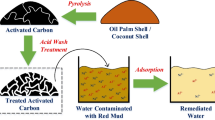
Abstract
Various human activities have led to multiple contamination of natural water systems. The present study investigated the effect of a novel multifunctional biochar to treat nutrients, oil, and harmful algae in water. Specifically, magnesium (Mg) and biosurfactant rhamnolipid (RL) were incorporated into biochar, including Mg-biochar, RL-biochar, and Mg-RL-biochar. Their adsorption efficiency on phosphate and total petroleum hydrocarbons (TPH) was evaluated in separate batch studies. Also, the inhibition effect of RL-modified biochars on cyanobacteria was investigated. The results showed that Mg-impregnated biochar showed high adsorption capacity on phosphate (118 mg g−1), while RL-modified biochar significantly reduced TPH (especially aromatic and light aliphatic fraction) with adsorption capacity of 44.4 mg g−1. The inhibition effects of biochar composites on algae in water without contaminants were in order of Mg-RL-biochar > RL-biochar > biochar with biomass reduction ranging 61–64%. Overall, Mg-RL-biochar was suggested based on this study due to its ability to remove PO43− and TPH, and inhibit the growth of toxic algae.
Graphical abstract

Highlights
-
Mg-impregnated biochar showed high adsorption ability on PO43−.
-
RL-modified biochar showed high adsorption ability on TPH.
-
Mg-RL-biochar could remove PO43−, TPH, and inhibit toxic algae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Various human activities have led to the deterioration in the environment including the pollution of aquatic systems. For instance, oil contamination caused by oil spill event (Atlas and Hazen 2011), nutrient (e.g. PO43− and NO3−) enrichment induced from agricultural or sewage runoff (Day et al. 2012), and subsequent stimulated harmful algae bloom were reported in Gulf region in the U.S. (Wang and Gaston 2014), and Lake Taihu in China (Guo et al. 2012; Wang et al. 2019). These issues posed risks to aquatic ecosystems due to the growth disruption of aquatic organisms, causing eutrophication and water hypoxia, affecting ecosystem function and service (Rastogi et al. 2015; Bhateria and Jain 2016), and influencing the safety of drinking water (Dittmann and Wiegand 2006). Particularly, in Persian Gulf and Bohai Bay (China), there is a co-existance of nutrients (especially PO43−) and petroleum hydrocarbons as well as aquatic algae (Gu et al. 2022; Mirsaeidi 2023; Shang et al. 2015). Therefore, removing these contaminants from water body is essential, which requires a highly efficient and ecosystem-friendly remediation strategy.
Different approaches have been used for treating contaminant-rich eutrophic wastewaters including biological, physical, and chemical or a combination of these methods (Escudero et al. 2015; Delgadillo-Mirquez et al. 2016; Xia et al. 2016). Compared with other adsorbents, biochar is derived from plant or animal waste biomass and is inexpensive and eco-friendly (Lalley et al. 2016; Wang et al. 2017; Li et al. 2018b). Biochar, with the properties of the significant presence of surface functional groups, large surface area, and high porosity structures, has shown high removal efficiency of various organic and inorganic contaminants such as dye (Yang et al. 2016; Thines et al. 2017; Park et al. 2019), estrogen hormone (Sangeeta et al. 2015; Jiang et al. 2017; Liu et al. 2020), polyaromatic hydrocarbons (PAHs) (de Jesus et al. 2017; Rao et al. 2017; Kim and Hyun 2018), and heavy metals (Xiao et al. 2018; Wu et al. 2019) from water systems.
In recent years, research work has been focused on engineered carbonaceous materials through surface modification to achieve advanced adsorption capacity for specific contaminants. As a result, various high-efficiency biochar or metal-biochar composites have been developed for particular environmental application (Zhou et al. 2013; Wang et al. 2017; Li et al. 2018b). For example, Mg-modified biochar was reported high removal efficiency of phosphate (PO43−) and nitrate (NO3−) from water through adsorption, electrostatic attraction, and precipitation (Zhang et al. 2012; Li et al. 2017b; Xiao et al. 2020). Biosurfactants rhamnolipid (RL)-modified graphene was able to remove dyes from water (Wu et al. 2014). Graphene coating over biochar also improved the removal efficiency of PAHs such as phenanthrene from water (Tang et al. 2015; Dai et al. 2023a).
However, most previous studies emphasized the use of biochar in the remediation of one type of pollutant (Yan et al. 2022; Zhao et al. 2023). As natural aquatic systems and wastewater often contain mixture of various contaminants, treating these water systems with multifunctional biochar may become necessary as it could avoid high costs due to multiple steps of treatments (Tao et al. 2016; Du et al. 2023). In removing petroleum hydrocarbons from aquatic systems, biosurfactants such as RL with the hydrophilic-lipophilic property are often used to help the dispersion of petroleum hydrocarbons and increase the contact of biochar with oil contaminants for removal (Karlapudi et al. 2018; Wei et al. 2020a). As a kind of amphiphilic molecule, biosurfactants produced by microorganisms can be suitable for all kinds of environments with lower toxicity than chemically synthesised surfactants (Sun et al. 2019). Dell’Anno et al. (2018) has reported that biosurfactants can effectively enhance the biodegradation of crude oil by increasing the solubility and bioavailability of hydrophobic pollutants. Recently, researchers have found that a bacterial consortium constructed from petroleum degraders and biosurfactant producers in the bioremediation of oil-polluted sediment can enhance the removal of C8–C40 n-alkanes (80.2 ± 2.8% removal efficiency) and aromatic compounds (34.4 ± 10.8% removal efficiency) within 10 weeks (Zhou et al. 2023). Similarly, Dai et al. (2023b) integrated RL biosurfactants with slow-release nutrients, and the study showed that the combined application reduced heavy oil by 39.4%, which was 17.0% higher than the natural attenuation. On the other hand, our previous study found that RL could inhibit the growth of algae due to cell lysis in saline water wetland environment (Wei et al. 2020b). This suggests that it is possible to synthesize biosurfactant-modified biochar to treat oil-contaminated water meanwhile to minimize algae growth. Moreover, through modification with biosurfactants with hydrophilic-lipophilic property (Karlapudi et al. 2018), the mobility of biochar in water could be enhanced, which likely otherwise increased contact to contaminants for removal. Therefore, it is plausible to develop a biosurfactant-modified Mg-biochar for simultaneously treating water contaminated with nutrients, algae and other organic pollutants.
Therefore, we hypothesized the development of multifunctional biochar integrated RL and Mg modification for removing nutrients (especially PO43−) and petroleum hydrocarbons from water as well as inhibiting the growth of aquatic algae. The objectives of this study were to synthesize and characterize RL-modified Mg-biochar composite with high efficiency to treat water with nutrients and oil contamination as well as harmful algae bloom.
