Abstract
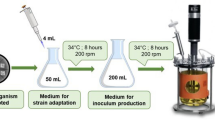
Eco-friendly natural pigment demand has ever-increasing popularity due to health and environmental concerns. In this context, the aim of this study was to evaluate the feasibility use of Saba banana peel as low-cost fermentable substrate for the production of pigments, xylanase and cellulase enzymes by Monascus purpureus. Among the strains tested, M. purpureus TISTR 3385 produced pigments better and had higher enzyme activities. Under the optimal pigment-producing conditions at the initial moisture content of 40% and initial pH of 6.0, the pigments comprising yellow, orange, and red produced by the fungi were achieved in the range of 0.40–0.93 UA/g/day. The maximum xylanase and cellulase activities of 8.92 ± 0.46 U/g and 4.72 ± 0.04 U/g were also obtained, respectively. More importantly, solid-state fermentation of non-sterile peel could be achieved without sacrificing the production of the pigments and both enzymes. These indicated the potential use of the peel as fermentable feedstock for pigment production by the fungi and an environmental-friendly approach for sustainable waste management and industrial pigment and enzyme application.




Similar content being viewed by others
References
Lagashetti AC, Dufossé L, Singh SK, Singh PN (2019) Fungal pigments and their prospects in different industries. Microorganisms 7(12):604. https://doi.org/10.3390/microorganisms7120604
Lopes FC, Ligabue-Braun R (2021) Agro-industrial residues: Eco-friendly and inexpensive substrates for microbial pigments production. Front Sustain Food Syst 5:1–16. https://doi.org/10.3389/fsufs.2021.589414
Kantifedaki A, Kachrimanidou V, Mallouchos A, Papanikolaou S, Koutinas AA (2018) Orange processing waste valorisation for the production of bio-based pigments using the fungal strains Monascus purpureus and Penicillium purpurogenum. J Clean Prod 185:882–890. https://doi.org/10.1016/j.jclepro.2018.03.032
Liu J, Luo Y, Guo T, Tang C, Chai X, Zhao W, Bai J, Lin Q (2020) Cost-effective pigment production by Monascus purpureus using rice straw hydrolysate as substrate in submerged fermentation. J Biosci Bioeng 129(2):229–236. https://doi.org/10.1016/j.jbiosc.2019.08.007
Venil C K, Devi P R, Ahmad W A (2020) Agro-industrial waste as substrates for the production of bacterial pigment. In: Zakaria Z., Boopathy R., Dib J. (eds) Valorisation of agro-industrial residues – Volume I: Biological Approaches. Applied Environmental Science and Engineering for a Sustainable Future. Springer, Cham https://doi.org/10.1007/978-3-030-39137-9_7
Terán Hilares R, de Souza RA, Marcelino PF, da Silva SS, Dragone G, Mussatto SI, Santos JC (2018) Sugarcane bagasse hydrolysate as a potential feedstock for red pigment production by Monascus ruber. Food Chem 245:786–791. https://doi.org/10.1016/j.foodchem.2017.11.111
de Almeida AB, Santos NH, de Lima TM, Santana RV, de Oliveira Filho JG, Peres DS, Egea MB (2021) Pigment bioproduction by Monascus purpureus using corn bran, a byproduct of the corn industry. Biocatal Agric Biotechnol 32:101931. https://doi.org/10.1016/j.bcab.2021.101931
Chen X, Yan J, Chen J, Gui R, Wu Y, Li N (2021) Potato pomace: an efficient resource for Monascus pigments production through solid-state fermentation. J Biosci Bioeng 132(2):167–173. https://doi.org/10.1016/j.jbiosc.2021.03.007
Ahmad M, Panda B P (2014) Optimization of red pigment production by Monascus purpureus MTCC 369 under solid-state fermentation using response surface methodology. Songklanakarin J. Sci. Technol. 36(4): 439–444. https://rdo.psu.ac.th/sjstweb/journal/36-4/36-4-8.pdf.
