Abstract
Hydrochar (HC), produced by hydrothermal carbonization, offers technical advantages over biochar (BC) produced by pyrolysis, and is suitable for soil amelioration, carbon sequestration, and enhanced plant growth. BC grain size has been shown to influence nutrient retention, microbial colonization and aggregate formation; however, similar research for HC is lacking. Pot trials were conducted to investigate the influence of HC grain size [coarse (6.3–2 mm), medium (2–0.63 mm) and fine (< 0.63 mm)], produced from biogas digestate, for soil improvement in three soils: loamy Chernozem, sandy Podzol, and clayey Gleysol, at a 5% HC application rate (w/w). All soils including two controls (with and without plants) were analysed for water holding capacity (WHC), cation exchange capacity (CEC), wet aggregate stability, pH, plant available nutrients (PO4–P, K and Nmin) and germination and biomass success using standard laboratory and statistical methods. Soil pH showed a compensatory shift toward the HC pH (7.2) in all soils over the course of the study. For example, the pH of the medium grained HC treatment for the Chernozem decreased from 7.9 to 7.2 and increased in the Podzol and Gleysol from 5.9 to 6.1 and 4.9 to 5.5, respectively. The nutrient-rich HC (2034 ± 38.3 mg kg−1 PO4–P and 2612.5 ± 268.7 mg kg−1 K content) provided only a short-term supply of nutrients, due to the relatively easily mineralized fraction of HC, which allowed for quick nutrient release. The pH and PO4–P effects were most pronounced in the fine grained HC treatments, with a ~ 87%, ~ 308% and ~ 2500% increase in PO4–P content in the Chernozem, Podzol and Gleysol, respectively, compared to the controls at the beginning of the study. The same trend was observed for the K and NH4+ content in the fine and medium grained HC treatments in all soils. No seed germination inhibition of Chinese cabbage was observed, with average germination rates > 50% in all soils. An effect on NO3− content was indeterminable, while there was little to no effect on biomass production, WHC, CEC and aggregate stability. In conclusion, the application of 5% fine grained HC significantly influenced the nutrient content over a short-term. However, the application rate was insufficient to substantially improve plant growth, nor to sustain a longer-term nutrients supply, regardless of grain size.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Intensified agricultural and energy production resulting from increasing global demands on resources such as energy, food and land have seen deleterious consequences for the global climate and soils through, inter alia, increased greenhouse gas (GHG) emissions and reduced soil quality. Poor agricultural practices and land use changes disturb the soil organic carbon (SOC) pool and liberate carbon dioxide (CO2) from the soil, thus contributing to the addition of GHGs into the atmosphere and decreasing soil quality and its resistance to stresses (Jones et al. 2015). A potential remedial measure to the problems of reduced soil quality may lie in the improvement and protection of the SOC pool through soil amendment.
The highly fertile Terra Preta de Indio soils of the Amazon Basin attribute their high carbon (C) content to the deliberate addition of charcoal-like materials over thousands of years by the prehistoric local inhabitants as well as by long-term mulching and frequent burning practices (Glaser et al. 2002; Lehmann and Rondon 2006; Sombroek et al. 1993; Steiner et al. 2009a, b; WinklerPrins 2014). The link between these practices and the improved soil quality of these previously infertile tropical soils has conceptualized the use of biochar (BC), a C-rich charcoal, as a popular soil amendment and management tool. BC is produced by pyrolysis which mimics the natural formation of coal, whereby biomass is heated at temperatures between 400 and 850 °C under dry and oxygen (O)-limited conditions. A large proportion (~ 50%) of the source materials’ C content remains in the final product (Lehmann et al. 2006), which is proven to have a high resistance to microbial decomposition and mineralization, and thus decomposes at a much slower rate than that of the raw biomass from which it is produced (Steiner et al. 2009b).
This recalcitrance lowers the rate at which C fixed by photosynthesis is returned to the atmosphere, which subsequently improves the aggregate stability, water holding capacity (WHC), and fertility of soils through the associated net increase in soil C stocks (Woolf et al. 2010). However, pyrolysis is limited in its use of dry biomasses (Libra et al. 2011; Monlau et al. 2015), and therefore, has a substantial energy requirement if pre-drying of feedstocks is deemed necessary. Furthermore, during pyrolysis, ~ 50% of the raw biomass C is released back into the atmosphere, thus contributing to GHG emissions (Lehmann et al. 2006). As such, the potential benefits of BC application for soil amendment are somewhat negated by the shortcomings of the production process. An alternative to BC by pyrolysis is hydrochar (HC) produced by hydrothermal carbonization (HTC).
HTC is a thermochemical conversion process capable of converting wet and dry feedstocks in a single, closed, water-saturated system under elevated pressure (≤ 20 bar) and lower temperatures (180–250 °C) compared to pyrolysis, with a residence time of ≤ 12 h. HTC produces a charcoal-like material referred to as HC and wastewater as the end-products (Fiori et al. 2014; Libra et al. 2011). The lower temperature requirement for HTC ensures that combustion does not occur during the conversion process, making it a C-neutral process. Depending on the HTC process parameters, namely temperature, HC may retain as much as 60–84% of the raw biomass C compared to 50% for BC. A potential limitation of the HTC process is that this higher C content typically comes at the expense of the total mass yield, which decreases with increasing C content at higher process temperatures (Basso et al. 2013). Furthermore, the higher pressure requirement of HTC means that the manufacturing costs of the HTC equipment may be more expensive, and it also increases the complexity and hazard risk of the process (Kambo and Dutta 2015).
The mass yield of char produced by HTC is also a function of the heating rate, whereby slower heating rates coupled with lower maximum temperatures and increased pressure result in greater mass yields (Zhang et al. 2008; Septien et al. 2018). The heating rate also strongly influences the pore size and specific surface area (SSA) of the resultant char. It has been shown that slower heating rates typically produce micropores and higher SSA, while faster rates increase the macropore content of the char (Downie et al. 2009; Septien et al. 2018). The consequent SSA is an important consideration, as it has been linked to a higher C mineralization due to increased availability to microorganisms, especially in digestate-derived HC (Eibisch et al. 2013).
The potential limitations of the HTC process (as mentioned above) are overshadowed by its ability to process wet feedstocks (with water contents of ≥ 70%), which broadens the possible biomass types that may be utilized to include unconventional feedstocks such as biogas digestate and wet animal manures (Libra et al. 2011). This advantage is particularly important in areas of high agricultural production, where excessive waste is problematic. A boom in the establishment of biogas plants over the past 20 years (Theuerl et al. 2019) has resulted in an excess of the by-product, biogas digestate (Marchetti and Castelli 2013), which is commonly used as fertilizer (Garlapalli et al. 2016). Consequently, the amount produced surpasses fertilizer demands, thus classifying it as agricultural waste. HTC of biogas digestate produces a solid product with a higher C content than the source materials, which allows for easier storage and transportation to areas where fertilization is required (Kambo and Dutta 2015; Eskandari et al. 2019).
Pyrolysis and HTC process conditions and source materials influence the characteristics of the final product, and consequently its recalcitrance and efficiency as a soil amendment tool. These determinant parameters (e.g. feedstock type and process conditions) for the intended application of BC can be manipulated to design a product with specific characteristics, i.e. structure and size. The application of BC to soils has shown to improve the properties of the soils by increasing the WHC and cation exchange capacity (CEC), with improved nutrient dynamics and microbial activity, which subsequently stimulates plant growth and improves plant health (Brodowski et al. 2005; Glaser et al. 2002; Hagemann et al. 2016; Liu et al. 2017; Schulz and Glaser 2012; Verheijen et al. 2009). A meta-analysis of over 300 studies by Biederman and Harpole (2013) found that, on average, the addition of BC increased above ground biomass production, soil phosphorous (P)-, potassium (K)- and total C- and nitrogen (N) content and increased the pH of acidic soils, regardless of soil type and climatic conditions.
The grain size of the applied BC holds implications for its intended effects on the soil, due to their arrangement within the soil particle matrix and the consequent alteration of the soil pore size distribution. The size of the BC particles applied to soil together with the inherent porosity and surface area influence the bioavailability of nutrients, the accessibility of microbial communities to the pores for colonization, water storage and mobility, and the degree of oxidation that takes place on the surface of the BC particles, which have further ramifications for aggregate formation and soil stability (Brodowski et al. 2005; Hagemann et al. 2016; Liu et al. 2017; Verheijen et al. 2009). The addition of small grained BC particles (< 0.5 mm) increases soil aggregation and water retention through a greater SSA, compared to larger BC particles (Blanco-Canqui 2017; de Jesus et al. 2019; Głąb et al. 2016).
