Abstract
Ecological communities of tropical regions respond to habitat configuration, being negatively affected by anthropogenic habitats. In the tropics, mountainous highland landscapes sustain ecological communities with contrasting responses to habitat transformation. This study assessed the effect of different habitats under different disturbance regimes on the dung beetle assemblage that inhabits a mountainous Andean landscape in Colombia. The habitat types surveyed were oak forest cores (low intervention), forest edges (medium intervention), and pastures (high intervention). A total of 3,810 dung beetles from ten species were collected. Dung beetle diversity was affected by habitat type, and forest cores and forest edges had higher diversity than pastures. Besides, each habitat type was characterized by a distinct dung beetle assemblage, with pastures showing the highest heterogeneity in the dung beetle assemblage. In conclusion, our study suggests that the dung beetles of a portion of Colombian Andean landscapes are sensitive to shifts of habitat quality. The higher dung beetle diversity in forested habitats indicates that pastures comprise limiting environments for their assemblages at this Andean region.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecological communities of tropical regions are negatively affected by anthropogenic habitats (e.g., Gascon et al. 1999; Pearman 2002; Tylianakis et al. 2006, 2007), which act as a filter for most species that dwell in native environments. Due to landscape transformations generated by human activities, there is a modification in the vegetation structure complexity and microclimatic conditions (Saunders et al. 1991; Magnano et al. 2015; Ezcurra 2016). Such habitat shifts affect ecological communities, especially in conserved tropical areas, which often comprise more diverse communities than disturbed sites (Tabarelli et al. 2012). Nevertheless, there are ecosystems in which anthropogenic and native habitats may sustain similar diversity levels (Gascon et al. 1999; Prevedello and Vieira 2010). In the tropics, mountainous highland landscapes sustain ecological communities with contrasting responses to habitat transformation. For example, Medina et al. (2002) and Larsen (2012) found that dung beetle assemblages from higher elevations in the Andean region may tolerate land cover changes. Conversely, other animal groups (i.e., moths and birds) are more sensitive to habitat transformation in these landscapes (Hill et al. 2006; Lloyd and Marsden 2008), presenting impoverished ecological communities. Such contrasting responses may be related to the climatic (e.g., sunlight exposure, environmental temperature) and food source requirements of the biota from such regions (Carvalho et al. 2020, 2022), which can have different proportions of eurytopic species. Thus, it is essential to understand how ecological communities responds to habitat transformation, which may present new perspectives regarding anthropogenic effects on tropical diversity.
Analysing the community structure makes it possible to understand how diversity is structured in different habitat types. Species richness, species composition, and abundance are critical parameters used to describe ecological communities (Gotelli 2001; Magurran 2004; Begon et al. 2006), being related to ecosystem functioning and environmental stability. Higher community parameters values are usually associated with more diverse and conserved environments (e.g., Kocher and Williams 2000; Glor et al. 2001; Lira et al. 2019). However, this may vary since some anthropogenic habitats comprise fauna from both conserved and disturbed areas, resulting in a high diversity not precisely related to a conserved environmental condition (e.g., Silva et al. 2008; Quintero et al. 2010; Souza et al. 2014). In addition, there are ecosystems in which disturbed habitats may sustain a high abundance of species tolerant to disturbance (‘winner species’, see McKinney and Lockwood 1999; Tabarelli et al. 2012), which could mask the effects of anthropogenic habitat transformation on native communities. Thus, it is important to consider each species’ identity (Santos et al. 2010; Davis et al. 2012; Villada-Bedoya et al. 2017), which may be related to the effects that different habitat types have on ecological communities.
Dung beetles (Coleoptera: Scarabaeinae) have been proposed as good bioindicators to analyze, assess and monitor biodiversity (Halffter and Favila 1993; Gardner et al. 2008). This group is considered important for evaluating changes produced by anthropogenic activity in natural ecosystems, responding to habitat loss and the effects of fragmentation (Spector 2006; Nichols et al. 2007; Villada-Bedoya et al. 2017; Salomão et al. 2020). Also, the dung beetles are strongly related to vertebrate feces, since such a resource constitutes the main food for its larvae and adults (Halffter and Matthews 1966; Halffter and Favila 1993). Due to their coprophagous habit, they play a significant role in nutrient recycling, soil fertilization, secondary seed dispersal, and the control of vertebrate parasites (Nichols et al. 2008). Dung beetles are a well-defined taxon that share morphological, ecological, functional, and behavioral characteristics (Halffter 1991). Worldwide, more than 250 genera with around 6,200 species are known (ScarabNet 2009; Vaz-de-Mello et al. 2011; Tarasov and Dimitrov 2016). Approximately 25% of the dung beetle species are found in South America (Halffter and Matthews 1966; Hanski and Cambefort 1991; Vaz-de-Mello 2000). In Colombia, there are approximately 300 registered species, with more than 100 species recorded in the Andean landscape of this country (Medina et al. 2001; Escobar et al. 2005; Pulido et al. 2007). Although ecological studies that use this group as a model to establish the impact of human activities are frequent, there have been few studies carried out in the highlands of the Andes (Amat et al. 1997; Escobar et al. 2005).
The natural ecosystems in Colombia suffers strong and chronic anthropogenic disturbances, which cause an impoverishment of the fauna diversity. According to the demographic dynamics of the country, especially the forested areas in the Andean region are in the process of accelerated transformation mainly by timber extraction, replacing the forested areas with agricultural and livestock systems (Etter 1993; Amat et al. 1997). Among the Andean forests, one of the most representative in Colombia is the oak forest dominated by Quercus humboldtii (Andean oak). Oak forests covered large areas in the three Cordilleras of the Colombian Andes but are currently regarded as a threatened ecosystem since the populations of Q. humboldtii have been reduced to about 42% due to logging (Cárdenas and Salinas 2007; Avella and Rangel 2016). This species is considered Least Concern (LC) According to IUCN (Gallagher 2018) and vulnerable (VU) according to the Colombian Red Book (Cárdenas and Salinas 2007), and its main fragments are found in the eastern Cordillera, where only 10% of forests is protected by state entities or civil society organizations (Cárdenas and Salinas 2007; Avella and Rangel 2016). Therefore, oak forests are an interesting model system to analyze the impacts of natural habitats and anthropogenic disturbances on ecological communities in the montane Andean region.
