feedstock used and the soil characteristics. During a year of incubation, the pH of mineral-poor oak wood biochar dropped from 4.9 to 4.7, whereas the pH of mineral-rich maize stover biochar rose from 6.7 to 8.1 (Nguyen and Lehmann 2009). The acidic carboxyl groups formed when carbon is oxidized mainly cause the pH to decrease (Cheng et al. 2006). Nevertheless, increasing the pH in biochar may be associated with the dissolution of alkaline minerals. A study on rice husk biochar revealed that glyphosate adsorption rose from 75% to 85% between pH 3 and 5, declined from 75% to 65% between pH 6 and 8, and then dropped significantly to 55% at pH 9 (Herath et al. 2017). Contrastingly, previous study indicated that biochar exhibited low adsorption of glyphosate at high and low pH levels (Cederlund et al. 2016). Ultimately, alterations in pH can impact how pesticides may dissolve, thereby affecting their ability to be adsorbed onto biochar.
5.1.2 Surface area and porosity
The sorption capacity of biochar with a large surface area and high porosity is typically high. The porous surface of biochar is created during pyrolysis when water loss increases during dehydration. Based on the International Union of Pure and Applied Chemistry (IUPAC), the sizes of biochar pores can range from micro (< 2 nm) to meso (≥ 2 and ≤ 50 nm) to macro (> 50 nm) (Leng et al. 2021). Biochar with a lower porosity cannot absorb the pesticide molecules, irrespective of their molecular charges or polarities. The surface area of biochar is the most crucial component in influencing its sorption capacity, whereas temperature is one of the critical aspects to consider when producing biochar. The surface area may differ from one material to another, between used and non-used materials and also between aged and fresh biochar. The surface area of activated carbon is larger. Biochar produced with no activation step has a smaller surface area and pores (Kim et al. 2012). Therefore, an activation process is necessary to raise biochar surface area and porosity. The activation process can entail physical and chemical activation processes.
5.1.3 Surface functional groups
Biochar sorption ability is contributed by functional groups including amide, hydroxyl, amine, and lactonic carboxyl groups (Rajapaksha et al. 2016; Antón-Herrero et al. 2018; Usman et al. 2019), which also influence pesticide adsorption in the soil. Feedstock and pyrolysis temperature affect the surface functional groups of biochar (El‐Naggar et al. 2018; Askeland et al. 2019; El-Naggar et al. 2019). When temperatures rise, the atomic ratios of hydrogen to carbon (H:C), oxygen to carbon (O:C), and nitrogen to carbon (N:C) decrease (Liu et al. 2017b), lowering the biochar surface and functional groups.
Fourier transform infrared spectroscopy analyzes the biochar functional groups, including phenolic, hydroxyl, and carboxyl (Igalavithana et al. 2017; Antoniadis et al. 2019). FTIR spectra indicate that varied pyrolysis temperatures result in biochar with noticeably varied structural compositions (Al-Wabel et al. 2013; Sun et al. 2014). Pyrolysis of raw feedstock at temperatures between 100 and 200 °C does not exhibit a substantial change in FTIR spectra. However, cellulose, lignin, and hemicellulose are dehydrated at 300 °C (3500–3200 cm−1). At 400 °C, various products produced from lignin and cellulose emerged owing to the breakdown of carboxylic and phenolic chemicals in lignin, with numerous peaks between 1600–700 cm−1. According to Keiluweit et al. (2010), the intensity of aromatic condensation might be lower at 1650–1500 cm−1 than between 885–752 cm−1. This is because aromatic condensation occurs at temperatures higher than 500 °C.
Biochar derived from manure within the temperature range of 350 to 700 °C exhibited minor carboxyl and hydroxyl groups (Cantrell et al. 2012). Compared with biochar produced at 300 °C, the FTIR spectra of biochar generated from peanut straw, maize, soybeans, and canola were similar when measured at 700 °C (Awad et al. 2019). Nuclear magnetic resonance (NMR) is typically utilized to describe biochar functional groups. In this context, 2D-13C NMR correlation spectroscopy indicated that pyrolysis of bran straw and rice biochar between 100 and 800 °C produces dehydrogenation/dihydroxylation and aromatization issues due to the creation of aromatic structures and cleavage of O-alkylated carbons, and anomeric O–C–O carbons (Li et al. 2013). Before aromatic structure formation, temperature increases cause mass breakage of O-alkylated and anomeric O–C–O carbon groups. Biochar generated at lowered and elevated pyrolysis temperatures (< 300 and > 300 °C, respectively) had more aromatic structures and aliphatic O-alkylated carbons (Li et al. 2013). NMR spectroscopy showed that the O-alkyl carbon groups in wheat straw biochar dropped from 20–56 and 6.9–13% at increasing pyrolysis temperatures of 200 and 600 °C, respectively. Despite this, Liu et al. (2016) showed that no alkyl carbons formed at 600 °C within biochar. Pesticide sorption on biochar surface was commonly reduced when functional groups were reduced. This is crucial for understanding how biochar regulates pesticides in the soil, thereby benefiting plants.
5.1.4 Chemical composition of biochar
Ashes, fixed carbon, and volatile materials are the components that make up the basic chemical composition of biochar. The proportion of volatile material released during biochar heating comprises carbon monoxide, hydrocarbons, and carbon dioxide molecules. However, another quantity of carbon stays passably intact since it is not lost with volatile materials and is termed "fixed carbon." The fixed carbon and volatile material can be determined by heating biochar at about 900 °C. On the other hand, ash is a mineral oxide residue left behind after biochar is completely burned. The quantity of the produced oxidized residue could be used to determine the amount of ash in the biochar. When heated to very high temperatures, biomass experiences a transformation in which its constituent parts are drastically altered (Tomczyk et al. 2020; Ćwieląg-Piasecka et al. 2023).
The manufactured biochar for particular pyrolysis settings and high lignin feedstocks is related to its immediate features, such as higher fixed carbon levels and lower ash and volatile content. Each range of temperature results in the production of a unique product; however, the final temperature significantly impacts the characteristics of biochar. Biochar produced from eucalyptus wood and leaves had a more significant total carbon fixed content than bovine dung (Singh et al. 2010). Higher carbon content levels in biochar evince an effect on the sorption capacity of pesticides. According to Cabrera et al. (2014), the quantity of organic carbon content in the materials improved the impact of several biochars on bentazone sorption. A greater sorption of aminocyclopyrachlor was found in biochar with a more extensive humification index and surface area (Cabrera et al. 2014). This evidence shows that biochar interacts with each pesticide to reduce its effects on the soil environment.
Biochar characteristics are typically linked to the feedstock, pyrolysis temperature, and time circumstances under which it is produced. The biomass used as a feedstock for biochar production is based on the accessibility of wood and its derivatives and agricultural wastes. These raw materials contain more lignin, cellulose, and hemicelluloses, the primary components to be converted into a carbon matrix during biochar production. Consequently, the aggregates produced during biochar production and the proportional abundance of those aggregates determine the characteristics of the manufactured biochar (Yavari et al. 2015). Biochar sorption capacity is governed by its carbon content and aromatic structure (de Jesus et al. 2019). Biochar has significant levels of mineral ash, recalcitrant carbon, and leachable or labile carbon (Jing et al. 2022). The principal chemical variation among biochar and organic materials is a substantially more significant proportion of aromatic C (Iacomino et al. 2022). These merged aromatic biochar structures can occur in various forms, like turbo-stratic C and amorphous C, which are predominant at low and elevated temperatures in pyrolysis (da Silva Amaral et al. 2022). At temperatures lower than 500 °C, biochar generated from lignocellulose materials has a higher carbon content (Liu et al. 2017a).
The carbon content of biochar produced at 600 °C is significantly greater than that generated at 400 °C (Heitkötter and Marschner 2015). For instance, pine chip biochar pyrolyzed at 400 and 600 °C had a C content of 79% and 92%, whereas maize digestate biochar at the same temperatures had a C content of 65% and 64%. Guo et al. (2021) showed that biochar from C-rich feedstocks showed a more remarkable sorption ability for organic pollutants. High carbonization and aromatization levels in the biochar structure make it highly stable so that it can remain in the soil for a long time (Wang et al. 2022). Biochar has lately shown the capacity to reduce soil contamination. Zhang and Shen (2022) evinced that biochar has a limited decomposition rate, indicating that soil microorganisms cannot access and use the nitrogen, carbon, and other nutrients in a biochar C structure.
5.1.5 Elemental ratios of biochar
Most biochar generated from nuts and wood evinces extremely high carbon-to-nitrogen and carbon-to-phosphorus ratios, while biochar derived from crops, manure, and food waste has substantially lower ratios (Joseph and Lehmann 2009; Askeland et al. 2019). Biochar produced from manure has a higher mineral content than its C content. The poor stability of biochar, which is high in minerals and ash, is connected to its high aromaticity and carbon content (Xie et al. 2023). The H:C and O:C correlate with the polarity and aromaticity of biochar (Kookana 2010). Despite its durability, biochar is subject to fragmentation, carboxylation, and biological and physical oxidation when contained in the soil. It has been found that the atomic ratios of H:C and O:C in biochar decline as the pyrolysis temperature rises. As an example, NMR data from Baldock and Smernik. (2002) showed that the aromaticity level increases as the temperature rises. Further, the heating time of the biomass also impacts the H:C and O:C ratios.