The above stated impacts on soil properties are all reported in studies pertaining to BC produced by pyrolysis. Presumably, the addition of HC would have similar effects, due to its similarities (namely, the high C content) with BC. However, HC differs from BC in its physical and chemical structure, which holds implications for their intended purposes (Busch and Glaser 2015; Libra et al. 2011). Compared with BC, HC has a higher hydrogen (H)/C and O/C ratio, lower porosity and poorer surface area, and the surface functionalities between the chars differ considerably (Kambo and Dutta, 2015; Libra et al. 2011). Such differences highlight the fact that the known impacts of BC cannot simply be inferred for HC as well.
In addition to the relatively limited studies of the impacts of HC on soil properties, its influence on plant growth dynamics is also an important consideration. A study by Bargmann et al. (2014b) showed that the addition of HC initially inhibited seed germination of barley, but this effect was diminished after nine weeks, likely due to microbial decomposition. It was suggested that this decomposition was indirectly facilitated by the dissolved organic carbon (DOC) content of the HC, which stimulated microbial growth (Gronwald et al. 2015; Hao et al. 2018). The transient nature of this effect was also evinced by Sun et al. (2014) and Mukherjee et al. (2016). Related studies by Prayogo et al. (2014) found that SOC mineralization was reduced in soils amended with HC, due to the preferential utilization of the soluble C fraction of the HC by soil microbes. Contrarily, Fang et al. (2015) saw no significant influence on seed germination by HC addition, while Puccini et al. (2018) found a significant reduction in the germination success of lettuce using fresh- and post-treated HC.
With regards to plant growth, Baronti et al. (2017) found a significant increase in the biomass production of poplar trees with HC addition after two consecutive years, while Bargmann et al. (2014b) showed no negative effects on spring barley at a 4% HC application rate. Furthermore, Schimmelpfennig et al. (2014) saw a substantial reduction in plant growth with the field application of HC, particularly in the first year of the experiment. However, HTC is a relatively modern carbonization method and research regarding the effectiveness of HC for soil amendment and plant growth is disproportionate compared to BC, and such information remains limited.
Based on existing and available information, we therefore hypothesize that the addition of HC will: (a) improve soil properties essential to plant growth; (b) impact the soil properties differently based on the grain size of the applied HC; (c) initially inhibit seed germination (based on Bargmann et al. 2014a; Röhrdanz et al. 2019; Steiner et al. 2009b); and (d) enhance biomass production due to an improved soil quality.
The objectives of this study test these hypotheses by analysing the overall influence of HC application on soil improvement, germination success and biomass production as well as with respect to different HC grain sizes. Three soil types were studied: Chernozem, Podzol and Gleysol, which were selected based on their dissimilar properties. To the authors’ best knowledge, no existing studies focus on the effect of grain size of HC on soil amendment, seed germination and biomass production.
To achieve these objectives, the following research questions were posed:
-
1.
Does the addition of HC improve the soil properties essential to plant growth, such as WHC, CEC, pH, aggregate stability and nutrient contents?
-
2.
Does the grain size of the applied HC affect its influence on soil properties?
-
3.
How does HC application affect seed germination and biomass production of Chinese cabbage?
By answering these questions, this study will substantially contribute to our relatively rudimentary understanding of HC as a promising amendment product.
2 Materials and methods
2.1 Experimental design
The soils used in this study were collected from conventionally operating farms in December 2017. The sampling sites of the Chernozem and Podzol are under long-term arable use, and are located within the Hohe Börde Municipality in Saxony-Anhalt, Germany (52° 10′ 35″ N, 11° 30′ 42″ E), and the Bruchhausen-Vilsen Municipality in Lower Saxony, Germany (52° 48′ 35″ N, 8° 59′ 41″ E), respectively. The Gleysol was sampled within the Ovelgönne Municipality in Lower Saxony, Germany (53° 15′ 45″ N, 8° 19′ 49″ E), and the site is under grassland conditions. The Chernozem may be classified as a silty clay (particle size distribution: 16% sand, 43% silt, 41% clay), the Podzol a sandy loam (66% sand, 22% silt, 13% clay), and the Gleysol a clay (1% sand, 6% silt, 93% clay). The samples were randomly collected from the upper 30 cm and were air-dried in the greenhouse before being ground to < 2 mm using a grinding mill.
The HC was provided by Grenol GmbH (Meiersberg [Ratingen], Germany) and was produced from biogas digestate; carbonized at ~ 200 °C, 18–20 bar pressure, with a residence time at the maximum temperature of approximately 3 h and a heating and cooling time of 1.5 h each. This equates to a heating rate of ca. 2–3 °C per min by constant heating, while the cooling rate could not be specified as it is dependent on the ambient temperature of the surroundings of the batch reactor. The digestate source material was derived from corn, sugar beets and beef- and liquid-swine manure. It was not pre- or post-treated for any specification and was air-dried and pressed into briquettes after conversion for transportation.
The HC briquettes were manually broken into smaller pieces using a hammer and then sieved into three grain size classes, namely, a coarse grained (6.3–2 mm), medium grained (2–0.63 mm) and fine grained (< 0.63 mm) fraction. At an application rate of 5% (w/w), the HC was then manually homogenously mixed with the soils, before placing the mixtures into pots of ~ 13 cm diameter (ca. 1 litre [l] volume). A mesh material was placed over openings at the bottom of the pots to reduce soil loss. Seven pots for each HC grain size-soil mixture was used, as well as five control pots of original soil (no HC added) which were sown with Chinese cabbage seeds. Additional five control pots of original soil were kept devoid of seeds. All experiments were conducted in a greenhouse in a non-climate controlled environment.
2.1.1 Seed germination experiment
Two rounds of seed germination experiments were carried out to determine whether any germination inhibition effects may result from the addition of HC, as reported in numerous studies (Bargmann et al. 2014a; Röhrdanz et al. 2019; Steiner et al. 2009b). The pots (except for the control devoid of plants for each soil) were planted with 25 Chinese cabbage seeds (Brassica rapa ssp. Pekinensis) and were watered manually based on plant requirement. Germination success was calculated as the percentage of germinated seeds (25 seeds = 100%). At the conclusion of each round (ca. 3 weeks each) of the germination experiment, the plants were harvested from the pots before being sown with new seeds.
2.1.2 Plant growth experiment
Subsequent to the germination experiment, the plant growth experiment was conducted, which involved sowing each pot with four Chinese cabbage seeds. After approx. 4 weeks of growth, the number of plants was reduced to one per pot. The plants were grown for approx. 6 weeks from the start of the plant growth experiment, after which they were harvested (above ground biomass exclusively), and weighed before and after oven-drying at 105 °C to constant weight to determine the total biomass for each treatment (HC grain size-soil mixture) and control.
2.2 Soil analyses
Control samples were collected from each soil prior to the addition of HC, which are hereinafter referred to as Time 0 (T0) samples. Samples were collected immediately after mixing the HC with the soils at the beginning of the study for soil property analyses, which are hereinafter referred to as Time 1 (T1) samples. At the end of the plant growth experiment, samples were collected destructively from each pot for each respective treatment and control. The seven samples from each grain size treatment and five samples from each of the controls were mixed into respective composite samples, which were transported to the laboratory for soil property analyses. These samples collected at the end of the experiment are hereinafter referred to as Time 2 (T2) samples. The samples were air-dried and sieved < 2 mm for all soil property analyses. Extractions and measurements for soil property analyses were conducted approx. 3–4 weeks after sampling at T2.
Particle size distribution was determined by a combination of mechanical sieving and sedimentation with Atterberg cylinders, after removal of carbonates, organic matter (OM) and iron (Fe) oxides. Soil textural classes were determined based on the Food and Agriculture Organization of the United Nations (FAO 2006) soil taxonomy system. The pH was determined in a 1 g: 2.5 ml soil-to-distilled water (H2O) solution, after manual stirring at 15 min intervals for 1 h.
Given the nature of the material (the presence of HC), procedural modifications were required for the determination of the WHC and CEC. The WHC was determined using a filtration method adapted from Alef (1991), as described in Röhrdanz et al. (2016). The potential CEC (CECpot) was determined using a modified extraction method, after Sänger-von-Oepen et al. (1993), Handbuch Forstliche Analytik (2005), and VDLUFA (2012a).
