Abstract
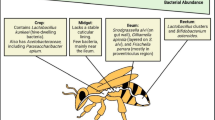
The honey bee, Apis mellifera, is the most critical managed agricultural pollinator and its recent colony losses is of global concern. Bacterial pathogens are one of the causes attributed to the decline of the bee population. Serratia marcescens is an opportunistic pathogen with a broad host range, capable of affecting various plants and animals. S. marcescens can invade various tissues of the honey bee, including the gut and hemolymph, leading to severe infections and compromising bee health. S. marcescens is often associated with secondary infections, and can rapidly multiply forming biofilms, making it particularly challenging to control and eliminate. Understanding the interactions between S. marcescens and the honey bee, as well as exploring potential solutions such as probiotics, is crucial for bee health improvement. In this study, we assessed the effect of Lactobacillus kunkeei, a bee gut associated bacterium on honey bee survival, protection against the opportunistic pathogen S. marcescens, and activation of bee immune responses. Newly emerged worker bees were divided into groups: MD- no treatment, GH-inoculated with hive bees’ gut homogenate and Lk- inoculated with L. kunkeei. After five days of inoculation, bees in each respective group were exposed to the opportunistic pathogen S. marcescens. Our results show that L. kunkeei increased bee survival after S. marcescens infection, inhibited its proliferation in the gut, and reduced antimicrobial peptide genes expression in the gut epithelium compared to microbe-deprived bees in a laboratory setting. Our findings highlight the potential use of the probiotic to enhance bee health.




Similar content being viewed by others
References
Arredondo D, Castelli L, Porrini MP, Garrido PM, Eguaras MJ, Zunino P et al (2018) Lactobacillus kunkeei strains decreased the infection by honey bee pathogens Paenibacillus larvae and Nosema ceranae. Benef Microbes 9:279–290. https://doi.org/10.3920/BM2017.0075
Audisio MC (2017) Gram-positive Bacteria with probiotic potential for the Apis mellifera L. Honey Bee: the experience in the Northwest of Argentina. Probiotics Antimicrob Proteins 9:22–31. https://doi.org/10.1007/s12602-016-9231-0
Bisson LF, Walker G, Ramakrishnan V, Luo Y, Fan Q, Wiemer E, Luong P, Ogawa M, Joseph L The Two Faces of Lactobacillus kunkeei: Wine Spoilage Agent and Bee Probiotic. Catalyst: Discovery into Practice, 1(1), 1–11., Rosso G, Engel P (2017) (2018). Functional roles and metabolic niches in the honey bee gut microbiota. Curr. Opin. Microbiol. 43, 69–76. https://doi.org/10.1016/j.mib.2017.12.009
Bonilla-Rosso G, Engel P (2018). Functional roles and metabolic niches in the honey bee gut microbiota. Curr Opin Microbiol 43:69–76. https://doi.org/10.1016/j.mib.2017.12.009
Broderick NA, Buchon N, Lemaitre B (2014) Microbiota-Induced Changes in Drosophila melanogaster Host Gene Expression and Gut Morphology. MBio 5, 1–13. https://doi.org/10.1128/mBio.01117-14
Burritt NL, Foss NJ, Neeno-Eckwall EC, Church JO, Hilger AM, Hildebrand JA et al (2016) Sepsis and hemocyte loss in Honey Bees (Apis mellifera) infected with Serratia marcescens strain Sicaria. PLoS ONE 11:e0167752. https://doi.org/10.1371/journal.pone.0167752
Cremer S, Armitage SAO, Schmid-Hempel P (2007) Social Immun Curr Biol 17:693–702. https://doi.org/10.1016/j.cub.2007.06.008
Daisley BA, Pitek AP, Chmiel JA, Al KF, Chernyshova AM, Faragalla KM et al (2019) Novel probiotic approach to counter Paenibacillus larvae infection in honey bees. ISME J 2019 1–16. https://doi.org/10.1038/s41396-019-0541-6
Danihlík J, Aronstein K, Petřivalský M (2015) Antimicrobial peptides: a key component of honey bee innate immunity. J Apic Res 54:123–136. https://doi.org/10.1080/00218839.2015.1109919
Engel P, James RR, Koga R, Kwong WK, McFrederick QS, Moran NA (2013) Standard methods for research on Apis mellifera gut symbionts. J Apic Res 52:1–24. https://doi.org/10.3896/IBRA.1.52.4.07
