Abstract
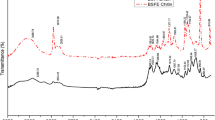
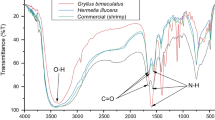
Insect chitin was isolated from different life stages of the black soldier fly Hermetia illucens, such as instar stages, prepupae and pupae, reared separately on fruit and vegetable waste substrates after removal of fat, protein and minerals. Chitin yield was high in prepupae fed with vegetable waste (11.78 ± 0.13%) followed by fruit waste (6.82 ± 0.36%). The extracted chitin was compared with a commercial chitin from shrimp by Fourier-transform infrared spectroscopy, X-ray diffraction, scanning electron microscopy and thermogravimetric analysis. The results revealed that both chitins from commercial shrimp source and the H. illucens had similar chemical structures and physicochemical properties indicating the H. illucens chitin samples to be of α- chitin orientation type. With regards to the H. illucens samples, small physiochemical changes were noticed. Furthermore, the polymers derived from BSF are equivalent to commercial polymers in terms of purity and structural morphology, indicating their utility for industrial and medical applications. Thus, H. illucens prepupae is a promising alternative source of chitin.
Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this publication will be made available without restriction to any competent researcher by the authors.
References
Al Sagheer FA, Al-Sughayer MA, Muslim S, Elsabee MZ (2009) Extraction and characterization of chitin and chitosan from marine sources in Arabian Gulf. Carbohydr Polym 77(2):410–419. https://doi.org/10.1016/j.carbpol.2009.01.032
Aranaz I, Acosta N, Civera C, Elorza B, Mingo J, Castro C, Gandía MD, Heras Caballero A (2018) Cosmetics and cosmeceutical applications of chitin, chitosan and their derivatives. Polymers 10(2):213. https://doi.org/10.3390/polym10020213
Battampara P, Sathish TN, Reddy R, Guna V, Nagananda GS, Reddy N, Ramesha BS, Maharaddi VH, Rao AP, Ravikumar HN, Biradar A (2020) Properties of chitin and chitosan extracted from silkworm pupae and egg shells. Int J Biol Macromol 161:1296–1304. https://doi.org/10.1016/j.ijbiomac.2020.07.161
Brusca RC (2009) Unraveling the History of Arthropod Biodiversification. Ann Mo Bot Gard 87(1):13–25
Casadidio C, Peregrina DV, Gigliobianco MR, Deng S, Censi R, Di Martino P (2019) Mar Drugs 17(6):369. https://doi.org/10.3390/md17060369
da Silva Lucas AJ, Oreste EQ, Costa HL, López HM, Saad CD, Prentice C (2021) Extraction, physicochemical characterization, and morphological properties of chitin and chitosan from cuticles of edible insects. Food Chem 343:128550. https://doi.org/10.1016/j.foodchem.2020.128550
Feás X, Vázquez-Tato MP, Seijas JA, Nikalje APG, Fraga-López F (2020) Extraction and physicochemical characterization of chitin derived from the Asian hornet Vespa velutina. Molecules 25(2):384. https://doi.org/10.3390/molecules25020384
Hahn T, Tafi E, Paul A, Salvia R, Falabella P, Zibek S (2020) Current state of chitin purification and chitosan production from insects. J Chem Tech Biotech 95(11):2775–2795. https://doi.org/10.1002/jctb.6533
Hamed N, Al K, Ali H, Al S, Amzad M, Abdullah M, Sibani A (2020) Optimization of the demineralization process for the extraction of chitin from Omani Portunidae segnis. Biochem Biophys Rep 23:100779. https://doi.org/10.1016/j.bbrep.2020.100779
Ibitoye EB, Lokman IH, Hezmee MNM, Goh YM, Zuki ABZ, Jimoh AA (2018) Extraction and physicochemical characterization of chitin and chitosan isolated from house cricket. Biomed Mater 13(2):025009. https://doi.org/10.1088/1748-605X/aa9dde
