Abstract
Anti-oviposition induced by Cordyceps fumosorosea FG340 to Spodoptera exigua and its persistence had been assessed on the Chinese cabbage seedlings sprayed with 0, 106, 107 and 108 conidia per mL in greenhouse and laboratory. In a randomized complete block, four couples (males and females) of moths were released in each cage. In the laboratory, the percentage of egg laid varied significantly between treatments with 59.8, 14.8, 11.9, and 13.5% on 0, 106, 107 and 108 conidia per mL; respectively. Similarly, in the greenhouse, the percentage of eggs laid differed significantly between suspensions with 88.18, 8.7, 0, and 3.13% on 0, 106, 107, and 108 conidia per mL; respectively. Repellence study showed that there is a major difference between suspensions with average 34.55, 35.21 and 40.02% for 106, 107 and 108 conidia per mL; respectively. Endophytic C. fumosorosea and its culture fluid affect the behavior of armyworm larvae. The concentration of 108 conidia per mL can affect the oviposition of the armyworm.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Chinese cabbage (Brassica rapa chinensis L.) is an important commodity in South Korea (Lee et al. 2014). This crop ranked third after rice and fresh vegetables in annual production; but nowadays, its demand increases because of its usage in preparing the native dish named Kimchi (Park et al. 2014). It has been remarkably proven that this vegetable crop has therapeutic effects on many human diseases (Islam and Choi 2009; Joo et al. 2016, 2018). Its planting in Korea is done in the high elevation areas of the country. Unfortunately, this crop is subject to abiotic and biotic constraints, which include fluctuating temperature, diseases and insect pests (Shim et al. 2016; Kim et al. 2012). One of the major problems that farmers are facing is the armyworm, Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae), which is devastating the Chinese cabbage crops. The reduction in quality and quantity of yield resulting from the pest attack represent major economic losses of cabbage in South Korea (Kabir et al. 2015). Armyworm feeds on 90 plant species, belonging to 18 families (Farahani et al. 2012). They cause serious damage various crop including asparagus, soybean, beet, broccoli, cabbage and wild hosts worldwide (Farahani et al. 2012). Their life survival, fecundity, longevity and even susceptibility to insecticides can be affected by host plants (Farahani et al. 2012; Saeed et al. 2017). These aforementioned authors (Kabir et al. 2015) revealed that hatching also depends on environmental conditions, such as variation in temperature, relative humidity (RH), insolation, among others. Farmers rely often on intensive chemical control that affects non-target arthropods, contributes to insecticide resistance and disrupts the ecological balance. Moreover, chemicals are harmful to human health (Pimentel 2005; Boateng and Kusi 2008). Therefore, an alternative control method is urgently required for the well-being of human and the environment (Muntz et al. 2016).
Microbial control that includes the use of the insect pathogenic agents such as viruses, bacteria and fungi, is nowadays seen as a large scale ecological management option. It requires the understanding of the entire agroecosystem, which includes soils, plants, and associated organisms from different trophic levels (Bale et al. 2007). Endophytic Cordyceps fumosorosea (formerly Isaria fumosorosea) is a potential microbial control agent for many insect pests (Hunter et al. 2011; Hussein et al. 2016; Qasim et al. 2018). Recent studies in Korea about the pathogenicity of the entomopathogenic fungus (EPF) C. fumosorosea FG340 revealed that this fungal isolate was virulent and can be considered as a valuable control option against S. exigua (Han et al. 2014).
The main objective of this study is to elucidate the anti-ovipositional and the repellence effects after spraying the EPF.
Materials and methods
Experimental sites
Two different studies were carried out both in the laboratory and acrylic cages at the headquarters of the National Institute of Agricultural Sciences (RDA) hosted at Jeonju (N 35°49’, E 127°09’ and 53.5 m sea levels) in the South-West of the Republic of Korea with an agricultural area of 50.0 km2. Its topography varies from steep forests to plains with four seasons, as in temperate climate (Kim et al. 2008). Studies in the laboratory and screen house were conducted in January and August 2016.
Insects used in the experiment
Larvae of S. exigua used for experiments were previously reared in the Crops Protection Division, at the RDA. Larvae were nourished on artificial diet (#F9219B, mixing direction; Bio-Serv, San Diego, CA, USA), maintained at 25 ± 1 °C, 75 ± 10% RH and 14: 10 h (L: D) photoperiod (Han et al. 2014). Both male and female insects were used during the different bioassays. At hatching, insects from same generation were enclosed in same cage for mating. Moths were fed with a 10% sugar solution dropped on hydrophilic cotton to avoid starvation and dehydration. All insects for experiment were used only once to avoid stress.
Plants materials
Single potted-plants of the commercial Chinese cabbage variety referred to as “Jang Mi Baechu” (Nong Woo Bio Co, Ltd.) were sown in the greenhouse. Except the use of pesticides, all cultural practices recommended for vegetable crops were performed, accordingly.