5.1.6 Feedstocks
The type of feedstock used to produce biochar significantly influences its chemical composition. Different feedstocks, such as woody and non-woody biomass, have distinct compositions, and these variations can impact the properties of biochar (Sorrenti et al. 2016; Pariyar et al. 2020). Wood-based biochar is commonly used and has been studied for its ability to adsorb pesticides. However, non-wood biomass biochar, derived from materials such as crop residues, agricultural waste, or other plant-based sources, can also be practical, depending on their specific properties. The carbon content of biochar is a fundamental factor influenced by the feedstock. Carbon-rich biochar is generally more effective in adsorbing pesticides due to the abundance of carbonaceous surfaces (Yavari et al. 2015). The presence of heteroatoms (such as oxygen, nitrogen, and sulfur) and functional groups in biochar, which play a role in the adsorption of pesticides through various chemical interactions, is influenced by the feedstock composition (Safaei Khorram et al. 2016; He et al. 2019). The feedstock influences the physical structure of biochar, including surface area and porosity. Feedstocks with high lignocellulose content can result in biochars with greater porosity, enhancing their ability to adsorb pesticides (Kumari et al. 2016; Liu et al. 2018). The mineral content of the feedstock influences the ash content in biochar. Higher ash content may affect the pH of the soil and impact pesticide behavior (Yadav et al. 2023). The biochar feedstock can influence microbial communities in the soil (Sadet-Bourgeteau et al. 2023), impacting pesticide degradation or persistence.
5.2 Indirect influences of biochar on the fate of pesticides in the soil
5.2.1 Influences of biochar on the soil properties
Besides improving soil fertility, biochar can be used to minimize concerns in agriculture activities, leading to improved crop production. For instance, El-Naggar et al. (2015) showed that when conocarpus wood waste biochar is applied to a soil lacking nutrients and soil organic matter (e.g., calcareous sandy soils (CSS) in dry and semi-arid climates), it improves these soils. Inal et al. (2015) evinced that the utilization of biochar associated with poultry manure in the soil improved the content of N, P, and K due to two significant occurrences: (1) increased N mineralization due to increased microbial activity and (2) increased cation exchange. Therefore, the addition of biochar to the CSS can be a proper strategy for enhancing soil quality and promoting plant growth. Biochar generated from rice hulls was productively used in the soil of reclaimed tidal lands to improve the unfavorable effects of salt on maize plants and increase production (Kim et al. 2016). When added at 27 and 67.5 t. ha−1, biochar improved maize yields by 52% and 101%, respectively (Palansooriya et al. 2019a). This occurred in salty conditions and may be traced back to an improvement in soil phosphate, saturated organic carbon, and particle-size fractions of water-stable aggregates.
Additionally, the high potassium content of biochar reduces the amount of sodium uptake by maize plants and the quantity of sodium that may be exchanged in salty soil (Kim et al. 2016). A further challenge for highland agriculture is irrigation with salted water. Biochar from conocarpus wood waste increased yield from 14.0% to 43.3% higher than the control sample when given to tomato plants cultivated in saline irrigation (Usman et al. 2016). The improved crop production occurred despite salt stress and may be attributable to enhanced nutrient availability because of biochar (Usman et al. 2016). Sorghum yields have improved in the two distinct kinds of sandy soil in desert environments (Laghari et al. 2015). In addition, dry matter production was increased by around 18.0–20.0% for both soils compared with the control group (Laghari et al. 2015).
Environmental, economic, and sustainability issues currently hamper the efficient utilization of biochar in farming. The synthesis of biochar from food, agricultural, and municipal waste has favorable financial implications. The economic viability of biochar in agricultural and environmental uses is determined by its life cycle costs and environmental implications. Ecological advantages and increases in agricultural yield must be weighed against the expenses of garbage pickup, feedstock preparation, pyrolysis, and biochar handling and application (Lehmann and Joseph 2015). Biochar also influences the soil microbial population by regulating pH, phosphorus level, nitrogen level, and dissolved organic matter, thereby impacting the biodegradation of pesticides (Noyce et al. 2015). The addition of biochar to the soil improves soil sorption capacity for certain pesticides. Biochar sorption capacity and the length of its residence period in soil rely on the physical and chemical characteristics of biochar (Twagirayezu et al. 2022; Deng et al. 2022). The addition of biochar to the soil affects pesticide transformation in several ways. The enzyme activity, microbe activity, and structure of the soil are affected by biochar. These characteristics lead to the enhanced biodegradation of pesticides, which stimulates soil fertility and crop productivity (Xie et al. 2013; Cheng et al. 2022).
Biochar influences not only the soil microbiological qualities but also its chemical and physical qualities. These property changes could affect how the indigenous microbial community in the soil gets started, yet they may only partially affect the entire amount of microbial biomass in the soil (Zhu et al. 2017). The addition of biochar in the soil environments lowers emissions of greenhouse gases and enhances climate change mitigation. This happens due to an increase in the activity of microbial functional groups, which ultimately leads to an improvement in the physiochemical properties of the soil (Lehmann and Joseph 2015; Cederlund et al. 2016; Kumar et al. 2022a). Biochar often consists of a sizeable fraction of aromatic compounds, and these molecules are resistant to microbial breakdown, making them persistent pollutants. Further, Noyce et al. (2015) and Wang et al. (2012) revealed that biochar improves long-term carbon sequestration in land environments.
5.2.2 Influences of biochar on the soil microbial community diversity and composition
In addition to the ecological services they provide, the population and activity of soil microbes have a considerable impact on the functioning of the soil (Díaz-López et al. 2019; Twagirayezu et al. 2023). Hence, any factor that affects soil microbial populations and activities can influence the persistence and retention of pesticides in the soil environment (Ali et al. 2019). The addition of biochar to the soil impacts the population and behavior of soil microbes. The impact of biochar on microbial population and abundance in the soil has been evaluated by several techniques such as culture and plate counting (Jiang et al. 2017), extraction of phospholipid fatty acid (Ali et al. 2019), total genomic DNA extracted (Grossman et al. 2010; Igalavithana et al. 2019), fumigation extraction ( Palansooriya et al. 2019b; Yuan et al. 2019), respiration induced by the substrate (Kolb et al. 2009). Other studies have also shown that amendment of biochar in the soil enhances the reproduction rate of microbial population and abundance (Qiu et al. 2019; Ge et al. 2019). However, the changes in microbial population and abundance in the soil that are mediated by biochar vary for various types (Lehmann et al. 2011). For instance, the addition of biochar enhanced the quantity of firmicutes, proteobacteria, actinobacteria, and gemmatimonadetes (Ali et al. 2019). On the contrary, biochar lowered the relative abundances of actinobacteria and chloroflexi. In addition, the addition of biochar in the soil led to an increase in the proportion of bacterial populations while simultaneously reducing the number of arbuscular mycorrhizal fungi (Song et al. 2019). The use of biochar typically has a positive impact on the population of microorganisms as well as the rate of reproduction of the ectomycorrhizal and arbuscular mycorrhizal fungus species, which are the two most frequent types of mycorrhizal fungus (Ali et al. 2019). It has been shown that 0.6–6 t ha–1 of eucalyptus wood biochar could increase the colonization of arbuscular mycorrhizal fungi in the roots of Triticum aestivum by 20–40% (Shukla et al. 2017). Similarly, biochar in the soil increased the number of root tips on larch plants and the rate of ectomycorrhizal infection by 19–157% (Shukla et al. 2017). However, other studies have indicated that biochar could reduce the quantity or amount of soil microbes (Lehmann et al. 2011). Biochar in the soil enhances the availability of water and nutrients to plants, leading to a lower necessity for mycorrhizal symbiosis and reducing the microbial population. This illustrates that addition of biochar in the soil may increase or decrease the quantity and population of microorganisms, which may alter pesticide adsorption, degradation, or mineralization.
6 The advance of biochar application in the remediation of pesticide polluted soil
6.1 Aging of biochar
Typically, biochar properties change in the soil during aging and weathering processes (Gámiz et al. 2019). Biotic and abiotic factors regulate the weathering or aging process, including microbial activity, crop types, tillage regimes, UV exposure, moisture, and temperature fluctuations (Sorrenti et al. 2016). When biochar ages, its pores get clogged by mineral particles, roots, organic matter, or microorganisms, thereby reducing the surface available for chemical reactions (Ren et al. 2018). Aging processes significantly modify the properties of biochar (Mia et al. 2017), which slightly reduces its adsorption capacity in the soil environment (He et al. 2019). For instance, Ren et al. (2018) revealed that aging altered biochar elemental concentration and surface area, affecting biochar adsorption capacities. In addition, Mia et al. (2017) and Cheng et al. (2023) have reported that the aging process may reduce the absorption capacity of biochar, leading to an increased leaching out and mineralization of simazine in the soil environment.