Jang MK, Kong BG, Yl J, Lee CH, Nah JW (2004) Physicochemical characterization of α-chitin, β-chitin, and γ-chitin separated from natural resources. J Polym Sci Part A Polym Chem 42(14):3423–3432. https://doi.org/10.1002/pola.20176
Kabalak M, Aracagök D, Torun M (2020) Extraction, characterization and comparison of chitins from large bodied four Coleoptera and rthoptera species. Int J Biol Macromol 145:402–409. https://doi.org/10.1016/j.ijbiomac.2019.12.194
Kamal M, Adly E, Alharbi SA, Khaled AS, Rady MH, Ibrahim NA (2020) Exploring simplified methods for insect chitin extraction and application as a potential alternative bioethanol resource. InSects 11(11):788. https://doi.org/10.3390/insects11110788
Kaya M, Baran T (2015) Description of a new surface morphology for chitin extracted from wings of cockroach (Periplaneta americana). Int J Biol Macromol 75:7–12. https://doi.org/10.1016/j.ijbiomac.2015.01.015
Kaya M, Baran T, Erdoğan S, Menteş A, Özüsağlam MA, Çakmak YS (2014a) Physicochemical comparison of chitin and chitosan obtained from larvae and adult Colorado potato beetle (Leptinotarsa decemlineata). Mater Sci Eng C 45:72–81. https://doi.org/10.1016/j.msec.2014.09.004
Kaya M, Karaarslan M, Baran T, Can E, Ekemen G, Bitim B, Duman F (2014b) The quick extraction of chitin from an epizoic crustacean species (Chelonibia patula). Nat Prod Res 28(23):2186–2190. https://doi.org/10.1080/14786419.2014.927469
Kaya M, Lelešius E, Nagrockaite R, Sargin I, Arslan G, Mol A, Baran T, Can E, Bitim B (2015) Differentiations of chitin content and surface morphologies of chitins extracted from male and female grasshopper species. PLoS ONE 10(1):e0115531. https://doi.org/10.1371/journal.pone.0115531
Kaya M, Seyyar O, Baran T, Erdoǧan S, Kar MA (2014c) A physicochemical characterization of fully acetylated chitin structure isolated from two spider species: With new surface morphology. Int J Biol Macromol 65:553–558. https://doi.org/10.1016/j.ijbiomac.2014.02.010
Kaya M, Sofi K, Sargin I, Mujtaba M (2016) Changes in physicochemical properties of chitin at developmental stages (larvae, pupa and adult) of Vespa crabro (wasp). Carbohydr Polym 145:64–70. https://doi.org/10.1016/j.carbpol.2016.03.010
Kim MW, Han YS, Jo YH, Choi MH, Kang SH, Kim SA, Jung WJ (2016) Extraction of chitin and chitosan from housefly, Musca domestica, pupa shells. Entomol Res 46(5):324–328. https://doi.org/10.1111/1748-5967.12175
King CA, Shamshina JL, Zavgorodnya O, Cutfield T, Block LE, Rogers RD (2017) Porous chitin microbeads for more sustainable cosmetics. ACS Sustain Chem Eng 5(12):11660-11667. https://doi.org/10.1021/acssuschemeng.7b03053
Lagat MK, Were S, Ndwigah F, Kemboi VJ, Kipkoech C, Tanga CM (2021) Antimicrobial activity of chemically and biologically treated chitosan prepared from black soldier fly (Hermetia illucens) pupal shell waste. Microorganisms 9(12):2417. https://doi.org/10.3390/microorganisms9122417
Liu S, Sun J, Yu L, Zhang C, Bi J, Zhu F, Qu M, Jiang C, Yang Q (2012) Extraction and characterization of chitin from the beetle Holotrichia parallela motschulsky. Molecules 17(4):4604–4611. https://doi.org/10.3390/molecules17044604
Liu X, Zhang J, Zhu KY (2019) Chitin in arthropods: Biosynthesis, modification, and metabolism. Targeting Chitin-Containing Organisms 1142:169–207. https://doi.org/10.1007/978-981-13-7318-3_9
