Abstract
Based on a quantitative understanding of the environmental factors effecting armoured bush cricket, Acanthoplus discoidalis population dynamics, a hypothesis was formulated to explain the occurrence of outbreaks in some years and locations. The principles, expressed using a rule-based or qualitative model, were that nymph and adult survival and fecundity were reduced in years with uneven rainfall and that egg survival was reduced in years with a wet late-season as this is associated with increased egg predation and/or water-logging. The implication was that large egg banks resulted either when a large number of adults were present or when neither low fecundity nor high egg mortality were constraints. Such large egg banks were however predicted only to lead to outbreaks when there was an adequate amount of food for nymph and adult survival in the following season. Model predictions were compared with observed outbreaks of A. discoidalis between 1988 to 2002 for the three climatic zones of the east, central and western parts of southern Botswana. There was significant agreement between model predictions and observed outbreaks in two of the three zones (95% confidence interval of the kappa coefficient of agreement > 0). Taking the data for all three zones together and compared to the average outbreak frequency, an outbreak was three times more likely to occur when the model predicted an outbreak and six times less likely to occur when it predicted no outbreak.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The armoured bush cricket (ABC), Acanthoplus discoidalis (Walker) (Orthoptera: Tettigoniidae: Hetrodinae) is an important insect pest of smallholder cereal crops in semi-arid regions of southern Africa (Musonda 2000; Wohlleber 1996; 2000; Minja 2000). Although a sporadic pest, it has been rated as the most damaging cereal panicle pest in northern Namibia (Leuschner, 1995) whilst in eastern Botswana farmers consider it the second most serious pest just behind Quelea birds in terms of crop damage caused in outbreak years (Matsaert et al. 2000). The species remains a widespread and sporadic pest particularly in smallholder cropping systems in the region (Bazelet and Naskrecki 2014). Acanthoplus discoidalis has outbreak characteristics which resemble migrant pests such as Quelea, armyworm and desert locust; though itself not migratory, A. discoidalis outbreaks can result in congregations of insects similar to locust hoppers and armyworm larvae. In common with migrant pests, A. discoidalis outbreaks are characterised by large numbers of individuals which are obvious to farmers (Green 2002). It is generally held that outbreaks occur when a relatively wet year follows several dry seasons although this has never been substantiated.
This paper aims to provide an explanation for the observed changes in outbreak patterns using a hypothesis which incorporates rainfall effects on ABC population dynamics including fecundity, development and mortality (Mviha 2005). Formulated as a rule-based or qualitative model (Holt et al. 2003), the interactions of the different processes involved were used to simulate ABC population dynamics and make outbreak predictions; the use of rule-based models has been described in wildlife management (Starfield and Bleloch 1986) and pest management (Holt and Day 1993). ABC population dynamics were simulated from one year to the next as affected by rainfall patterns and other related environmental factors. The intention was to explore if this hypothesis explaining ABC population dynamics expressed by the model was consistent with the occurrence of outbreaks. Model predictions of outbreaks were compared with outbreak patterns over a 14-year cropping period in three zones of southern Botswana.
Aspects of ABC biology
Egg hatching commences as early as October in the central and northern regions of Botswana and as late as January in the southwest of the country (Mviha 2005). Comparing southwestern and central/northern populations, nymphs take about two and three months to reach the adult stage respectively. Eggs are laid towards the end of the rainy season. The reproductive capacity of adults, which varies between these two areas, is dependent on food quantity and quality, factors that, in themselves, are largely dependent on adequate rain (Green et al. 2003a, b). High and timely rainfall produces high quality crop plants that improve food supply for adult ABC at the time when wild grasses dry up (Mosupi et al. 2003), resulting in an improvement of both reproductive capacity and adult survival. If the start of the main rains is late then ABC development is delayed. In such circumstances, low temperatures at the end of the season may reduce adult survival significantly and hence lead to low fecundity. An early or average onset of the rainy season improves the food supply and subsequent survival and fecundity of adults (Mviha et al. 2003; Mviha 2005).