On the other hand, even if aged biochar cannot work efficiently, it should be used in soil remediation. Kumari et al. (2016) found that after 7–19 months, soil amended with wood biochar heated to 500 ℃ had a higher cation exchange capacity and greater specific surface area, leading to a rise in glyphosate sorption in the soil environment. Trigo et al. (2016) found that the sorption of the chemical metolachlor increased with time in the soil amended with various types of biochar (macadamia: fresh = 2.4-fold, 1 year = 2.5-fold, 4 years = 1.9-fold, wood: fresh = twofold, and 5 years = 14-fold). In addition, Martin et al. (2012) conducted a study using biochar aged for 32 months in the soil at a rate of 10 t.ha−1 to assess its sorption capacity for atrazine and diuron. In this research, soil with biochar had two to five times more pesticide sorption than soil without biochar. Aging processes may reduce the average pore diameter, and the biochar surface becomes more amenable to the leaching of materials. The sorption capacity of diuron was shown to be reduced by 47–68% due to the gradual pore clogging.
Moreover, Cheng et al. (2023) conducted research using biochar that had naturally aged for 9 years in the field. The results of the study revealed that the average leaching and mineralization of simazine in the soil amended with aged biochar were significantly higher by 4.8% and 1.66%, respectively, compared to those in the soil amended with fresh biochar. Rice husk biochar generated at 500 °C for 30 min in the soil for 13 months lost carbon and nitrogen content, pH values dropped to nearly neutral, and specific surface area and porosity decreased (Cao et al. 2017). The weathering degrades the biochar particles, changing their mass and affecting soil pesticides. Based on the findings of Dong et al. (2017), there was a drop in biochar mass of around 40% over 5 years, irrespective of the added quantity (30, 60, or 90 t ha−1). A decrease in the abundance of biochar mass with time has also been noticed by other researchers as well (Maestrini et al. 2014; Obia et al. 2017). The natural aging process of biochar may also alter the material structure. Trigo et al. (2014) analyzed the surface of biochar that had been aged for 1 and 2 years. They found that the addition of that biochar to the soil caused clay minerals to stick to it, carboxylic acids to cover the structure, and fatty acids to disappear, resulting in a change in the possible connections to the surface and its ability to hold pesticides. In their research, biochar particles had an even greater specific surface area after 2 years of incubation, and the holes were filled with mineral material. The organic carbon content of the biochar decreased from 80.4 mg L−1 to 31.6 mg.L−1 after 1 year of incubation due to weathering processes. However, the changes in the chemical and structural properties that influence the adsorption capacity of biochar depend on the type of material, pyrolysis temperature, conditions, and time incubated in the soil.
6.2 Modification of biochar
Modification of biochar to remediate soil polluted with pesticides involves enhancing its properties to improve effectiveness in adsorbing, degrading, or immobilizing pesticides. Concerning the content of Fig. 7, biochar can be prepared with or without pretreatment. There are four different sorts of modification techniques (Wang et al. 2017; Sizmur et al. 2017): (1) controlling general production conditions (pyrolytic conditions and feedstock); (2) physical modification; (3) chemical modification (pre-treatments of feedstock and post-treatments of pristine biochar); and (4) biochar-based organic composites (co-composting with organic waste). By the modification strategies mentioned above, increasing the surface area and optimizing the pore structure of biochar can enhance its adsorption capacity (Cabrera et al. 2014; Cheng et al. 2018). This involves controlling pyrolysis conditions during biochar production or post-treatment processes to create a more porous structure. Introducing or enhancing of functional groups on the biochar surface can improve its affinity for pesticides. Oxygen-containing functional groups, such as hydroxyl (–OH), carboxyl (–COOH), and phenolic groups, can interact with pesticides through hydrogen bonding and other interactions.
Modifying the surface charge of biochar can influence its ability to attract and retain charged pesticide molecules. Incorporating metal oxides or nanoparticles into biochar can improve its catalytic properties, enhancing pesticide degradation (Cheng et al. 2022). Mixing biochar with organic amendments, such as compost or manure, can enhance soil fertility and microbial activity, contributing to the degradation of pesticides through microbial activity. Subjecting biochar to post-treatment processes like steam activation or thermal treatment at elevated temperatures can modify its properties, including surface area and porosity, thus enhancing its effectiveness in pesticide remediation. The hydrophobic nature of biochar can be changed to improve its interaction with hydrophobic pesticides. Controlling biochar particle size can influence its distribution in the soil matrix. Fine particles may have higher reactivity and surface area, impacting the ability of biochar to interact with and adsorb pesticides.
Recently, modified biochar (biochar + FeOS, biochar + FeCl3, biochar + Fe) enhanced the adsorption capacity of soil for simazine and changed the microbial community and structures that regulate the fate of simazine in the soil environment (Cheng et al. 2022). In the aforementioned study, the modified biochar decreased the quantity of microorganisms in the soil, thereby limiting pesticide decomposition. The effectiveness of pesticide removal is directly linked to pesticide concentration in the leachate and biochar adsorption capacity (Liu et al. 2018). Biochar addition results in a considerable decrease in the leachate pesticides (e.g., simazine). However, biochar modification results in an increase in improves the ability of biochar to adsorb simazine, thereby reducing the potential danger of simazine movement into watercourses (Cheng et al. 2022). Inevitably, the procedure mentioned above increases the adsorption of pesticides by biochar and can alter the nature and quantity of the microbial population and community, thereby altering the fate of pesticides in the soil.
7 The current situation, challenges, and mitigation measures for excessive pesticides in the soil
7.1 Current situation
Pesticides are intended to control weeds in crop farming for a certain amount before or during planting. This aids farmers in enhancing crop yield by minimizing the presence of undesirable plants and pests in the field. For efficient weed control, pesticides should be used consistently and at the appropriate rates. However, their lengthy persistence, often known as carryover, may harm the future crop. Pesticides may exert their effects by preventing cell division, photosynthesis, or the formation of amino acids, or they may cause abnormalities in plants by imitating the actions of natural plant growth hormones. The effects of excessive pesticides in the soil are a concerning issue. However, most countries, particularly developing countries, still need to be aware of the severe problems caused by the overuse and misuse of pesticides in the soil environment. Therefore, pesticide adsorption is crucial when there is a problem of pesticide soil pollution.
7.2 Challenges and mitigation measures for excessive pesticides in the soil
Many pesticides are produced and added to the soil in each agricultural season to protect crops from pest infestations. When pesticides are applied, they may endure in the sprayed area. This may cause a large concentrations of pesticides in other environmental compartments, such as surface and groundwater. In Australia, the United States, and Europe, the pesticide simazine was detected in water at concentrations reaching hundreds of micrograms per liter (µg L–1) (Cox et al. 2000; Troiano et al. 2001). This has generated several environmental concerns, including those regarding human health and the health of terrestrial and aquatic environments (Regitano et al. 2006; Rico et al. 2012). There are several techniques available for remediating pesticide-polluted soil. Among these methods, commonly employed approaches include biodegradation, phytoremediation, the use of pesticide-degrading microorganisms, bioremediation of pesticide-contaminated soils, and the application of soil additives for pesticide remediation. However, choosing the most effective technique to remove pesticides from the environment is still a big challenge. As displayed in different literature, the time required for the remediation process is still challenging. Existing methods cannot completely remove or degrade pesticides from the soil environment. In addition, there are some limitations, such as poor effectiveness, a long time, and even the production of new pollution, which lead to a variety of problems in the process of removing pesticide contamination from the environment (Meriam Suhaimy et al. 2020; Cheng et al. 2022). Hence, workers engaged in remediating polluted areas must be equipped with appropriate personal protective equipment, including gloves, splash goggles, boots, respirators, and coveralls. Protective equipment must be carefully stored and disinfected during the complete remediation activities. Most importantly, a systematic surveillance system should be established at the national level, including regular, continuous measurements of pesticides utilized in agricultural activities. Therefore, it is advisable to utilize surveillance methods to assess the state of pesticides in the soil.
8 Conclusions and future perspectives
This review paper shows that biochar can influence the fate of pesticides differently, impacting pesticide sorption, biodegradation, and leaching. The temperature during the pyrolysis process, feedstocks, and techniques used for the pyrolysis substantially affect biochar for controlling soil pesticides. However, using biochar as a primary source for remediating soil polluted by pesticides is possible. Surface functional groups of biochar, like carboxyl and hydroxyl groups, are primarily responsible for the ability of biochar to adsorb pesticides. Even though the effectiveness of biochar in remediating soil polluted with pesticides depends on various factors, future efforts should prioritize refining its qualities. This can be achieved by enhancing its capacity through modifications or combining it with another promising remediation technique, such as phytoremediation. These enhancements could position biochar as a promising option for remediating soil contaminated with pesticides.
On the other hand, ongoing studies are essential to explore optimal methods for recovering biochar from the soil, particularly in the context of diverse environmental applications. These should be done by balancing the positive and negative influence of biochar in the soil. It is of the utmost significance to categorize biochar according to its potential for remediating soil polluted by pesticides. This can be done by checking the physicochemical characteristics of the biochar and soil and the kind of pesticide. In addition, positive and negative influences should be balanced during biochar application in the soil. For future research endeavors, the adsorption processes of pesticides on biochar have to be well explored. The influence that feedstock and pyrolysis of biochar could have on the activity of soil microorganisms and plants must be evident. Further, a comprehensive understanding of the sorption and adsorption processes of pesticides on biochar in various types of soil is still needed.
Availability of data and materials
Some of the data and materials used in this critical review are available on Web of Science, while others are from the critics of the authors.