Engel P, Moran NA (2013) Functional and evolutionary insights into the simple yet specific gut microbiota of the honey bee from metagenomic analysis. Gut Microbes 4:60–65. https://doi.org/10.4161/gmic.22517
Evans JD (2006) Beepath: an ordered quantitative-PCR array for exploring honey bee immunity and disease. J Invertebr Pathol 93:135–139. https://doi.org/10.1016/j.jip.2006.04.004
Evans JD, Schwarz RS (2011) Bees brought to their knees: microbes affecting honey bee health. Trends Microbiol 19:614–620. https://doi.org/10.1016/j.tim.2011.09.003
Evans JD, Spivak M (2010) Socialized medicine: individual and communal disease barriers in honey bees. J Invertebr Pathol 103:S62–S72. https://doi.org/10.1016/j.jip.2009.06.019
Fünfhaus A, Ebeling J, Genersch E (2018) Bacterial pathogens of bees. Curr Opin Insect Sci 26:89–96. https://doi.org/10.1016/j.cois.2018.02.008
Forsgren E, Olofsson TC, Vásquez A, Fries I (2010) Novel lactic acid bacteria inhibiting Paenibacillus larvae in honey bee larvae. Apidologie 41:99–108. https://doi.org/10.1051/apido/2009065
Goulson D, Nicholls E, Botías C, Rotheray EL (2015) Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Sci (80-) 347. https://doi.org/10.1126/science.1255957
Iorizzo M, Lombardi SJ, Ganassi S, Testa B, Ianiro M, Letizia F et al (2020) Antagonistic Activity against Ascosphaera apis and Functional Properties of Lactobacillus kunkeei Strains. antibiotics 9, 1–16. https://doi.org/10.3390/antibiotics9050262
Iwaya A, Nakagawa S, Iwakura N, Taneike I, Kurihara M, Kuwano T et al (2005) Rapid and quantitative detection of blood Serratia marcescens by a real-time PCR assay: its clinical application and evaluation in a mouse infection model. FEMS Microbiol Lett 248:163–170. https://doi.org/10.1016/j.femsle.2005.05.041
Janashia I, Alaux C (2016) Specific immune stimulation by endogenous bacteria in honey bees (Hymenoptera: Apidae). J Econ Entomol 109:1474–1477. https://doi.org/10.1093/jee/tow065
Janashia I, Choiset Y, Rabesona H, Hwanhlem N, Bakuradze N, Chanishvili N et al (2016) Protection of honeybee Apis mellifera by its endogenous and exogenous lactic flora against bacterial infections. Ann Agrar Sci 14:177–181. https://doi.org/10.1016/j.aasci.2016.07.002
Kešnerová L, Emery O, Troilo M, Liberti J, Erkosar B, Engel P (2019) Gut microbiota structure differs between honeybees in winter and summer. ISME J 14:801–814. https://doi.org/10.1038/s41396-019-0568-8
Kwong WK, Moran NA (2016) Gut microbial communities of social bees. Nat Rev Microbiol 14:374–384. https://doi.org/10.1038/nrmicro.2016.43
Lamei S, Stephan JG, Riesbeck K, Vasquez A, Olofsson T, Nilson B et al (2019) The secretome of honey bee-specific lactic acid bacteria inhibits Paenibacillus larvae growth. J Apic Res 58:405–412. https://doi.org/10.1080/00218839.2019.1572096
Martinson VG, Danforth BN, Minckley RL, Rueppell O, Tingek S, Moran NA (2011) A simple and distinctive microbiota associated with honey bees and bumble bees. Mol Ecol 20:619–628. https://doi.org/10.1111/j.1365-294X.2010.04959.x
Moran NA, Hansen AK, Powell JE, Sabree ZL (2012) Distinctive gut microbiota of honey bees assessed using deep sampling from individual worker bees. PLoS ONE 7:1–10. https://doi.org/10.1371/journal.pone.0036393
Motta EVS, Powell JE, Leonard SP, Moran NA (2022) Prospects for probiotics in social bees. Philosophical Trans Royal Soc B: Biol Sci 377(1853):0–3. https://doi.org/10.1098/rstb.2021.0156
Motta EVS, Raymann K, Moran NA (2018) Glyphosate perturbs the gut microbiota of honey bees. Proc. Natl. Acad. Sci. U. S. A. 115, 10305–10310. https://doi.org/10.1073/pnas.1803880115
Olofsson TC, Butler È, Markowicz P, Lindholm C, Larsson L, Vásquez A (2016) Lactic acid bacterial symbionts in honeybees – an unknown key to honey’s antimicrobial and therapeutic activities. Int Wound J 13:668–679. https://doi.org/10.1111/iwj.12345
Paredes JC, Welchman DP, Poidevin M, Lemaitre B (2011) Negative regulation by Amidase PGRPs shapes the Drosophila antibacterial response and protects the fly from innocuous infection. Immunity 35:770–779. https://doi.org/10.1016/j.immuni.2011.09.018