Preparation of conidial suspensions
This EPF known as KACC93199P was isolated from soil of the agricultural field (31.2641431 N, 126.9863588 E) using insect-bait method on Galleria mellonella in Korea, and selected after bioassays because of its huge pathogenicity in controlling armyworm. For this purpose, the EPF was cultivated in an incubator (Bionex Multi-Room Incubator VS-1203PFC-L, VISION SCIENTIFIC CO; LTD) set at 25 ± 1° C, 75 ± 10% RH; and 12:12 h (L: D photoperiod) on potato dextrose agar (PDA) medium for 14 days (Qasim et al. 2021). Conidia were harvested and suspended in 0.05% Tween 80 solution made with sterilized water; and vortexed for 3 min to homogenize the mixture. Then, it was filtered through several layered of sterile cheesecloth to remove hyphae and conidial concentrations were determined using a hemocytometer (Merch KGaA; Bright-Line Model No. Z375357, Darmstadt, Germany) as described by Han et al. (Han et al. 2014).
During this study, both the “No-choice” and “Free choice” bioassays, filtrated EPF and its crude repellent activities were assessed using third, fourth, and fifth instar larvae of S. exigua. The three assessed treatments used comprised control (0.05% Tween 80 or 0), filtrate of EPF, and crude. During “No-choice” bioassay, 5 mL of each treatment was sprayed in each hole of the detached leaf. For the “Free choice” one, 2.5 mL was sprayed on each half leaf blade. Each treatment was paired with control such as filtrate vs control and crude vs control.
Other materials
The screen cage (60 × 60 × 60 cm) was used to keep the insects on the plants and to exclude the attack of other pests that would compromise the quality of results. A stereoscopic binocular microscope (Leica M60 Stereo Microscope, Max Schmidheiny Str. 201 CH-9435 Heerbrugg Switzerland) was used to visualize and record the eggs laid. The hemocytometer was used to prepare different suspensions. A core borer was used to cut leaf samples (leaf discs) in order to assess the daily conidial density. Finally, a watering can had been used to prevent the leaching and dispersing of spores.
Inoculum used on plants
Different concentrations (0, 106, 107 and 108 conidia per mL) of the EPF were sprayed on two sides of each leaf for the behavioral study of the moth. The control treatment was composed of 0.05% Tween 80 solution only.
Anti-oviposition effects of EPF
Three choice trials were carried out to show the effects of different concentrations of the EPF on the oviposition of S. exigua. Two trials were completed in the laboratory (Temp of 25 ± 1 °C; and at a RH of 95.8 ± 1%) while the last trial conducted in the greenhouse. In all the trials S. exigua oviposition was studied at 0, 106, 107, and 108 conidia per mL. Individual trial comprised four acrylic cages (length: 40 cm; width: 40 cm; height: 35 cm) which contained separately four clean potted plant. A 2-L pot (top Diam: 17.0 cm, base Diam: 12.0 com, and height: 13.0 cm) was used. The distance between consecutive plants in the same cage was 15 cm. Each cage was considered as a plot, where four plants were treated with different concentrations of EPF suspensions (0, 106, 107, and 108 conidia per mL). A volume of 15 mL was sprayed per plot. After an hour after spraying four couples of moths were released into the cage. For these two trials, per laboratory requirements, eight acrylic cages were used.
Persistence of EPF after spraying
Two bioassays were carried out under the plastic greenhouse with recorded weather patterns as follows: temperature (35.6- 25.1 °C); RH (99.7- 57.8%); light intensity (372.4- 0.4 lum/ft2); and photoperiod (12: 12 L: D) using an Onset HOBO (U12 Temp/RH/2 External Channel Logger). In the beginning, 10 plants per screen cage were individually stored. The plants were six weeks old, about 10 cm height, and grew 5 leaves. Four screen cages were used per trial, corresponding to the treatments (0, 106, 107 and 108 conidia per mL). The plants under the same screen cage were individually sprayed once with 20 ml of the suspension for evaluation.
Daily assessment consisted of moving four plants from each initial screen cage to a new cage. After that, four couples of armyworms were released on each of them and the screen cage was kept closed. The next day, the number of eggs laid by armyworm on each plant was recorded. Each trial lasted for five successive days. Every day, with a cork borer, nine leaf discs were sampled per plant (0.5 cm-diameter leaf disc was cut), from the top, middle, and bottom of each plant (three discs from each plant part), respectively. The discs were then placed in a tube that contained five milliliters of 0.05% Tween 80® solution and vortexed for 1 min to promote detachment of the adhered spores. After using hemocytometer, they were observed under a microscope to determine the density of conidia present per mm2. This data allows determining the correlations between the density of conidia per leaf and the number of eggs on the leaf surface after spraying.
No-choice and free-choice tests after applying suspensions on detached leaves
The cabbage plants were grown up in the greenhouse and assessed under controlled conditions like in the anti-oviposition study. One clean detached leaf cut from plant with scissors was immediately placed in vertical position into plastic cup (4.5 × 11.5 cm diameter), which contains moistened hydrophilic cotton, and used sterile distilled water (Fig. 1).