In the present study, we assessed the effect of different habitats (i.e., forest cores, forest edges, and pasture) on the dung beetle assemblage that inhabits a region of the Cordillera Oriental of the Colombian Andes. Since forest edges represent the ecotone between pastures and forest cores, it may be an intermediate disturbance habitat. Therefore, following the intermediate disturbance hypothesis (Connel 1978), we expect that forest edges will encompass a more diverse dung beetle assemblage than those observed in forest cores and pastures. In addition, pastures represent a barrier for many dung beetles in the Neotropics, resulting in low diversity compared to native forests (Horgan 2008; Alvarado et al. 2018; Salomão et al. 2020). Thus, we expect pastures to encompass the most impoverished beetle assemblage of our study region.
Materials and methods
Study site
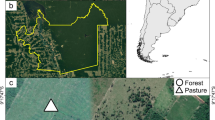
The study was carried out in a high Andean landscape located in the eastern mountain range, adjacent to the Peñas Blancas town, in the municipality of Arcabuco, department of Boyacá, Colombia (5°46’N, 73°26’W; Fig. 1), with elevations ranging between 2,483 and 2,729 m.a.s.l. The climate is classified as oceanic (Cfb) according to Köppen classification, with annual mean temperature ranges between 6 and 14.6 ºC, and the annual rainfall ranges between 600 and 1,800 mm/year. Annual rainfall presents a bimodal regime, with two precipitation periods between April-May and between October-November (IDEAM : Instituto de Hidrología, Meteorología y Estudios Ambientales 2010). The landscape is a mosaic of cattle ranching pastures and highly fragmented Andean cloud forests, restricted to the steepest portions of this landscape. The vegetation is dominated by Q. humboldtii (Andean oak), accompanied by species of Rubiaceae, Melastomataceae, Myrsinaceae, Clusiaceae, among others (Galindo 2005). The study area is part of the buffer zone of the “Corredor biológico Guántiva-La Rusia-Iguaque”, where the most important fragments of oak forests in Colombia are found. In this region, the main anthropic processes are livestock and agriculture (Cárdenas and Salinas 2007). High mammal diversity is associated with these oak forests. Nearly 55 species from 10 orders and 14 families have been recorded, where the most representative species are from the orders Chiroptera and Rodentia (Otálora 2003; Vargas-Ramirez et al. 2018). In addition, in the study area, it was possible to observe or have traces of Red Squirrel (Sciurus granatensis), White-tailed deer (Odocoileus goudotii), and common opossum (Didelphis marsupialis).
Three main habitat types in the study area (forest cores, forest edges, and pastures) were selected for this study (Fig. 1). The forest edges represent the extension of forest surrounding the main forest patch, approximately 50-meter-wide, bordering with the cattle pastures. The pastures are close to the forest edges, in which cattle-rearing activities are maintained. Pastures had different sizes (i.e., 21 ha, 36 ha, and 410 ha), had a low density of cattle (2.1, 3.6, and 0.04 cattle/ha), which were active during different periods of the experiment (P1 = during all the experiment; P2 = only during June 2007; P3 = only during March 2007, Fig. 1). The pastures were composed of exotic grasses (e.g., Paspalum sp., Axonopus sp., and Danthonia sp.) introduced and cultivated in the Andean landscapes. Pastures of this region are used for consumption by cattle in rotational systems, in which cattle consume one sector of the pasture and are moved to another sector, allowing the grass to grow. Three sampling sites were established, each comprising a forest fragment with different sizes (i.e., 15 ha, 44 ha, and 129 ha, see Fig. 1), which were ca. 1 km apart from each one. Except for one additional forest patch on an inaccessible steep slope, the three studied remnants were the only ones in an 8 km radius, limiting the number of forest remnants available for this study.
Data collection
In each sampling site, six surveys were carried out monthly from January to June 2007. In each survey, a 450 m linear transect was defined. In each transect, we installed ten baited pitfall traps (Escobar and Chacón-Ulloa 2001) distant 50 m each other (Larsen and Forsyth 2005; but see Silva and Hernández 2015). We had a total of 18 samples per habitat type (6 months * three sampling sites), totalling 54 samples in the study (following Iannuzzi et al. 2016; Ribeiro et al. 2022). Pitfall traps consisted of 500 ml plastic containers with perforations in the base to avoid flooding due to the rain. A plastic funnel was adapted to each recipient, allowing the individuals to enter the trap but reducing the probability of its exit. The traps were baited with human excrement (ca. 30 g) placed in a 25 ml plastic cup suspended with an inverted L-shaped wire. The dung beetles were collected and stored 48 h after deploying the traps for subsequent identification.
The beetles were identified at species-level using the specialized keys of Cook (2002); Génier (1996, 2009); Medina and Lopera (2000); Vaz-de-Mello et al. (2011), and corroborated by experts. The specimens were deposited in the following entomological collections: Universidad Pedagógica y Tecnológica de Colombia (UPTC-E) (Tunja, Colombia), Instituto de Investigación de Recursos Biológicos Alexander Von Humboldt (IAVH) (Villa de Leyva, Colombia), Instituto de Ciencias Naturales-Museo de Historia Natural, Universidad Nacional de Colombia (ICN-MHN) (Bogotá, Colombia), Escarabajos coprófagos de Colombia (CALT-ECC) (Bogotá, Colombia) and Seção de Entomologia da Coleção Zoológica da Universidade Federal de Mato Grosso (CEMT) (Cuiabá, Brazil).
Data analysis
To assess sampling efficiency in each habitat (i.e., forest cores, forest edges, pastures), we used the method developed by Chao and Jost (2012). Such estimations were conducted based on the number of collected individuals. Sampling efficiency was performed using iNEXT online software (Chao et al. 2016).