References
Abdollahdokht D, Gao Y, Faramarz S et al (2022) Conventional agrochemicals towards nano-biopesticides: an overview on recent advances. Chem Biol Technol Agric 9:1–19. https://doi.org/10.1186/s40538-021-00281-0
Abelmann K, Kleineidam S, Knicker H et al (2005) Sorption of HOC in soils with carbonaceous contamination: influence of organic-matter composition. J Plant Nutr Soil Sci 168:293–306. https://doi.org/10.1002/jpln.200421622
Ahmed K, Hasan M, Haider J (2021) Electrical and mechanical properties of sugarcane bagasse pyrolyzed biochar reinforced polyvinyl alcohol biocomposite films. J Compos Sci 5:249. https://doi.org/10.3390/jcs5090249
Al-Ahmadi MS (2019) Pesticides, anthropogenic activities, and the health of our environment safety. In: Pesticides-use and misuse and their impact in the environment. IntechOpen. https://doi.org/10.5772/intechopen.84161
Albalasmeh A, Gharaibeh MA, Mohawesh O et al (2020) Characterization and Artificial Neural Networks Modelling of methylene blue adsorption of biochar derived from agricultural residues: effect of biomass type, pyrolysis temperature, particle size. J Saudi Chem Soc 24:811–823. https://doi.org/10.1016/j.jscs.2020.07.005
Ali N, Khan S, Li Y et al (2019) Influence of biochars on the accessibility of organochlorine pesticides and microbial community in contaminated soils. Sci Total Environ 647:551–560. https://doi.org/10.1016/j.scitotenv.2018.07.425
Alrashidi SH, Sallam AS, Usman ARA (2020) Acid-modified and unmodified natural clay deposits for in situ immobilization and reducing phytoavailability of molybdenum in a sandy loam calcareous soil. Sustainability 12:8203. https://doi.org/10.3390/su12198203
Al-Wabel MI, Al-Omran A, El-Naggar AH et al (2013) Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour Technol 131:374–379. https://doi.org/10.1016/j.biortech.2012.12.165
Antón-Herrero R, García-Delgado C, Alonso-Izquierdo M et al (2018) Comparative adsorption of tetracyclines on biochars and stevensite: looking for the most effective adsorbent. Appl Clay Sci 160:162–172. https://doi.org/10.1016/j.clay.2017.12.023
Antoniadis V, Shaheen SM, Levizou E et al (2019) A critical prospective analysis of the potential toxicity of trace element regulation limits in soils worldwide: are they protective concerning health risk assessment? - A review. Environ Int 127:819–847. https://doi.org/10.1016/j.envint.2019.03.039
Arora S, Arora S, Sahni D et al (2019) Pesticides use and its effect on soil bacteria and fungal populations, microbial biomass carbon and enzymatic activity. Curr Sci 116:643. https://doi.org/10.18520/cs/v116/i4/643-649
Askeland M, Clarke B, Paz-Ferreiro J (2019) Comparative characterization of biochars produced at three selected pyrolysis temperatures from common woody and herbaceous waste streams. PeerJ 7:e6784. https://doi.org/10.7717/peerj.6784
Awad YM, Vithanage M, Niazi NK et al (2019) Potential toxicity of trace elements and nanomaterials to Chinese cabbage in arsenic- and lead-contaminated soil amended with biochars. Environ Geochem Health 41:1777–1791. https://doi.org/10.1007/s10653-017-9989-3
Awasthi MK, Sarsaiya S, Wainaina S et al (2021) Techno-economics and life-cycle assessment of biological and thermochemical treatment of bio-waste. Renew Sustain Energy Rev 144:110837. https://doi.org/10.1016/j.rser.2021.110837
Bai T, Twagirayezu G, Wang Z et al (2023) Biochar amendment in vermi-wetland for enhancing nitrification during excess sludge recycling. Sustainability 15:16551. https://doi.org/10.3390/su152416551
Baldock JA, Smernik RJ (2002) Chemical composition and bioavailability of thermally altered Pinus resinosa (Red pine) wood. Org Geochem 33:1093–1109. https://doi.org/10.1016/S0146-6380(02)00062-1
Borowik A, Wyszkowska J, Zaborowska M, Kucharski J (2023) The impact of permethrin and cypermethrin on plants, soil enzyme activity, and microbial communities. Int J Mol Sci 24:2892. https://doi.org/10.3390/ijms24032892
Cabrera A, Cox L, Spokas KA et al (2011) Comparative sorption and leaching study of the herbicides fluometuron and 4-chloro-2-methylphenoxyacetic acid (MCPA) in a soil amended with biochars and other sorbents. J Agric Food Chem 59:12550–12560. https://doi.org/10.1021/jf202713q
Cabrera A, Cox L, Spokas K et al (2014) Influence of biochar amendments on the sorption–desorption of aminocyclopyrachlor, bentazone and pyraclostrobin pesticides to an agricultural soil. Sci Total Environ 470–471:438–443. https://doi.org/10.1016/j.scitotenv.2013.09.080
Cantrell KB, Hunt PG, Uchimiya M et al (2012) Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar. Bioresour Technol 107:419–428. https://doi.org/10.1016/j.biortech.2011.11.084
Cao X, Ma L, Gao B, Harris W (2009) Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ Sci Technol 43:3285–3291. https://doi.org/10.1021/es803092k
Cao T, Chen W, Yang T et al (2017) Surface characterization of aged biochar incubated in different types of soil. BioResources 12:6366–6377. https://doi.org/10.15376/biores.12.3.6366-6377
Cao N, Ji J, Li C et al (2023) Rapid and efficient removal of multiple aqueous pesticides by one-step construction boric acid modified biochar. RSC Adv 13:8765–8778. https://doi.org/10.1039/d2ra07684e
Cederlund H, Börjesson E, Lundberg D, Stenström J (2016) Adsorption of pesticides with different chemical properties to a wood biochar treated with heat and iron. Water, Air, Soil Pollut 227:203. https://doi.org/10.1007/s11270-016-2894-z
Cederlund H, Börjesson E, Stenström J (2017) Effects of a wood-based biochar on the leaching of pesticides chlorpyrifos, diuron, glyphosate and MCPA. J Environ Manage 191:28–34. https://doi.org/10.1016/j.jenvman.2017.01.004
Chaudhary S (2017) Progress on Azadirachta indica based biopesticides in replacing synthetic toxic pesticides. Front Plant Sci 8:610. https://doi.org/10.3389/fpls.2017.00610
Cheng K, Hu C, Liu Y et al (2006) Effects of dietary calcium, phosphorus and calcium/phosphorus ratio on the growth and tissue mineralization of Litopenaeus vannamei reared in low-salinity water. Aquaculture 251:472–483. https://doi.org/10.1016/j.aquaculture.2005.06.022
Cheng H, Hill PW, Bastami MS, Jones DL (2016) Biochar stimulates the decomposition of simple organic matter and suppresses the decomposition of complex organic matter in a sandy loam soil. GCB Bioenergy 9:1110–1121. https://doi.org/10.1111/gcbb.12402
Cheng H, Jones DL, Hill P et al (2018) Influence of biochar produced from different pyrolysis temperature on nutrient retention and leaching. Arch Agron Soil Sci 64:850–859. https://doi.org/10.1080/03650340.2017.1384545
Cheng H, Xing D, Lin S et al (2022) Iron-modified biochar strengthens simazine adsorption and decreases simazine decomposition in the soil. Front Microbiol 13:1–10. https://doi.org/10.3389/fmicb.2022.901658
Cheng H, Xing D, Twagirayezu G, Lin S, Shangyi Gu, Chenglong Tu, Hill PW, Chadwick DR, Jones DL (2023) Effects of field-aging on the impact of biochar on herbicide fate and microbial community structure in the soil environment. Chemosphere. https://doi.org/10.1016/j.chemosphere.2023.140682
Cox L, Celis R, Hermosín MC, Cornejo J (2000) Natural soil colloids to retard simazine and 2,4-D leaching in soil. J Agric Food Chem 48:93–99. https://doi.org/10.1021/jf990585k
Ćwieląg-Piasecka I, Jamroz E, Medyńska-Juraszek A et al (2023) Deashed wheat-straw biochar as a potential superabsorbent for pesticides. Materials (basel) 16:1–14. https://doi.org/10.3390/ma16062185
Cybulak M, Sokołowska Z, Boguta P, Tomczyk A (2019) Influence of pH and grain size on physicochemical properties of biochar and released humic substances. Fuel 240:334–338. https://doi.org/10.1016/j.fuel.2018.12.003
da Silva Amaral G, Ix-Balam MA, Mendes KF, et al (2022) Impacts of Biochar Addition on Herbicides’ Efficacy for Weed Control in Agriculture. In: Interactions of Biochar and Herbicides in the Environment. CRC Press, Boca Raton, pp 219–249. https://doi.org/10.1201/9781003202073-8
de Jesus JHF, da Matosda Cunha TTSCG et al (2019) Adsorption of aromatic compounds by biochar: influence of the type of tropical biomass precursor. Cellulose 26:4291–4299. https://doi.org/10.1007/s10570-019-02394-0
de Souza ES, Dias YN, da Costa HSC et al (2019) Organic residues and biochar to immobilize potentially toxic elements in soil from a gold mine in the Amazon. Ecotoxicol Environ Saf 169:425–434. https://doi.org/10.1016/j.ecoenv.2018.11.032