“No-choice test” consisted of spraying entire single leaf blade (both sides) with 5 mL of EPF suspensions at 0, 106, 107, and 108 conidia per mL; respectively. Then, the leaf was allowed to dry under a laminar flow cabinet (CHC LAB; Model No. CLB-202D, Daedeok Techno Valley, 520–1, Yongsan-dong, Yuseong-gu, Daejeon 305–500, Korea). Larvae from third, fourth, and fifth instar were placed on the leaf petiole at 4–8 cm below the treated leaf blade. The loading point was 4 cm for the assessment of the third instar larval assessment; while one of 8 cm below the leaf blade was considered during the fourth and the fifth instar larvae bioassays. Those distances where the larvae had been placed were determined after a mock assessment (authors personal communication). Single larva was assessed once to avoid any stress because of repetitive uses. The only one data recorded during those assessments was the time that each larva took to reach the leaf blade.
“Free-choice test” method was similar to that implemented during the “No-choice” test, with minor modifications. Each leaf was treated with two different suspensions; midrib represented the border between two treatments applied on the same leaf. Half of each leaf blade was separated by midrib, while each side was sprayed with 2.5 mL of different suspension combinations as follows (0 vs 106), (0 vs 107), and (0 vs 108) conidia per mL (Fig. 1b). An individual larva was placed at the center of the midrib; and its stay time on a chosen side recorded for 15 min. The 0 level or negative control comprised 0.05% Tween 80 (T.W) solution added to sterilized water; and then vortexed for 3 min. The remaining treatments of the suspensions of the EPF consisted of 106, 107, and 108 conidia per mL.
Fungal fluid culture
Mycelium from the EPF was cultivated in a 250 mL Erlenmeyer flask that contains 100 mL of PDB (Potato Dextrose Broth, BD Difco™ Dehydrated Culture Media, Catalog No. DF0549-17–9) medium incubated on a rotary shaker (Vision Scientific Co., Ltd; Model No.VS-37SIF-L, 260 Techno 2Ro, Yuseong-Gu, Daejeon_Si, Korea) settled at 150 rpm and 25 °C for 2 weeks. After two weeks, fungal fluid were centrifuged at 10,000 × g for 20 min at 4 °C, and then the precipitated cells and the supernatants were separated. The supernatants were filtered through a membrane filter paper (Advantec No. 2, Advantec, Tokyo, Japan) to separate the mycelial and spore masses, and The culture filtrate re-filtered through a 0.2 μm membrane filter(28 mm syringe filter, Corning, New York, USA).
Processing of assessed data
Data relating to the number of eggs per treatment had not been transformed but calculated to satisfy the assumption of normality. The percentage of oviposition (%OV), and the oviposition deterrence indices (ODI) recorded on each suspension of EPF are calculated by the following formulas:
where NEgg is the number of eggs laid per treatment and Tot eggs are the total eggs laid per day in all treatments.
\(\mathrm{ODI}=100(\mathrm C-\mathrm T)/(\mathrm C+\mathrm T)\), according to Abbott (1925); where C is the number of eggs in control treatment; T is the number of eggs per EPF suspension on a specific date.
Statistical analyses
Data were statically analyzed for significance using one-way ANOVA (p < 0.05). If significant, treatment post hoc means were separated using Tukey’s HSD test (p < 0.05) and Duncan’s multiple range test (p < 0.05). Statistical analyses were conducted using SAS Proc GLM procedures and executed on a SAS 9.2 version.
Data analyzed were %OV., ODI, time spent before making choices, number of EPF conidia per mm2 of the EPF against S. exigua, both in the laboratory and greenhouse. At the end, a linear regression analysis was performed to figure out the correlation between daily density of conidia on leaf surface and the concentration of suspensions.
Results
Anti-oviposition effects after spraying of EPF
Laboratory study of oviposition under different concentrations
Oviposition was inhibited by the EPF and significant variations were found among sprayed conidial concentrations (F = 8.80, df = 3, 45, p < 0.0001). Average percentage of eggs laid on leaves decreased while the concentration of the suspension increased (Fig. 2).
Greenhouse study of anti-oviposition effects at different concentrations
The %OV was inhibited by the EPF and significantly varied according to different conidial concentrations (F = 78.76, df = 3, 24, p < 0.0001). Similarly, females laid more eggs on the control plantlets than on the treated ones. Average percentage of eggs laid on leaves decreased while the concentration of the suspension increased (Fig. 3).
Persistence effects after spraying of EPF
Ovipositional deterrence by residual conidia
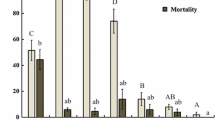
Conidia adhered to leaf surface significantly affected the oviposition of S. exigua for five days after spraying (Fig. 4). Day 1 after spraying, there was a significant difference between eggs laid on control leaves and treated ones (F = 3.99, df = 3, 31, P = 0.0175); but there was no significant discrepancy between %OV on treated plants (Fig. 4). Day 2 after spraying, data did not show any major difference between controlled, 106, 107 and 108 conidia per mL (Fig. 4). Day 3 (F = 1.16, df = 3, 31, P = 0.3430) and day 4 (F = 3.76, df = 3, 31, P = 0.0143) after spraying, data showed significant difference between %OV on controlled plants and treated ones, but there was no major variation between treated ones (Fig. 4). Day 5 after spraying, there were highly differences between %OV on controlled plants and on treated ones (F = 4.38, df = 3, 31, P = 0.0186). There was also a significant gap between %OV recorded on plants treated with 106 conidia per mL and those recorded on 107 and 108 conidia per mL. %OV did not show any crucial difference between 107 and 108 conidia per mL. At the end, the lowest %OV value was recorded on 106 conidia per mL (Fig. 4).