The greatest proportion of the life cycle of the armoured bush cricket is spent in the egg stage. Eggs are contained in a tough, porous egg pod containing 6 – 12 sibling eggs, laid some 2 – 5 cm below the soil surface. During this stage, the majority of the eggs experience a complex diapause syndrome, while they are exposed to a range of factors that affect both their development and survival (Green 2002). Moisture beyond certain critical limits is an important egg mortality factor. In particular, egg flooding immediately after oviposition promotes egg damage resulting from water logging. Thus, prolonged rains, while resulting in regeneration of some of wild grasses and availability of fresh grass food may reduce the survival rate of the new-laid eggs which are particularly sensitive to soil water saturation. Similarly, very high moisture levels during incubation result in reduced egg hatch possibly following death of the developing embryos due to drowning. Low moisture conditions lead to egg desiccation and when this persists to the hatching phase, the number of eggs that finally hatch is greatly reduced (Mviha et al. 2003; Mviha 2005).
In this paper we test the hypothesis that the regional-scale population dynamics of ABC can be modelled by what is known about the effects of rainfall on the insect’s biology and ecology. The objective is to compare the predictions of the model with reported ABC outbreak patterns, and to establish the predictive capability of the model as a potential pest forecasting tool to aid future ABC management efforts.
Materials and methods
Modelling the factors driving ABC population dynamics
Based on what is known of ABC biology and ecology, the population model was based on the following principles:
-
(1)
Fecundity in a season is positively related to the total rainfall in that season, since rainfall improves food supply availability, which has a direct effect on female reproductive capacity.
-
(2)
Failure of rains at any stage in the season will affect the proportion of hatching individuals that survive to reproduce successfully because of the resulting constraint imposed by limited food availability.
-
(3)
High egg mortality results when prolonged rainfall occurs late in the season.
-
(4)
Diet quality affects ABCs’ survival and fecundity. In Botswana, like most semi-arid regions, the supply of quality food as influenced by rainfall availability is the most limiting ecological factor (Pike 1971); low rainfall in the mid and late parts of the season is therefore assumed to reduce ABC fecundity.
-
(5)
The number of viable eggs in the egg bank is constrained by both female fecundity and by the subsequent survival of those eggs laid.
-
(6)
Both recruitment through previous egg laying and egg survival between seasons are critical determinants of the size of the egg bank.
-
(7)
The numbers of adult ABC is constrained by either poor conditions for survival during the season or a limited egg bank at the start of the season.
The expression of these principles in the form of a rule-based or qualitative model allowed our understanding that certain conditions are better or worse than others, in their effects on particular aspects of ABC life history (e.g. survival and fecundity), to be used to predict ABC population dynamics. A description of all model components on a three-point ordinal scale: high, medium and low, was sufficient to express ‘better’, ‘worse’ and ‘similar’.
Two kinds of relationships between model components were employed. In the first, the value of a component is constrained by any one of a set of other components, and so the value is given by a relationship of the form a = Min {b, c}, meaning that the value of component a is determined by the smaller of the values of b and c. In the second type of relationship, any one of a set of components is sufficient to determine the value of the component concerned, and so the relationship has the form a = Max {b, c} meaning the value of component 'a' is determined by the larger of b and c. The identity, high > medium > low holds throughout, and for computational convenience high, medium and low were coded 3, 2 and 1, respectively.
A qualitative description of rainfall in southern Botswana
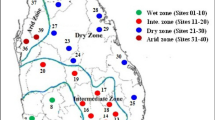
Based on the quantity and temporal pattern of the seasonal rainfall, seasons were characterised for the period 1988 to 2002 for Zones 5, 6 and 7 (Fig. 1), located in the east, central and western part of southern Botswana, respectively.
The geographical situation of southern Botswana is such that in most cases it receives only attenuated effects of rain-bearing air masses from the eastern coast of southern Africa and from the Inter-Tropical Convergence Zone (ITCZ) (Pike 1971). This causes a high degree of variability in rainfall with erratic seasonal rainfall distribution that worsens from east to west. Thus, zone 7 normally registers the most erratic rainfall distribution compared to zones 5 and 6.
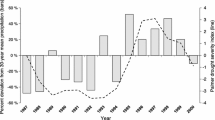
Directly or indirectly, the population dynamics of ABC are widely believed to be determined by the quantity and pattern of rainfall, both within and between seasons. The rainy season occurs from October to April and the cold cloud duration (CCD) profile for that period was used to provide an indication of both the pattern and amount of rainfall over the course of the season. The use of quite detailed temporal and spatial CCD information has proved of great value predicting armyworm outbreaks whose population dynamics are similarly rainfall-driven (Tucker 1997; Holt et al. 2000); here it was used more simply, to categorise season type. Originally accessed via the FAO/Artemis project (Hielkema 2008), CCD data, interpreted as rainfall in mm per day, is now freely available at daily resolution (Maidmant et al. 2017).