Dechene A, Rosendahl I, Laabs V, Amelung W (2014) Sorption of polar herbicides and herbicide metabolites by biochar-amended soil. Chemosphere 109:180–186. https://doi.org/10.1016/j.chemosphere.2014.02.010
Deng R, Huang D, Wan J et al (2019) Chloro-phosphate impregnated biochar prepared by co-precipitation for the lead, cadmium and copper synergic scavenging from aqueous solution. Bioresour Technol 293:122102. https://doi.org/10.1016/j.biortech.2019.122102
Deng Z, Gu S, Cheng H et al (2022) Removal of phosphate from aqueous solution by zeolite-biochar composite: adsorption performance and regulation mechanism. Appl Sci 12:5334. https://doi.org/10.3390/app12115334
Díaz-López M, García C, Garrido I et al (2019) Solarization-based pesticide degradation results in decreased activity and biomass of the soil microbial community. Geoderma 354:113893. https://doi.org/10.1016/j.geoderma.2019.113893
Diez MC, Levio M, Briceño G et al (2013) Biochar as a partial replacement of peat in pesticide-degrading biomixtures formulated with different soil types. J Biobased Mater Bioenergy 7:741–747. https://doi.org/10.1166/jbmb.2013.1376
Dong X, Li G, Lin Q, Zhao X (2017) Quantity and quality changes of biochar aged for 5 years in soil under field conditions. CATENA 159:136–143. https://doi.org/10.1016/j.catena.2017.08.008
Dong X, Singh BP, Li G et al (2019) Biochar increased field soil inorganic carbon content five years after application. Soil Tillage Res 186:36–41. https://doi.org/10.1016/j.still.2018.09.013
dos Araujo RS, LopesBarbosa MPWF et al (2019) Spinosad-mediated effects on survival, overall group activity and the midgut of workers of Partamona helleri (Hymenoptera: Apidae). Ecotoxicol Environ Saf 175:148–154. https://doi.org/10.1016/j.ecoenv.2019.03.050
Duke SO, Cantrell CL, Meepagala KM et al (2010) Natural toxins for use in pest management. Toxins (basel) 2:1943–1962. https://doi.org/10.3390/toxins2081943
Egamberdieva D, Jabbarov Z, Arora NK et al (2021) Biochar mitigates effects of pesticides on soil biological activities. Environ Sustain 4:335–342. https://doi.org/10.1007/s42398-021-00190-w
Eibisch N, Schroll R, Fuß R et al (2015) Pyrochars and hydrochars differently alter the sorption of the herbicide isoproturon in an agricultural soil. Chemosphere 119:155–162. https://doi.org/10.1016/j.chemosphere.2014.05.059
El-Naggar AH, Usman ARA, Al-Omran A et al (2015) Carbon mineralization and nutrient availability in calcareous sandy soils amended with woody waste biochar. Chemosphere 138:67–73. https://doi.org/10.1016/j.chemosphere.2015.05.052
El-Naggar A, Awad YM, Tang X-Y et al (2018) Biochar influences soil carbon pools and facilitates interactions with soil: a field investigation. L Degrad Dev 29:2162–2171. https://doi.org/10.1002/ldr.2896
El-Naggar A, El-Naggar AH, Shaheen SM et al (2019) Biochar composition-dependent impacts on soil nutrient release, carbon mineralization, and potential environmental risk: a review. J Environ Manage 241:458–467. https://doi.org/10.1016/j.jenvman.2019.02.044
Faber D (2020) Poisoning the world for profit: petro-chemical capital and the global pesticide crisis. Capital Nat Soc 31:1–17. https://doi.org/10.1080/10455752.2020.1829794
Frontera A, Quiñonero D, Deyà PM (2011) Cation–π and anion–π interactions. Wires Comput Mol Sci 1:440–459. https://doi.org/10.1002/wcms.14
Gámiz B, Velarde P, Spokas KA et al (2019) Changes in sorption and bioavailability of herbicides in soil amended with fresh and aged biochar. Geoderma 337:341–349. https://doi.org/10.1016/j.geoderma.2018.09.033
Ge X, Cao Y, Zhou B et al (2019) Biochar addition increases subsurface soil microbial biomass but has limited effects on soil CO2 emissions in subtropical moso bamboo plantations. Appl Soil Ecol 142:155–165. https://doi.org/10.1016/j.apsoil.2019.04.021
Grossman JM, O’Neill BE, Tsai SM et al (2010) Amazonian anthrosols support similar microbial communities that differ distinctly from those extant in adjacent, unmodified soils of the same mineralogy. Microb Ecol 60:192–205. https://doi.org/10.1007/s00248-010-9689-3
Guo J, Zheng L, Li Z et al (2021) Effects of various pyrolysis conditions and feedstock compositions on the physicochemical characteristics of cow manure-derived biochar. J Clean Prod 311:127458. https://doi.org/10.1016/j.jclepro.2021.127458
Hall KE, Ray C, Ki SJ et al (2015) Pesticide sorption and leaching potential on three Hawaiian soils. J Environ Manage 159:227–234. https://doi.org/10.1016/j.jenvman.2015.04.046
Hall KE, Spokas KA, Gamiz B et al (2018) Glyphosate sorption/desorption on biochars – interactions of physical and chemical processes. Pest Manag Sci 74:1206–1212. https://doi.org/10.1002/ps.4530
He Y, Liu C, Tang X-Y et al (2019) Biochar impacts on sorption-desorption of oxytetracycline and florfenicol in an alkaline farmland soil as affected by field ageing. Sci Total Environ 671:928–936. https://doi.org/10.1016/j.scitotenv.2019.03.414
Heitkötter J, Marschner B (2015) Interactive effects of biochar ageing in soils related to feedstock, pyrolysis temperature, and historic charcoal production. Geoderma 245–246:56–64. https://doi.org/10.1016/j.geoderma.2015.01.012
Herath HMMTB, Pahalagamage SP, Yogendranathan N et al (2017) Amitraz poisoning: a case report of an unusual pesticide poisoning in Sri Lanka and literature review. BMC Pharmacol Toxicol 18:6. https://doi.org/10.1186/s40360-016-0114-5
Hu B, Ai Y, Jin J et al (2020) Efficient elimination of organic and inorganic pollutants by biochar and biochar-based materials. Biochar 2:47–64. https://doi.org/10.1007/s42773-020-00044-4
Huang K, Jingyang C, Mengxin G et al (2020) Effects of biochars on the fate of antibiotics and their resistance genes during vermicomposting of dewatered sludge. J Hazard Mater 397:1–10. https://doi.org/10.1016/j.jhazmat.2020.122767
Iacomino G, Sarker TC, Ippolito F et al (2022) Biochar and compost application either alone or in combination affects vegetable yield in a volcanic Mediterranean soil. Agronomy 12:1996. https://doi.org/10.3390/agronomy12091996
Igalavithana AD, Mandal S, Niazi NK et al (2017) Advances and future directions of biochar characterization methods and applications. Crit Rev Environ Sci Technol 47:2275–2330. https://doi.org/10.1080/10643389.2017.1421844
Igalavithana AD, Kim K-H, Jung J-M et al (2019) Effect of biochars pyrolyzed in N2 and CO2, and feedstock on microbial community in metal(loid)s contaminated soils. Environ Int 126:791–801. https://doi.org/10.1016/j.envint.2019.02.061
Inal A, Gunes A, Sahin O et al (2015) Impacts of biochar and processed poultry manure, applied to a calcareous soil, on the growth of bean and maize. Soil Use Manag 31:106–113. https://doi.org/10.1111/sum.12162
Itoh T, Fujiwara N, Iwabuchi K et al (2020) Effects of pyrolysis temperature and feedstock type on particulate matter emission characteristics during biochar combustion. Fuel Process Technol 204:106408. https://doi.org/10.1016/j.fuproc.2020.106408
Jiang L, Han G, Lan Y et al (2017) Corn cob biochar increases soil culturable bacterial abundance without enhancing their capacities in utilizing carbon sources in Biolog Eco-plates. J Integr Agric 16:713–724. https://doi.org/10.1016/S2095-3119(16)61338-2
Jing F, Sun Y, Liu Y et al (2022) Interactions between biochar and clay minerals in changing biochar carbon stability. Sci Total Environ 809:151124. https://doi.org/10.1016/j.scitotenv.2021.151124
Joseph S, Lehmann J (2009) Biochar for environmental management: science and technology. Earthscan, London. https://doi.org/10.1016/j.forpol.2009.07.001
Karimi H, Mahdavi S, Asgari Lajayer B et al (2022) Insights on the bioremediation technologies for pesticide-contaminated soils. Environ Geochem Health 44:1329–1354. https://doi.org/10.1007/s10653-021-01081-z
Karpouzas DG, Tsiamis G, Trevisan M et al (2016) ” LOVE TO HATE” pesticides: felicity or curse for the soil microbial community? An FP7 IAPP Marie Curie project aiming to establish tools for the assessment of the mechanisms controlling the interactions of pesticides with soil microorganisms. Environ Sci Pollut Res 23:18947–18951. https://doi.org/10.1007/s11356-016-7319-4
Khiari B, Jeguirim M, Limousy L, Bennici S (2019) Biomass derived chars for energy applications. Renew Sustain Energy Rev 108:253–273. https://doi.org/10.1016/j.rser.2019.03.057