2 Materials and methods
2.1 Biochar preparation
2.1.1 Mg impregnation
The sugarcane harvest residue biomass used in this study was collected from Louisiana State University AgCenter Sugar Research Station at St. Gabriel (LSU Agcenter, LA, USA) and was cut into < 5 cm small pieces, washed with deionized (DI) water to remove dust, and dry in oven at 60 °C for overnight. The dry residue biomass was crushed by a high-speed rotary cutting mill, passed through a screen (0.12 mm), and used for biochar preparation. To prepare Mg-biochar, 30 g of biomass was added to a 500 mL flask of magnesium chloride (MgCl2) solution in desired concentrations and shaken at 120 rpm overnight. After that, the mixtures were heated at 80 °C until completely dry. Before pyrolysis, dried material was transferred into a porcelain crucible and placed in a muffle furnace under N2 flow of 400 mL min−1 for 30 min to remove oxygen (See Additional file 1: Fig. S1). After that, the mixture in the muffle furnace was heated at 10 °C min−1 and maintained the temperature at 550 °C for 1 h under N2 flow of 200 mL min−1 (Jeong et al. 2016). The biochar products were gently crushed and passed through a nylon sieve (0.12 mm), washed with DI water, and oven-dried at 105 °C to completely dry.
The Mg as well as other elements in biochar was analyzed after digestion with concentrated sulfuric acid and hydrogen peroxide using inductively coupled plasmaatomic emission spectrometry (ICP-AES, SPECTRO Plasma 3200, Germany) by ICP detection. Surface morphologies of the composites were characterized using a field emission gun scanning electron microscopy (FEG-SEM, JEOL 6335F, Japan) equipped with a transmission electron microscopy (JEOL 200CX TEM, Japan). The Brunauer–Emmett–Teller specific surface area (SBET) and pore size distribution were characterized using a Quantachrome surface analyzer which was evaluated by multipoint BET analysis of adsorption data points with relative pressures of 0.05–0.3 by BJH method, and surface elemental composition was determined using x-ray photoelectron spectroscopy (XPS, Japan) by a Kratos AXIS Ultra DLD spectrophotometer with an Al K X-ray source in the condition of 1486.6 eV of photons. Additional Fourier-transform infrared spectroscopy (FT-IR) (Nicolet iS50, USA) measurement was carried out to verify the addition of RL to biochar particle surface. X-ray diffraction (XRD) analysis was also carried out to identify any crystallographic structure in the biochar/composite samples by a computer-controlled X-ray diffractometer (Philips Electronic Instruments, Mahwah, NJ, USA) with a scan rate (2θ) of 0.02 per second.
2.1.2 Rhamnolipid biosurfactant functionalization
A portion of biochar and Mg-biochar were further modified with biosurfactant RL. Rhamnolipid functionalization was modified based on a procedure for synthesizing RL-graphene oxide composite residues described by Wu et al. (2014). Specifically, 100 mg biochar materials were added to 50 mL N, N-dimethylformamide (DMF) solvent and bath sonicated for 1 h to dissolve. With stirring, 300 mg RL was added to the biochar suspension with continuing sonication until RL was completely dissolved. Following the dissolution of biochar and RL, 0.5 g of N-(3-dimethylaminopropyl-N-ethylcarbodiimide) hydrochloride (EDC) and 100 mg of 4-(dimethylamino) pyridine (DMAP) were introduced to catalyze the reaction of RL to biochar particle surface through dehydration. The dehydration reaction was conducted with stirring and ultra-sonication. Once the reaction was complete, the RL-biochar suspension was precipitated with methanol, and the black solid precipitates were centrifuged, washed with ethanol and deionized water, and freeze-dried.
To determine the amount of RL on biochar, the concentration of RL in solvent system after modification was measured followed by methylene blue-based analysis (Pinzon and Ju 2009). Briefly, the sample pH was first adjusted to 2.3 ± 0.2 using 1 N HCl. The acidified sample was then extracted with fivefold volume of chloroform. The chloroform extract of 4 mL was carefully removed and put in contact with a freshly prepared methylene blue solution containing 200 μL of the 1 g L−1 methylene blue reagent, and 4.9 mL of distilled water. The pH of this aqueous solution of methylene blue was pre-adjusted to 8.6 ± 0.2 by adding 50 mM borax buffer (~ 15 μL). After being vigorously mixed for 4 min, the samples were left to stand for 15 min. The chloroform phase was transferred into a cuvette and measured for absorbance at 638 nm using a Shimadzu UV/V spectrophotometer (Model 1601). All chemicals were purchased from Sigma-Aldrich. All biochar composites were sterilized before adsorption experiment.
2.2 Synthesized biochar composites on PO4 3− adsorption
Batch experiments were conducted to examine the adsorption efficiency of PO43− from aqueous solutions by Mg-modified biochar. In this way, biochar of 0.05 g was added to a polyethylene centrifuge tube containing 20 mL of 50 mg L−1 solution diluted from a 1000 mg L−1 PO43− stock solution. All the experiments were performed in triplicate. After 24 h mixing, sample mixture was centrifuged and the concentration of PO43− in the supernatant was analyzed using a Thermo Scientific EVO 60 spectrophotometer (USA) at wavelength of 880 nm (USEPA 1983). Freundlich (Eq. 1) and Langmuir Eq. (2) were used to describe PO43− adsorption isotherm data, which are expressed as:
where qe (μg g−1) is the equilibrium sorbed concentration, Ce (μg L−1) is the equilibrium aqueous concentration, KF (μg g−1 / (μg L−1)n) is the Freundlich affinity coefficient, n is the Freundlich exponential coefficient, Qmax (μg g−1) is the maximum capacity, and KL (L g−1) is the bonding energy constant.