Mineral nitrogen (Nmin), in the form of ammonium (NH4+) and nitrate (NO3−), was determined by the standard Nmin laboratory method (DIN 19746), according to VDLUFA (2002). The plant available P and K were determined using the calcium (Ca)-acetate-lactate extraction method (VDLUFA 2012b), at a concentration of 0.6%. This solution was buffered to a pH of 3.6. The P concentration was determined colorimetrically as phosphate (PO4–P) using a coloring agent of 0.5% ascorbic acid and 1% ammonium heptamolybdate, measured by a spectrophotometer (Shimadzu UVmin-1240). The plant available K was measured by atomic absorption spectrometry. The water aggregate stability was determined using the wet sieving method, as described in Kalinina et al. (2011), and which is derived from Six et al. (2000).
2.3 Elemental analysis
The elemental composition of the HC was determined for C, H, N and sulphur (S) using a Euro Elemental Analyzer (Eurovector; HEKAtech GmbH), after oven drying at 105 °C (Schröter 2018). The ash content of the HC was determined as the amount of material remaining after dry oxidation at 550 °C, according to DIN EN 14775:2010-04. The O content of the HC was calculated as the difference in the C, H, N, S and ash content of the sample to 100%, as illustrated in equation below:
The elemental composition and ash content of the HC is provided in Table 1. These results were used to calculate the molar element ratios.
2.4 Statistical analyses
A normal distribution is a critical assumption in the use of a one-way analysis of variance (ANOVA). Due to the small number of replicates (n = 3) in this study, the normal distribution of the data could not be determined with confidence. Therefore, the non-parametric Kruskal–Wallis H test was applied for all residuals, following tests for normal distribution (Shapiro–Wilk test) and homogeneity of variance (Levene’s test). Significant differences between groups were determined by the pairwise comparisons post-hoc test, using the procedure prescribed by Dunn (1964), with a Bonferroni correction for multiple comparisons (p < 0.05) (Laerd Statistics 2015). These statistical analyses were performed to determine significant differences between HC grain size treatments at each phase of the study (T1 and T2). The biomass results were subjected to the ANOVA statistical test, and the normally distributed residuals further underwent a comparison of means post-hoc test (Scheffe test) to indicate significant differences (p < 0.01). It must be noted that only two replicates were measured for the WHC and CEC analysis, and therefore statistical analyses for these properties could not be performed.
To determine if the differences over the course of the study (T0 to T2) between HC grain size treatments and controls were statistically significant, the independent-samples t-test was employed, following the Shapiro–Wilk test for normal distribution, and Levene’s test of homogeneity of variances. If the assumption of homogeneity of variances was violated, the independent-samples t-test was calculated using separate variances and the Welch–Satterhwaite correction to the degrees of freedom (Laerd Statistics 2015). Significant differences are based on mean values. Statistical analyses were performed using SPSS 25.
3 Results and discussion
3.1 Effect of hydrochar application on soil properties
3.1.1 Water holding capacity
A sufficient water supply is essential for optimum plant growth. Hence, increasing WHC is a major driver for soil improvement, especially for coarse grained soils (e.g. Podzol), for which WHC is naturally notably low. Several studies stated a positive effect of HC addition on WHC, and furthermore, positive correlations between finer grained BC particles (< 0.25 mm) and water content and retention have been noted by Liu et al. (2017) and de Jesus et al. (2019).
Our results for the controls (at T0) show an initial average WHC of 0.9 ± 0.1 g H2O/g dry soil (g/g) for the Chernozem; 0.7 ± 0.1 g/g for the Podzol; and 1.2 ± 0.1 g/g for the Gleysol. The minimum and maximum WHC values for the controls and HC amended soils are provided in Table 2. The average WHC of the HC was measured at 1.47 g/g, which is in line with other findings (Libra et al. 2011). A tendency of improved WHC shortly after the addition of HC (at T1) was revealed in the Podzol. However, this initial increasing tendency did not persist throughout the course of the study. Post the three-month plant growth experiment (T2), no distinguishable trend between HC grain size and WHC was apparent for all soils, although a general decreasing tendency in WHC was observed for the amended soils from T1 to T2.
The increasing tendency in WHC observed mainly in the Podzol (sandy loam) at T1 is supported by Abel et al. (2013) and Röhrdanz et al. (2016), who have shown that the potential for increasing WHC is greater in soils with lower WHC, such as sandy soils, due to the smaller SSA and lower microporosity of sandy (coarse) textured soils compared to more clay-rich soils (Blanco-Canqui 2017). Liu et al. (2017) attributed this improvement (in sandy soils especially) to the change in pore structure and pore space dynamics induced by the addition of BC, which may also be relevant to HC. However, the application rate of 5% HC with a WHC of 1.47 g/g, as performed in this study, was not sufficient to significantly alter the pore distribution of the soils, and as such, could not substantially increase the WHC of the soils. The unexpected lack of HC influence on WHC may also be due to the hydrophobicity of the HC, as shown by Berge et al. (2013), which may result when HC is air-dried (as in this study).
3.1.2 Aggregate stability
Aggregation alters the pore size distribution in soils and consequently, the air and water capacities. Aggregation is particularly prevalent in fine grained clayey soils, resulting in the formation of bigger pores and thus, substantially improving air and water balances. For BC, it was found that its addition improved aggregation (Pituello et al. 2018; Blanco-Canqui 2017).
The best proxy for aggregation is the Mean Weight Diameter (MWD), which indicates the relation between aggregate stability and size of aggregates, whereby a greater proportion of larger aggregates implies greater stability (Nimmo and Perkins 2002; Sun and Lu 2014). Of the controls at T0, the Chernozem had an average MWD of 0.3 ± 0.01 mm; the Podzol, 0.2 ± 0.01 mm; and the Gleysol, 0.5 ± 0.03 mm (Fig. 1). Shortly after HC application (T1), the MWD for the HC amended Gleysol showed no directed trend, but the HC amended Chernozem and Podzol appeared to increase compared to the respective controls. However, the variation in statistically significant differences between treatments and controls indicates the absence of a clear trend between the grain size of the added HC and aggregate stability at T1. At the end of the study (T2), there were no significant differences between the controls and HC amended soils for all soils. However, over the course of the study (T1 to T2), the MWD of stable aggregates increased for all soils, but was only significantly different for all Chernozem and Gleysol samples, except the Chernozem coarse grained treatment (Chernozemcoarse) and Gleysolfine. It is likely that the increase in MWD in the controls and HC amended soils during the course of the study (from T1 to T2) was the result of other factors, and therefore, no clear effect of HC on MWD was indicated in the results.
Average Mean Weight Diameter (MWD) for the controls and hydrochar grain size treatments in a Chernozem, Podzol and Gleysol over the course of the plant growth experiment. Error bars represent standard deviation of the means. Different letters indicate significant differences in means at p < 0.05 level between treatments at the respective time period. n.s = nonsignificant. Solid bars indicate significant differences and patterned bars nonsignificant differences in means (p < 0.05) between treatments at the beginning (T0 and T1) and end of the study (T2). Control_pl = control with plant
The lack of (a definitive) HC influence on soil aggregation at T1 may not be surprising, as aggregate formation is dependent on, inter alia, biological activity and time. The increase in MWD of aggregates over the course of the study for the controls, as well as most of the HC amended soils, indicates that the HC is likely not responsible for the improved aggregation. More likely, the results point to the probability that the increase in MWD at T2 was due to the formation of macroaggregates as part of the initial phases of soil restoration, following semi-destructive sampling (Kalinina et al. 2011).
3.1.3 Soil pH
The management and readjustment of pH are critical in soil amelioration practises. Where pH values are too acidic or alkaline, toxicities and limited nutrient availabilities may result. Due to general acidification processes in temperate soils, a good pH regulation may be achieved by the addition of liming materials. Similarly, the addition of HC has been found to alter soil pH (Malghani et al. 2015; Biederman and Harpole 2013), and hence, HC could considerably contribute to pH readjustment.
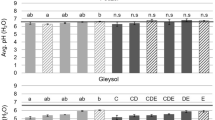
In this study, the initial average pH of the Chernozemcontrol was slightly alkaline (7.9 ± 0.1), while the Podzolcontrol and Gleysolcontrol were more acidic (5.8 ± 0.1 and 4.9 ± 0.0, respectively). Shortly after the addition of HC with pH 7.2 ± 0.1 at T1, the Podzol and Gleysol showed a slight increasing shift in pH, most pronouncedly in the fine grained HC treatments, while the pH of the Chernozem remained relatively unchanged. At the end of the study (T2), the pH of the HC amended Chernozem was lower than the controls, and furthermore, the Chernozemmedium and Chernozemfine had reached the same pH as the HC (7.2) (see Fig. 2). Simultaneously, the trend of increasing pH in the amended Podzol and Gleysol continued, resulting in significant differences between the controls and amended soils, namely, the Podzolfine (p = 0.040) and the Gleysolmedium (p = 0.010). Therefore, it would appear that the addition of HC resulted in a shift of soil pH to the pH of the HC, which was more prominent in the finer grained fractions.