Kim KH, Kim J-Y, Cho T-S, Choi JW (2012) Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Bioresour Technol 118:158–162. https://doi.org/10.1016/j.biortech.2012.04.094
Kim H-S, Kim K-R, Yang JE et al (2016) Effect of biochar on reclaimed tidal land soil properties and maize (Zea mays L.) response. Chemosphere 142:153–159. https://doi.org/10.1016/j.chemosphere.2015.06.041
Kolb SE, Fermanich KJ, Dornbush ME (2009) Effect of charcoal quantity on microbial biomass and activity in temperate soils. Soil Sci Soc Am J 73:1173–1181. https://doi.org/10.2136/sssaj2008.0232
Kookana RS (2010) The role of biochar in modifying the environmental fate, bioavailability, and efficacy of pesticides in soils: a review. Soil Res 48:627. https://doi.org/10.1071/SR10007
Kumar A, Singh E, Mishra R, Kumar S (2022a) Biochar as environmental armour and its diverse role towards protecting soil, water and air. Sci Total Environ 806:150444. https://doi.org/10.1016/j.scitotenv.2021.150444
Kumar M, Bolan N et al (2022b) Mobilization of contaminants: potential for soil remediation and unintended consequences. Sci Total Environ 839:156373. https://doi.org/10.1016/j.scitotenv.2022.156373
Kumari KGID, Moldrup P, Paradelo M et al (2016) Soil properties control glyphosate sorption in soils amended with birch wood biochar. Water, Air, Soil Pollut 227:174. https://doi.org/10.1007/s11270-016-2867-2
Kwak J-H, Islam MS, Wang S et al (2019) Biochar properties and lead(II) adsorption capacity depend on feedstock type, pyrolysis temperature, and steam activation. Chemosphere 231:393–404. https://doi.org/10.1016/j.chemosphere.2019.05.128
Laghari M, Mirjat MS, Hu Z et al (2015) Effects of biochar application rate on sandy desert soil properties and sorghum growth. CATENA 135:313–320. https://doi.org/10.1016/j.catena.2015.08.013
Lehmann J, Joseph S (2015) Biochar for environmental management. Routledge. https://doi.org/10.4324/9780203762264
Lehmann J, Rillig MC, Thies J et al (2011) Biochar effects on soil biota – a review. Soil Biol Biochem 43:1812–1836. https://doi.org/10.1016/j.soilbio.2011.04.022
Leng L, Xiong Q, Yang L et al (2021) An overview on engineering the surface area and porosity of biochar. Sci Total Environ 763:144204. https://doi.org/10.1016/j.scitotenv.2020.144204
Li X, Shen Q, Zhang D et al (2013) Functional groups determine biochar properties (pH and EC) as studied by two-dimensional 13C NMR correlation spectroscopy. PLoS ONE 8:e65949. https://doi.org/10.1371/journal.pone.0065949
Li H, Dong X, da Silva EB et al (2017) Mechanisms of metal sorption by biochars: biochar characteristics and modifications. Chemosphere 178:466–478. https://doi.org/10.1016/j.chemosphere.2017.03.072
Liu Y, Lu H, Yang S, Wang Y (2016) Impacts of biochar addition on rice yield and soil properties in a cold waterlogged paddy for two crop seasons. F Crop Res 191:161–167. https://doi.org/10.1016/j.fcr.2016.03.003
Liu W, Huo R, Xu J et al (2017a) Effects of biochar on nitrogen transformation and heavy metals in sludge composting. Bioresour Technol 235:43–49. https://doi.org/10.1016/j.biortech.2017.03.052
Liu Y, Yao S, Wang Y et al (2017b) Bio- and hydrochars from rice straw and pig manure: Inter-comparison. Bioresour Technol 235:332–337. https://doi.org/10.1016/j.biortech.2017.03.103
Liu Y, Lonappan L, Brar SK, Yang S (2018) Impact of biochar amendment in agricultural soils on the sorption, desorption, and degradation of pesticides: a review. Sci Total Environ 645:60–70. https://doi.org/10.1016/j.scitotenv.2018.07.099
Loganathan VA, Feng Y, Sheng GD, Clement TP (2009) Crop-residue-derived char influences sorption, desorption and bioavailability of atrazine in soils. Soil Sci Soc Am J 73:967–974. https://doi.org/10.2136/sssaj2008.0208
Maestrini B, Abiven S, Singh N et al (2014) Carbon losses from pyrolysed and original wood in a forest soil under natural and increased N deposition. Biogeosciences 11:5199–5213. https://doi.org/10.5194/bg-11-5199-2014
Martin SM, Kookana RS, Van Zwieten L, Krull E (2012) Marked changes in herbicide sorption–desorption upon ageing of biochars in soil. J Hazard Mater 231–232:70–78. https://doi.org/10.1016/j.jhazmat.2012.06.040
Mayakaduwa SS, Herath I, Ok YS et al (2017) Insights into aqueous carbofuran removal by modified and non-modified rice husk biochars. Environ Sci Pollut Res 24:22755–22763. https://doi.org/10.1007/s11356-016-7430-6
Meriam Suhaimy SH, Ghazali N, Arith F, Fauzi B (2020) Enhanced simazine herbicide degradation by optimized fluoride concentrations in TiO2 nanotubes growth. Optik (stuttg) 212:164651. https://doi.org/10.1016/j.ijleo.2020.164651
Merola VM, Eubig PA (2012) Toxicology of avermectins and milbemycins (macrocylic lactones) and the role of P-glycoprotein in dogs and cats. Vet Clin North Am Small Anim Pract 42:313–333. https://doi.org/10.1016/j.cvsm.2011.12.005
Mia, S. Dijkstra FA, Singh B (2017) Long-term aging of biochar: a molecular understanding with agricultural and environmental implications, 1st edn. Elsevier Inc. https://doi.org/10.1016/bs.agron.2016.10.001
Millar NS, Denholm I (2007) Nicotinic acetylcholine receptors: targets for commercially important insecticides. Invertebr Neurosci 7:53–66. https://doi.org/10.1007/s10158-006-0040-0
Narayanan M, Ma Y (2022) Influences of biochar on bioremediation/phytoremediation potential of metal-contaminated soils. Front Microbiol 13:1–11. https://doi.org/10.3389/fmicb.2022.929730
Nguyen BT, Lehmann J (2009) Black carbon decomposition under varying water regimes. Org Geochem 40:846–853. https://doi.org/10.1016/j.orggeochem.2009.05.004
Nie C, Yang X, Niazi NK et al (2018) Impact of sugarcane bagasse-derived biochar on heavy metal availability and microbial activity: a field study. Chemosphere 200:274–282. https://doi.org/10.1016/j.chemosphere.2018.02.134
Noyce GL, Basiliko N, Fulthorpe R et al (2015) Soil microbial responses over 2 years following biochar addition to a north temperate forest. Biol Fertil Soils 51:649–659. https://doi.org/10.1007/s00374-015-1010-7
Obia A, Børresen T, Martinsen V et al (2017) Vertical and lateral transport of biochar in light-textured tropical soils. Soil Tillage Res 165:34–40. https://doi.org/10.1016/j.still.2016.07.016
Ogura AP, Lima JZ, Marques JP et al (2021) A review of pesticides sorption in biochar from maize, rice, and wheat residues: current status and challenges for soil application. J Environ Manag. https://doi.org/10.1016/j.jenvman.2021.113753
Owens DK, Nanayakkara NPD, Dayan FE (2013) In planta mechanism of action of leptospermone: impact of its physico-chemical properties on uptake, translocation, and metabolism. J Chem Ecol 39:262–270. https://doi.org/10.1007/s10886-013-0237-8
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg. https://doi.org/10.1016/j.ijsu.2021.105906
Palansooriya KN, Wong JTF, Hashimoto Y et al (2019a) (2019b) Response of microbial communities to biochar-amended soils: a critical review. Biochar 1:3–22. https://doi.org/10.1007/s42773-019-00009-2
Palansooriya KN, Ok YS, Awad YM et al (2019b) Impacts of biochar application on upland agriculture: a review. J Environ Manage 234:52–64. https://doi.org/10.1016/j.jenvman.2018.12.085
Pariyar P, Kumari K, Jain MK, Jadhao PS (2020) Evaluation of change in biochar properties derived from different feedstock and pyrolysis temperature for environmental and agricultural application. Sci Total Environ 713:136433. https://doi.org/10.1016/j.scitotenv.2019.136433
Peshin R (2014) Environmental and economic costs of the application of pesticides primarily in the United States. Integr Pest Manag Pestic Probl 3:1–474. https://doi.org/10.1007/978-94-007-7796-5
Pignatello JJ, Mitch WA, Xu W (2017) Activity and reactivity of pyrogenic carbonaceous matter toward organic compounds. Environ Sci Technol 51:8893–8908. https://doi.org/10.1021/acs.est.7b01088
Qi Z, Miller GW, Voit EO (2014) Rotenone and paraquat perturb dopamine metabolism: a computational analysis of pesticide toxicity. Toxicology 315:92–101. https://doi.org/10.1016/j.tox.2013.11.003
Qiu X, Zhou G, Zhang J, Wang W (2019) Microbial community responses to biochar addition when a green waste and manure mix are composted: a molecular ecological network analysis. Bioresour Technol 273:666–671. https://doi.org/10.1016/j.biortech.2018.12.001