We compared the patterns of dung beetle diversity among each habitat using Hill numbers (Jost 2006). We used the diversity components (qD) of orders 0D (species richness, which is insensitive to species abundance and assigns high weight to rare species), 1D (exponential of Shannon entropy, which considers the relative abundance of species), and 2D (inverse of Simpson diversity, assigns a higher weight to abundant species than 0D and 1D) (Hill 1973; Jost 2006). We calculated diversity numbers by using the entire species-abundance data of the dung beetles obtained in each habitat type and compared their diversities according to their confidence intervals. We calculated diversity numbers using the iNEXT online software (Chao et al. 2016).
We performed non-metric multidimensional scaling (NMDS) for a visual and explorative evaluation of the spatial effects on dung beetle assemblage. In addition, we performed Permutation Analysis of Variance (PERMANOVA) to assess statistical differences in the dung beetle assemblages across the habitats. To analyze the heterogeneity of multivariate dispersions among habitat types, we ran the Permutational Multivariate Analysis of Dispersion (PERMDISP) with 999 permutations (Anderson 2006). Habitat type was used as a factor, and the dung beetle abundances and species composition, as the attribute. We calculated Bray-Curtis indices for the samples (n = 18 for each habitat type), and NMDS ordination was performed with 2,500 repetitions. One of the samples of pastures had no dung beetles collected, and was excluded from NMDS, PERMANOVA, and PERMDISP to avoid bias in the analyses. PERMANOVA and PERMDISP were performed using the vegan and lattice libraries, respectively, in R software version 3.2.0 (R Core Development Team 2015; Oksanen et al. 2016; Sarkar 2018; Simpson et al. 2018), and NMDS were performed using Primer software version 6.0 (Clarke and Gorley 2006).
Results
We collected 3,810 dung beetles from ten species at the forest cores, forest edges, and pastures in Arcabuco. From the collected species, Uroxys coarcatus Harold, 1867, Ontherus brevicollis Kirsch, 1870, and Canthidium sp. Were the dominant species, which together comprised 89% of the total beetles collected (Table 1). Five species (Canthon arcabuquensis (Molano and Medina 2010), Cryptocanthon foveatus Cook 2002, Dichotomius aff. satanas, Dichotomius protectus (Harold, 1867), and Eurysternus marmoreus Castelnau, 1840 were rare in this study, representing less than 1% of the total dung beetle abundance. Still, only one species, D. protectus, was a singleton. Eight species were collected at forest cores and forest edges, and only four species were recorded at pasture. The sampling coverage in the three habitats was high (forest cores –100% of the expected number of species; forest edges –99%; pastures –99%).
There were marked differences in beetle diversity regarding habitat type among forest cores, forest edges, and pastures. In total, we collected eight species (n = 1,463 beetles) in forest cores, eight (n = 1,355) in forest edges, and four (n = 992) in pastures (Table 1), and there was a statistically higher species richness in forest cores and forest edges than in pastures (Fig. 2A). Regarding the number of abundant species and the number of dominant species, there was a gradient of diversity, ranging from forest cores (highest diversity) to the pastures (lowest diversity), with forest edges being the intermediate habitat in terms of diversity (Fig. 2B, C).
Among the dung beetles collected, three species co-occurred at the three habitats. Two of them (O. brevicollis and Onthophagus curvicornis Latreille, 1811) had markedly distinct abundances in each habitat (Table 1). Two species were recorded exclusively at forest cores (C. arcabuquensis and C. foveatus), one at forest edges (D. protectus), and none at pastures. Dominant species were distinct in each habitat. At the forest cores, O. brevicollis, Canthidium sp., and Deltochilum hypponum (Buquet, 1844) were dominant, accounting for 97% of the dung beetles in this habitat. At the forest edges, U. coarcatus, O. brevicollis, and D. hypponum were the dominant ones (95% of the total beetle abundance). At pastures, U. coarcatus, O. curvicornis, and O. brevicollis were the dominant species (99% of the total abundance). The dung beetle assemblage was statistically different among the habitat types (F2 = 24.61, p < 0.01, Fig. 3). Besides, habitat types showed statistically distinct dispersion of the variance of assemblage data (PERMDISP, F2 = 3.50, p = 0.03). Pastures had a statistically higher dispersion than forest edges (p = 0.04) and had a marginally higher dispersion than forest cores (p = 0.09) (Fig. 4).
Discussion
Understanding how habitat change affects biodiversity in the tropics remains an important goal for establishing conservation strategies. The effects of habitat type have been extensively studied in ecological communities from the tropics (e.g., Tylianakis et al. 2005; Tscharntke et al. 2008; Kiatoko et al. 2017); however, such studies on dung beetles are rare in the Colombian Andes. Herein, we assessed how habitat type affects dung beetle assemblages from a region of the Andean landscape in Colombia. As expected, habitat type features as an important parameter that drives ecological communities in the tropics, even under the temperate climatic conditions of the Andean landscapes. Also following our predictions, pastures comprised the most impoverished habitat. Nonetheless, contrary to our predictions, forest edges were not the most diverse habitat; instead of that, we found a gradient forest-edge-pasture regarding dung beetle diversity.
As expected, habitat type modulated dung beetle assemblage, with a higher diversity in forest cores and forest edges than in pastures. In the Neotropics, dung beetle species differ in their habitat specificity, with some species being more generalist and some species being more specific in their distribution (Hanski and Cambefort 1991; Larsen et al. 2006; Nichols et al. 2007; Scholtz et al. 2009). Such differences in distribution patterns are possibly related to their microclimatic requirements (Larsen et al. 2006; Larsen 2012; Barretto et al. 2020), and that forest-specific species apparently cannot cross open canopy sites. Likewise, native vegetation structure is mainly related to higher levels of dung beetle diversity (e.g., Korasaki et al. 2010; Louzada et al. 2010; Filgueiras et al. 2015). Besides, the vegetation dissimilarity between disturbed and conserved habitats may negatively influence the dung beetle assemblage structure inhabiting disturbed areas (Nichols et al. 2007). The reason for this may be that dung beetle assemblages are adapted to the microclimatic conditions of native habitats.