Majtán J, Bíliková K, Markovič O, Gróf J, Kogan G, Šimúth J (2007) Isolation and characterization of chitin from bumblebee (Bombus terrestris). Int J Biol Macromol 40(3):237–241. https://doi.org/10.1016/j.ijbiomac.2006.07.010
Manditsera FA, Luning PA, Fogliano V, Lakemond CM (2019) The contribution of wild harvested edible insects (Eulepida mashona and Henicus whellani) to nutrition security in Zimbabwe. J Food Compos Anal 75:17–25. https://doi.org/10.1016/j.jfca.2018.09.013
Marei NH, Abd El-Samie E, Salah T, Saad GR, Elwahy AH (2015) Isolation and characterization of chitosan from different local insects in Egypt. Int J Biol Macromol 82:871–877. https://doi.org/10.1016/j.ijbiomac.2015.10.024
Merzendorfer H, Zimoch L (2003) Chitin metabolism in insects: Structure, function and regulation of chitin synthases and chitinases. J Exp Biol 206(24):4393–4412. https://doi.org/10.1242/jeb.00709
Mirwandhono E, Nasution MIA, Yunilas, (2022) Extraction of chitin and chitosan black soldier fly (Hermetia illucens) prepupa phase on characterization and yield. IOP Conf Ser Earth Environ Sci 1114(1):012019. https://doi.org/10.1088/1755-1315/1114/1/012019
Moussian B (2019) Chitin: Structure, chemistry and biology. Targeting Chitin-Containing Organisms 1142:5–18. https://doi.org/10.1007/978-981-13-7318-3_2
No HK, Meyers SP (2000) Application of chitosan for treatment of wastewaters. Reviews of Environmental Contamination and Toxicology: Continuation of Residue Reviews. 2000:1–27. https://doi.org/10.1007/978-1-4757-6429-1_1
Paulino AT, Simionato JI, Garcia JC, Nozaki J (2006) Characterization of chitosan and chitin produced from silkworm crysalides. Carbohydr Polym 64(1):98–103. https://doi.org/10.1016/j.carbpol.2005.10.032
Psarianos M, Dimopoulos G, Ojha S, Cavini ACM, Bußler S, Taoukis P, Schlüter OK (2022) Effect of pulsed electric fields on cricket (Acheta domesticus) flour: Extraction yield (protein, fat and chitin) and techno-functional properties. Innov Food Sci Emerg Technol 76:102908. https://doi.org/10.1016/j.ifset.2021.102908
Purkayastha D, Sarkar S (2020) Physicochemical structure analysis of shitin extracted from pupa exuviae and dead imago of wild black soldier sly (Hermetia illucens). J Polym Environ 28(2):445–457. https://doi.org/10.1007/s10924-019-01620-x
Raghu HS, Raghavendra SN, Achur RN (2018) Isolation and characterization of chitin from Millipede (Spirobolida). J Basic Appl Zool 79(30):1–4. https://doi.org/10.1186/s41936-018-0043-5
Ramos A, Mahowald A, Jacobs-Lorena M (1994) Peritrophic matrix of the black fly Simulium vittatum: Formation, structure, and analysis of its protein components. J Exp Zool A Ecol Integr Physiol 268(4):269–281. https://doi.org/10.1002/jez.1402680403
Rudall KM, Kenchington WJ (1973) The chitin system. Biol Rev 48(4):597–633. https://doi.org/10.1111/j.1469-185x.1973.tb01570.x
Sajomsang W, Gonil P (2010) Preparation and characterization of α-chitin from cicada sloughs. Mater Sci Eng C 30(3):357–363. https://doi.org/10.1016/j.msec.2009.11.014
Sharbidre A, Sargar S, Gogoi H, Patil R (2021) Characterization of chitin content extracted from edible insect, Coridius nepalensis (Westwood, 1837)(Hemiptera: Dinidoridae). Int J Trop Insect Sci 41(2):1893–1900. https://doi.org/10.1007/s42690-020-00386-3
Shin CS, Kim DY, Shin WS (2019) Characterization of chitosan extracted from Mealworm beetle (Tenebrio molitor, Zophobas morio) and Rhinoceros beetle (Allomyrina dichotoma) and their antibacterial activities. Int J Biol Macromol 125:72–77. https://doi.org/10.1016/j.ijbiomac.2018.11.242