Comparison of seasonal profiles of CCD’s with their long-term average was used to show differences between seasons. These differences were summarised by making a distinction between early, mid and late periods of the season: before mid-December, mid-December to mid-February, and after mid-February, respectively. As explained below, these periods were selected in order to appropriately describe and model the effects of rainfall on ABC life history characteristics. For example, rainfall after mid-February is influential for egg survival. Thus, each season was characterised by three periods, (early, mid, late) and each could have one of three rainfall values, low (1) when CCD values were below average, medium (2) when the CCD values were close to the average, and high (3) when the CCD values were above average. For example, monthly rainfall ± 25% of the long-term average can be categorised as close to average (Meteorological Office 2021). A monthly rainfall total greater than 125% of the long-term average would then be categorised as a positive anomaly ‘high’, and less than 75%, a negative anomaly, ‘low’. Whether the rains were low, medium or high for each of the three periods is shown in Tables 1 – 3. This can be summarised using the following notation:
For example, the year 2000/01 in Botswana Zone 5 (eastern Botswana) had wet conditions in the early part of the season, a dry mid season and a wet late season, so Season 2000/01 = {3,1,3} (Table 1).
Biological reasoning and assumptions underlying the model
All the parameters determining the population dynamics of ABC are related to this seasonal rainfall distribution. The survival of newly hatched ABC nymphs, and the later survival of older nymphs and adults, depends on continuity of food supply throughout the long life cycle. Failure of rains at any stage in the season will affect the proportion of hatching individuals that survive to reproduce successfully. Because survival is constrained by periods of water and food stress, survival is related to the period in the season with the worst conditions, so
Fecundity is also constrained by sub-optimal rainfall conditions but it is assumed that it is the mid and late parts of the season that influence fecundity, and so
If high rainfall occurs in the late part of the season, it is assumed that water logging is increased, and therefore mortality would be higher. This leads to the expression,
To clarify the form of Expression 4 with a numeric example, egg survival = 1 (low) if late = 3 (high). At the other extreme, egg survival = 3 (high) if late = 1 (low).
The contribution of each adult female to the egg bank is constrained both by her fecundity and by the subsequent survival of the eggs laid. If either is low, then the contribution to the egg bank is low and therefore,
Total egg recruitment to the egg bank can be high either because there are many adults or because reproductive success is high. This leads to the expression,
Adult numbers are constrained either by poor conditions for survival during the season or by lack of eggs in the egg bank at the start of the season (previous season’s eggs). This is expressed as,
This series of expressions (Eqs. 1 to 7) together define the model. One season is linked to the next by Eq. 7 because the number of ABC emerging in a new season depends on the eggs previously accrued in the egg bank. Thus Eqs. 1 to 7 were used to simulate ABC population dynamics over a sequence of years.
Evaluation of agreement between predicted and actual ABC outbreaks
The kappa coefficient (Ќ) (Sim and Wright 2005) was used to assess pair-wise agreement between predicted and actual outbreak years for each zone. Kappa coefficient in this case was a test ratio for the observed variability involving predicted and actual data. Estimates of (Ќ) were obtained as
where Po was the proportion of years in which the predicted and actual outbreaks were in agreement and Pc was the proportion of years for which agreement was expected only by chance. The numerator was the observed improvement over chance agreement while the denominator was the maximum possible improvement over chance agreement. Thus, the kappa coefficient was intended to assess how much agreement was observed beyond that expected by chance. The Kappa coefficient has a range of − 1 (indicating perfect disagreement) through 0 (representing the agreement expected by chance) to + 1 (indicating perfect agreement). The 95% confidence intervals of the kappa coefficients were calculated (McHugh 2012) and whether or not the interval encompassed zero indicated whether model predictions demonstrated significantly more than chance agreement with observed outbreaks.
The prior probability of outbreaks was also calculated for the three zones as was the conditional probability of an outbreak given predictions of both ‘outbreak’ and ‘no outbreak’ by the model. The increase or decrease in outbreak probability, given the model prediction was therefore determined.