Qiu M, Liu L, Ling Q et al (2022) Biochar for the removal of contaminants from soil and water: a review. Biochar 4:19. https://doi.org/10.1007/s42773-022-00146-1
Rajapaksha AU, Chen SS, Tsang DCW et al (2016) Engineered/designer biochar for contaminant removal/immobilization from soil and water: potential and implication of biochar modification. Chemosphere 148:276–291. https://doi.org/10.1016/j.chemosphere.2016.01.043
Rajmohan KS, Chandrasekaran R, Varjani S (2020) A review on occurrence of pesticides in environment and current technologies for their remediation and management. Indian J Microbiol 60:125–138. https://doi.org/10.1007/s12088-019-00841-x
Rasool S, Rasool T, Gani KM (2022) A review of interactions of pesticides within various interfaces of intrinsic and organic residue amended soil environment. Chem Eng J Adv. https://doi.org/10.1016/j.ceja.2022.100301
Regitano JB, Koskinen WC, Sadowsky MJ (2006) Influence of soil aging on sorption and bioavailability of simazine. J Agric Food Chem 54:1373–1379. https://doi.org/10.1021/jf052343s
Ren X, Wang F, Zhang P et al (2018) Aging effect of minerals on biochar properties and sorption capacities for atrazine and phenanthrene. Chemosphere. https://doi.org/10.1016/j.chemosphere.2018.04.125
Rico A, Satapornvanit K, Haque MM et al (2012) Use of chemicals and biological products in Asian aquaculture and their potential environmental risks: a critical review. Rev Aquac 4:75–93. https://doi.org/10.1111/j.1753-5131.2012.01062.x
Sadet-bourgeteau S, Pr NC (2023) Effects of biochar on soil microbial communities: a meta-analysis. Sci Total Environ 902:166079. https://doi.org/10.1016/j.scitotenv.2023.166079
Safaei Khorram M, Zhang Q, Lin D et al (2016) Biochar: a review of its impact on pesticide behavior in soil environments and its potential applications. J Environ Sci 44:269–279. https://doi.org/10.1016/j.jes.2015.12.027
Sakhiya AK, Anand A, Kaushal P (2020) Production, activation, and applications of biochar in recent times. Biochar. https://doi.org/10.1007/s42773-020-00047-1
Salgado VL (2017) Insect TRP channels as targets for insecticides and repellents. J Pestic Sci 42:1–6. https://doi.org/10.1584/jpestics.D16-104
Satapute P, Kamble MV, Adhikari SS, Jogaiah S (2019) Influence of triazole pesticides on tillage soil microbial populations and metabolic changes. Sci Total Environ 651:2334–2344. https://doi.org/10.1016/j.scitotenv.2018.10.099
Shahid M, Pinelli E, Dumat C (2012) Review of Pb availability and toxicity to plants in relation with metal speciation; role of synthetic and natural organic ligands. J Hazard Mater 219–220:1–12. https://doi.org/10.1016/j.jhazmat.2012.01.060
Shukla V, Kumar S, Kumar N (2017) Plant adaptation strategies in changing environment. Springer Singapore, Singapore. https://doi.org/10.1007/978-981-10-6744-0
Silver KS, Du Y, Nomura Y, et al (2014) Voltage-gated sodium channels as insecticide targets. In: Advances in insect physiology. Elsevier, pp 389–433. https://doi.org/10.1016/B978-0-12-417010-0.00005-7
Singh B, Singh BP, Cowie AL (2010) Characterisation and evaluation of biochars for their application as a soil amendment. Soil Res 48:516. https://doi.org/10.1071/SR10058
Sizmur T, Fresno T, Akgül G et al (2017) Biochar modification to enhance sorption of inorganics from water. Bioresour Technol 246:34–47. https://doi.org/10.1016/j.biortech.2017.07.082
Song D, Xi X, Zheng Q et al (2019) Soil nutrient and microbial activity responses to two years after maize straw biochar application in a calcareous soil. Ecotoxicol Environ Saf 180:348–356. https://doi.org/10.1016/j.ecoenv.2019.04.073
Song B, Zhou C, Qin M et al (2023) When biochar is involved in rhizosphere dissipation and plant absorption of pesticides: a meta-analysis. J Environ Manage 345:118518. https://doi.org/10.1016/j.jenvman.2023.118518
Sopeña F, Semple K, Sohi S, Bending G (2012) Assessing the chemical and biological accessibility of the herbicide isoproturon in soil amended with biochar. Chemosphere 88:77–83. https://doi.org/10.1016/j.chemosphere.2012.02.066
Sorrenti G, Masiello CA, Dugan B, Toselli M (2016) Biochar physico-chemical properties as affected by environmental exposure. Sci Total Environ 563:237–246. https://doi.org/10.1016/j.scitotenv.2016.03.245
Spokas KA, Koskinen WC, Baker JM, Reicosky DC (2009) Impacts of woodchip biochar additions on greenhouse gas production and sorption/degradation of two herbicides in a Minnesota soil. Chemosphere 77:574–581. https://doi.org/10.1016/j.chemosphere.2009.06.053
Sun K, Gao B, Ro KS et al (2012) Assessment of herbicide sorption by biochars and organic matter associated with soil and sediment. Environ Pollut 163:167–173. https://doi.org/10.1016/j.envpol.2011.12.015
Sun Y, Gao B, Yao Y et al (2014) Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem Eng J 240:574–578. https://doi.org/10.1016/j.cej.2013.10.081
Sun B, Lian F, Bao Q et al (2016) Impact of low molecular weight organic acids (LMWOAs) on biochar micropores and sorption properties for sulfamethoxazole. Environ Pollut 214:142–148. https://doi.org/10.1016/j.envpol.2016.04.017
Sun Y, Xiong X, He M et al (2021) Roles of biochar-derived dissolved organic matter in soil amendment and environmental remediation: a critical review. Chem Eng J 424:130387. https://doi.org/10.1016/j.cej.2021.130387
Sun W, Zhang S, Su C (2018) Impact of biochar on the bioremediation and phytoremediation of heavy metal(loid)s in Soil. In: Advances in bioremediation and phytoremediation. InTech. https://doi.org/10.5772/intechopen.70349
Susilawati A, Maftuah E, Fahmi A (2020) The utilization of agricultural waste as biochar for optimizing swampland: a review. IOP Conf Ser Mater Sci Eng 980:012065. https://doi.org/10.1088/1757-899X/980/1/012065
Tatarková V, Hiller E, Vaculík M (2013) Impact of wheat straw biochar addition to soil on the sorption, leaching, dissipation of the herbicide (4-chloro-2-methylphenoxy)acetic acid and the growth of sunflower (Helianthus annuus L.). Ecotoxicol Environ Saf 92:215–221. https://doi.org/10.1016/j.ecoenv.2013.02.005
Tomczyk A, Sokołowska Z, Boguta P (2020) Biochar physicochemical properties: pyrolysis temperature and feedstock kind effects. Rev Environ Sci Bio/technology 19:191–215. https://doi.org/10.1007/s11157-020-09523-3
Trigo C, Spokas KA, Cox L, Koskinen WC (2014) Influence of soil biochar aging on sorption of the herbicides MCPA, Nicosulfuron, Terbuthylazine, Indaziflam, and Fluoroethyldiaminotriazine. J Agric Food Chem 62:10855–10860. https://doi.org/10.1021/jf5034398
Trigo C, Spokas KA, Hall KE et al (2016) Metolachlor sorption and degradation in soil amended with fresh and aged biochars. J Agric Food Chem 64:3141–3149. https://doi.org/10.1021/acs.jafc.6b00246
Tripathi S, Srivastava P, Devi RS, Bhadouria R (2020) Influence of synthetic fertilizers and pesticides on soil health and soil microbiology. In: agrochemicals detection, treatment and remediation. Elsevier, pp 25–54. https://doi.org/10.1016/B978-0-08-103017-2.00002-7
Troiano J, Weaver D, Marade J et al (2001) Summary of well water sampling in California to detect pesticide residues resulting from nonpoint-source applications. J Environ Qual 30:448–459. https://doi.org/10.2134/jeq2001.302448x
Tudi M, Li H, Li H et al (2022) Exposure routes and health risks associated with pesticide application. Toxics 10:335. https://doi.org/10.3390/toxics10060335
Twagirayezu G, Huang K, Sang C et al (2022) Performance and mechanisms of biochar for promoting the removal efficiency of organic solids in the vermi-wetland during the recycling of excess sludge. J Clean Prod 360:132172. https://doi.org/10.1016/j.jclepro.2022.132172
Twagirayezu G, Huang K, Xia H (2023) Effects of bio-contaminants in organic waste products on the soil environment. In: Fate of biological contaminants during recycling of organic wastes. Elsevier, pp 187–212. https://doi.org/10.1016/B978-0-323-95998-8.00013-3
Uchimiya M (2014) Influence of pH, ionic strength, and multidentate ligand on the interaction of Cd II with biochars. ACS Sustain Chem Eng 2:2019–2027. https://doi.org/10.1021/sc5002269
Usman ARA, Al-wabel MI, Ok YS et al (2016) Conocarpus biochar induces changes in soil nutrient availability and tomato growth under saline irrigation. Pedosphere 26:27–38. https://doi.org/10.1016/S1002-0160(15)60019-4