Jampala P, Tadikamalla S, Preethi M, Ramanujam S, Uppuluri KB (2017) Concurrent production of cellulase and xylanase from Trichoderma reesei NCIM 1186: enhancement of production by desirability-based multi-objective method. Biotech 7(1) https://doi.org/10.1007/s13205-017-0607-y
Lim T K (2012) Musa acuminata × balbisiana (ABB Group) ‘Saba’. In: Edible Medicinal And Non Medicinal Plants. Springer, Dordrecht https://doi.org/10.1007/978-94-007-2534-8_70
Reginio FC, Ketnawa S, Ogawa Y (2020) In vitro examination of starch digestibility of Saba banana [Musa ‘saba’(Musa acuminata × Musa balbisiana)]: impact of maturity and physical properties of digesta Sci. Rep 10:1811. https://doi.org/10.1038/s41598-020-58611-5
Khamsucharit P, Laohaphatanalert K, Gavinlertvatana P, Sriroth K, Sangseethong K (2018) Characterization of pectin extracted from banana peels of different varieties. Food Sci Biotechnol 27:623–629. https://doi.org/10.1007/s10068-017-0302-0
Qiu LP, Zhao GL, Wu H, Jiang L, Li XF, Liu JJ (2010) Investigation of combined effects of independent variables on extraction of pectin from banana peel using response surface methodology. Carbohydr Polym 80(2):326–331. https://doi.org/10.1016/j.carbpol.2010.01.018
Rehman S, Aslam H, Ahmad A, Khan SA, Sohail M (2014) Production of plant cell wall degrading enzymes by monoculture and co-culture of Aspergillus niger and Aspergillus terreus under SSF of banana peels. Braz J Microbiol 45(4):1485–1492. https://doi.org/10.1590/s1517-83822014000400045
Yu H, Yan M, Huang X (2018) Cell counting. In: CMOS Integrated Lab-on-a-Chip System for Personalized Biomedical Diagnosis. John Wiley & Sons https://doi.org/10.1002/9781119218333
Yu X, Liu Y, Cui Y, Cheng Q, Zhang Z, Lu JH, Meng Q, Teng L, Ren X (2016) Measurement of filter paper activities of cellulase with microplate-based assay. Saudi J Biol Sci 23:S93–S98. https://doi.org/10.1016/j.sjbs.2015.06.018
Katsimpouras C, Dedes G, Thomaidis NS, Topakas E (2019) A novel fungal GH30 xylanase with xylobiohydrolase auxiliary activity. Biotechnol Biofuels 12:120. https://doi.org/10.1186/s13068-019-1455-2
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428. https://doi.org/10.1021/ac60147a030
Endo A (1979) Monacolin K, a new hypocholesterolemic agent produced by a Monascus species. J Antibiot (Tokyo) 32(8):852–854. https://doi.org/10.7164/antibiotics.32.852
Pisareva E, Savov V, Kujumdzieva A (2005) Pigments and citrinin biosynthesis by fungi belonging to genus Monascus. Zeitschrift fur Naturforsch. - Sect. C J Biosci 60:116–120. https://doi.org/10.1515/znc-2005-1-221
Dikshit R, Tallapragada P (2018) Development and screening of mutants from Monascus sanguineus for secondary metabolites production. Beni-Suef Univ J Basic Appl Sci 7(2):235–240. https://doi.org/10.1016/j.bjbas.2018.03.001
Prakash H, Chauhan PS, General T, Sharma AK (2018) Development of eco-friendly process for the production of bioethanol from banana peel using inhouse developed cocktail of thermo-alkali-stable depolymerizing enzymes. Bioprocess Biosyst Eng 41:1003–1016. https://doi.org/10.1007/s00449-018-1930-3
Huang L, Sun N, Ban L, Wang Y, Yang H (2019) Ability of different edible fungi to degrade crop straw. AMB Express 9(4) https://doi.org/10.1186/s13568-018-0731-z
Lv J, Zhang BB, Liu XD, Zhang C, Chen L, Xu GR, Cheung PCK (2017) Enhanced production of natural yellow pigments from Monascus purpureus by liquid culture: The relationship between fermentation conditions and mycelial morphology. J Biosci Bioeng 124:452–458. https://doi.org/10.1016/j.jbiosc.2017.05.010
Hajjaj H, Klaébé A, Goma G, Blanc P J, Barbier E, François J (2000) Medium-chain fatty acids affect citrinin production in the filamentous fungus Monascus ruber. Appl Environ Microbiol 66 https://doi.org/10.1128/AEM.66.3.1120-1125.2000
Agboyibor C, Kong WB, Chen D, Zhang AM, Niu SQ (2018) Monascus pigments production, composition, bioactivity and its application: a review. Biocatal Agric Biotechnol 16:433–447. https://doi.org/10.1016/j.bcab.2018.09.012
Wang M, Huang T, Chen G, Wu Z (2017) Production of water-soluble yellow pigments via high glucose stress fermentation of Monascus ruber CGMCC 10910. Appl Microbiol Biotechnol 101:3121–3130. https://doi.org/10.1007/s00253-017-8106-y