2.3 Synthesized biochar composites on oil removal
The effect of RL-functionalized biochar composites on oil adsorption was evaluated with comparison to that without RL modification. In this way, the synthesized biochar composites with different levels of RL were mixed with water containing different amounts oil (0.1–5.0%) in 50 mL Teflon tubes at solid: liquid ratio of 1:100. After 24 h shaking, the mixtures were centrifuged at 5000 rpm for 5 min and the liquid phase was transferred to 250-mL separation funnel, followed by adding DCM (v/v = 1:1) solvent to break the phase. After 1 h separation, the organic phase of DCM that contains petroleum hydrocarbons was collected from the bottom of the funnel. The DCM extracts were concentrated using a N2 evaporator (Organomation; Mode 11645) in a 50 °C water bath until completely dry. Dichloromethane was added to re-dissolve the dry material and bring the final volume to 1.0 mL before transferring into GC vials for analysis. Petroleum hydrocarbons in the extracts were quantified using a Shimadzu 2010 GC by flame ionization detection (FID) following a procedure described by Wei et al. (2020a). Total flow rate for carrier gas was set at 6.7 mL min−1 with ultra-pure helium (99.9%, Airgas, USA). The injection port was held at 290 °C and a 1.0 µL injection volume was used. The column was 30 m × 0.25 mm × 0.50 µm capillary column (Rxi®-5Sil MS, Restek, USA) with temperature programmed from 50 to 280 °C at 6 °C min−1 after a 2-min hold, and then increased to 300 °C at 1.5 °C min−1after a 2-min hold. The total analysis time was 57.67 min. The FID detector was set at 300 °C. The oil standard with 44 hydrocarbon compounds ranging from C10 to C35 (Absolute Standard Inc., CT; Part# 90311) was used as an external standard with concentration ranges from 0 to 10 ppm for each compound to calibrate the GC before sample analysis. The oil sorption isotherms were fitted by Langmuir and Freundlich models as described in 2.2.
2.4 Synthesized biochar composites on the removal of cyanobacteria
The effectiveness of algal inhibition of the prepared biochar composites was evaluated by algal bioassays (method ISO 8692-2004). Cyanobacteria Microcystis aeruginosa, a species causing harmful algae blooms, was used for this experiment. Microcystis aeruginosa was purchased from the UTEX Culture Collection of Algae of University of Texas at Austin. In brief, Microcystis aeruginosa was inoculated into a culture medium (bold 3N Medium from UTEX) in a 500-mL flask. Approximately 0.5 g of each prepared biochar composite sample sealed in a polythene mesh bag was added to a 500-mL flask with the initial algal density (overall final composite solid: liquid ratio of 1:500). The algae culture without biochar composite sample was labeled as the control. All flasks were cultured at 25 ± 0.2 °C under photosynthetically active radiation (PAR) at 350 mmol m−2 s−1 lm direct light. The content of chlorophyll-a in Microcystis aeruginosa was assayed according to the classical ethanol extracting method (Ni et al. 2012). Algal cells were harvested by suction filtration of 10 mL algae mixture with a 0.45 mm cellulose membrane. All the cell pellets entrapped by the membrane were re-suspended in a centrifuge tube with 10 mL ethanol (95%) and then transferred into a 75 °C water bath for 10 min followed by moving into a 4 °C refrigerator without light. After 24 h extraction, the mixtures in the tubes were centrifuged at 8000 rpm for 10 min, and the supernatants were collected to measure absorbance (A) at 630, 645, and 663 nm with a spectronic 501 spectrophotometer, respectively (Bausch and Lomb, Milton Roy LLC, USA). The contents of chlorophyll were calculated by:
where A1, A2, and A3 are absorbance at 663 nm, 645 nm and 630 nm, respectively, and V1 and V2 are volumes (mL) of acetone used in the extraction and the filtered sample, respectively (Lee et al. 2013).
2.5 Statistics
All experiments in this study were set three replications. The significance of differences among treatments was compared using Tukey’s honest significant difference test (SAS 9.4, SAS Institute, Cary, NC). The parameters of isotherm models were obtained through MATLAB 7.0©.
3 Results and discussion
3.1 Characterization of biochars
Elemental composition and textural properties of the prepared biochar samples are given in Table 1. The total element analysis showed that about 12–13% Mg content was detected in Mg-modified biochars (Mg-biochar and Mg-RL biochar). The carbon content decreased from 51 to 33% after Mg impregnation. The surface area analysis showed the SBET were higher for Mg-biochar as compared to pristine biochar without metal impregnation, indicating significant enhancement of metal impregnation on surface area. The surface morphological features of biochars were characterized by SEM analysis (Fig. 1). In general, the pristine biochar showed smooth surface as well as sharp edges and corners (Fig. 1a), while the surfaces of Mg-biochar particles were very coarse with some metal oxides covered in the surface homogeneously (Fig. 1d). The SEM scans reveal that the pore size of the Mg-biochar is larger and more developed. The Mg-biochar showed high EDX peaks for carbon, oxygen, chloride, and magnesium (Additional file 1: Fig. S4b), which are from the MgCl2 used in biomass impregnation. Besides, silicon, a common element within sugarcane residue, was also found high peak in EDX pattern of Mg-biochar sample. The XRD analysis of Mg-biochar also showed the peaks at 2θ values of 36.9°, 42.9°, and 62.2° (Fig. 2), which indicate the presence of MgO (Wetteland et al. 2018). Additionally, the peak of Mg 2p with binding energies of 50.72 and 51.55 were likely attributed to MgO (Chen and Zhu 2019), which is suggested formed by thermally decomposition of Mg-preloaded sugarcane residue powder during pyrolysis (Li et al. 2016). In TEM results (Fig. 3), the incorporation of Mg increased the crystalline diameter of biochar from 8.3 nm (biochar) to 26 nm (Mg-biochar) and 30 nm (Mg-RL biochar). As clearly seen in the figure, flake-like structure was observed in biochar incorporated with Mg, further indicating the formation of MgO crystal on biochar.
Rhamnolipid modification increased the surface area of biochar with SBET 2.1 times higher than original biochar (Table 1). The SEM images show that there are many crumplings on the surface of original biochar which come from the scrolling of biochar sheets (Fig. 1a). In contrast, the RL-biochar (Fig. 1d) has a relative smooth surface. It was likely that the existence of biosurfactant RL can effectively inhabit the agglomeration of biochar (Wu et al. 2014). In TEM results (Fig. 3), the incorporation of Mg and RL increased the crystalline diameter of biochar from 8.3 nm (biochar) to 10 nm (RL-biochar), and 30 nm (Mg-RL biochar). There was no obvious difference found with RL modification. The FT-IR spectrums of biochar with and without RL modification were shown in Fig. 4. Several characteristic FT-IR peaks of biochar were observed, such as, aromatic C=C (1624 cm−1), and aromatic C-H (751 cm−1). Similarly, in the FT-IR spectrum of RL-biochar, the same functional groups mentioned above were found around the corresponding wavenumber. The appearance of the peaks of C=O (1402 cm−1), C–O–C (1053 cm−1) were likely due to the formation of ester or –COOH, which indicated the successful binding of RL to biochar (Wu et al. 2014). Additionally, the mole ratio of carbon content to oxygen content (C/O) for biochar is 6.02 and for RL-biochar is 4.93 (Table 1), which implies that more oxygen-containing functional groups, such as C–OH, C=O were introduced. Additionally, a computational multi-peak resolution method for XPS has been applied to the C1s band of biochar and RL-biochar (Fig. 5). The full scan XPS spectrums showed the presence of C1s band and O1s at the binding energy of 284.80 eV and 533.09 eV for biochar (Fig. 5a, b), and 284.80 eV and 533.29 eV for biochar (Fig. 5e, f), respectively, which is corresponding to the C=C/C–C, C–O, carbonyl and carboxyl groups, respectively (Tiwari et al. 2013). The C1s band of RL-biochar (Fig. 5e) also could be deconvoluted into four peaks, corresponding to C=C/C–C (284.66 eV), C–O (286.36 eV), C=O (287.66 eV) and COOH (289.42 eV) (Wu et al. 2014). These results further confirmed the bonding between RL and biochar, and were agree with characterization results by FT-IR.