The model was based both on information from the literature detailed above and on studies conducted in Botswana’s Zone 5 (Mviha 2005) and tested in the three zones using CCD averages for each zone. The information regarding which years were outbreak or non-outbreak was obtained through a survey conducted in 2000 (Marsaert et al. 2000). This was followed by both oral and questionnaire interviews conducted with farmers and extension specialists respectively (Green 2002).
Results
Model predictions
At the start of the 14-year simulation, a low-density egg bank was assumed to be present following the 1987 season. If the simulation resulted in a medium or high adult number in any season then this was taken as equating to an outbreak and a low number to no outbreak for the season in question. In practice only two solutions to Eq. 5, which determines the number of surviving eggs per adult, proved possible, low and medium. This was because seasonal conditions favoured either high fecundity or high egg survival but not both in the same season. Outbreaks were predicted when two factors coincided: (1) A large egg bank from previous year dependent on wetness during the oviposition period and (2) Good nymph and adult survival dependent on an adequate amount of food being available in the current year.
Seasons with large egg banks
A large egg bank results when either a large number of adults were present or when neither low fecundity nor high egg mortality constrained egg production. Since it is not possible for both to be high, rainfall conditions most conducive to large egg bank were those that resulted in medium values of both fecundity (f) and egg survival (c) or those that result in high values of one variable and low value of the other. Such conditions occurred in 1990/1991, 1991/92, 1992/93, 1993/94, 1994/95, 1995/96, 1996/97, 1997/98, 1998/99,1999/00 and 2001/02 in zone 5, 1988/89, 1989/90, 1990/91, 1991/92, 1992/93, 1993/94, 1995/96, 1997/98, 1998/99 and 2000/01 in Botswana zone 6 and 1989/90, 1992/93, 1993/94, 1994/95, 1995/96, 1996/97, 1997/98, 1998/99 and 1999/00 in Botswana zone 7 (Tables 1 –3). These seasonal conditions however, did not always lead to an outbreak prediction since survival of the emerging nymphs was constrained by the rainfall conditions in the current season.
Outbreak seasons
According to the principles expressed in the model, outbreaks should only have occurred in 1991/92, 1995/96, 1996/97, 1997/98, 1998/99 and 1999/00 in Zone 5 (Table 1), in 1989/1990, 1998/99 and 1999/00 in Zone 6 (Table 2) and in 1996/97, 1998/99 and 1999/00 in Zone 7 (Table 3). When compared with the actual occurrence of ABC outbreak years in the three zones, the kappa coefficient of agreement between observed and model-predicted outbreaks was greater than zero in all three zones. In two of the three zones, neither did zero fall within the 95% confidence interval of the Ќ values (Table 4) indicating, that in two of the three zones, there is a significant measure of agreement between model and observations.
A practical measure of the predictive capability of the model can be obtained from the increase or decrease in the likelihood of an outbreak given the model predictions. The prior probability of an outbreak occurring in any year was 0.29, 0.14 and 0.14 in Zones 5, 6 and 7, respectively. If we now consider the conditional probability of an outbreak, given that the model predicts an outbreak, these values increased to 0.5, 0.67 and 0.67, respectively. Conversely, they decreased to 0.125, 0 and 0, in zones 5, 6 and 7, respectively, when the model predicted no outbreak. The predictions of the model therefore made a large difference to outbreak expectation, particularly in Zones 6 and 7. To take an overall figure derived from data for all zones together, an outbreak was 19 times more likely to occur when an outbreak was forecast than when it was not.
Discussion
There was good consistency amongst farmers and research and extension staff who reported outbreaks to have occurred in 1992/93, 1993/94, 1998/99 and 1999/00 in zone 5 (Marsaert et al. 2000; Green 2002). Of the four outbreak seasons which were experienced, three of these, 1991/1992, 1998/99 and 1999/2000 were correctly predicted. The model also gave correct prediction for 7 of the 10 non-outbreak years. In zone 6, outbreaks were reported in 1989/90, 1998/99 and 1999/00. The model gave correct outbreak predictions for 1998/99 and the 1999/00 seasons. All but one non-outbreak season were correctly predicted. Again, three outbreak seasons were predicted in zone 7 and of these, 1998/99 and 1999/00 were correctly predicted. Again, the model correctly predicted all but one of the non-outbreak seasons in this zone.