Usually, ecological studies of dung beetle assemblages are focused on general data and statistical trends, but it is also important to consider the role of species natural and evolutive history. In this sense, we call the attention for distribution of the most abundant species recorded in this study. Among them, we could draw four distinct spatial distribution patterns: (1) there were species distributed exclusively in forested habitats (forest cores and edges) that were absent in pastures (Canthidium sp. and D. hypponum); some species occurred in all habitats, but (2) represented a gradual decrease of in the direction forest cores – forest edges – pastures (O. brevicollis), or (3) the opposite (O. curvicornis); also, (4) there was one abundant species (U. coarcatus) being recorded at pastures and forest edges but being absent in forest cores. The different patterns observed by those species can be related to the strategies presented among the species of those genera. For example, Canthidium Erichson, 1847 and Deltochilum Eschscholtz, 1822 are Neotropical genera and most of their species have specialized in forested ecosystems (Hanski and Cambefort 1991). Regarding the higher abundances of O. curvicornis and U. coarctatus in pastures compared to forest cores, the body traits and the evolutive history of these species may explain our findings. It is well known that the conversion of native forests to pastures in Neotropical region filters large-bodied species, favoring the smaller ones (Hanski and Cambefort 1991; Amat et al. 1997). Most of Uroxys Westwood, 1842 and Onthophagus Latreille, 1802 species are small bodied, which could favor their distribution in pastures compared to forest cores, explaining our findings. Besides, Onthophagus species are highly diverse in Neotropical highlands, with eurytopic species that are adapted to both closed-canopy and open-canopy ecosystems (Moctezuma 2021). The distribution of the most abundant species throughout the different habitats of this reinforces the importance of analyzing species natural history traits in order to present a finer scenario regarding species spatial distribution in the Anthropocene.
In our study, pastures comprise the most heterogeneous habitat, which was confirmed by the higher dispersion in the variances of the data when compared to forest edges. Such high dispersion may be explained by the heterogeneous distribution of dung beetle species among samples. Although there are marked differences between the dung beetle fauna from native forests when compared to anthropogenic matrices (Escobar 2004; Nichols et al. 2007; Davis et al. 2012), the environmental instability in anthropogenic habitats may explain the high variation in species distribution per sample found in pastures of the current study. Dung beetle species in the tropics may cross pastures when moving between the main habitats, which are usually the forested sites (Cultid-Medina et al. 2015; Barretto et al. 2021), resulting in more unstable and unpredictable species distribution in pastures (Salomão et al. 2020). Our results reinforce the idea that the use of pastures as secondary habitats may establish assemblages that change abruptly through space and time.
In the studied region, three of the ten species co-occurred in all studied habitats (E. marmoreus, O. brevicollis, and O. curvicornis), suggesting a certain level of species permeability among forest cores, forest edges, and pastures. As canopy height decreases and canopy openness increases with altitude in Andean vegetation (Asner et al. 2014), we suggest that the landscape conditions observed in the current study area present conditions that favor open-habitat species such as U. coarctatus. According to previous studies in the Andean region, high elevation species (as observed in this study) may be more tolerant of land-use changes than lowland species (Medina et al. 2002; Larsen 2012) suggests that the broad environmental tolerance of highland species may be related to a wide range of physiological tolerance or a broad availability of ecological niche due to the species’ poor assemblages of highlands. However, the patterns of species diversity observed herein may indicate that at 2,500 m a.s.l. the dung beetle assemblage is still composed of species with different degrees of permeability across different habitat types. This idea is supported by the dominant species of each habitat, which had similar abundances (from 800 to 958 individuals) but comprised distinct species (O. brevicollis was dominant in forest cores, and U. coarctatus was dominant in forest edges and pastures). Such a result is interesting, considering that dung beetle ecosystem services (e.g., secondary seed dispersion, dung burial, soil aeration) are related to their abundances (Larsen et al. 2005; Nichols et al. 2008). According to our results, we may suggest that the studied region of Andean highlands comprise impoverished dung beetle assemblages at human-made habitats.
Ten species were recorded in this study, and three of them (U. coarcatus, O. brevicollis, and Canthidium sp.) represented almost 90% of the collected beetles. The Andean landscape of Colombia comprises a considerable rich dung beetle diversity, with more than 100 species dwelling in this mountain range (Escobar et al. 2005). It is important to note that altitude is one of the critical factors that determine dung beetle assemblages in the Andes (Escobar 2000; Escobar et al. 2005; Larsen 2012) and in other mountain ranges of the Neotropical region (Alvarado et al. 2014; Domínguez et al. 2015; Nunes et al. 2016). According to Escobar et al. (2005), the low species richness in Andean highlands is due to the adaptation of dung beetles from this region to warm temperatures; therefore, the highlands of this region represent a barrier for most dung beetle species. Compared to other studies in the Andean mountains, we observed a relatively low species richness of dung beetles (e.g., s = 22 in Medina et al. (2002); s = 20 in Escobar (2004); s = 52 in Horgan (2005)). However, when comparing dung beetle diversity from similar altitudinal ranges of the Andes, dung beetle species richness was close to our findings (e.g., s = from 3 to 10 in Amat et al. (1997); s = 8 in Medina et al. (2002); s = from 2 to 9 in Escobar et al. (2005); s = 12 in Lopera-Toro and Cardenas-Bautista (2019)). Thus, we conclude that dung beetle species dwelling in this region of Andean highlands are part of species-poor assemblages conformed by Neotropical species that tolerate the temperate climatic conditions of such regions.
In conclusion, our study indicates that habitat type affects dung beetle assemblages at a portion of Andean highlands in Colombia. Dung beetle assemblages in tropical ecosystems are often sensitive to habitat transformation (Hanski and Cambefort 1991; Nichols et al. 2007; Scholtz et al. 2009). Here we present cues that introduced pastures in Andean landscapes maintain impoverished dung beetle diversity – also, this anthropogenic habitat is characterized by a distinct assemblage compared to forested area and its edges. Our results reinforce the importance to maintain native forest patches in the Andean region, in order to maintain the native species diversity and consequently by the ecosystem services they provide. Based on the results of this study, we believe that it is important to analyze patterns of ecological communities in each different habitat carefully.