3.2 Effect of biochars on PO4 3− adsorption
The batch data for PO43− adsorption with biochar with and without Mg impregnation were fitted using Freundlich isotherm model (Fig. 6a) and the parameters are shown in Additional file 1: Table S1a. The PO43− adsorption by both biochar was well fitted by Freundlich model with R2 ranging 0.971–0.987. Compared with biochar, both Mg-biochar and Mg-RL-biochar have larger KF (0.25 vs. 12.59 and 10.83, respectively). The high values of KF indicate that Mg-modified biochar has a high adsorption capacity and affinity for PO43−. Since original biochar exhibited very limited ability to absorb anionic due to the net negatively charged surface (Chen et al. 2011), Mg impregnation could enhance the adsorption of PO43− by the formation of MgO during pyrolysis (Figs. 4, 6a). The adsorption mechanisms were suggested as the formation of outer-sphere surface complex between surface MgO and phosphate (Mg–O–P) and precipitation of insoluble P salt (MgHPO4 and Mg (H2PO4)2). Based on our results, the scanning electron microscope showed the significant surface morphology change of Mg-biochar, and indicated the precipitation of MgHPO4 and Mg(H2PO4)2 by the formation of prism-like crystals (Fig. 6a, c), and the EDS spectrums confirmed the presence of elements Mg, P and O in biochar after adsorption (Additional file 1: Fig. S4), further verifying the precipitation of MgHPO4 and Mg (H2PO4)2 on biochar surface (Fig. 6b, d). On the other hand, among the biochars with Mg impregnation, biochars with RL co-treatment showed a lower adsorption ability towards PO43− with slightly lower KF values (Mg-biochar > Mg-RL-biochar). This result indicates that the presence of RL may interfere the binding of Mg oxide on biochar surface. Overall, the results above showed the effectiveness of Mg-decorated biochar on the adsorption of PO43− in water.
The Langmuir maximum adsorption capacity of Mg-biochar was 118.31 μg g−1 (Additional file 1: Table S1a). The maximum adsorption capacity of biochar with about 13% Mg (Table 1) in this study was slightly less than that of 121.25 mg P/g of biochar with 20% Mg that reported by Li et al. (2016), suggesting the comparable adsorption ability of biochar with 15%, 20%, and 25% Mg as the results of our preliminary study (Additional file 1: Fig. S2).
Due to the insufficient affinity of pristine biochar to anions (Li et al. 2018c), impregnation of metals to form metal oxide-biochar composites are one of the efficient ways to improve anion adsorption. Among various metal oxide-biochar composites, MgO-biochar composites were found to yield the highest adsorption capacity of phosphate with surface precipitation as MgHPO4 and Mg(H2PO4)2 and surface electrostatic attraction as the main retention mechanisms (Yao et al. 2013; Li et al. 2018a). Li et al. (2016) also prepared magnetic MgO-biochar composites with high phosphate removal efficiency. Besides surface electrostatic attraction and surface precipitation of PO43− with MgO, in treating with real livestock wastewater, MgO-biochar composites also demonstrated significant removal of ammonium beside phosphate through the formation of struvite (MgNH4PO4·6H2O) at biochar surface, strong adsorption of dissolved organic carbon, and clarified water (Li et al. 2017a).
3.3 Effect of biochars on TPH adsorption
The batch data for phosphate adsorption with biochar with and without RL treatment were fitted using the Freundlich isotherm model (Fig. 6b) and the parameters are shown in Additional file 1: Table S1b. The phosphate adsorption by both biochar could be well fitted with R2 ranging 0.985–0.994. Since this experiment was conducted in sterilized water, the dissipation of TPH should be attribute to adsorption process without microbial degradation. Compared with biochar, RL-biochar and Mg-RL-biochar showed much higher adsorption ability with greater KF (0.03 vs. 0.23 and 0.15, respectively). There was no significant difference (p > 0.05) between the adsorption capacities of RL-biochar and Mg-RL-biochar. The Langmuir maximum adsorption capacity of RL-biochar was 44.42 μg g−1 (Additional file 1: Table S1b). Additionally, the removal efficiency of TPH at around 50% RL addition was close to that of 75–100% addition (Additional file 1: Fig. S3), suggesting strong oil removal of Mg-biochar composite with surface modification of RL.
The fractions of petroleum hydrocarbons in water after adsorption by RL-modified biochar were grouped into heavy fraction aliphatic hydrocarbons (HFA; C26-C35), light fraction aliphatic hydrocarbons (LFA; C10-C25), and aromatic hydrocarbons (Fig. 7). The results show that RL modified-biochar significantly reduced the petroleum hydrocarbons in water especially for aromatic compounds and LFA but had little change on HFA. Specifically, higher surface area and aromaticity with lower polarity was suggested higher adsorption on polyaromatic hydrocarbons (PAHs) (Chen et al. 2011). Compared with original biochar, biochar with RL modification has higher surface area (Table 1) as well as higher aromaticity with higher H/C molar ratio (0.42 vs. 1.06; Table 1), therefore providing greater PAHs removal. The bonding between biochar aromatic rings and PAHs compounds was suggested as π–π electron donor–acceptor interactions (Wang et al. 2016; Kim and Hyun 2018). For aliphatic compounds in petroleum with low polarity, pore-filling was suggested as one of the dominant mechanisms for sorption to biochar (Chun et al. 2004). Apart from these, hydrophobic partition (de Jesus et al. 2017; Fu et al. 2018; Kandanelli et al. 2018), and pore filling (Hale et al. 2011) were also suggested responsible for TPH sorption of these biochars.