Average pH (measured in distilled H2O) for the controls and hydrochar grain size treatments in a Chernozem, Podzol and Gleysol over the course of the plant growth experiment. The solid line represents the pH of the HC (7.2). Error bars represent standard deviation of the means. Different letters indicate significant differences in means at p < 0.05 level between treatments at the respective time periods. n.s = nonsignificant. Solid bars indicate significant differences in means (p < 0.05) between treatments at the beginning and end of the study. Patterned bars = nonsignificant. Control_pl = control with plant
These results are consistent with that of Malghani et al. (2015), where HC addition significantly increased the pH of a coarse grained acidic soil. A related study by Liao and Thomas (2019) found that the addition of small grained (0.06–0.5 mm) sieved BC to a granitic sand increased the pH by 0.3 units, compared to a larger grain size (2–4 mm), which showed no significant difference; thus confirming our findings in respect of grain size effects. Liao and Thomas (2019) further suggested that BC produced from feedstocks of smaller particle size, such as the decomposed plant material which constitutes part of the digestate from which the HC in this study was produced (Greenberg et al. 2019), have a higher ash content, thus promoting alkalinity.
The equilibration of the pH values of the soil and (finer grained) BC are suggested to be the result of increased physical contact between the soil and BC particles, as well as the high ash content of the BC, which promotes the liming effect of the BC and its capacity to buffer soil acidity (Domingues et al. 2017) and provides additional cations to the soil, which increases pH, particularly in sandy and loam textured soils (Glaser et al. 2002). As found for the Chernozem in this study, a reduction in soil pH was also possible when HC was added to a soil that had a higher pH than that of the HC. As such, this study’s findings confirm that the conclusion of Biederman and Harpole (2013) may be inferred for HC as well, whereby the pH response is dependent on the initial pH of the soil, as well as the pH of the BC/HC material.
3.1.4 Potential cation exchange capacity (CECpot)
CEC is a major controlling factor in the availability of plant nutrients. Hence, increasing the CEC of soils is of particular interest in the context of soil improvement, especially since increased CEC is coincident with higher fertilizer efficiency and the reduced entry of nutrient loads into groundwater systems (Bargmann et al. 2014a). BC typically has a high CEC, and its addition to soils has shown to increase the soil CEC (Garlapalli et al. 2016). However, contradictory findings have been reported for HC. For example, Puccini et al. (2018) observed a very low CEC for fresh HC, while Salem (2013) report increased CEC of soils by HC addition. Additionally, Libra et al. (2011) hypothesized that HC surface functional groups should improve soil CEC. Such variability leaves open questions regarding the sorption capacity of HC and its ability to improve soil CEC.
In this investigation, the CECpot of the Chernozemcontrol at the beginning of the study (T0) was 39.5 ± 2 cmol kg−1, for the Podzolcontrol, 18.8 ± 2 cmol kg−1, and for the Gleysolcontrol, 85.8 ± 0.4 cmol kg−1 (Table 3). Shortly after HC addition (T1), the CECpot remained relatively unchanged for all HC grain size treatments compared to the controls for all soils. The same result was found at the end of the study (T2), whereby a difference in CECpot between controls and HC amended soils remained indistinct. Hence, the results show no impact on CECpot by HC addition.
Schulz and Glaser (2012) argued that improved CEC by BC addition to soils is unlikely to occur in the short-term, and that the CEC of BC can only be increased when the BC is microbially activated, such as via compositing. Therefore, the addition of an “un-activated” BC would not elevate the CEC. Hale et al. (2013) maintained that soils which are most likely to respond to BC (and HC) addition have a CEC of ≤ 10 cmol kg−1. The initial CEC of the soils of this study were all well above 10 cmol kg−1. Furthermore, the addition of 5% HC with a CEC of 28.2 ± 2.3 cmol kg−1 was insufficient to distinctly increase soil CEC. According to Glaser et al. (2002), a sustainable increase in CEC, as well as the formation of organo-mineral complexes, can be achieved through the formation of carboxylic functional groups on the edges of the aromatic structure of charcoal by means of slow oxidation. It is plausible that the duration of this study may not have been sufficient to allow for such processes to take place.
3.1.5 Phosphorous (PO4–P)
An adequate supply of all the nutrients plants require is essential for optimum plant growth. However, fertilization practices are concentrated almost exclusively on the supply of N, P and K, as these are quantitatively in the highest demand. Fertilization can be achieved by applying mineral and/or organic fertilizers, and additionally, by HC application. A study by Bento et al. (2019) showed that HC released nutrients, however it was dependent on the soil type to which it was applied, as well as the HC application rate. Although research on this topic is comparatively less than for BC, it can be expected that the fertilizer effect of HC is even stronger, because the HC structure is less stable (Basso et al. 2013), presumably resulting in greater alteration and enhanced nutrient release.
The results for the beginning of the study (T0) showed the PO4–P content of the controls as 172.4 ± 7.8 mg kg−1 for the Chernozem; 431.9 ± 8 mg kg−1 for the Podzol; and 20.9 ± 1.5 mg kg−1 for the Gleysol (Fig. 3). The PO4–P content of the HC was 2034.6 ± 38.3 mg kg−1. Shortly after the addition of HC (T1), the PO4–P content increased in all amended soils, and furthermore, the fine grained HC treatments had significantly higher PO4–P contents (p < 0.05) compared to their respective controls, increasing by ~ 87%, ~ 308% and ~ 2500% in the Chernozem, Podzol and Gleysol, respectively. At the end of the study (T2), the PO4–P content of the HC amended soils was mostly higher than the controls for all soils, however this was not statistically confirmed, except for the Gleysolfine (p = 0.034). Over the course of the study (T1 to T2), the PO4–P content mostly decreased for all soils in both the controls, as well as HC amended samples. This was statistically significant (p < 0.05) for all HC amended soils, except for the Chernozemfine, Podzolcoarse, Gleysolcontrol_pl and Gleysolcoarse. The results evince a clear HC effect on PO4–P content, suggesting the HC acted as a short-term source of PO4–P to the soils, with the greatest release of PO4–P occurring in the fine grained fraction.
Average phosphate content (PO4–P) for the controls and hydrochar grain size treatments in a Chernozem, Podzol and Gleysol over the course of the plant growth experiment. Error bars represent standard deviation of the means. Different letters indicate significant differences in means at p < 0.05 level between treatments at the respective time periods. n.s = nonsignificant. Solid bars indicate significant differences in means (p < 0.05) between treatments at the beginning and end of the study. Patterned bars = nonsignificant. Control_pl = control with plant. *Statistically significantly difference (not visible)
The significant initial increase in PO4–P content in all soils compared to the controls was likely derived directly from the HC. This finding is supported by Gronwald et al. (2015), whereby HC produced from digestate feedstock, as in this study, contained 10 times more P compared to the other feedstocks. It has been shown that BC produced from crop residues and manure feedstocks are more easily decomposable due to their fine nature and high nutrient content. In line with this, a decreased resistance to degradation is most evident in the fine grained HC treatments in this study, which have an inherently larger surface area (compared with larger grain sizes), and consequently showed the highest PO4–P release from the HC at T1 and T2 in all soils.
Gronwald et al. (2015) and Verheijen et al. (2009) stated that HC shows a general structurally and chemically induced lower decomposition resistance. The relative ease at which the mineralization of HC takes place, and the (suggested) ensuing temporary increase in soil microbial activity, is proposed to only occur for as long as the easily mineralized fraction (as DOC) is present in the soil, hence, potentially only over a short-term (Gronwald et al. 2015). This may be a contributing factor to the decreased PO4–P content observed from T1 to T2. A similar finding was reported by Buss et al. (2018), where a reduction in P release from BC over a series of extractions was observed. Almost 17% of the total P content (258 ± 2.9 mg kg−1) was initially released in the first extraction step, and successively less P at the second (137 ± 2.4 mg kg−1) and sixth (34 ± 0.8 mg kg−1) extraction steps.