Soetemans L, Uyttebroek M, Bastiaens L (2020) Characteristics of chitin extracted from black soldier fly in different life stages. Int J Biol Macromol 165: 3206–3214. https://doi.org/10.1016/j.ijbiomac.2020.11.041
Son YJ, Hwang IK, Nho CW, Kim SM, Kim SH (2021) Determination of carbohydrate composition in mealworm (Tenebrio molitor L.) larvae and characterization of mealworm chitin and chitosan. Foods 10(3):640. https://doi.org/10.3390/foods10030640
Song YS, Kim MW, Moon C, Seo DJ, Han YS, Jo YH, Noh MY, Park YK, Kim SA, Kim YW, Jung WJ (2018) Extraction of chitin and chitosan from larval exuvium and whole body of edible mealworm Tenebrio molitor. Entomol Res 48(3):227–233. https://doi.org/10.1111/1748-5967.12304
Soon CY, Tee YB, Tan CH, Rosnita AT, Khalina A (2018) Extraction and physicochemical characterization of chitin and chitosan from Zophobas morio larvae in varying sodium hydroxide concentration. Int J Biol Macromol 108:135–142. https://doi.org/10.1016/j.ijbiomac.2017.11.138
Tolaimate A, Desbrieres J, Rhazi M, Alagui A (2003) Contribution to the preparation of chitins and chitosans with controlled physico-chemical properties. Polymer 44(26):7939–7952. https://doi.org/10.1016/j.polymer.2003.10.025
Triunfo M, Tafi E, Guarnieri A, Salvia R, Scieuzo C, Hahn T, Zibek S, Gagliardini A, Panariello L, Coltelli MB, De Bonis A (2022) Characterization of chitin and chitosan derived from Hermetia illucens, a further step in a circular economy process. Sci Rep 12(1):1–17. https://doi.org/10.1038/s41598-022-10423-5
Triunfo M, Tafi E, Guarnieri A, Scieuzo C, Hahn T, Zibek S, Salvia R, Falabella P (2021) Insect chitin-based nanomaterials for innovative cosmetics and cosmeceuticals. Cosmetics 8(2):1–20. https://doi.org/10.3390/cosmetics8020040
Wang H, ur Rehman K, Feng W, Yang D, ur Rehman R, Cai M, Zhang J, Yu Z, Zheng L (2020) Physicochemical structure of chitin in the developing stages of black soldier fly. Int J Biol Macromol 149:901–147. https://doi.org/10.1016/j.ijbiomac.2020.01.293
Waśko A, Bulak P, Polak-Berecka M, Nowak K, Polakowski C, Bieganowski A (2016) The first report of the physicochemical structure of chitin isolated from Hermetia illucens. Int J Biol Macromol 92:316–320. https://doi.org/10.1016/j.ijbiomac.2016.07.038
Younes I, Rinaudo M (2015) Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar Drugs 13(3):1133–1174. https://doi.org/10.3390/md13031133
Złotko K, Waśko A, Kamiński DM, Budziak-Wieczorek I, Bulak P, Bieganowski A (2021) Isolation of chitin from black soldier fly (Hermetia illucens) and its usage to metal sorption. Polymers 13(5):1–16. https://doi.org/10.3390/polym13050818
Acknowledgements
The authors wish to thank the Department of Life sciences, CHRIST (Deemed to be University), for providing the facilities to carry out the research, Mr Lexu Jose, Instrumentation Engineer, for his support in XRD analysis, Mr Jyothis Devasia, lab instructor for his help in FTIR analysis, Mr Hemanth, Analyst, CIF, UASB for the support in the use of scanning electron microscope.
Funding
This research received no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rampure, S.M., Velayudhannair, K. & Marimuthu, N. Characteristics of chitin extracted from different growth phases of black soldier fly, Hermetia illucens, fed with different organic wastes. Int J Trop Insect Sci 43, 979–987 (2023). https://doi.org/10.1007/s42690-023-00997-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-00997-6