Data availability
All data related to this study are present in the paper.
References
Alvarado FA, Escobar F, Montero-Muñoz J (2014) Diversity and biogeographical makeup of the dung beetle communities inhabiting two mountains in the mexican transition zone. Org Divers Evol 14:105–114. https://doi.org/10.1007/s13127-013-0148-0
Alvarado FA, Escobar F, Williams DR, Arroyo-Rodríguez V, Escobar-Hernández F (2018) The role of livestock intensification and landscape structure in maintaining tropical biodiversity. J Appl Ecol 55:185–194. https://doi.org/10.1111/1365-2664.12957
Amat G, Lopera-Toro A, Amezquita S (1997) Patrones de distribución de escarabajos coprófagos (Coleoptera: Scarabaeidae) en relicto de bosque altoandino, cordillera oriental de Colombia. Caldasia 19:191–204
Anderson MJ (2006) Distance based tests for homogeneity of multivariate dispersions. Biometrics 62:245–253
Asner GP, Anderson CB, Martin RE et al (2014) Landscape-scale changes in forest structure and functional traits along an Andes-to-Amazon elevation gradient. Biogeosciences 11:843–856. https://doi.org/10.5194/bg-11-843-2014
Avella A, Rangel O (2016) Los Robledales. Diversidad y Conservación. In: Moreno LA, Andrade GI, Ruiz-Contreras LF (eds) Biodiversidad 2016. Estado y tendencias de la biodiversidad continental de Colombia. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá, D. C., p 105
Barretto JW, Baena ML, Domínguez IH, Escobar F (2021) Spatiotemporal variation in the adult sex ratio, male aggregation, and movement of two tropical cloud forest dung beetles. Curr Zool zoab101. https://doi.org/10.1093/cz/zoab101
Barretto J, Salomão RP, Iannuzzi L (2020) Diversity of dung beetles in three vegetation physiognomies of the Caatinga dry forest. Int J Trop Insect Sc 40:385–392. https://doi.org/10.1007/s42690-019-00089-4
Begon M, Townsend CR, Harper JL (2006) Ecology from individuals to ecosystems, fourth edition. Blackwell publishing, Oxford
Cárdenas L, Salinas N (2007) Libro rojo de plantas de Colombia, volumen 4. Especies maderables amenazadas: Primera parte. Instituto Amazónico de Investigaciones Científicas SINCHI – Ministerio de Ambiente, Vivienda y Desarrollo Territorial, Bogotá
Carvalho RL, Andersen AN, Anjos DV, Pacheco R, Chagas L, Vasconcelos HL (2020) Understanding what bioindicators are actually indicating: linking disturbance responses to ecological traits of dung beetles and ants. Ecol Ind 108:105764. https://doi.org/10.1016/j.ecolind.2019.105764
Carvalho RL, Vieira J, Melo C, Silva AM, Tolentino VCM, Neves K, Vaz-de-Mello F, Andersen AN, Vasconcelos HL (2022) Interactions between land use, taxonomic group and aspects and levels of diversity in a brazilian savanna: implications for the use of bioindicators. J Appl Ecol 59:2642–2653. https://doi.org/10.1111/1365-2664.14270
Chao A, Jost L (2012) Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533–2547. https://doi.org/10.1890/11-1952.1
Chao A, Ma KH, Hsieh TC (2016) User’s guide for iNEXT online: software for interpolation and extrapolation of species diversity. http://140.114.36.3/wordpress/wp-content/uploads/software/iNEXTOnline_UserGuide.pdf
Clarke KR, Gorley RN (2006) Primer v6: user manual/tutorial. PRIMER-E, Albany
Connell ML (1978) Diversity in tropical rain forests and coral reefs. Science 199:1302–1310. https://doi.org/10.1126/science.199.4335.1302
Cook J (2002) A revision of the neotropical genus Cryptocanthon Balthasar (Coleoptera: Scarabaeidae: Scarabaeinae). Coleops Bull 1:1–96. https://doi.org/10.1649/0010-065X(2002)56[3:AROTNG]2.0.CO;2
Cultid-Medina CA, Martínez-Quintero BG, Escobar F, Ulloa PC (2015) Movement and population size of two dung beetle species in an Andean agricultural landscape dominated by sun-grown coffee. J Insect Conserv 19:617–626. https://doi.org/10.1007/s10841-015-9784-3
Davis ALV, Scholtz CH, Swemmer AS (2012) Effects of land usage on dung beetle assemblage structure: Kruger National Park versus adjacent farmland in South Africa. J Insect Cons 16:399–411. https://doi.org/10.1007/s10841-011-9426-3
Domínguez D, Marín-Armijos D, Ruiz C (2015) Structure of dung beetle communities in an altitudinal gradient of neotropical dry forest. Neotrop Entomol 44:40–46. https://doi.org/10.1007/s13744-014-0261-6
Escobar F (2000) Diversidad y distribución de los escarabajos del estiercol (Coleoptera: Scarabaeidae: Scarabaeinae) de Colombia. In: Martin-Piera JJ, Morrone A, Melic (eds) Hacia un proyecto CYTED para el inventario y estimaci6n de la diversidad entomológica en Iberoambrica: PrIBES-2000. Monografias Tercer Milenio, Zaragoza, pp 197–210
Escobar F, Chacón-Ulloa P (2001) Distribución espacial y temporal en un gradiente de sucesión de la fauna de escarabajos del coleópteros coprófagos (Scarabaeinae, Aphodiinae) en un bosque tropical montano, Nariño – Colombia. Rev Biol Trop 48:961–975
Escobar F (2004) Diversity and composition of dung beetle (Scarabaeinae) assemblages in a heterogeneous Andean landscape. Trop Zool 17:123–136. https://doi.org/10.1080/03946975.2004.10531202
Escobar F, Lobo JM, Halffter G (2005) Altitudinal variation of dung beetle (Scarabaeidae: Scarabaeinae) assemblages in the colombian Andes. Global Ecol Biogeogr 14:327–337. https://doi.org/10.1111/j.1466-822X.2005.00161.x