Conidial persistence on foliage
Conidial number on leaf surface gradually decreased as days went by after spraying. The daily number of conidia on the leaf surface varied according to the concentration. There were daily significant differences between suspensions of EPF. The number of residual conidia on the leaves considerably decreased two days after spraying. A high number of conidia was found on 108 conidia per mL (F = 102.46, df = 3, 35, P < 0.0001; Fig. 5).
Effect of treatments on average conidia per leaf surface, oviposition, and anti-oviposition
Residual conidial density on each leaf surface after a five-day assessment did not show any significant variation between 106 and 107 conidia per mL. There was a major difference between conidial density found on leaf surface sprayed with 108 conidia per mL and that found both with 106 and 107 conidia per mL. The residual conidia number increased according to the concentration of the suspensions (F = 119.39, df = 3, 35, p < 0.0001; Table 1).
Percent of egg laid did not show any significant difference between treatments on control, 106 and 107 conidia per mL for average %OV during the five days after spraying. There was a significant difference between %OV found on leaf surface treated with 108 conidia per mL and the remaining ones (control, both 106 and 107 conidia per mL). The %OV decreased as the concentration of the suspensions increased (F = 4.38, df = 3, 31, P = 0.0186; Table 1).
There was no significant difference between average ODI on 106, 107 and 108 conidia per mL during the five days of study. Results showed there was a significant difference between average ODI on controlled leaves and that recorded on those treated with 106, 107 and 108 conidia per mL. If the EPF concentration increased, the average ODI increased as well (F = 5.8, df = 3, 31, p = 0.0051; Table 1).
Linear regression studies between conidial density at leaf surface and suspensions sprayed
The figure below shows that there was a consistent relationship between concentration of EPF and persistence of conidia on leaf surface after spraying. The regression line is y = 0.0011x + 2298.9 with r2 = 0.9974 as the coefficient of determination. There were fewer applied concentration points to characterize the line as a consistent linear one for a consistent conclusion.
Larval repellence effects against EPF
Larval repellence study on detached leaves via no-choice test
Results showed that there were remarkable differences between treatments for the time took by the third instar larvae before getting in contact with the treated leaf surface (F = 4.38, df = 3, 254, p < 0.005). The third instar larvae avoided leaves treated with the EPF suspensions. As the EPF concentration increased, third instar larvae took much time to reach out the treated leaves (Fig. 6a).
Figure 6b shows that there was no major difference between EPF treatments for the time took by the fourth instar larvae before making a choice.
Figure 6c shows that there was a significant discrepancy between EPF treatments for the time took by the fifth instar larvae before making a choice (F = 4.13, df = 3, 309, p = 0.0069). There was a significant difference between 106 and 108 conidia per mL. Therefore, there was no significant difference between the controlled and the treated ones.
Larval repellence study on detached leaves via free-choice test
Results showed significant differences between the times the 3rd instar larvae stayed on the leaf sides treated with the EPF (106, 107, and 108 conidia per mL) and untreated one (F = 8.22, df = 1, 139, p = 0.0048), (F = 26.69, df = 1, 139, p = 0.0001), and (F = 35.41, df = 1, 139, p = 0.0001), respectively.
Results showed significant differences between the times the 4rd instar larvae spent on the leaf sides treated with the EPF (106, 107, and 108 conidia per mL) and untreated one (F = 10.32, df = 1, 139, p < 0.0016), (F = 11.78, df = 1, 139, p = 0.0008), and (F = 42.64, df = 1, 139, p < 0.0001), respectively.
Results showed significant differences between the times the 5rd instar larvae stayed on the leaf sides treated with the EPF at each of the following concentrations 106, 107, and 108 conidia per mL and untreated ones (F = 8.24, df = 1, 139, p = 0.0047), (F = 5.96, df = 1, 139, p < 0.0159), and (F = 25.11, df = 1, 139, p < 0.0001), respectively.
Larvae spent much time on controlled sides than on treated ones. Larvae avoided leaves treated with high suspension concentrations (Table 2).
The Comparisons of stay time on treated leaves showed that the 4th instar larvae spent the lowest time, while the 5th and the 3rd instar larvae spent the moderate and highest time, respectively (Table 2).
There was a remarkable difference between the time the larvae spent to reach the leaf blade treated with crude and the time spent to reach controlled ones (F = 14.42, df = 1, 89, p = 0.0003). Many larvae were attracted by the controlled leaf blade. There is no major difference between time spent by larvae on leaf blades treated with filtrated and controlled ones (Table 2).