The Kappa coefficients of agreement between predicted and actual outbreaks were all positive and clearly distinguishable from chance agreement in two of the three zones. The model appeared to perform better in the two more arid zones (6 and 7) where there were also fewer outbreaks than in the wetter Zone 5. In general, therefore, the model over-predicted outbreaks to some extent: only once did it fail to predict an outbreak which did occur but it predicted outbreaks which did not occur on five occasions. This over-prediction was most evident in the wetter Zone 5, suggesting that other constraining factors may be at work when rainfall conditions otherwise appear conducive to ABC outbreaks.
Two important elements of the of the overall hypothesis to explain ABC population dynamics are that egg mortality is high in high-rainfall years and that fecundity is also high in high-rainfall years. High rainfall therefore has both positive and negative effects but on different aspects of the organism’s population processes. High rainfall increases fecundity (Eq. 3) but reduces egg survival (Eq. 4), and as a result there is an optimum level of rainfall, neither too much nor too little, in which an optimum compromise exists between fecundity and egg survival to create maximum egg bank numbers (Mviha 2005).
The modelled hypothesis comprises the above two processes, together with the expectation of high nymph and adult mortality when prolonged dry conditions occur at any period during the cropping season. There are a number of reasons why predictions based on this logic may not always be correct. Two of these concern the spatial heterogeneity of the environment. Firstly, the reduction in egg survival due to prolonged rainfall rests on soil moisture saturation and water logging regardless of the nature and property of the soils. Depending on the locality, different soils may have different water retention capabilities related to organic matter content, soil structure and particle size. In addition, experiments have illustrated that ABC are selective in their choice of oviposition sites, avoiding certain soil types and conditions e.g. areas of wet soil that are indicative of potential water logging (Mviha et al. 2003; Mviha 2005).
Secondly, whether low rainfall is insufficient for adequate plant growth to maintain and ABC population depends on a number of factors such as soil water-holding capacity and plant water economy (Joern and Gaines 1990) as well as topography. In addition, some plant species require less water for their growth than others so that such plants can survive with limited water availability and may be able to provide more food to maintain ABC numbers than areas dominated by plants with a greater water requirement.
The ecology and behaviour of A. discoidalis is influenced by soil type (Mbata 2004) and by cropping system (Mhiva 2007; Franke et al. 2018) as well as by rainfall pattern. Rainfall has an influence on a large scale which differs both from season to season and place to place. Rainfall effects are complex, influencing crops such as sorghum in Botswana directly (Matsuokwane 2018) and indirectly due to A. discoidalis. Sorhum-legume intercropping has effects on A. discoidalis behaviour (Mviha 2007) and on insect pests and insect predators, more generally (Karabo et al. 2019).
Variation in soil type and topography between and within zones may therefore lead to different ABC population dynamics in different locations. In addition, if aspects of the spatial heterogeneity occur on a small scale, it may be that sub-optimal rainfall conditions and their effects on plant growth could be mitigated by the feeding and oviposition behaviour of the insect. Individuals appear to communicate effectively by calling (Kowalski and Lakes-Harlem 2011) which may enhance aggregation in favourable locations within a habitat. The assumption that ABC population is equally constrained by food shortages throughout its active life stages is a simplification and such shortages in the adult stage may constrain populations more than food shortage during nymphal stages (Joern and Gaines 1990).
The model is driven solely by seasonal rainfall but other climatic variables may be of significant importance. In particular cool, overcast, as opposed to sunny conditions, may slow egg development (Mbata 1993). In turn, this may result in delayed nymph and adult development, leading to a reduction in survival and fecundity if ABC experience food shortage as a result of an extended development period. In sunny conditions, ABC bask actively during the morning and late in the afternoon, thereby elevating their body temperature above ambient. Temperature affects fitness indirectly through increased growth rates, better physiological functions and more predator avoidance (Joern and Gaines 1990). There may therefore be negative effects correlated with high rainfall if it is also accompanied by prolonged overcast conditions.
In southern Africa, Acanthoplus discoidalis is mainly a pest of grain crops. Favouring semi-arid regions, sorghum is particularly affected (Guo 2011). It has also been investigated on other crops such as the oil-seed, Jatropha curcas (Masukujane et al. 2018). In the light of a growing interest in insects as food sources, Acanthoplus spp have also been explored as an animal and human food source so their effect may not be entirely negative (Yen 2009; Green 2002).