Pfaffl WM (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29(9):e45. https://doi.org/10.1093/nar/29.9.e45
Powell JE, Carver Z, Leonard SP (2021) Field-realistic Tylosin exposure impacts Honey Bee Microbiota and Pathogen susceptibility, which is ameliorated by native gut probiotics. Micribiology Spectr, 9(1)
Rangberg A, Mathiesen G, Amdam GV, Diep DB (2015) The paratransgenic potential of Lactobacillus kunkeei in the honey bee Apis mellifera. Benef Microbes 6:513–523. https://doi.org/10.3920/BM2014.0115
Raymann K, Bobay LM, Moran NA (2018a) Antibiotics reduce genetic diversity of core species in the honeybee gut microbiome. Mol Ecol 27:2057–2066. https://doi.org/10.1111/mec.14434
Raymann K, Coon KL, Shaffer Z, Salisbury S, Moran NA (2018b) Pathogenicity of serratia marcescens strains in honey bees. MBio 9(5):343–349. https://doi.org/10.1128/mbio.01649-18
Raymann K, Moran NA (2018) The role of the gut microbiome in health and disease of adult honey bee workers. Curr Opin Insect Sci 26:97–104. https://doi.org/10.1016/j.cois.2018.02.012
Raymann K, Shaffer Z, Moran NA (2017) Antibiotic exposure perturbs the gut microbiota and elevates mortality in honeybees. PLOS Biol 15:e2001861. https://doi.org/10.1371/journal.pbio.2001861
Schwarz RS, Moran NA, Evans JD (2016) Early gut colonizers shape parasite susceptibility and microbiota composition in honey bee workers. Proc. Natl. Acad. Sci. U. S. A. 113, 9345–9350. https://doi.org/10.1073/pnas.1606631113
Simone M, Evans JD, Spivak M (2009) Resin Collection and Social Immunity in Honey Bees. Evolution (N. Y). 63:3016–3022. https://doi.org/10.1111/j.1558-5646.2009.00772.x
Steele MI, Motta EVS, Tejashwini G, Moran NA (2021) The gut microbiota protects bees from Invasion by a bacterial Pathogen. Microbiol Spectr 9(2):1–11. https://doi.org/10.1128/Spectrum.00394-21
Tola YH, Waweru JW, Hurst GDD, Slippers B, Paredes JC (2020) Characterization of the kenyan Honey Bee (Apis mellifera) Gut Microbiota: A First look at Tropical and Sub-Saharan African Bee Associated Microbiomes. Microorganisms 8:1721. https://doi.org/10.3390/microorganisms8111721
Vásquez A, Forsgren E, Fries I, Paxton RJ, Flaberg E, Szekely L et al (2012) Symbionts as major modulators of Insect Health: lactic acid Bacteria and honeybees. PLoS ONE 7. https://doi.org/10.1371/journal.pone.0033188
Yoshiyama M, Wu M, Sugimura Y, Takaya N, Kimoto-Nira H, Suzuki C (2013) Inhibition of Paenibacillus larvae by lactic acid bacteria isolated from fermented materials. J Invertebr Pathol 112(1):62–67. https://doi.org/10.1016/j.jip.2012.09.002
Acknowledgements
We are also grateful to Yosef Hamba and Marlin Udo for their support in the experimental setup in the laboratory and our field experts James Ng’ang’a and Joseph Kilonzo.
Funding
This research was funded by the Newton-Utafiti grant from the British Council (grant 275898413), the Swiss National Science Foundation Postdoc Mobility (grants P300PA_174329 and P2ELP3_165168) and icipe core funding provided by UK Aid from the UK Government, Swedish International Development Cooperation Agency (Sida), Swiss Agency for Development and Cooperation (SDC), Federal Democratic Republic of Ethiopia, and the Government of Kenya. The views expressed herein do not necessarily reflect the official opinion of the donors.
Author information
Authors and Affiliations
Contributions
Conceptualization, J.C.P.; investigation, M.C.; data curation, formal analysis, funding acquisition, methodology, visualization, and writing—original draft preparation M.C. and J.C.P., writing—review and editing M.C., J.K. and J.C.P., and project administration: J.C.P.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chege, M., Kinyua, J. & Paredes, J.C. Lactobacillus kunkeei impacts the health of honey bees, Apis mellifera scutellata, and protects the bees against the opportunistic pathogen Serratia marcescens. Int J Trop Insect Sci 43, 1947–1955 (2023). https://doi.org/10.1007/s42690-023-01103-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-01103-6