No-choice tests using the fungal filtrate and its crude
There was no significant gap between amount of time spent by larvae to reach leaf treated with the culture fluid and the times took to go on controlled and culture filtrated ones. There was no significant difference between the time took by larvae to reach controlled and culture filtrated one. However, it took them too much time to reach leaves treated with culture fluid than controlled and culture filtrated ones (Fig. 7).
Discussion
The EPF showed ability to interfere with the normal development of the insect as it can inhibit the eggs-laying behavior. This statement is supported by different experiments under laboratory and greenhouse conditions and it confirmed anti-oviposition and the repellence effects of EPF. Our findings confirmed early results of both Lacey and Neven 2006 and Martinuz et al. 2012, which revealed that endophytic fungi can be a good defense agent to reduce insect oviposition (Lacey and Neven 2006; Martinuz et al. 2012). Other researchers found that EPF Beauveria bassiana has caused considerable deterrence to Ceratitis capitata, in terms of reduction in the number of female visits per fruit; and of oviposition punctures per fruit, on treated plants than on untreated ones (Falchi et al. 2015). That event could be accounted for in anti-ovipositional behavior. Certain biochemical (antixenosis) properties of conidia of EPF may impair the ability of S. exigua to detect cues, which dramatically reduced its oviposition ability. Hussain et al. (2010) found out that behavioral responses of workers Coptotermes formosanus (termites) were affected by entomopathogenic fungal volatiles using an electroantennograph, but the major proportion of EPF profile comprised ramification of cyclic alkanes (84.41%). Many scientific theories have highlighted the vital role of fungal volatiles in mediation of fungal–insect and plant interactions (Halls et al. 1994; Wang et al. 2020). For Pagans et al. (2006), volatile organic compounds (VOCs) started out as solids and liquids, then changed into gas phase by vaporizing at 20 °C and 0.01 kpa. They are intermediate or end products of metabolisms and belong to many classes such as terpenoids, phenols, heterosides, alkaloids, mucilages and reducing sugars (Bocco et al. 2017a); and can be found in endophytic fungi (Tan and Zou 2001). They produce a range of volatiles to interact with their environment and insects (Yanagawa et al. 2009; Müller et al. 2013). Secondary metabolites are not essential for normal plant growth, but they play key roles in chemical defense against insects by killing, repelling, inhibiting feeding, or inhibiting insect growth (Zhu and Gu 2000). Secondary metabolites also have important effects on host selection by insects (Wang and Qin 2007). Steroids can regulate plant growth and development (Fang et al. 2003; Fujioka and Yokota 2003; Fujioka et al. 2006) but they can also affect the choice of plant hosts by insects (Behmer and Elias 2000; Yang et al. 2001). Some specific sterols are involved in the process of hormone synthesis; and are specific components of the insect cell membranes (Hannich et al. 2011). Insects on their side have adapted themselves and developed detoxification enzymes to overcome the system of chemical defense of plants (Peng et al. 2010). These volatile organic compounds do not appear to be the only ones responsible for the behavior of the insects when facing the pathogenic fungus.
Our study showed that female insects laid more eggs on the control plants than on treated ones. EPF application heavily reduced the oviposition under different conidial concentrations. Daily average percentage of eggs laid on the surface of leaves were affected by conidia (Fig. 4). Early findings revealed that egg laying is also regulated by the physical and chemical information received by the tarsi and ovipositors (Maher et al. 2006; Calas et al. 2006). The process of egg-laying in the female insects, as well as the number of eggs per leaf depend on several factors such as contact-based cues, visual cues, stiffness and the texture of the substrate, substrate preference, among others (Karageorgi et al. 2017; Lerner et al. 2008; Becher et al. 2012; Sumethasorn and Turner 2016). There is selection deterrence in insects because some larvae are predators or cannibals and may kill each other when closer to each other (Labrie 2007; Bocco et al. 2017b). The process to optimize plant–insect interaction during host selection by insects for progenies is a complex mechanism.
Based on the linear regression model, the ideal suspension for microbiological control under our experimental conditions is 108 conidia per mL. This concentration exhibited a high value of persisting conidia/mm2 several days after spraying and was exactly on the regression line, unlike the other suspensions. Observations in Fig. 8 showed that suspension at 108 conidia per mL recorded the lowest percentage of oviposition for three days. These results confirm those of Allegrucci et al. (Allegrucci et al. 2017) on Beauveria bassiana using different inoculation techniques; and their efficacy on the tomato leaf miner (Tuta absoluta). The aforementioned researchers revealed that the suspension at 108 conidia per mL in 0.01% (v/v) Tween 80 is effective in controlling the pest than other suspensions. Other studies showed that the efficiency of the suspensions to be sprayed varied according to insect species (Bugti et al. 2017). Rahim et al. (2013) mentioned that the whitefly (Bemisia tabaci) was infected at all stages of development with a concentration of EPF at 106 conidia per mL. The same concentration of 106 conidia per mL was lower compared to those between 108 and 1010 conidia per mL, which are often used in commercial formulations and relates to real-life applicability or applications (Soliman et al. 2019; Chao et al. 2020).