Etter A (1993) Diversidad ecosistémica en Colombia hoy. (ed) Nuestra diversidad biológica. Fundación Alejandro Ángel Escobar, Bogotá, pp 43–61. CEREC
Ezcurra E (2016) Anthropogenic disturbances infiltrate forest fragments. PNAS 113:5150–5152. https://doi.org/10.1073/pnas.1604829113
Filgueiras BKC, Tabarelli M, Leal IR, Vaz-de-Mello FZ, Iannuzzi L (2015) Dung beetle persistence in human-modified landscapes: combining indicator species with anthropogenic land use and fragmentation-related effects. Ecol Ind 55:65–73. https://doi.org/10.1016/j.ecolind.2015.02.032
Galindo H (2005) Esquema de Ordenamiento Territorial del municipio de Arcabuco – EOTMA. CORPOBOYACA, Tunja
Gallagher G (2018) Quercus humboldtii. The IUCN red list of threatened species 2018:e.T194139A2302449. https://dx.doi.org/10.2305/IUCN.UK.2018-2.RLTS.T194139A2302449.en. Accessed 1 Dec 2022
Gardner TA, Barlow J, Araújo IS et al (2008) The cost-effectiveness of biodiversity surveys in tropical forests. Ecol Lett 11:139–150. https://doi.org/10.1111/j.1461-0248.2007.01133.x
Gascon C, Lovejoy TE, Bierregaard-Jr RO et al (1999) Matrix habitat and species richness in tropical forest remnants. Biol Conserv 91:223–229. https://doi.org/10.1016/S0006-3207(99)00080-4
Génier F (1996) A revision of the neotropical genus Ontherus Erichson. Mem Entomol Soc Can 170:1–168. https://doi.org/10.4039/entm128170fv
Génier F (2009) Le genre Eurysternus Dalman, 1824 (Scarabaeidae: Scarabaeinae: Oniticellini), révision taxonomique et clés de détermination illustrées. Pensoft, Sofia
Glor RE, Flecke AS, Benard MF, Power AG (2001) Lizard diversity and agricultural disturbance in a caribbean forest landscape. Biodivers Cons 10:711–723. https://doi.org/10.1023/A:1016665011087
Gotelli NJ (2001) A primer of ecology, third edition. Sinauer Associates, Sunderland
Halffter G (1991) Historical and ecological factors determining the geographical distribution of beetles (Coleoptera: Scarabaeidae: Scarabaeinae). Folia Entomol Mex 82:195–238. https://doi.org/10.21426/B615110376
Halffter G, Matthews E (1966) The natural history of dung beetles of the subfamily Scarabaeinae (Coleoptera, Scarabaeidae). Folia Entomol 12–14:1–313
Halffter G, Favila M (1993) The Scarabaeinae (Insecta: Coleoptera) an animal group for analysing, inventorying and monitoring biodiversity in tropical rainforest and modified landscapes. Biol Int 27:15–21
Hanski I, Cambefort Y (1991) Dung beetle ecology. Princeton University Press, Princeton
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–431. https://doi.org/10.2307/1934352
Hill N, Brehm G, Fiedler K (2006) Diversity and ensemble composition of geometrid moths along a successional gradient in the ecuadorian Andes. J Trop Ecol 22:155–166
Horgan FG (2005) Effects of deforestation on diversity, biomass and function of dung beetles on the eastern slopes of the peruvian Andes. For Ecol Manag 216:117–133. https://doi.org/10.1016/j.foreco.2005.05.049
Horgan FG (2008) Dung beetle assemblages in forests and pastures of El Salvador: a functional comparison. Biodivers Conserv 17:2961–2978. https://doi.org/10.1007/s10531-008-9408-2
Iannuzzi L, Salomão RP, Costa FC, Liberal CN (2016) Environmental patterns and daily activity of dung beetles (Coleoptera: Scarabaeidae) in the Atlantic Rainforest of Brazil. Entomotropica 31:196–207
IDEAM : Instituto de Hidrología, Meteorología y Estudios Ambientales (2010) Consulta y descarga de datos hidrometeorológicos. http://dhime.ideam.gov.co/atencionciudadano/
Jost L (2006) Entropy and diversity. Oikos 113:363–375. https://doi.org/10.1111/j.2006.0030-1299.14714.x
Kiatoko N, Raina SK, Langevelde FV (2017) Impact of habitat degradation on species diversity and nest abundance of five african stingless bee species in a tropical rainforest of Kenya. Int J Trop Insect Sc 37:189–197. https://doi.org/10.1017/S174275841700011X
Kocher SD, Williams EH (2000) The diversity and abundance of north american butterflies vary with habitat disturbance and geography. J Biogeogr 27:785–794. https://doi.org/10.1046/j.1365-2699.2000.00454.x
Korasaki V, Vaz-de-Mello FZ, Braga RF, Zanetti R, Louzada J (2010) Taxocenosis of the Scarabaeinae (Coleoptera: Scarabaeidae) in Benjamin constant, AM. Acta Amz 42:423–432. https://doi.org/10.1590/S0044-59672012000300015
Larsen TH, Forsyth A (2005) Trap spacing and transect design for dung beetle biodiversity studies. Biotropica 37:322–325. https://doi.org/10.1111/j.1744-7429.2005.00042.x
Larsen TH, Williams NM, Kremen C (2005) Extinction order and altered community structure rapidly disrupt ecosystem functioning. Ecol Lett 8:538–547. https://doi.org/10.1111/j.1461-0248.2005.00749.x
Larsen TH, Lopera A, Forsyth A (2006) Extreme trophic and habitat specialization by peruvian dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae). Coleopts Bull 60:315–324. https://doi.org/10.1649/0010-065X(2006)60[315:ETAHSB]2.0.CO;2
Larsen TH (2012) Upslope range shifts of andean dung beetles in response to deforestation: compounding and confounding effects of microclimatic change. Biotropica 44:82–89. https://doi.org/10.1111/j.1744-7429.2011.00768.x