Usman M, Hao S, Chen H et al (2019) Molecular and microbial insights towards understanding the anaerobic digestion of the wastewater from hydrothermal liquefaction of sewage sludge facilitated by granular activated carbon (GAC). Environ Int 133:105257. https://doi.org/10.1016/j.envint.2019.105257
Uwamungu JY, Nartey OD, Uwimpaye F et al (2019) Evaluating biochar impact on topramezone adsorption behavior on soil under no-tillage and rotary tillage treatments: Isotherms and kinetics. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph16245034
Wang H, Lin K, Hou Z et al (2010) Sorption of the herbicide terbuthylazine in two New Zealand forest soils amended with biosolids and biochars. J Soils Sediments 10:283–289. https://doi.org/10.1007/s11368-009-0111-z
Wang J, Pan X, Liu Y et al (2012) Effects of biochar amendment in two soils on greenhouse gas emissions and crop production. Plant Soil 360:287–298. https://doi.org/10.1007/s11104-012-1250-3
Wang P, Yin Y, Guo Y, Wang C (2015) Removal of chlorpyrifos from waste water by wheat straw-derived biochar synthesized through oxygen-limited method. RSC Adv 5:72572–72578. https://doi.org/10.1039/C5RA10487D
Wang B, Gao B, Fang J (2017) Recent advances in engineered biochar productions and applications. Crit Rev Environ Sci Technol 47:2158–2207. https://doi.org/10.1080/10643389.2017.1418580
Wang H, Nan Q, Waqas M, Wu W (2022) Stability of biochar in mineral soils: assessment methods, influencing factors and potential problems. Sci Total Environ 806:150789. https://doi.org/10.1016/j.scitotenv.2021.150789
Wani I, Kushvaha V, Garg A et al (2022) Review on effect of biochar on soil strength: towards exploring usage of biochar in geo-engineering infrastructure. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-022-02795-5
Wołejko E, Jabłońska-Trypuć A, Wydro U et al (2020) Soil biological activity as an indicator of soil pollution with pesticides – a review. Appl Soil Ecol 147:103356. https://doi.org/10.1016/j.apsoil.2019.09.006
Xie S, Wan R, Wang Z, Wang Q (2013) Atrazine biodegradation by Arthrobacter strain DAT1: effect of glucose supplementation and change of the soil microbial community. Environ Sci Pollut Res 20:4078–4084. https://doi.org/10.1007/s11356-012-1356-4
Xie S, Wang Y, Ma C et al (2023) Pyrolysis of antibiotic mycelial residue for biochar: kinetic deconvolution, biochar properties, and heavy metal immobilization. J Environ Manage 328:116956. https://doi.org/10.1016/j.jenvman.2022.116956
Xing D, Cheng H, Ning Z et al (2022) Field aging declines the regulatory effects of biochar on cadmium uptake by pepper in the soil. J Environ Manage 321:115832. https://doi.org/10.1016/j.jenvman.2022.115832
Xu C, Liu W, Sheng GD (2008) Burned rice straw reduces the availability of clomazone to barnyardgrass. Sci Total Environ 392:284–289. https://doi.org/10.1016/j.scitotenv.2007.11.033
Xu D, Cao J, Li Y et al (2019) Effect of pyrolysis temperature on characteristics of biochars derived from different feedstocks: a case study on ammonium adsorption capacity. Waste Manag 87:652–660. https://doi.org/10.1016/j.wasman.2019.02.049
Yadav S, Singh SR, Bahadur L et al (2023) Sugarcane trash ash affects degradation and bioavailability of pesticides in soils. Sugar Tech 25:77–85. https://doi.org/10.1007/s12355-022-01197-1
Yang Y, Sheng G (2003) Enhanced pesticide sorption by soils containing particulate matter from crop residue burns. Environ Sci Technol 37:3635–3639. https://doi.org/10.1021/es034006a
Yang Y, Sheng G, Huang M (2006) Bioavailability of diuron in soil containing wheat-straw-derived char. Sci Total Environ 354:170–178. https://doi.org/10.1016/j.scitotenv.2005.01.026
Yang Y, Wang X, Wang Y et al (2023) Pesticide contamination remediation by biochar-immobilized microorganisms: a review. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-023-05204-0
Yavari S, Malakahmad A, Sapari NB (2015) Biochar efficiency in pesticides sorption as a function of production variables—a review. Environ Sci Pollut Res 22:13824–13841. https://doi.org/10.1007/s11356-015-5114-2
Younis U, Malik SA, Rizwan M et al (2016) Biochar enhances the cadmium tolerance in spinach (Spinacia oleracea) through modification of Cd uptake and physiological and biochemical attributes. Environ Sci Pollut Res 23:21385–21394. https://doi.org/10.1007/s11356-016-7344-3
Yu X-Y, Ying G-G, Kookana RS (2006) Sorption and desorption behaviors of diuron in soils amended with charcoal. J Agric Food Chem 54:8545–8550. https://doi.org/10.1021/jf061354y
Yuan H, Zhu Z, Wei X et al (2019) Straw and biochar strongly affect functional diversity of microbial metabolism in paddy soils. J Integr Agric 18:1474–1485. https://doi.org/10.1016/S2095-3119(18)62102-1
Zhang J, Shen JL (2022) Effects of biochar on soil microbial diversity and community structure in clay soil. Ann Microbiol 72:1–14. https://doi.org/10.1186/s13213-022-01689-1
Zhang P, Sun H, Yu L, Sun T (2013) Adsorption and catalytic hydrolysis of carbaryl and atrazine on pig manure-derived biochars: impact of structural properties of biochars. J Hazard Mater 244–245:217–224. https://doi.org/10.1016/j.jhazmat.2012.11.046
Zhang A, Li X, Xing J, Xu G (2020a) Adsorption of potentially toxic elements in water by modified biochar: a review. J Environ Chem Eng 8:104196. https://doi.org/10.1016/j.jece.2020.104196
Zhang H, Yuan X et al (2020b) Bioremediation of co-contaminated soil with heavy metals and pesticides: influence factors, mechanisms and evaluation methods. Chem Eng J 398:125657. https://doi.org/10.1016/j.cej.2020.125657
Zhao L, Cao X, Mašek O, Zimmerman A (2013) Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J Hazard Mater 256–257:1–9. https://doi.org/10.1016/j.jhazmat.2013.04.015
Zheng W, Guo M, Chow T et al (2010) Sorption properties of greenwaste biochar for two triazine pesticides. J Hazard Mater 181:121–126. https://doi.org/10.1016/j.jhazmat.2010.04.103
Zheng H, Zhang Q, Liu G et al (2019) Characteristics and mechanisms of chlorpyrifos and chlorpyrifos-methyl adsorption onto biochars: influence of deashing and low molecular weight organic acid (LMWOA) aging and co-existence. Sci Total Environ 657:953–962. https://doi.org/10.1016/j.scitotenv.2018.12.018
Zhou J, Chen H, Thring RW, Arocena JM (2019) Chemical pretreatment of rice straw biochar: effect on biochar properties and hexavalent chromium adsorption. Int J Environ Res 13:91–105. https://doi.org/10.1007/s41742-018-0156-1
Zhu X, Chen B, Zhu L, Xing B (2017) Effects and mechanisms of biochar-microbe interactions in soil improvement and pollution remediation: a review. Environ Pollut 227:98–115. https://doi.org/10.1016/j.envpol.2017.04.032
Acknowledgements
The authors of this review paper deeply acknowledge the State Key Laboratory of Environmental Geochemistry, the Chinese Academy of Sciences, the Guizhou Provincial Department of Science and Technology, and the Alliance of International Science Organizations (ANSO) Scholarship for Young Talents for supporting this work.
Funding
This review paper was financed by the "Light of West China" Program, Opening Fund of the State Key Laboratory of Environmental Geochemistry (SKLEG 2022216), and the Guizhou Provincial Department of Science and Technology (E2DF028).
Author information
Authors and Affiliations
Contributions
All authors collaborated in conceiving the idea and planning the execution of this review paper. GT, HC, and YW collected and analyzed information. GT, HL, SH, XF, and OI created the figures. GT drafted the initial version of the manuscript, which underwent review by HC, YW, HL, SH, XF, and OI. The final manuscript received thorough scrutiny and approval from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors do not have any conflicts of interest to disclose that would be pertinent to the content presented in this review paper.
Additional information
Handling Editor: Baoshan Xing.
Supplementary Information
Additional file 1: Fig. S1.
Classification of pesticides in the environment.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Twagirayezu, G., Cheng, H., Wu, Y. et al. Insights into the influences of biochar on the fate and transport of pesticides in the soil environment: a critical review. Biochar 6, 9 (2024). https://doi.org/10.1007/s42773-024-00301-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-024-00301-w