Huang T, Wang M, Shi K, Chen G, Tian X, Wu Z (2017) Metabolism and secretion of yellow pigment under high glucose stress with Monascus ruber. AMB Express 7:79. https://doi.org/10.1186/s13568-017-0382-5
Arantes V, Saddler JN (2011) Cellulose accessibility limits the effectiveness of minimum cellulase loading on the efficient hydrolysis of pretreated lignocellulosic substrates. Biotechnol Biofuels 4 https://doi.org/10.1186/1754-6834-4-3
Li J, Zhou P, Liu H, et al (2014) Synergism of cellulase, xylanase, and pectinase on hydrolyzing sugarcane bagasse resulting from different pretreatment technologies. Bioresour Technol 155 https://doi.org/10.1016/j.biortech.2013.12.113
Kongbangkerd T, Tochampa W, Chatdamrong W, Kraboun K (2014) Enhancement of antioxidant activity of monascal waxy corn by a 2-step fermentation. Int J Food Sci Technol 49 https://doi.org/10.1111/ijfs.12479
Kraboun K, Kongbangkerd T, Rojsuntornkitti K, Phanumong P (2019) Factors and advances on fermentation of Monascus sp. for pigments and monacolin K production: A review. Int. Food Res. J. 26(3): 751–761. http://www.ifrj.upm.edu.my
da Delabona PS, Pirota RDPB, Codima CA, Tremacoldi CR, Rodrigues A, Farinas CS (2013) Effect of initial moisture content on two Amazon rainforest Aspergillus strains cultivated on agro-industrial residues: Biomass-degrading enzymes production and characterization. Ind Crops Prod 42:236–242. https://doi.org/10.1016/j.indcrop.2012.05.035
Yazid NA, Barrena R, Komilis D, Sánchez A (2017) Solid-state fermentation as a novel paradigm for organic waste valorization: a review. Sustain 9(2):224. https://doi.org/10.3390/su9020224
Velmurugan P, Hur H, Balachandar V, Kamala-Kannan S, Lee KJ, Lee SM, Chae JC, Shea PJ, Oh BT (2011) Monascus pigment production by solid-state fermentation with corn cob substrate. J Biosci Bioeng 112(6):590–594. https://doi.org/10.1016/j.jbiosc.2011.08.009
Haque MA, Kachrimanidou V, Koutinas A, Lin CSK (2016) Valorization of bakery waste for biocolorant and enzyme production by Monascus purpureus. J Biotechnol 231(10):55–64. https://doi.org/10.1016/j.jbiotec.2016.05.003
Zhang BB, Xing HB, Jiang BJ, Chen L, Xu GR, Jiang Y, Zhang DY (2018) Using millet as substrate for efficient production of monacolin K by solid-state fermentation of Monascus ruber. J Biosci Bioeng 125(3):333–338. https://doi.org/10.1016/j.jbiosc.2017.10.011
Pirota RDPB, Tonelotto M, da Delabona PS, Fonseca RF, Paixão DAA, Baleeiro FCF, BertucciNeto V, Farinas CS (2013) Enhancing xylanases production by a new Amazon Forest strain of Aspergillus oryzae using solid-state fermentation under controlled operation conditions. Ind Crops Prod 45:465–471. https://doi.org/10.1016/j.indcrop.2013.01.010
Wen Q, Cao X, Chen Z, Xiong Z, Liu J, Cheng Z, Zheng Z, Long C, Zheng B, Huang Z (2020) An overview of Monascus fermentation processes for monacolin K production. Open Chem 18 https://doi.org/10.1515/chem-2020-0006
Panda BP, Javed S, Ali M (2010) Optimization of fermentation parameters for higher lovastatin production in red mold rice through co-culture of Monascus purpureus and Monascus ruber. Food Bioprocess Technol 3:373–378. https://doi.org/10.1007/s11947-008-0072-z
Valera HR, Gomes J, Lakshmi S, Gururaja R, Suryanarayan S, Kumar D (2005) Lovastatin production by solid state fermentation using Aspergillus flavipes. Enzyme Microb Technol 37(5):521–526. https://doi.org/10.1016/j.enzmictec.2005.03.009
Shaligram NS, Singh SK, Singhal RS, Szakacs G, Pandey A (2008) Compactin production in solid-state fermentation using orthogonal array method by P. brevicompactum. Biochem Eng J 41(3):295–300. https://doi.org/10.1016/j.bej.2008.05.011
Orak T, Caglar O, Ortucu S, Ozkan H, Taskin M (2018) Chicken feather peptone: a new alternative nitrogen source for pigment production by Monascus purpureus. J Biotechnol 271(10):56–62. https://doi.org/10.1016/j.jbiotec.2018.02.010
Embaby AM, Hussein MN, Hussein A (2018) Monascus orange and red pigments production by Monascus purpureus ATCC16436 through co-solid state fermentation of corn cob and glycerol: an eco-friendly environmental low cost approach. PLoS One 13 https://doi.org/10.1371/journal.pone.0207755