Lira AFA, Pordeus L, Salomão RP, Badillo-Montaño R, Albuquerque CMR (2019) Effects of anthropogenic land-use on scorpions (Arachnida: Scorpiones) in neotropical forests. Int J Trop Insect Sc 39:211–218. https://doi.org/10.1007/s42690-019-00029-2
Lloyd H, Marsden SJ (2008) Bird community variation across Polylepis woodland fragments and matrix habitats: implications for biodiversity conservation within a high Andean landscape. Biodivers Conserv 17:2645–2660. https://doi.org/10.1007/s10531-008-9343-2
Lopera-Toro A, Cárdenas-Bautista JE (2019) Escarabajos coprófagos (Coleoptera – Scarabaeinae) de los ecosistemas exocársticos de Peñón (Andes), Santander, Colombia. In: Lasso CA, Barriga JC, Fernández-Auderset J (eds) Biodiversidad subterránea y epigea de los sistemas cársticos de El Peñón (Andes), Santander, Colombia. VII. Serie Fauna Silvestre Neotropical. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá, pp 377–399
Louzada J, Lima AP, Matavelli R, Zambaldi L, Barlow J (2010) Community structure of dung beetles in amazonian savannas: role of fire disturbance, vegetation and landscape structure. Landsc Ecol 25:631–641. https://doi.org/10.1007/s10980-010-9448-3
Magnano LFS, Rocha MFR, Meyer L, Martins SV, Meira-Neto JAA (2015) Microclimatic conditions at forest edges have significant impacts on vegetation structure in large Atlantic forest fragments. Biodivers Conserv 24:2305–2318. https://doi.org/10.1007/s10531-015-0961-1
Magurran AE (2004) Measuring biological diversity. Blackwell publishing, Oxford
McKinney ML, Lockwood JL (1999) Biotic homogenization: a few winners replacing many losers in the next mass extinction. Trends Ecol Evol 14:450–453. https://doi.org/10.1016/S0169-5347(99)01679-1
Medina CA, Lopera A (2000) Clave ilustrada para la identificación de géneros de escarabajos coprófagos (Coleoptera: Scarabaeinae) de Colombia. Caldasia 22:299–315
Medina CA, Lopera A, Vítolo A, Gill B (2001) Escarabajos Coprófagos (Coleoptera: Scarabaeidae: Scarabaeinae) de Colombia. Biota Colombiana 2:131–144
Medina CA, Escobar F, Kattan GH (2002) Diversity and habitat use of dung beetles in a restored Andean landscape. Biotropica 34:181–187
Moctezuma V (2021) El género Onthophagus Latreille, 1802 (Coleoptera: Scarabaeidae) de México. Dugesiana 28:175–220. https://doi.org/10.32870/dugesiana.v28i2.7166
Molano FR, Medina CAU (2010) BA new species of Scybalocanthon (Coleoptera: Scarabaeinae: Canthonini) and description of the variation of the male genitalia. Rev Mex Biodiv 81:689–699
Nichols E, Larsen T, Spector S et al (2007) Global dung beetle response to tropical forest modification and fragmentation: a quantitative literature review and meta-analysis. Biol Conserv 137:1–19. https://doi.org/10.1016/j.biocon.2007.01.023
Nichols E, Spector S, Louzada J, Larsen T, Amezquita S, Favila ME (2008) Ecological functions and ecosystem services provided by Scarabaeinae dung beetles. Biol Conserv 141:1461–1474. https://doi.org/10.1016/j.biocon.2008.04.011
Nunes CA, Braga RF, Figueira JEC, Neves FS, Fernandes GW (2016) Dung beetles along a tropical altitudinal gradient: environmental filtering on taxonomic and functional diversity. PlosOne 11:e0157442. https://doi.org/10.1371/journal.pone.0157442
Oksanen J, Blanchet FG, Kindt R et al (2016) Vegan: community ecology package. https://CRAN.R-project.org/package=vegan
Otálora A (2003) Mamíferos de los bosques de roble. Acta Biol Colombiana 8:57–71
Pearman PB (2002) The scale of community structure: habitat variation and avian guilds in tropical forest understory. Ecol Monogr 72:19–39. https://doi.org/10.1890/0012-9615(2002)07[0019:TSOCSH]2.0.CO;22
Prevedello JA, Vieira MV (2010) Does the type of matrix matter? A quantitative review of the evidence. Biodivers Conserv 19:1205–1223. https://doi.org/10.1007/s10531-009-9750-z
Pulido L, Medina C, Riveros R (2007) Nuevos registros de escarabajos coprófagos (Scarabaeidae: Scarabaeinae) para la región andina de Colombia. Rev Acad Col Cienc Físicas Exactas 31:305–310
Quintero C, Morales CL, Aizen MA (2010) Effects of anthropogenic habitat disturbance on local pollinator diversity and species turnover across a precipitation gradient. Biodivers Conserv 19:257–274. https://doi.org/10.1007/s10531-009-9720-5
R Core Development Team (2015) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna
Ribeiro PHO, Togni PHB, Frizzas MR (2022) Spatial and temporal segregation in dung beetles (Coleoptera: Scarabaeinae) in the Cerrado of Central Brazil. Biodivers Conserv 31:2723–2274. https://doi.org/10.1007/s10531-022-02453-2
Salomão RP, Favila ME, González-Tokman D (2020) Spatial and temporal changes in the dung beetle diversity of a protected, but fragmented, landscape of the northernmost neotropical rainforest. Ecol Ind 111:105968. https://doi.org/10.1016/j.ecolind.2019.105968
Santos BA, Arroyo-Rodríguez V, Moreno CE, Tabarelli M (2010) Edge related loss of tree phylogenetic diversity in the severely fragmented brazilian Atlantic Forest. PLoS ONE 5:e12625. https://doi.org/10.1371/journal.pone.0012625