Results from our study show the importance of EPF in the mechanism of anti-oviposition; and the choice of host plants by S. exigua. Female insects chose the non-treated plants, rather than treated ones. The number of eggs laid on each plant decreased when the EPF concentrations increased. The larvae also exhibited different behaviors when exposed to treated leaves (Table 2). Results from free choice and no-choice tests on detached leaves confirmed that larvae spent fewer times on treated leaves than on controlled ones. Under no-choice and free choice tests, fungal filtrate and its crude on leaf blade showed that crude has a deterrent, repellent, and anti-feeding action on larvae. The discovery of these repellent properties in the EPF offers an opportunity for further research, especially on development of fumigants and repellents to protect agricultural commodities. EPF suspension at 108 conidia per mL was more efficient than any other ones. Concentration of 108 conidia per mL can be recommended to farmers in a field control against S. exigua. In sight of the results on fungal repellence and anti-oviposition activities via its suspensions and metabolic crude, we encourage pest managers to introduce EPF isolate in a biological control program after completing the field assessment.
Data availability
Not applicable.
References
Abbott WS (1925) A Method of computing the effectiveness of an insecticide. J Econ Entomol 18:265–267
Allegrucci N, Velazquez MS, Russo ML, Perez E, Scorsetti AC (2017) Endophytic colonisation of tomato by the entomopathogenic fungus Beauveria bassiana: the use of different inoculation techniques and their effects on the tomato leafminer Tuta absoluta (Lepidoptera: Gelechiidae). J Plant Prot Res 57(4):2015–2211
Bale JS, van Lenteren JC, Bigler F (2007) Biological control and sustainable food production. Philos Trans R Soc Lond B Biol Sci 363:761–776
Becher PG, Flick G, Rozpędowska E, Schmidt A, Hagman A, Lebreton S, Larsson MC, Hansson BS, Piškur J, Witzgall P, Bengtsson M (2012) Yeast, not fruit volatiles mediate Drosophila melanogaster attraction, oviposition and development. Funct Ecol 26:822–828
Behmer ST, Elias DO (2000) Sterol metabolic constraints as a factor contributing to the maintenance of diet mixing in grasshoppers (Orthoptera: Acrididae). Physiol Biochem Zool 73:219–230
Boateng BA, Kusi F (2008) Toxicity of jathropha seed oil to Callosobruchus maculatus (Coleoptera: Bruchidae) and its parasitoid, Dinarmus basalis (Himenoptera: Pteromalidae). J Appl Sci Res 4:945–951
Bocco R, Gandonou CB, Dannon AE, Zanklan AS (2017) Diopsids (Diopsis thoracica and D. apicalis) damaging rice production in Africa: A review. Int J Curr Res Biosci Plant Biol 4:33–41
Bocco R, Gandonou CB, Gbaguidi F, Ahouansou AC (2017a) Phytochemical screening and quantitative variation of some secondary metabolites in five cultivated rice. J Appl Biosci 113:11146–11157
Bugti GA, Na C, Bin W, Feng LH (2017) Control of plant sap-sucking insects using entomopathogenic fungi Isaria fumosorosea strain (Ifu13a). Plant Protect Sci 52:7
Calas D, Thiery D, Marion-Poll F (2006) 20-Hydroxyecdysone deters oviposition and larval feeding in the European grapevine moth, Lobesia botrana. J Chem Ecol 32:2443–2454
Chao Y, Wang M, Dai W, Dong F, Wang X, Zhang F (2020) Synergism between hydramethylnon and Metarhizium anisopliae and their influence on the gut microbiome of Blattella germanica (L.). Insects 11:538. https://doi.org/10.3390/insects11080538
Falchi G, Giovanni M, Maria G, Ruiu L (2015) Hydrophobins from aerial conidia of Beauveria bassiana interfere with Ceratitis capitata oviposition behavior. Biol Control 81:37–43
Fang N, Yu S, Badger TM (2003) Characterization of triterpene alcohol and sterol ferulates in rice bran using LC-MS/MS. J Agric Food Chem 51:2239–2243
Farahani S, Talebi Asghar A, Fathipour Y (2012) Life table of Spodoptera exigua (Lepidoptera: Noctuidae) on five soybean cultivars. Hindawi, Publishing Corporation Psyche Volume, Article ID 513824, 7 pages https://doi.org/10.1155/2012/513824
Fujioka S, Ohnishi T, Watanabe B (2006) Arabidopsis CYP90B1 catalyses the early C-22 hydroxylation of C27, C28 and C29 sterols. Plant J 45:765–774
Fujioka S, Yokota T (2003) Biosynthesis and metabolism of brassinosteroids. Annu Rev Plant Biol 54:137–164