Subsaendee T, Kitpreechavanich V, Yongsmith B (2014) Growth, glucoamylase, pigments and monacolin K production on rice solid culture in flask and koji chamber using Monascus sp. KB9. Chiang Mai J. Sci. 41(5.1): 1044–1057. http://epg.science.cmu.ac.th/ejournal/index.php
Zhang JC, Wang CT, Wang CT, Wang SX, Sun BG (2019) Changes in components of aqueous and non-aqueous extracts from sea buckthorn seed residues through solid state fermentation of Monascus purpureus. BioResources 14(1):2197–2215 (ojs.cnr.ncsu.edu)
Chen Z, Wan C (2017) Non-sterile fermentations for the economical biochemical conversion of renewable feedstocks. Biotechnol Lett 39:1765–1777. https://doi.org/10.1007/s10529-017-2429-8
Koutinas AA, Chatzifragkou A, Kopsahelis N, Papanikolaou S, Kookos IK (2014) Design and techno-economic evaluation of microbial oil production as a renewable resource for biodiesel and oleochemical production. Fuel 116:566–577. https://doi.org/10.1016/j.fuel.2013.08.045
Moustogianni A, Bellou S, Triantaphyllidou I E, Aggelis G (2015) Feasibility of raw glycerol conversion into single cell oil by zygomycetes under non-aseptic conditions. Biotechnol Bioeng 112 https://doi.org/10.1002/bit.25482
Cruz AF, Barka GD, Blum LEB, et al. (2019) Evaluation of microbial communities in peels of Brazilian tropical fruits by amplicon sequence analysis. Braz J Microbiol 50(3) https://doi.org/10.1007/s42770-019-00088-0
Vasco-Correa J, Ge X, Li Y (2016) Fungal pretreatment of non-sterile miscanthus for enhanced enzymatic hydrolysis. Bioresour Technol 203:118–123. https://doi.org/10.1016/j.biortech.2015.12.018
Yafetto L (2018) Protein enrichment of cassava pulp by solid-state fermentation using Aspergillus niger. Stud Fungi 3(1):7–18. https://doi.org/10.5943/sif/3/1/2
Pan S, Chen G, Wu R, Cao X, Liang Z (2019) Non-sterile submerged fermentation of fibrinolytic enzyme by marine bacillus subtilis harboring antibacterial activity with starvation strategy. Front Microbiol 10 https://doi.org/10.3389/fmicb.2019.01025
Singhania RR, Sukumaran RK, Patel AK, Larroche C, Pandey A (2010) Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases. Enzyme Microb Technol 46:541–549. https://doi.org/10.1016/j.enzmictec.2010.03.010
Vk P, Savitha J (2012) Solid state fermentation: An effective method for Lovastatin production by fungi over submerged fermentation. E3 J. Biotechnol. Pharm. Res. 3(2): 15–21. http://www.e3journals.org/JBPR
Carvalho JC, Pandey A, Babitha S, Soccol CR (2003) Production of Monascus biopigments: An overview. Agro Food Ind Hi Tech 14(6):37–43. https://doi.org/10.21608/bfsa.2021.73860.1100
Wonganu B, Kongruang S (2010) Red bacterial cellulose production by fermentation of Monascus purpureus. In: International Conference on Chemistry and Chemical Engineering 137–141 IEEE
Kongruang S (2011) Growth kinetics of biopigment production by Thai isolated Monascus purpureus in a stirred tank bioreactor. J Ind Microbiol Biotechnol 38(1):93–99. https://doi.org/10.1007/s10295-010-0834-2
Acknowledgements
Thanks to the Biology Laboratory, Faculty of Science Technology and Agriculture, Yala Rajabhat University for providing the resources and facilities. Thanks also to Mr. Andrew Barclay for his assistance with the English.
Funding
The research was financially supported by Yala Rajabhat University. The first and fourth authors were supported by Thailand Research Fund [grant no. RTA6280014].
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, investigation, data curation, and writing—original draft, review and editing: Yasmi Louhasakul; Investigation and data curation: Hindol Wado and Rohana Lateh; Funding acquisition, supervision, and writing—review and editing: Benjamas Cheirsilp.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any experiments with human participants or animals.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor:Luis Augusto Nero
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Louhasakul, Y., Wado, H., Lateh, R. et al. Solid-state fermentation of Saba banana peel for pigment production by Monascus purpureus. Braz J Microbiol 54, 93–102 (2023). https://doi.org/10.1007/s42770-022-00866-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-022-00866-3