Sarkar D (2018) Package ‘lattice’. https://cran.r-project.org/web/packages/lattice/lattice.Pdf. Accessed 1 May 2022
Saunders DA, Hobbs RJ, Margules CR (1991) Biological consequences of ecosystem fragmentation – a review. Conserv Biol 5:18–32. https://doi.org/10.1111/j.1523-1739.1991.tb00384.x
ScarabNet (2009) ScarabNet global taxon database. http://scarabnet.myspecies.info/scarabnet-welcome. Accessed 1 Aug 2020
Scholtz ADL, Davis AL, Kryger U (2009) Evolutionary biology and conservation of dung beetles. Pensoft Publishers, Sofia-Moscow
Silva PM, Aguiar CAS, Niemela J, Sousa JP, Serrano ARM (2008) Diversity patterns of ground-beetles (Coleoptera: Carabidae) along a gradient of land-use disturbance. Agr Ecosyst Environ 124:270–274. https://doi.org/10.1016/j.agee.2007.10.007
Silva PG, Hernández MIM (2015) Spatial patterns of movement of dung beetle species in a tropical forest suggest a new trap spacing for dung beetle biodiversity studies. PlosOne 10:e0126112. https://doi.org/10.1371/journal.pone.0126112
Simpson GL, Core Development R, Team, Bates DM, Oksanen J (2018) Package ‘permute’. https://cran.r-project.org/web/packages/permute/permute.pdf
Spector S (2006) Scarabaeine dung beetles (Coleoptera: Scarabaeidae: Scarabaeinae): an invertebrate focal taxon for biodiversity research and conservation. Coleopts Bull 60:71–83. https://doi.org/10.1649/0010-065X(2006)60[71:SDBCSS]2.0.CO;2
Souza JM, Marinoni RC, Marinoni L (2014) Open and disturbed habitats support higher diversity of Syrphidae (Diptera)? A case study during three yr of sampling in a fragment of araucaria forest in Southern Brazil. J Insect Sc 14:1–8. https://doi.org/10.1093/jisesa/ieu098
Tabarelli M, Peres CA, Melo FPL (2012) The ‘few winners and many losers’ paradigm revisited: emerging prospects for tropical forest biodiversity. Biol Conserv 155:136–140. https://doi.org/10.1016/j.biocon.2012.06.020
Tarasov S, Dimitrov D (2016) Multigene phylogenetic analysis redefines dung beetles relationships and classification (Coleoptera: Scarabaeidae: Scarabaeinae). BMC Evol Biol 16:257. https://doi.org/10.1186/s12862-016-0822-x
Tscharntke T, Sekercioglu CH, Dietsch TV, Sodhi NS, Hoehn P, Tylianakis JM (2008) Landscape constraints on functional diversity of birds and insects in tropical agroecosystems. Ecology 89:944–951. https://doi.org/10.1890/07-0455.1
Tylianakis JM, Klein A, Tscharntke T (2005) Spatiotemporal variation in the diversity of hymenoptera across a tropical habitat gradient. Ecology 86:3296–3302. https://doi.org/10.1890/05-0371
Tylianakis JM, Tscharntke T, Klein AM (2006) Diversity, ecosystem function, and stability of parasitoid–host interactions across a tropical habitat gradient. Ecology 87:3047–3057. https://doi.org/10.1890/0012-9658(2006)8[3047:DEFASO]2.0.CO;2
Tylianakis JM, Tscharntke T, Lewis OT (2007) Habitat modification alters the structure of tropical host–parasitoid food webs. Nature 445:202–205. https://doi.org/10.1038/nature05429
Vargas-Ramírez L, Colmenares-Pinzón J, Serrano-Cardozo V (2018) Uso de microhábitat por un ensamble de pequeños mamíferos no voladores en un bosque de roble sobre la Cordillera Oriental de los Andes colombianos. Rev Mex Biodivers 89:479–487. https://doi.org/10.22201/ib.20078706e.2018.2.1933
Vaz-de-Mello FZ (2000) Present degree of knowledge of the brazilian Scarabaeidae s. str. (Coleoptera: Scarabaeoidea). In: Martín-Piera P, Morrone JJ, Melic A (eds) Trabajos del 1er taller iberoamericano de entomología sistemática. Monografías del Tercer Milenio, Zaragoza, pp 183–195
Vaz-de-Mello FZ, Edmonds W, Ocampo F, Schoolmeesters P (2011) A multilingual key to the genera and subgenera of the subfamily Scarabaeinae of the New World (Coleoptera: Scarabaeidae). Zootaxa 2854:1–73. https://doi.org/10.11646/zootaxa.2854.1.1
Villada-Bedoya S, Cultid-Medina CA, Escobar F, Guevara R, Zurita G (2017) Edge effects on dung beetle assemblages in an andean mosaic of forest and coffee plantations. Biotropica 49:195–205. https://doi.org/10.1111/btp.12373
Acknowledgements
We are grateful to the communities of the Peñas Blancas Town, especially Rosa Bautista, Gabriel Robles, and their families. Thanks to Fernando Vaz-de-Mello, François Génier, Bruce Gill, and Joyce Cook for the corroboration of the determination and comments of the species. To Fredy Molano (Posthumous tribute) and Claudia Medina for their collaboration in the study’s first phase. To Juana Andrade López and Mónica Ospina for their comments. We also thank the biologists Paola Delgado and Vladimir Páez for their valuable cooperation and support in the field phase. Renato Portela Salomão thanks CAPES and DGAPA/UNAM for his postdoctoral scholarships.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest/Competing interests
The study does not have any conflict of interests or competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salomão, R.P., Lopera-Toro, A., Pulido-Herrera, L.A. et al. Habitat type affects the diversity of dung beetle (Coleoptera: Scarabaeidae) assemblages in a neotropical mountainous region of Colombia. Int J Trop Insect Sci 43, 793–803 (2023). https://doi.org/10.1007/s42690-023-00987-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-00987-8