Additionally, the PO4–P decrease may be related to physicochemical immobilization processes. It is well known that P contributes to numerous precipitation, mineralization, immobilization and fixation processes, which are further influenced by pH (Zhang et al. 2016). It has been proposed that increased PO4–P content may be caused by the liberation of P that is affixed to Fe- and aluminium (Al) oxides in soils as a result of increased pH due to BC addition, which effectively increases the availability of P to microbial communities and plants (Alling et al. 2014; Marmiroli et al. 2018). This liberation is particularly prevalent in acidic soils, whereby the liming effect of the added char acts to lower the Al and Fe content of the soil (Biederman and Harpole 2013). This is evident (by the greater increase in PO4–P content with HC addition at T1) in the acidic Podzol and Gleysol (T0 control pH: 5.8 and 4.9, respectively) in this study compared to the smaller increase observed in the more alkaline Chernozem (T0 control pH: 7.9).
The optimal pH range for the availability of P to plants is 6.0–6.5 (Buss et al. 2018), which all soil types were approaching during the course of this study i.e. from T1 to T2 (see Fig. 3). However, immobilization processes with solid soil compounds cannot be excluded, including those by soil microbes. The transformation of available P into the microbial biomass (and therefore its immobilization) is strongly dependent on the C:P ratio of the soil, whereby an increase in the soil C content increases P immobilization by microbes (Zhang et al. 2017). It is reasonable to assume that the provision of labile C compounds to soils by HC addition likely promoted the incorporation of P into the microbial biomass by stimulating microbial activity, thus reducing its concentration in the soils.
It is unlikely that precipitation processes played a dominant role in the decreased PO4–P content of the soils in this study, since the pH ranges achieved in all the soils by the addition of HC favour a reduced solubility of those sorbent ions (Fe, Al, and Ca), thus also reducing the precipitation of P (Penn and Camberato 2019; Huang et al. 2005; Ifansyah 2013). Therefore, the increased availability of PO4–P due to shifting pH caused by HC addition during this study, and its subsequent uptake by plants and microbes, are the most likely explanations for the observed decrease in PO4–P content from T1 to T2.
The roles and extent to which the processes of precipitation, inclusion, fixation and adsorption play a part remains open for further research. It can only be stated that the investigated soils in this study should react differently in respect of these processes. However, with regards to the fertilization efficiency of HTC, the results indicate that the HC did not have a medium- or long-term fertilization effect.
3.1.6 Potassium (K)
The initial K content of the controls was 565 ± 28.2 mg kg−1 for the Chernozem, 69.9 ± 14.2 mg kg−1 for the Podzol, and 43.9 ± 0.29 mg kg−1 for the Gleysol (Fig. 4). The K content of the HC was measured as 2612.5 ± 268.7 mg kg− 1. Shortly after adding HC (T1), the K content increased in all soils compared to the controls, but only the difference between the Podzolcontrol and Podzolmedium was statistically significant (p = 0.034). At the end of the study (T2), there were no significant differences in K content between the controls and HC amended soils or between the HC grain size treatments for all soils. Over the course of the study (T1 to T2), the K content decreased significantly (p < 0.05) in all treatments for all soils, except for the Podzolcontrol and both Gleysol controls (with and without plants). The addition of HC resulted in a short-term increase in K content, thus indicating HC as a K source.
Average potassium (K) content for the controls and hydrochar grain size treatments in a Chernozem, Podzol and Gleysol over the course of the plant growth experiment. Error bars represent standard deviation of the means. Different letters indicate significant differences in means at p < 0.05 level between treatments at the respective time periods. n.s = nonsignificant. Solid bars indicate significant differences in means (p < 0.05) between treatments at the beginning and end of the study. Patterned bars = nonsignificant. Control_pl = control with plant
The increase in K content in all HC amended soils compared to the controls at T1 is corroborated by Alling et al. (2014). As argued for PO4–P content (see above), it is reasonable to assume that the relatively easily degradable fraction of the HC is accountable for the quick release of K. As already shown for PO4–P, the data showed a K depletion at T2. Causation by immobilization via fixation to clays can be excluded in this study, since all soils reacted similarly, and furthermore, contain effectively no clays (Podzol) or consist mainly of micas (Gleysol) (Brümmer and Schroeder 1976), a non-fixing clay mineral. The same may be said for fixation to HC, and hence, plant uptake and/or microbial immobilization are the most probable K depleting processes, although a definitive explanation cannot be given at this point.
3.1.7 Mineral nitrogen (Nmin)
At the beginning of the study (T0), the Chernozemcontrol had an average ammonium (NH4+) content of 1.7 ± 0.6 mg kg−1; the Podzolcontrol: 3.1 ± 1.5 mg kg−1; and the Gleysolcontrol: 6.2 ± 0.3 mg kg−1. After initially adding the HC (T1), which contained 2.7% N (Table 1), the NH4+ content increased substantially for all soils (Fig. 5), however this increase was only significant (p < 0.05) for the Chernozemmedium (p = 0.034), Podzolfine (p = 0.049), and Gleysolcoarse (p = 0.034). At the end of the study (T2), no significant differences in NH4+ content between controls and HC amended soils were observed, except for the Gleysolfine (p = 0.009). Subsequent to the initial increase, the NH4+ content decreased significantly for all soils, except for the Chernozemcontrol_pl and Podzolcontrol_pl. From the results, it appears that HC acted as a source of NH4+ for a short time period after application.
Average ammonium content (expressed as NH4+–N) for the controls and hydrochar grain size treatments in a Chernozem, Podzol and Gleysol over the course of the plant growth experiment. Error bars represent standard deviation of the means. Different letters indicate significant differences in means at p < 0.05 level between treatments at the respective time periods. n.s = nonsignificant. Solid bars indicate significant differences in means (p < 0.05) between treatments at the beginning and end of the study. Patterned bars = nonsignificant. Control_pl = control with plant. *Statistically significantly difference (not visible on graphic)
At the beginning of the study (T0), the average nitrate (NO3−) of the Chernozemcontrol was 25 ± 2 mg kg−1; 17 ± 0.2 mg kg−1 for the Podzolcontrol; and 36.5 ± 0.6 mg kg−1 for the Gleysolcontrol (Fig. 6). All soils showed a varied response shortly after HC addition (T1), however no significant differences were observed. The same varied response between controls and HC grain size treatments was observed at T2. Significant differences in NO3− content occurred from T1 to T2 for all soils, except for the Chernozemcoarse, Podzolcontrol_pl and Gleysolfine. However, the soils reacted inconsistently with increasing and decreasing NO3− content. Consequently, a relationship between HC and NO3− content was indeterminable.
Average nitrate content (expressed as NO3−-N) for the controls and hydrochar grain size treatments in a Chernozem, Podzol and Gleysol over the course of the plant growth experiment. Error bars represent standard deviation of the means. Different letters indicate significant differences in means at p < 0.05 level between treatments at the respective time periods. n.s = nonsignificant. Solid bars indicate significant differences in means (p < 0.05) between treatments at the beginning and end of the study. Patterned bars = nonsignificant. Control_pl = control with plant
Tambone and Adani (2017) compared a digestate produced by anaerobic digestion to compost and sewage sludge, and found that the digestate had the highest N content (157.2 k kg−1 dry mass), of which ~ 80% occurred as NH4+. The HC used in this study may therefore have a similarly high initial NH4+ content, which, as is suggested for PO4–P and K, may have been released directly into the soil solution; thereby substantially increasing the NH4+ content of the HC amended soils at T1. This is substantiated by the C/N ratio of the HC used in this study (15.2) (refer to Table 1), whereby a C/N ratio < 20 indicates N mineralization (ammonification), and therefore the release of N from the HC (Dieguez-Alonso et al. 2018; Hagemann et al. 2016). However, as already stated for PO4–P and K, the relatively easily mineralized fraction of HC resulted in only a short-term release of nutrients (Gronwald et al. 2015), which was subsequently further rapidly processed within the food chain (Libra et al. 2011). In Bargmann et al. (2013a), the N content measured in the microbial biomass present in HC amended soils was found to be proportionate to the decreased N content of the incubated soils.
According to Tambone and Adani (2017), nitrification is likely responsible for the decreasing NH4+ concentration in soils, however, the associated NO3− results of this study do not comply with this explanation. Although it is probable that nitrification took place from T1 to T2, the non-continuous analysis during the course of the study resulted in this process going undetected. The results imply that the NO3− content, which has a higher susceptibility to change compared to the other measured plant available nutrients, responded more strongly to the micro-environmental conditions within the pots, such as the microbial activity and communities, temperature and moisture content, and the original soil organic matter (SOM) content of the different soil types (Hagemann et al. 2016). This finding is supported by Sun et al. (2014), who found that the interaction between BC and nitrification is multifactorial.