Without taking account of the potentially complicating factors discussed above, the fairly straight-forward hypothesis presented here to explain ABC outbreaks had reasonable predictive power. Economic analysis of the costs and benefits of responding to an ABC outbreak forecast of this accuracy would be valuable future work. The model has the potential to play a role to provide an advance warning of increased likelihood of outbreaks to alert farmers to examine natural ABC habitats and take action to protect fields (Green et al. 2003a, b; Green et al. 2004). However, particularly in Zone 5 where the model predicted several outbreak seasons that did not occur it may be that soil, vegetation type or more subtle weather effects had a role. Zone 5, where the model was least accurate, was characterised by hardvelt, rather than the sandvelt of the other zones, and by generally wetter environment than the other zones (de Wit and Bekker 1990). The negative effects of rainfall and soil type on ABC ecology may be a more significant feature in Zone 5 than in the other zones.
References
Bazelet C, Naskrecki P (2014) Acanthoplus discoidalis. The IUCN Red List of Threatened Species 2014: e.T20636721A43266507.http://dx.doi.org/https://doi.org/10.2305/IUCN.UK.2014-1.RLTS.T20636721A43266507.en. [Accessed on 27/11/2017]
Franke AC, van den Brand GJ, Vanlauwe B, Giller KE (2018) Sustainable intensification through rotations with grain legumes in Sub-Saharan Africa: A review. Agr Ecosyst Environ 261:172–185
Green SV, Ekandjo SJ, Musond EM, Mosupi POP, Mitchell J, Mviha PJZ, Minja EM, Cumbi JS (2003) Armoured bush crickets (Orthoptera: Tettigoniidae: Hetrodinae) as agricultural pests - a geographic overview. In: Proceedings of the 14th Entomological Congress. Entomological Society of Southern Africa, Pretoria
Green SV, Mosupi POP, Mosinkie K (2003) Setotojane! - The Armoured Bush Cricket Farmers can provide early warning of an Armoured Bush Cricket outbreak (poster in English and Setswana). Department of Agricultural Research
Green SV, Holt J, Mazhani L, Mitchell J, Mosopi POP, Mviha PJZ, Scholtz C, van den Berg J, Wadhams L (2004) Trench warfare to combat crickets in southern Africa Control of Armoured Bush Cricket in southern Africa. DfID Crop Protection Programme Project Ref: CPP68. Natural Resources Institute
Green SV (2002) Biology and control of armoured bush crickets in Southern Africa. Final Technical Report NRRD Project No. R7428, London: Department for International Development Crop Protection Programme. https://assets.publishing.service.gov.uk/media/57a08d3240f0b649740016e6/R7428_FTR.pdf. [Accessed 10/11/2017]
Guo C, Cui W, Feng X, Zhao J, Guihua Lu (2011) Sorghum insect problems and management. J Integr Plant Biol 53(3):178–192
Hielkema JU (2008) Operational environmental satellite remote sensing for food security and locust control by FAO: The ARTEMIS and DIANA Systems. Food and Agriculture Organization of the United Nations, Rome. https://gcmd.nasa.gov/records/GCMD_CIESIN0122.html. [Accessed 10/11/2017]
Holt J, Day RK (1993) Rule-based models. In: Norton GA, Mumford JD (eds) Decision tools for pest management. CABI, Wallingford, pp 147–158
Holt J, Mushobozi WL, Tucker MR, Venn JF (2000) Modelling African Armyworm Population Dynamics to Forecast Outbreaks. Workshop on Research Priorities for Migrant Pests of Agriculture in Southern Africa, Plant Protection Research Institute, Pretoria, South Africa, 24–26 March 1999 R. Cheke, J. Rosenberg and M Kieser (Eds.) Natural Resources Institute, Chatham
Holt J, Mviha PJZ, Green SV (2003) Prediction of armoured bush cricket (Orthoptera: Tettigoniidae: Hetrodinae) outbreaks. In: Proceedings of the 14th Entomological Congress. Entomological Society of Southern Africa, Pretoria