However, the surface modification of RL largely reduced the hydrophobicity of biochar, which may affect the partition of hydrophobic petroleum hydrocarbons. From SEM the images of biochar and RL-biochar before and after oil adsorption (Fig. 8b, d), the surface coverage by oil on RL-biochar was much larger than biochar, indicating the increased contact due to RL. The hydrophilic-lipophilic property of biosurfactant RL helped the dispersion of petroleum hydrocarbons and increased the contact of biochar with oil contaminants for removal (Karlapudi et al. 2018; Wei et al. 2020b). With biochar as carrier, RL could disperse oil in water while biochar assists the adsorption of oil. In addition, RL could improve the mobility of biochar in water and increase the contact of organic contaminants with biochar. Other studies have reported the integration use of surfactant with carbon material could improve the removal of organic contaminants. For example, RL-modified graphene oxide (Wu et al. 2014) and RL-modified biochar (Zhen et al. 2018) were found to increase the adsorption of methylene blue, with improved electrostatic attraction, π–π interaction, and hydrogen bond formation. Muhammad et al. (2015) showed the eggshell modified by chemical surfactant hexadecyl-trimethyl-ammonium-bromide (HDTMA-Br) for the effective removal of crude oil from water. The findings in current study further indicate the effectiveness of biosurfactant modification on biochar in treating oil contaminated water.
As chemical removal approach, surfactants are commonly used to enhance the processes of remediation due to the hydrophobicity property of many organic pollutants by reducing the surface and interfacial tension between organic phase and water phase, and enhance the processes of remediation by means of emulsification, solubilization and mobilization (Uzoigwe et al. 2015). Recently, biosurfactants have been found effective in improving the adsorption efficiency of organic contaminants by surface modification onto or integrated use with carbon-based materials (Wu et al. 2014; Zhen et al. 2018). For example, RL-modified graphene oxide was found to increase the adsorption of methylene blue, and the adsorption mechanisms were attributed to increased functional groups, which improved electrostatic attraction, π–π interaction, and hydrogen bond formation (Wu et al. 2014; Zhen et al. 2018).
3.4 Inhibition effect of biochars on algae growth
The change of algal biomass with various biochar amendment during three week incubation is shown in Fig. 9. Generally, compared to original biochar, RL-modified biochar showed the anti-algal effect after 3-week incubation with biomass reduction of 61% for RL-biochar, and 64% for Mg-RL-biochar. Additionally, the suppression effect of RL-biochar on algae was likely due to the cell lysis or damage to the plasma membrane and organelles caused by RL (Wang et al. 2005; Invally and Ju 2017). The consistent result was also found in our previous study that RL could reduce algae biomass in wetland (Wei et al. 2020b). Current study further indicates the effectiveness of RL biosurfactant on algae inhibition when attached on biochar through engineering. Yet, when co-treated with Mg, Mg-RL-biochar showed little change. Moreover, when RL-biochar was applied with double doses, biomass loss increased to 98%. Thus, based on the findings in this study, double doses of RL-biochar are recommended when treating algae in water.
Biosurfactant RL with hydrophilic-lipophilic property could cause cell lysis or damage to the plasma membrane and organelles (Wang et al. 2005; Invally and Ju 2017), and has been also used as algaecide (Dashtbozorg et al. 2019). Several studies reported the inhibition effect of RL on water algae growth (Wang et al. 2005; Gustafsson et al. 2009; Dashtbozorg et al. 2019). In our results, it is interesting that RL with 1 g L−1 and 2 g L−1 showed great difference (The cultivation time of algae was two weeks). To further investigate the effect of RL-biochar, a series of application dose were tested (Additional file 1: Fig. S5). The results showed the inhibition effect on algae was increased with the addition concentration of RL-biochar.
3.5 Integration effect of biochars in water with multiple contaminates
In eutrophic water with multiple contaminates including PO43− and oil, modified biochar showed different algae inhibition effects, which depended on the relative of ratio of PO43− and oil. Compared to water with low relative level phosphorous (P:Oil = 1:5), algal biomass in water with high level of PO43− (P:Oil = 5:1) was significantly (p < 0.05) lower with about 50% chlorophyll-a content, suggesting the promotion effect of PO43− on algal growth (Fig. 10). On the other hand, compared to water with low level of oil (P:Oil = 5:1), algal biomass in water with high level of oil (P:Oil = 1:5) was significantly (p < 0.05) lower, suggesting that oil had a toxic effect on algal growth (Fig. 10). The decrease in photosynthetic activity, chlorophyll content, and carbon uptake of algae after exposure to petroleum hydrocarbons was also reported other studies (Batterton et al. 1978; Gonzalez et al. 2009; Carrera Martinez et al. 2011; Jaiswar et al. 2013). These results suggested that activity and biomass of algae are likely higher in high P-impacted water than in oil-impacted water.
Additionally, compared with oil-dominant water (P:Oil = 1:5), Mg-biochar showed stronger inhibition effect in phosphorous-dominant water (P:Oil = 5:1) with 37% algae biomass reduced, indicating the reduced nutrient P in water due to Mg-biochar adsorption. Overall, these findings gave suggestions in choosing modification of biochar when treating water with different contamination.
3.6 Implications
When Cu-containing materials are among the most commonly used strategies to control algae, biosurfactants is rarely applied as algalcide in field scale. Although algicidal activity of single use of RL on species Heterosigma akashiwo, Phagotrophic Alga Ochromonas danica have been found by several studies (Wang et al. 2005; Invally and Ju 2017), and natural algae in wetland environment (Wei et al. 2020b). The results in current study indicated that as attached to surface of carbon material, RL is also capable of suppressing the growth of harmful algae cyanobacteria species. The effect could be further enhanced especially when applied with high dose (Fig. 9). When RL was added on Mg-biochar, the inhibition effect was further enhanced. The application of RL has low safety concern due to its high biodegradability in natural environment.
For TPH removal, Mg-RL-biochar showed comparable effect as RL-biochar (Fig. 6b), suggesting that as surface modification on biochar, Mg-impregnation did not reduce the effect of RL. On the other hand, a preliminary experiment was conducted to evaluate the potential of MgO-biochar composite modified with different levels of RL on phosphate sorption (Additional file 1: Fig. S2). Rhamnolipid modification would only affect the performance of Mg-biochar composite on phosphorous sorption capacity when its concentration is over 50% of Mg-biochar amount, suggesting that the potential interference of RL on biochar surface for the function of nutrient removal is relatively low.
It is noticeable that although biochar could also be a source of PAHs (Domene et al. 2015), the overall significant reduction of aromatic petroleum hydrocarbons (Fig. 4) likely indicates the major adsorption by biochar. Particularly, a positive interaction of RL and biochar in oil remediation in wetland was suggested by our previous study (Wei et al. 2020a), high potential of RL-biochar in removing oil in water could be expected. Further, this experiment was conducted in sterilized environment, thus dissipation of oil by RL-biochar could be underestimated under real eutrophic water matrices with natural oil degrading microorganisms, which attributed to both adsorption and biodegradation processes. Overall, Mg-RL-biochar were suggested based on this study due to its ability on removal of PO43− and TPH, as well as inhibition on growth of toxic algae. These results provide strong support of potential successful development of multifunctional biochar composites for remediation of water with different types of pollutants.