3.2 Effect of hydrochar application on plant growth dynamics of Chinese cabbage
3.2.1 Seed germination
The germination success of the controls was, on average, 53% for the Chernozem, 84% for the Podzol, and 76% for the Gleysol (Table 4). Despite a varied response of the soils to HC addition between round 1 and 2, and independent of HC grain size; the average germination success for the HC amended soils over the course of the experiment was in a similar range as the controls, with the Chernozem showing a germination rate of 56%, the Podzol 75%, and the Gleysol 60%. Hence, on average, the addition of HC did not inhibit the germination of Chinese cabbage seeds in any soils.
The reduction in germination rate in the Chernozem and Podzol from round one to round two, and the increase in the Gleysol, occur in both the controls and the HC amended soils, which indicates an environmental influence in combination with soil specific factors. The differences in the properties of the three soil types, such as texture, aggregate stability and WHC resulted in varied responses to potential environmental fluctuations, including increasing temperature. In the area in which the study was conducted, the temperature increased from an avg. of ~ 1 °C in February 2018 at the beginning of the study to ~ 13 °C by the end of the germination experiment in April 2018 (temperatures measured at 32 m height; Kock 2019). Such increased temperatures over this time may have caused a greater level of desiccation in the Chernozem and Podzol due to lower WHC compared to the Gleysol (see Table 2), which reduced the rate of seed germination in the Chernozem and Podzol in the second round of the experiment.
However, the overall lack of germination inhibition exhibited in this study contradicts existing literature, which states that the addition of a fresh and/or non-pretreated HC to soils delays or severely inhibits seed germination and plant growth. This inhibition is attributed to the organic contaminants inherent in the HC, such as phenols and organic acids (Bargmann et al. 2013b, 2014a; Röhrdanz et al. 2019), particularly at concentrations above 10% (w/w) (Bargmann et al. 2013b, 2014a; Busch et al. 2012). The HTC temperature and reaction time used in this study (~ 200 °C for ca. 3 h) may have been sufficiently high and long in duration to remove the volatile compounds that are potentially harmful for germination and plant growth (Reza et al. 2014), or the free gas exchange at the soil–atmosphere interface allowed for the quick release of these substances. The latter is supported by Sun et al. (2014), in which the potential to improve soil aeration was evinced by an enhanced soil porosity following BC addition, which further increased gas diffusion.
This study’s finding are corroborated by Fang et al. (2015), where the addition of HC produced at 200 °C yielded similar results for seedling growth to that of the control treatment. Puccini et al. (2018) found a reduction in the volatile fatty acid (98%), total polyphenol (41%) and total tannins (37%) contents of a HC after aging for 4 months under room temperature and free air exchange conditions. This aged HC showed the highest seed germination success of lettuce, compared to the fresh and washed HC. As such, the findings of this study may be the result of the relatively advanced age of the HC (at least > 3 months) and storage time (Bargmann et al. 2013b, 2014a), as well as the relatively low concentration of HC (5%) applied to the soils, which supposedly rendered the typically adverse impacts of HC on germination ineffective.
3.2.2 Biomass
With the exception of the Gleysolfine, there were no significant differences in biomass production between HC amended soils and the controls, nor between HC grain size treatments in all soils (Fig. 7). Therefore, it can be generalized from the results that the addition of HC to the soils showed no positive or negative influence on plant growth.
The results are corroborated by Bargmann et al. (2014a) and Röhrdanz et al. (2019), who also found that the addition of untreated HC had little influence on the growth of Chinese cabbage. The age of the HC, its low concentration in the soils, the release of volatile compounds in open-air conditions (Bargmann et al. 2013b) and/or HTC production conditions may all contribute to these findings. Furthermore, Bargmann et al. (2014a) postulated that the absence of negative HC influence may have been due to microbial activity, which acted to decompose the harmful phytotoxic compounds in the HC, and thereby allow for favourable plant growth conditions. The significantly higher biomass production in the Gleysolfine may be due to a combined effect of the higher WHC of the Gleysol compared to the other soil types, and the relatively easily degradable fraction of HC, as previously mentioned, which allowed for the release of essential nutrients for plant growth, particularly for the fine grained HC particles due to their inherently larger surface area, which is more vulnerable to degradation.
4 Conclusion
The addition of 5% HC from digestate feedstock resulted in a short-term supply of nutrients (PO4–P, K and NH4+) to all the soils, which was most significant in the finer grained fractions. This finding exemplifies the fertilization effect of HC. Importantly, the quick release and transient nature of this effect was likely due to the increased rate of decomposition as a result of the less stable C structure of the HC and its more labile C species relative to BC. However, the effects of HC on the biomass production of Chinese cabbage, WHC, aggregate stability and CEC were minor or intangible.
The fertilization effect of the HC was associated with a persistent compensatory shift in soil pH toward the pH of the HC, which was most pronounced in the finer grained HC treatments. Hence, the application of HC could partially substitute lime supply. Additionally, the observed pH shift provoked an intensified PO4–P solubility. On the other hand, the increased pH might favour SOM mineralization, a rather undesired impact, especially for sandy soils. A positive effect was found for seed germination. Contrary to other studies (Bargmann et al. 2013b, 2014a; Busch et al. 2012), no germination inhibition occurred which means that 5% HC application can be implemented without inducing harmful consequences for plant growth. The Chinese cabbage grew satisfyingly, however there was no further improvement compared to the controls.
It may therefore be concluded that the digestate feedstock was successfully converted into a useful product for soil amendment. The 5% HC application did not induce detrimental pH shifts and acted as a short-term fertilizer. However, it was unable to sustain the supply of nutrients at levels necessary to enhance plant growth and would therefore not be suitable as a long-term fertilizer in its present form. Nonetheless, its readily available high nutrient content, particularly in fine grained fractions, could have the potential as a replacement for traditional P and K fertilizers. Therefore, further research is necessary for this potential to be realised.
References
Abel S, Peters A, Trinks S, Schonsky H, Facklam M, Wessolek G (2013) Impact of biochar and hydrochar addition on water retention and water repellency of sandy soil. Geoderma 202–203:183–191
Alef K (1991) Methodenhandbuch bodenmikrobiologie: aktivitäten, biomasse, differenzierung. Ecomed Verlagsgesellschaft mbH, Landsberg/Lech
Alling V, Hale SE, Martinsen V, Mulder J, Smebye A, Breedveld GD, Cornelissen G (2014) The role of biochar in retaining nutrients in amended tropical soils. J Plant Nutr Soil Sci 177:671–680
Bargmann I, Martens R, Rillig MC, Kruse A, Kücke M (2013a) Hydrochar amendment promotes microbial immobilization of mineral nitrogen. J Plant Nutr Soil Sci 177:59–67
Bargmann I, Rillig MC, Buss W, Kruse A, Kücke M (2013b) Hydrochar and biochar effects on germination of spring barley. J Agron Crop Sci 199:360–373
Bargmann I, Rillig MC, Kruse A, Greef J-M, Kücke M (2014a) Effects of hydrochar application on the dynamics of soluble nitrogen in soils and on plant availability. J Plant Nutr Soil Sci 177:48–58