Halls SC, Gang DR, Weber DJ (1994) Seasonal variation in volatile secondary compounds of Chrysothamnus nauseosus (Pallas) britt.; Asteraceae ssp. Hololeucus (Gray) hall. & clem. influences herbivory. J Chem Ecol 20:2055–2063
Han JH, Jin BR, Kim JJ, Lee SY (2014) Virulence of entomopathogenic fungi Metarhizium anisopliae and Paecilomyces fumusorosea for the microbial control of Spodoptera exigua. Microbiol 42:385–390
Hannich JT, Umebayashi K, Riezman H (2011) Distribution and functions of sterols and sphingolipids. Cold Spring Harb Perspect Biol 3:a004762
Hunter WB, Avery PB, Pick D, Powell CA (2011) Broad spectrum potential of Isaria fumosorosea against insect pests of citrus. Fla Entomolog 94:1051–1054
Hussain A, Tian M-Y, He Y-R, Bland J, Gu W-X (2010) Behavioral and electrophysiological responses of Coptotermes formosanus Shiraki towards entomopathogenic fungal volatiles. Biol Control 55:166–173
Hussein HM, Habuštová OS, Puza V, Zemek R (2016) Laboratory evaluation of Isaria fumosorosea CCM8367 and Steinernema feltiaev Ustinov against immature stages of the Coloraldo potato beetle. PLoS ONE 11:e0152399
Islam MS, Choi H (2009) Antidiabetic effect of Korean traditional baechu (Chinese cabbage) kimchi in a type 2 diabetes model of rats. J Med Food 12:292–297
Joo HK, Choi S, Lee YR, Lee EO, Park MS, Lim YP, Park JT, Jeon BH (2016) Ethanol extract of Brassica rapa spp. pekinensis suppresses tumor necrosis factor-α-induced inflammatory response in human umbilical vein endothelial cells. J Med Food 20:511–518
Joo HK, Choi S, Lee YR, Lee EO, Park MS, Park KB, Kim C-S, Lim YP, Park J-T, Jeon BH (2018) Anthocyanin-rich extract from red Chinese cabbage alleviates vascular inflammation in endothelial cells and Apo E-/- mice. Int J Mol Sci 19:816
Kabir MF, Shin JH, Kweon OG, Lee DW (2015) Temperature and root extract effect on hatching and development of Heterodera schachtii. Korean J Soil Zool 19:22–27
Karageorgi M, Bracker LB, Lebreton S, Minervino C, Cavey M, Siju KP, GrunwaldKadow IC, Gompel N, Prud’homme B (2017) Evolutionof multiple sensory systems drives novel egg-laying behavior in the fruit pest Drosophila suzukii. Curr Biol 27:847–853
Kim M-K, Jung K-H, Yun S-G, Kim C-S (2008) Region-scaled soil erosion assessment using USLE and WEPP in Korea. Korean J Environ Agric 27:314–320
Kim MJ, Shim CK, Kim YK, Hong SJ, Park JH, Han EJ, Jee HJ (2012) Screening of resistance cultivar to club root caused by Plasmodiophora brassicae for organic cultivation of Chinese cabbage. Res Plant Dis 18:123–128
Labrie G (2007) Les mécanismes d’invasion de la coccinelle asiatique Harmonia axyridid Pallas au Québec. Université du Québec à Montréal, Thèse de Doctorat en Biologie, p 256
Lacey LA, Neven LG (2006) The potential of the fungus, Muscodor albus, as a microbial control agent of potato tuber moth (Lepidoptera: Gelechiidae) in stored potatoes. J Invertebr Pathol 91:195–198. https://doi.org/10.1016/j.jip.2006.01.002
Lee M-H, Lim CJ, Lee IH, Song J-H (2014) High-purity seed production of doubled-haploid chinese cabbage [Brassica rapa L. ssp. Pekinensis (Lour.)] through microspore culture. Plant Breed Biotech 2:167–175
Lerner A, Meltser N, Sapir N, Erlick C, Shashar N, Broza M (2008) Reflected polarization guides chironomid females to oviposition sites. J Exp Biol 211(3536):3543
Maher K, Steefel CI, DePaolo DJ, Viani BE (2006) The mineral dissolution rate conundrum: Insights from reactive transport modeling of U isotopes and pore fluid chemistry in marine sediments. Geochim Cosmochim Acta 70:337–363
Martinuz A, Schouten A, Sikora R (2012) Systemically induced resistance and microbial competitive exclusion: implications on biological control. Phytopathol 102:260–266. https://doi.org/10.1094/PHYTO-04-11-0120
Müller A, Faubert P, Hangen M, zu Castel W, Polle A, Schnitzler JP, Rosenkranz M (2013) Volatile profiles of fungi- chemotyping of species and ecological functions. Fungal Genet Biol 54:25–33
Muntz H, Miller R, Alston D (2016) Alternative pest control methods for homeowners. all agricultural extension, UtahState University, AG/Pesticides/2016–02pr
Pagans E, Font X, Sánchez A (2006) Emission of volatile organic compounds from composting of different solid wastes: abatement by biofiltration. J Hazard Mater 131:179–186
Park KY, Jeong JK, Lee YE, Daily JW III (2014) Health benefits of kimchi (Korean fermented vegetables) as a probiotic food. J Med Food 17:6–20