4 Conclusions
This study demonstrated the proposed hypothesis that the development of multifunctional biochar integrated by RL and Mg modification for removing nutrients (especially PO43−) and petroleum hydrocarbons from water as well as inhibiting the growth of aquatic algae. To sum up, Mg-impregnated biochar showed high adsorption ability on PO43− through surface electrostatic attraction with MgO and Mg-phosphate precipitation reactions, while RL-modified biochar showed high adsorption ability on TPH and algae inhibition effect by π-π interactions and hydrophobic effects. In water with PO43− and oil, Mg-RL-biochar showed the highest inhibition effect on algae. Thus, Mg-RL-biochar was suggested based on this study due to its ability to remove PO43− and TPH, as well as inhibiting the growth of toxic algae. Overall, our study developed efficient, multifunctional, environment-friendly biochar composites that can efficiently treat water contaminated with multiple types of pollutants, especially nutrients, hydrophobic organic contaminants and algae with the potential to be used in both natural and waste water systems. It is expected that these improved biochar composites will help recover nutrients, enhance water quality and maintain ecosystem service and stability.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Atlas RM, Hazen TC (2011) Oil biodegradation and bioremediation: a tale of the two worst spills in U.S. history. Environ Sci Technol 45(16):6709–6715
Batterton JC, Winters K, Van Baalen C (1978) Sensitivity of three microalgae to crude oils and fuel oils. Mar Environ Res 1(1):31–41
Bhateria R, Jain D (2016) Water quality assessment of lake water: a review. Sustain Water Resour Manag 2(2):161–173
Carrera-Martinez D, Mateos-Sanz A, Lopez-Rodas V, Costas E (2011) Adaptation of microalgae to a gradient of continuous petroleum contamination. Aquat Toxicol 101:342–350
Chen J, Zhu J (2019) A query on the Mg 2p binding energy of MgO. Res Chem Intermed 45:947–950
Chen BL, Chen ZM, Lv SF (2011) A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour Technol 102:716–723
Chun Y, Sheng G, Chiou CT, Xing B (2004) Compositions and sorptive properties of crop residue-derived chars. Environ Sci Technol 38:4649–4655
Costas E, LopezeRodas V (2006) Copper sulfate and DCMU-herbicide treatments increase asymmetry between sister cells in the toxic cyanobacteria Microcystis aeruginosa: implications for detecting environmental stress. Water Res 40:2447–2451
Dai J, Wang Z, Chen K, Ding D, Yang S, Cai T (2023a) Applying a novel advanced oxidation process of biochar activated periodate for the efficient degradation of bisphenol A: two nonradical pathways. Chem Eng J 453:139889
Dai X, Lv J, Zhang Z, Wang H (2023b) Bioremediation of heavy oil-contaminated intertidal zones using slow-release nutrients and rhamnolipid biosurfactants. J Environ Chem Eng 11(2):109323
Dashtbozorg SS, Invally K, Sancheti A, Ju LK (2019) Antimicrobial Applications of Rhamnolipids in Agriculture and Wound Healing. In Microbial Biosurfactants and their Environmental and Industrial Applications (pp. 56–80). CRC Press
Day JW, Hunter R, Keim RF, DeLaune R, Shaffer G, Evers E, Reed DJ, Brantley C, Kemp P, Day J et al (2012) Ecological response of forested wetlands with and without Large-Scale Mississippi River input: implications for management. Ecol Eng 2012(46):57–67
de Jesus JHF, Da G, Cardoso EMC, Mangrich AS, Romão LPC (2017) Evaluation of waste biomasses and their biochars for removal of polycyclic aromatic hydrocarbons. J Environ Manage 200:186–195
Delgadillo-Mirquez L, Lopes F, Taidi B, Pareau D (2016) Nitrogen and phosphate removal from wastewater with a mixed microalgae and bacteria culture. Biotechnol Rep (amst) 11:18–26
Dell’Anno F, Sansone C, Ianora A, Dell’Anno A (2018) Biosurfactant-induced remediation of contaminated marine sediments: current knowledge and future perspectives. Mar Environ Res 137:196–205
Dittmann E, Wiegand C (2006) Cyanobacterial toxins—occurrence, biosynthesis and impact on human affairs. Mol Nutr Food Res 50:7–17
Domene X, Enders A, Hanley K, Lehmann J (2015) Ecotoxicological characterization of biochars: role of feedstock and pyrolysis temperature. Sci Total Environ 512(513):552–561
Du C, Yang S, Ding D, Cai T, Chen R (2023) Origin of synergistic effect between Fe/Mn minerals and biochar for peroxymonosulfate activation. Chem Eng J 453:139899
Escudero A, Blanco F, Lacalle A, Pinto M (2015) Struvite precipitation for ammonium removal from anaerobically treated effluents. J Environ Chem Eng 3:413–419
Fu H, Wei C, Qu X, Li H, Zhu D (2018) Strong binding of apolar hydrophobic organic contaminants by dissolved black carbon released from biochar: a mechanism of pseudomicelle partition and environmental implications. Environ Pollut 232:402–410
Gonzalez J, Figueiras FG, Aranguren-Gassis M, Crespo BG, Fernandez E, Moran XAG, Nieto-Cid M (2009) Effect of a simulated oil spill on natural assemblages of marine phytoplankton enclosed in microcosms. Estuar Coast Shelf Sci 83(3):265–276
Gu R, Xu Y, Li Z, Jian S, Tu J, He S, Sun J (2022) PSR-FCCLP model based total maximum allocated loads optimization of TN and TP in Bohai Bay. Mar Pollut Bull 185(Pt A):114249
Guo J, Fang J, Cao J (2012) Characteristics of petroleum contaminants and their distribution in Lake Taihu, China. Chem Central J 6:92
Gustafsson S, Hultberg M, Figueroa RI, Rengefors K (2009) On the control of HAB species using low biosurfactant concentrations. Harmful Algae 8(6):857–863
Hale SE, Hanley K, Lehmann J, Zimmerman AR, Cornelissen G (2011) Effects of chemical, biological, and physical aging as well as soil addition on the sorption of pyrene to activated carbon and biochar. Environ Sci Technol 45:10445–10453
Invally K, Ju LK (2017) Biolytic effect of rhamnolipid biosurfactant and dodecyl sulfate against phagotrophic Alga Ochromonas Danica. J Surfactants Deterg 20(6):1161–1171