Bargmann I, Rillig MC, Kruse A, Greef J-M, Kücke M (2014b) Initial and subsequent effects of hydrochar amendment on germination and nitrogen uptake of spring barley. J Plant Nutr Soil Sci 177:68–74
Baronti S, Alberti G, Camin F, Criscuoli I, Genesio L, Mass R, Vaccari FP, Ziller L, Miglietta F (2017) Hydrochar enhances growth of poplar for bioenergy while marginally contributing to direct soil carbon sequestration. GCB Bioenergy 9:1618–1626
Basso D, Castello D, Baratieri M, Fiori L (2013) Hydrothermal carbonization of waste biomass: progress report and prospects. In: 21st European Biomass Conference and Exhibition, 3–7 June, Copenhagen, Denmark, pp 1478–1487
Bento LR, Castro AJR, Moreira AB, Ferreira OP, Bisinoti MC, Melo CA (2019) Release of nutrients and organic carbon in different soil types from hydrochar obtained using sugarcane bagasse and vinasse. Geoderma 334:24–32
Berge ND, Kammann C, Ro K, Libra J (2013) Environmental applications of hydrothermal carbonization technology: biochar production, carbon sequestration, and waste conversion. In: Titirici M-M (ed) Sustainable carbon materials from hydrothermal processes. Wiley, London, pp 295–329
Biederman LA, Harpole SW (2013) Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. GCB Bioenergy 5:202–214
Blanco-Canqui H (2017) Biochar and soil physical properties. Soil Sci Soc Am J 81:687–711
Brodowski S, Amelung W, Haumaier L, Abetz C, Zech W (2005) Morphological and chemical properties of black carbon in physical soil fractions as revealed by scanning electron microscopy and energy- dispersive X-ray spectroscopy. Geoderma 128:116–129
Brümmer G, Schroeder D (1976) Bestand, Umwandlung und Neubildung von Tonmineralen in küstennahen Sedimenten der Nordsee. Meyniana 28:19–28
Busch D, Glaser B (2015) Stability of co-composted hydrochar and biochar under field conditions in a temperate soil. Soil Use Manage 31:251–258
Busch D, Kammann C, Grünhage L, Müller C (2012) Simple biotoxicity tests for evaluation of carbonaceous soil additives: establishment and reproducibility of four test procedures. J Environ Qual 41:1023–1032
Buss W, Assavavittayanon K, Shepherd JG, Heal KV, Sohi S (2018) Biochar phosphorous release is limited by high pH and excess calcium. J Environ Qual 47:1298–1303
de Jesus Duarte S, Glaser B, Pellegrino Cerri CE (2019) Effect of biochar particle size on physical, hydrological and chemical properties of loamy and sandy tropical soils. Agronomy 9:165
Dieguez-Alonso A, Funke A, Anca-Couce A, Rombolà AG, Ojeda G, Bachmann J, Behrendt F (2018) Towards biochar and hydrochar engineering-influence of process conditions on surface physical and chemical properties, thermal stability, nutrient availability, toxicity and wettability. Energies 11:496
Domingues RR, Trugilho PF, Silva CA, de Melo ICNA, Melo LCA, Magriotis ZM, Sánchez-Monedero MA (2017) Properties of biochar derived from wood and high-nutrient biomasses with the aim of agronomic and environmental benefits. PLoS One 12:e0176884
Downie A, Crosky A, Munroe P (2009) Physical properties of biochar. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, pp 13–32
Dunn OJ (1964) Multiple comparisons using rank sums. Technometrics 6:241–252
Eibisch N, Helfrich M, Don A, Mikutta R, Kruse A, Ellerbrock R, Flessa H (2013) Properties and degradability of hydrothermal carbonization products. J Environ Qual 42:1565–1573
Eskandari S, Mohammadi A, Sandberg M, Eckstein RL, Hedberg K, Granström (2019) Hydrochar-amended substrates for production of containerized pine tree seedlings under different fertilization regimes. Agronomy 9:350
Fang J, Gao B, Chen J, Zimmerman AR (2015) Hydrochars derived from plant biomass under various conditions: characterization and potential applications and impacts. Chem Eng J 267:253–259
Fiori L, Basso D, Castello D, Baratieri M (2014) Hydrothermal carbonization of biomass: design of a batch reactor and preliminary experimental results. Chem Eng Trans 37:55–60
Garlapalli RK, Wirth B, Reza MT (2016) Pyrolysis of hydrochar from digestate: effect of hydrothermal carbonization and pyrolysis temperatures on pyrochar formation. Bioresour Technol 220:168–174
Głąb T, Palmowska J, Zaleski T, Gondek K (2016) Effect of biochar application on soil hydrological properties and physical quality of sandy soil. Geoderma 281:11–20
Glaser B, Lehmann J, Zech W (2002) Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—a review. Biol Fertil Soils 35:219–230
Greenberg I, Kaiser M, Gunina A, Ledesma P, Polifka S, Wiedner K, Mueller CW, Glaser B, Ludwig B (2019) Substitution of mineral fertilizers with biogas digestate plus biochar increases physically stabilized soil carbon but not crop biomass in a field trial. Sci Total Environ 680:181–189
Gronwald M, Don A, Tiemeyer B, Halfrich M (2015) Effects of fresh and aged chars from pyrolysis and hydrothermal carbonization on nutrient sorption in agricultural soils. Soil 1:475–489
Hagemann N, Harter J, Behrens S (2016) Elucidating the impacts of biochar applications on nitrogen cycling microbial communities. In: Ralebitso-Senior TK, Orr CH (eds) Biochar application: essential soil microbial ecology. Elsevier, Amsterdam, pp 163–198
Hale SE, Alling V, Martinsen V, Mulder J, Breedveld GD, Cornelissen G (2013) The sorption and desorption of phosphate-P, ammonium-N and nitrate-N in cacao shell and corn cob biochars. Chemosphere 91:1612–1619
Handbuch Forstliche Analytik (2005) Potentielle Kationenaustauschkapazität, DIN ISO 13536:1997-04, Eine Loseblatt-Sammlung der Analysemethoden im Forstbereich, Gutachterausschuss Forstliche Analytik (Hg), 1. Ergänzung (June 2005). https://www.bmel.de/SharedDocs/Downloads/Landwirtschaft/Wald-Jagd/Bodenzustandserhebung/Handbuch/Z-Ergaenzung1.pdf?__blob=publicationFile. Accessed 27 June 2019
Hao S, Zhu X, Liu Y, Qian F, Fang Z, Shi Q, Zhang S, Chen J, Ren ZJ (2018) Production temperature effects on the structure of hydrochar-derived dissolved organic matter and associated toxicity. Environ Sci Technol 52:7486–7495
Huang Q, Wang Z, Wang C, Wang S, Jin X (2005) Phosphorus release in response to pH variation in the lake sediments with different ratios of iron-bound P to calcium-bound P. Chem Spec Bioavailabil 17(2):55–61
Ifansyah H (2013) Soil pH and solubility of aluminum, iron, and phosphorus in Ultisols: the roles of humic acid. J Trop Soils 18(3):203–208
Jones NF, Pejchar L, Kiesecker JM (2015) The energy footprint: how oil, natural gas, and wind energy affect land for biodiversity and the flow of ecosystem services. Bioscience 65:290–301
Kalinina O, Krause S-E, Goryachkin SV, Karavaeva NA, Lyuri DI, Giani L (2011) Self-restoration of post-agrogenic Chernozems of Russia: soil development, carbon stocks, and dynamics of carbon pools. Geoderma 162:196–206
Kambo HS, Dutta A (2015) A comparative review of biochar and hydrochar in terms of production, physico-chemical properties and applications. Renew Sust Energ Rev 45:359–378
Kock D (2019) Aktuelles Wetter/Klimawerte. Carl Von Ossietzky Universität Oldenburg. https://uol.de/wetter. Accessed 26 Nov 2019
Laerd Statistics (2015) Kruskal-Wallis H Test using SPSS Statistics. Statistical tutorials and software guides. http://statistics/laerd.com/. Accessed 16 Aug 2019
Lehmann J, Rondon M (2006) Bio-char soil management on highly weathered soils in the humid tropics. In: Uphoff N, Ball AS, Fernandes E, Herren H, Husson O, Laing M, Palm C, Pretty J, Sánchez P, Sanginga N, Thies J (eds) Biological approaches to sustainable soil systems. CRC, Boca Raton, pp 517–530
Lehmann J, Gaunt J, Rondon M (2006) Bio-char sequestration in terrestrial ecosystems—a review. Mitigation adapt. Strateg Glob Change 11:403–427
Liao W, Thomas SC (2019) Biochar particle size and post-pyrolysis mechanical processing affect soil pH, water retention capacity, and plant performance. Soil Syst 3:14
Libra JA, Ro KS, Kammann C, Funke A, Berge ND, Neubauer Y, Titirici M-M, Fühner C, Bens O, Kern J, Emmerich K-H (2011) Hydrothermal carbonisation of biomass residuals: a comparative review of the chemistry, processes and applications of wet and dry pyrolysis. BioFuels 2:71–106
Liu Z, Dugan B, Masiello CA, Gonnermann HM (2017) Biochar particle size, shape, and porosity act together to influence soil water properties. PLoS One 12:1–19