Peng XL, Xu WT, Wang Y, Huang KL, Liang ZH, Zhao WW, Luo YB (2010) Mycotoxin ochratoxin A-induced cell death and changes in oxidative metabolism of Arabidopsis thaliana. Plant Cell Rep 29:153–161
Pimentel D (2005) Environmental and economic costs of the application of pesticides primarily in the United States. Environ Dev Sustain 7:229–252
Qasim M, Lin Y, Dash CK, Bamisile BS, Ravindra K, Islam SU, Ali H, Wang F, Wang L (2018) Temperature-dependent development of Asian citrus psyllid on various hosts, and mortality by two strains of Isaria. Microb Pathogen 119:109–118. https://doi.org/10.2016/j.micpath.2018.04.019
Qasim M, Xiao H, He K, Omar MAA, Hussain D, Noman A, Rizwan M, Khan KA, Al-zoubi OM, Alharbi SA, Wang L, Li F (2021) Host-pathogen interaction between Asian citrus psyllid and entomopathogenic fungus (Cordyceps fumosorosea) is regulated by modulations in gene expression, enzymatic activity and HLB-bacterial population of the host. Comp Biochem Physiol C 248:109–112. https://doi.org/10.2016/j.cbpc.2021.109112
Rahim E, Ahmad SS, Dzolkhifli O, Nur AA (2013) First record of Isaria fumosorosea (Deuteromycotina: Hyphomycetes) infecting Bemisia tabacci (Gennadius) (Hemiptera: Aleyrodidae) in Malaysia. J Entomol 10:182–219
Saeed Q, Saeed S, Ahmad F (2017) Switching among natal and auxiliary hosts increases vulnerability of Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae) to insecticides. Ecol Evol 7:2725–2734. Published 19 Mar 2017. https://doi.org/10.1002/ece3.2908
Shim J-Y, Kim D-G, Park J-T, Kandpal LM, Hong S-J, Cho B-K, Lee W-H (2016) Physiochemical quality changes in Chinese cabbage with storage period and temperature: Review. J Biosyst Eng 41:373–388
Soliman EP, Castro BM, Wilcken CF, Firmino AC, Pogetto MH, Barbosa LR, Zanuncio JC (2019) Susceptibility of Thaumastocoris peregrinus (Hemiptera: Thaumastocoridae), a eucalyptus pest, to entomopathogenic fungi. Sci Agric 76:255–260
Sumethasorn M, Turner TL (2016) Oviposition preferences for ethanol depend on spatial arrangement and differ dramatically among closely related Drosophila species. Biol Open 5:1642–1647
Tan RX, Zou WX (2001) Endophytes: a rich source of functional metabolites. Nat Prod Rep 18:448–459
Wang CZ, Qin JD (2007) Insect-plant co-evolution: multitrophic interactions concerning Helicoverpa species. Chin Bull Entomol 44:311–319
Wang F, Cale JA, Hussain A, Erbilgin N (2020) Exposure to fungal volatiles can influence volatile emissions from other ophiostomatoid fungi. Front Microbiol 11:567462. https://doi.org/10.3389/fmicb.2020.567462
Yanagawa A, Yokohari F, Shimizu S (2009) The role of antennae in removing entomopathogenic fungi from cuticle of the termite, Coptotermes formosanus. J Insect Sci 9:6
Yang B, Karlsson RM, Oksman PH, Kallio HP (2001) Phytosterols in sea buckthorn (Hippophae rhamnoides L.) berries: identification and effects of different origins and harvesting time. J Agric Food Chem 44:3260–3267
Zhu L, Gu D (2000) The adaptive strategies of insects to plant alleochemicals. Chin J Ecol 19:36–45
Acknowledgements
We acknowledge the National Academy of Agricultural Science and the Rural Development Administration (RDA) of the Republic of Korea which funded Postdoctoral Fellowship Program that hired R.B. in 2019.
Funding
This research was funded by National Academy of Agricultural Science and the Rural Development Administration (RDA) of the Republic of Korea, research grant number PJ0125801 and The APC was funded by the RDA.
Author information
Authors and Affiliations
Contributions
Conceptualization, R.B., H.Y.J. and J.H.H.; methodology, R.B. and J.H.H.; software, J.H.H., S-Y.L.; validation, D.K. and S.A.; formal analysis, R.B. and J.H.H.; investigation, R.B. and H.Y.J.; resources, J.H.H.; data curation, R.B.; writing original draft preparation, R.B.; writing review and editing, R.B. and J.H.H.; visualization, J.H.H.; S-Y.L.; supervision, J.H.H.; project administration, J.H.H.; funding acquisition, J.H.H. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Institutional review board statement
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bocco, R., Jeong, H.J., Han, J.H. et al. Anti-oviposition and repellence of Cordyceps fumosorosea against Spodoptera exigua. Int J Trop Insect Sci 43, 1059–1069 (2023). https://doi.org/10.1007/s42690-023-01013-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-01013-7