Jaiswar JRM, Ram A, Rokade MA, Karangutkar S, Yengal B, Dalvi S, Acharya D, Sharma S, Gajbhiye SN (2013) Phytoplankton dynamic responses to oil spill in Mumbai Harbour. Int J Innovat Biol Res 2(1):30–50
Jeong C, Dodla SK, Wang JJ (2016) Fundamental and molecular composition characteristics of biochars produced from sugarcane and rice crop residues and their by-products. Chemosphere 142:4–13
Jiang L, Liu Y, Liu S, Zeng G, Hu X, Hu X, Guo Z, Tan X, Wang L, Wu Z (2017) Adsorption of estrogen contaminants by graphene nanomaterials under natural organic matter preloading: comparison to carbon nanotube, biochar, and activated carbon. Environ Sci Technol 51(11):6352–6359
Kandanelli R, Meesala L, Kumar J, Raju CSK, Peddy VCR, Gandham S, Kumar P (2018) Cost effective and practically viable oil spillage mitigation: comprehensive study with biochar. Mar Pollut Bull 128:32–40
Karlapudi AP, Venkateswarulu TC, Tammineedi J, Kanumuri L, Ravuru BK, Dirisala V, Kodali VP (2018) Role of biosurfactants in bioremediation of oil pollution-a review. Petroleum 4(3):241–249
Kim J, Hyun S (2018) Sorption of ionic and nonionic organic solutes onto giant Miscanthus-derived biochar from methanol-water mixtures. Sci Total Environ 615:805–813
Lalley J, Han C, Li X, Dionysiou DD, Nadagouda MN (2016) Phosphate adsorption using modified iron oxide−based sorbents in lake water: Kinetics, equilibrium, and column tests. Chem Eng J 284:1386–1396
Lee MK, Park HJ, Kwon SH, Jung YJ, Lyu HN, Lee DG, Song NY, Choi HK, Park S, Baek NI, Lee YH (2013) Tellimoside, a flavonol glycoside from Brasenia schreberi, inhibits the growth of cyanobacterium (Microcystis aeruginosa LB 2385). J Korean Soc Appl Bi 56(1):117–121
Li R, Wang JJ, Zhou B, Awasthi MK, Ali A, Zhang Z, Lahori AH, Mahar A (2016) Recovery of phosphate from aqueous solution by magnesium oxide decorated magnetic biochar and its potential as phosphate-based fertilizer substitute. Bioresour Technol 215:209–214
Li J, Cao L, Yuan Y, Wang R, Wen Y, Man J (2017a) Comparative study for microcystin-LR sorption onto biochars produced from various plant- and animal-wastes at different pyrolysis temperatures: influencing mechanisms of biochar properties. Bioresour Technol 247:794–803
Li R, Wang JJ, Zhou B, Zhang Z, Liu S, Lei S, Xiao R (2017b) Simultaneous capture removal of phosphate, ammonium and organic substances by MgO-impregnated biochar and its potential use in swine wastewater treatment. J Cleaner Prod 147:97–107
Li R, Liang W, Wang JJ, Gaston LA, Huang D, Huang H, Lei S, Awasthi MK, Zhou B, Xiao R, Zhang Z (2018a) Facilitative capture of As(V), Pb(II) and methylene blue from aqueous solutions with MgO hybrid sponge-like carbonaceous composite derived from sugarcane leafy trash. J Environ Manage 212:77–87
Li R, Wang JJ, Gaston LA, Zhou B, Li M, Xiao R, Wang Q, Zhang Z, Huang H, Liang W, Huang H (2018b) An overview of carbothermal synthesis of metal–biochar composites for the removal of oxyanion contaminants from aqueous solution. Carbon 129:674–687
Li R, Wang JJ, Zhang Z, Awasthi MK, Du D, Dang P, Huang Q, Zhang Y, Wang L (2018c) Recovery of phosphate and dissolved organic matter from aqueous solution using a novel CaO-MgO composite and its feasibility in phosphorous recycling. Sci Total Environ 642:526–536
Liu N, Liu Y, Zeng G, Gong J, Tan X, Wen J, Liu S, Jiang L, Li M, Yin Z (2020) Adsorption of 17β-estradiol from aqueous solution by raw and direct/pre/post-KOH treated lotus seedpod biochar. J Environ Sci 87:10–23
Mirsaeidi A (2023) The effects of nitrogen and phosphorus nutrients on the bioremediation of oil-contaminated waters by Gracilariopsis persica in the coastal areas of Bandar Abbas. Mar Pollut Bull 188:114660
Muhammad IM, El-Nafaty UA, Surajudeen A, Makarfi YI (2015) Oil Removal from Produced Water Using Surfactant Modified Eggshell. 2015 4th International Conference on Environmental, Energy and Biotechnology 85: 84–92
Ni LX, Acharya K, Hao XY, Li SY (2012) Isolation and identification of an antialgal compound from Artemisia annua and mechanisms of inhibitory effect on algae. Chemosphere 88:1051–1057
Park JH, Wang JJ, Meng Y, Wei Z, DeLaune RD, Seo DC (2019) Adsorption/desorption behavior of cationic and anionic dyes by biochar prepared at normal and high pyrolysis temperatures. Colloids Surf a: Physicochem Eng Asp 572:274–282
Pinzon NM, Ju LK (2009) Analysis of rhamnolipid biosurfactants by methylene blue complexation. Appl Microbiol Biotechnol 82(5):975–981
Rao M, Simeone G, Scelza R, Conte P (2017) Biochar based remediation of water and soil contaminated by phenanthrene and pentachlorophenol. Chemosphere 186:193–201
Rastogi RP, Madamwar D, Incharoensakdi A (2015) Bloom dynamics of cyanobacteria and their toxins: environmental health impacts and mitigation strategies. Front Microbiol 6:1254
Sangeeta P, Han J, Gao W (2015) Sorption of 17β-estradiol from aqueous solutions on to bone char derived from waste cattle bones: Kinetics and isotherms. J Environ Chem Eng 3:1562–1569
Shang X, Lou A, Sun X, Sun Y (2015) Numerical simulation of petroleum hydrocarbons transport in Bohai Sea and the influences on water quality. Marine Environ Sci 34(1):58–65
Sun S, Wang Y, Zang T, Wei J, Wu H, Wei C, Qiu G, Li F (2019) A biosurfactant-producing Pseudomonas aeruginosa S5 isolated from coking wastewater and its application for bioremediation of polycyclic aromatic hydrocarbons. Bioresource Technol 281:421–428
Tang J, Lv G, Gong Y, Huang Y (2015) Preparation and characterization of a novel graphene/biochar composite for aqueous phenanthrene and mercury removal. Bioresour Technol 196:355–363
Tao W, Fattah KP, Huchzermeier MP (2016) Struvite recovery from anaerobically digested dairy manure: a review of application potential and hindrances. J Environ Manage 169:46–57
Thines KR, Abdullah EC, Mubarak NM (2017) Effect of process parameters for production of microporous magnetic biochar derived from agriculture waste biomass. Microporous Mesoporous Mater 253:29–39