Malghani S, Jüschke E, Baumert J, Thuille A, Antonietti M, Trumbore S, Gleixner G (2015) Carbon sequestration potential of hydrothermal carbonization char (hydrochar) in two contrasting soils; results of a 1-year field study. Biol Fertil Soils 51:123–134
Marchetti R, Castelli F (2013) Biochar from swine solids and digestate influence nutrient dynamics and carbon dioxide release in soil. J Environ Qual 42:893–901
Marmiroli M, Bonas U, Imperiale D, Lencioni G, Mussi F, Marmiroli N, Maestri E (2018) Structural and functional features of chars from different biomasses as potential plant amendments. Front Plant Sci 9:1119
Monlau F, Sambusiti C, Ficara E, Aboulkas A, Barakat A, Carrère H (2015) New opportunities for agricultural digestate valorization: current situation and perspectives. Energy Environ Sci 8:2600–2621
Mukherjee S, Weihermueller L, Tappe W, Vereechen H, Burauel P (2016) Microbial respiration of biochar- and digestate-based mixtures. Biol Fertil Soils 52:151–164
Nimmo JR, Perkins KS (2002) Aggregation stability and size distribution. In: Dane JH, Topp GC (eds) Methods of soil analysis, Part 4-Physical methods. Soil Sci Soc Am, Madison, pp 317–328
Penn CJ, Camberato JJ (2019) A Critical review on soil chemical processes that control how soil pH affects phosphorous availability to plants. Agriculture 9(6):120
Pituello C, Dal Ferro N, Francioso O, Simonetti G, Berti A, Piccoli I, Pisi A, Morari F (2018) Effects of biochar on the dynamics of aggregate stability in clay and sandy loam soils. Eur J Soil Sci 69(5):827–842
Prayogo C, Jones JE, Baeyens J, Bending GD (2014) Impact of biochar on mineralisation of C and N from soil and willow litter and its relationship with microbial community biomass and structure. Biol Fertil Soils 50:695–702
Puccini M, Ceccarini L, Antichi D, Seggiani M, Tavarini S, Latorre MH, Vitolo S (2018) Hydrothermal carbonization of municipal woody and herbaceous prunings: hydrochar valorisation as soil amendment and growth medium for horticulture. Sustainability. 10:846
Reza MT, Andert J, Wirth B, Busch D, Pielert J, Lynam JG, Mumme J (2014) Hydrothermal carbonisation of biomass for energy and crop production. Appl Bioenergy 1:11–29
Röhrdanz M, Rebling T, Ohlert J, Jasper J, Greve T, Buchwald R, von Frieling P, Wark M (2016) Hydrothermal carbonisation of biomass from landscape management- Influence of process parameters on soil properties of hydrochars. J Environ Manage 173:72–78
Röhrdanz M, Greve T, de Jager M, Buchwald R, Wark R (2019) Co-composted hydrochar substrates as growing media for horticultural crops. Sci Hortic 252:96–103
Salem MSA (2013) Soil biota interactions with hydrochar. PhD thesis, Freie Universität Berlin, Germany
Sänger-von Open P, Nack T, Nixdorf J, Mencke B (1993) Vorstellung der SrCl2-Methode nach Bach zur Bestimmung der effektiven Austauschkapazität und Vergleich mit der NH4Cl-Methode, in Zeitschrift für Pflanzenernährung und Bodenkunde, 156, pp 311–316
Schimmelpfennig S, Müller C, Grünhage L, Koch C, Kammann C (2014) Biochar, hydrochar and uncarbonized feedstock application to permanent grassland-Effects on greenhouse gas emissions and plant growth. Agric Ecosyst Environ 191:39–52
Schröter F (2018) The production of porous hydrochars. Unpublished PhD thesis, Carl von Ossietzky Universität, Germany
Schulz H, Glaser B (2012) Effects of biochar compared to organic and inorganic fertilizers on soil quality and plant growth in a greenhouse experiment. J Plant Nutr Soil Sci 175:410–422
Septien S, Sanz FJE, Salvador S, Valin S (2018) The effect of pyrolysis heating rate on the steam gasification reactivity of char from woodchips. Energy 142:68–78
Six J, Paustian K, Elliot ET, Combrink C (2000) Soil structure and organic matter. Soil Sci Soc Am J 64:681–689
Sombroek WG, Nachtergaele FO, Hebel A (1993) Amounts, dynamics and sequestering of carbon in tropical and subtropical soils. Ambio 22:417–426
Steiner C, Garcia M, Zech W (2009a) Effects of charcoal as slow release nutrient carrier on N-P-K dynamics and soil microbial population: pot experiments with ferralsol substrate. In: Woods WI, Teixeira WG, Lehmann J, Steiner C, WinklerPrins A, Rebellato L (eds) Amazonian dark earths: wim Sombroek’s vision. Springer, Dordrecht, pp 325–328
Steiner C, Teixeira WG, Woods WI, Zech W (2009b) Indigenous knowledge about terra preta formation. In: Woods WI, Teixeira WG, Lehmann J, Steiner C, WinklerPrins A, Rebellato L (eds) Amazonian dark earths: Wim Sombroek’s vision. Springer, Dordrecht, pp 193–204
Sun F, Lu S (2014) Biochars improve aggregate stability, water retention, and pore-space properties of clayey soil. J Plant Nutr Soil Sci 177:26–33
Sun Z, Bruun EW, Arthur E, de Jonge LW, Moldrup P, Hauggaard-Nielsen H, Elsgaard L (2014) Effect of biochar on aerobic processes, enzyme activity, and crop yields in two sandy loam soils. Biol Fertil Soils 50:1087–1097
Tambone F, Adani F (2017) Nitrogen mineralization from digestate in comparison to sewage sludge, compost and urea in a laboratory incubated soil experiment. J Plant Nutr Soil Sci 180:335–365
Theuerl S, Herrmann C, Heiermann M, Grundmann P, Landwehr N, Kreidenweis U, Prochnow A (2019) The future agricultural biogas plant in Germany: a vision. Energies 12(3):396
VDLUFA (Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten (Hg) (2002) Bestimmung von mineralischem Stickstoff (Nitrat und Ammonium) in Bodenprofilen (Nmin-Labormethode), Die Untersuchung von Böden, A 6.1.4.1, VDLUFA Methodenbuch I, 3. Teillieferung, VDLUFA-Verlag. https://www.vdlufa.de/Methodenbuch/index.php?option=com_content&view=article&id=2065&lang=de. Accessed 27 June 2019
VDLUFA (Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten (Hg) (2012a) Bestimmung der Kationenaustauschkapazität und der austauschbaren Kationen mit Calcit-gesättigter Haxammincobalt-Trichlorid-Lösung, Die Untersuchung von Böden, A 9.1.1.1, VDLUFA Methodenbuch I, 6. Teillieferung, VDLUFA-Verlag. https://www.vdlufa.de/Methodenbuch/index.php?option=com_content&view=article&id=7&Itemid=108&lang=de. Accessed 27 June 2019
VDLUFA (Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten (Hg) (2012b) Bestimmung von Phosphor und Kalium im Calcium-Acetate-Lactat-Auszug, Die Untersuchung von Böden, A 6.2.1.1, VDLUFA Methodenbuch I, 6. Teillieferung, VDLUFA-Verlag. https://www.vdlufa.de/Methodenbuch/index.php?option=com_content&view=article&id=2072&lang=de. Accessed 27 June 2019
Verheijen FGA, Jeffrey S, Bastos AC, van der Velde M, Diafas I (2009) Biochar application to soils: A critical scientific review of effects on soil properties, processes and functions. EUR 24099 EN, Office for the Official Publications of the European Communities, Luxembourg
WinklerPrins AMGA (2014) Terra Preta: the mysterious soils of the Amazon. In: Churchman GJ, Landa ER (eds) The soils underfoot: infinite possibilities for a finite resource. Taylor & Francis, Florida, pp 235–245
Woolf D, Amonette JE, Street-Perrott FA, Lehmann J, Joseph S (2010) Sustainable biochar to mitigate global climate change. Nat Commun 1:1–9
Zhang B, von Keitz M, Valentas K (2008) Thermal effects on hydrothermal biomass liquefaction. Appl Biochem Biotechnol 147:143–150
Zhang H, Chen C, Gray EM, Boyd SE, Yang H, Zhang D (2016) Roles of biochar in improving phosphorous availability in soils: a phosphate adsorbent and a source of available phosphorous. Geoderma 276:1–6
Zhang X, Zhang L, Li A (2017) Hydrothermal co-carbonization of sewage sludge and pinewood sawdust for nutrient-rich hydrochar production: synergistic effects and products characterization. J Environ Manage 201:52–62
Acknowledgements
Open Access funding provided by Projekt DEAL. This study was conducted under the auspices of the Circular BIOmass CAScade to 100% (BIOCAS) project, which is funded by the European Union Interreg North Sea Region Project 38-2-4-17. We would like to thank the students of the Soil Science working group who provided analytical assistance, as well as Thomas Pollmann for his support and consultation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Jager, M., Röhrdanz, M. & Giani, L. The influence of hydrochar from biogas digestate on soil improvement and plant growth aspects. Biochar 2, 177–194 (2020). https://doi.org/10.1007/s42773-020-00054-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42773-020-00054-2