Tiwari JN, Mahesh K, Le NH, Kemp KC, Timilsina R, Tiwari RN, Kim KS (2013) Reduced graphene oxide-based hydrogels for the efficient capture of dye pollutants from aqueous solutions. Carbon 56:173–182
USEPA (1983) Methods for chemical analysis of water and wastes. 2nd ed. Method 365.2. U.S. Environmental Protection Agency, Washington, DC
Uzoigwe C, Burgess JG, Ennis CJ, Rahman PKSM (2015) Bioemulsifiers are not biosurfactants and require different screening approaches. Front Microbiol 6:245
Wang JJ, Gaston LA (2014) Nutrient chemistry of manure and manure-impacted soils as influenced by application of bauxite. In: He Z, Zhang H (eds) Applied manure and nutrient chemistry for sustainable agriculture and environment. Springer, Dordrecht Heidelberg, New York, pp 239–266
Wang X, Gong L, Liang S, Han X, Zhu C, Li Y (2005) Algicidal activity of rhamnolipid biosurfactants produced by Pseudomonas aeruginosa. Harmful Algae 4:433–443
Wang Z, Han L, Sun K, Jin J, Ro KS, Libra JA, Liu X, Xing B (2016) Sorption of four hydrophobic organic contaminants by biochars derived from maize straw, wood dust and swine manure at different pyrolytic temperatures. Chemosphere 144:285–291
Wang B, Gao B, Fang J (2017) Recent advances in engineered biochar productions and applications. Crit Rev Environ Sci Technol 47:2158–2207
Wang M, Strokal M, Burek P, Kroeze C, Ma L, Janssen ABG (2019) Excess nutrient loads to Lake Taihu: opportunities for nutrient reduction. Sci Total Environ 664:865–873
Wei Z, Wang JJ, Gaston LA, Li J, Fultz LM, DeLaune RD (2020a) Integrated effect of biochar, biosurfactant, and nitrogen application on crude oil remediation in a contaminated coastal marsh. J Hazard Mater 396(2020):122596
Wei Z, Wang JJ, Meng Y, Li J, Gaston LA, Fultz LM, DeLaune RD (2020b) Potential use of biochar and rhamnolipid biosurfactant for remediation of crude oil-contaminated coastal wetland soil: Ecotoxicity assessment. Chemosphere 253:126617
Wetteland CL, de Jesus Sanchez J, Silken CA, Nguyen NYT, Mahmood O, Liu H (2018) Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids. J Nanopart Res 20:215
Wu Z, Zhong H, Yuan X, Wang H, Wang L, Chen X, Zeng G, Wu Y (2014) Adsorptive removal of methylene blue by rhamnolipid-functionalized graphene oxide from wastewater. Water Res 67:330–344
Wu Q, Xian Y, He Z, Zhang Q, Wu J, Yang G, Zhang X, Qi H, Ma J, Xiao Y, Long L (2019) Adsorption characteristics of Pb(II) using biochar derived from spent mushroom substrate. Sci Rep 9(1):15999
Xia P, Wang XJ, Wang X, Song JK, Wang H, Zhang J, Zhao JF (2016) Struvite crystallization combined adsorption of phosphate and ammonium from aqueous solutions by mesoporous MgO– loaded diatomite. Colloid Surf A Physicochem Eng Asp 506:220–227
Xiao R, Wang JJ, Li R, Park JH, Meng Y, Zhou B, Pensky S, Zhang Z (2018) Enhanced sorption of hexavalent chromium [Cr(VI)] from aqueous solutions by diluted sulfuric acid-assisted MgO-coated biochar composite. Chemosphere 208:408–416
Xiao R, Zhang H, Tu Z, Li R, Li S, Xu Z, Zhang Z (2020) Enhanced removal of phosphate and ammonium by MgO-biochar composites with NH3·H2O hydrolysis pretreatment. Environ Sci Pollut Res 27(7):7493–7503
Yan J, Zuo X, Yang S, Chen R, Cai T, Ding D (2022) Evaluation of potassium ferrate activated biochar for the simultaneous adsorption of copper and sulfadiazine: Competitive versus synergistic. J Hazard Mater 424(Pt B):127435
Yang G, Wu L, Xian Q, Shen F, Wu J, Zhang Y (2016) Removal of congo red and methylene blue from aqueous solutions by vermicompost-derived biochars. PLoS ONE 11:0154562
Yao Y, Gao B, Chen JJ, Zhang M, Inyang M, Li YC, Alva A, Yang LY (2013) Engineered carbon (biochar) prepared by direct pyrolysis of Mg-accumulated tomato tissues: characterization and phosphate removal potential. Bioresour Technol 138:8–13
Zhang M, Gao B, Yao Y, Xue Y, Inyang M (2012) Synthesis of porous MgO-biochar nanocomposites for removal of phosphate and nitrate from aqueous solutions. Chem Eng J 210:26–32
Zhao H, Wang Z, Liang Y, Wu T, Chen Y, Yan J, Zhu Y, Ding D (2023) Adsorptive decontamination of antibiotics from livestock wastewater by using alkaline-modified biochar. Environ Res 226:115676
Zhen M, Tang J, Song B, Liu X (2018) Decontamination of methylene blue from aqueous solution by rhamnolipid-modified biochar. BioRes 13(2):3061–3081
Zhou Y, Gao B, Zimmerman AR, Fang J, Sun Y, Cao X (2013) Sorption of heavy metals on chitosan-modified biochars and its biological effects. Chem Eng J 231:512–518
Zhou H, Liu Q, Jiang L, Shen Q, Chen C, Zhang C, Tang J (2023) Enhanced remediation of oil-contaminated intertidal sediment by bacterial consortium of petroleum degraders and biosurfactant producers. Chemosphere 330:138763
Acknowledgements
The authors are grateful to the editor and the reviewers for their valuable comments and constructive suggestions.
Funding
This research is, in part, support from Yunnan Fundamental Research Projects (Grant # 202201BE070001-043), the National Natural Science Foundation of China (Grant #41967039), and the Yunnan Provincial Excellent Young Scientists Fund (Grant #202201AW070006), Yunnan Foundation Research Projects (202301AU070074).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ZW, YX, YW, YL, SN, and JJW. The first draft of the manuscript was written by ZW and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Handling editor: Wenfu Chen.
Supplementary Information
Additional file 1.
Supplementray table and figures.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wei, Z., Xu, Y., Wei, Y. et al. Application of magnesium and biosurfactant functionalized biochar composite in treating cyanobacteria in phosphorus and crude oil contaminated water. Biochar 5, 50 (2023). https://doi.org/10.1007/s42773-023-00249-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-023-00249-3