Joern A, Gaines SB (1990) Population dynamics and regulation in grasshoppers. In: Chapman RF, Joern A (eds) Biology of Grasshoppers. Wiley
Karabo O, Obopile M, Tiroesele B (2019) Insect diversity and population dynamics of natural enemies under sorghum–legume intercrops. Trans Royal Soci South Africa 74(3):258–267
Kowalski K, Lakes-Harlan R (2011) Temporal patterns of intra- and interspecific acoustic signals differ in two closely related species of Acanthoplus (Orthoptera: Tettigoniidae: Hetrodinae). Zoology (jena) 114:29–35
Leuschner K (1995) Insect pests of Sorghum panicles in eastern and southern Africa. In: Proceedings of an International Consultative Workshop 4–7 Oct 1993, Nwanze and Youm (Eds) ICRISAT, Niamey
Maidment R, Black E, Young M (2017) TAMSAT Daily rainfall estimates (Version 3.0). University of Reading. Dataset. https://doi.org/10.17864/1947.112. [Accessed 10/11/2017]
Marsaert H, Mosupi POP, Mviha PJ, Green SV, Minja EM (2000) Survey of farmers indigenous knowledge and control practices against the armoured bush cricket (Setotojane) in central and Francistown agricultural regions of Biotswana. Natural Resources Institute
Masukujane M, Coetzee T, Ngwanathebe RB, Ishimoto Y, Akashi K (2018) Diversity and seasonal variation of insect pests of Jatropha in Gaborone, Botswana. Int J Trop Insect Sci 38(4):294–298
Matsuokwane T (2018) The impact of climate variability and climate change on sorgum grain yield in the Goodhope sub-district of Botswana. MSc Thesis, Department Earth and Environmental Sciences, Faculty of Science, Botswana International University of Science and Technology
Mbata KJ (1993) A Thermal unit model for the development of Acanthoplus Speiseri Brancsik (Orthoptera: Tettigoniidae: Hetrodinae). Int J Trop Insect Sci 14:355–360
Mbata KJ (2004) Soil type selection for oviposition by gravid females of the armoured ground cricket Acanthoplus speiseri Brancsik (Orthoptera: Tettigoniidae), a pest of grain crops in Zambia. Int J Trop Insect Sci 24(4):271–279
McHugh ML (2012) Interrator reliability: the kappa statistic. Biochem Med (zagreb) 22:276–282
Minja EJ (2000) Management of the Armoured bush Cricket in Namibia and Zambia: farmer’s methods. In: Proceedings of the workshop on the management of sorghum and pearl millet pests in the SADC region 10–13 February 1998, EM Minja and J van den Berg, (Eds.) Matopos Research Station and ICRISAT, Zimbabwe
Mosupi POP, van den Berg J, Green SV (2003) Control options against armoured bush crickets (Orthoptera: Tettigoniidae: Hetrodinae). In: Proceedings of the 14th Entomological Congress. Entomological Society of Southrn Africa, Pretoria
Musonda EM (2000) Developing control methods for armoured bush cricket (Acanthoplus speiseri Brancsik) with minimum input of insecticides in Zambia. In: Proceedings of the workshop on the management of sorghum and pearl millet pests in the SADC region, 10–13 February 1998, EM Minja and J van den Berg, (Eds) Matopos Research Station and ICRISAT Zimbabwe
Mviha PJZ, Scholtz CH, Holt J, Green SV (2007) Range of movement of Acanthoplus discoidalis (Walker) (Orthoptera: Ettigoniidae: Hetrodinae) in sole and mixed cropping systems of sorghum (Sorghum bicolor) and cowpeas (Vigna unguiculata L.) in south eastern Botswana, southern Africa. Malawi J Agr Sci 3:17–23
Mviha PJZ, Holt J, Green SV, Mitchell JD (2003) The ecology of the armoured bush cricket Acanthoplus discoidalis (Orthoptera: Tettigoniidae: Hetrodinae), with reference to habitat, behaviour, oviposition site selection and egg mortality. In: Proceedings of the 14th Entomological Congress. Entomological Society of Southern Africa, Pretoria
Mviha PJZ (2005) The ecology of the armoured bush cricket, Ancanthoplus discoidalis (Walker)(Orthoptera:Tetigoniidae). PhD thesis, University of Pretoria URI: http://hdl.handle.net/20.500.11892/175301
Meteorological Office (2021) https://www.metoffice.gov.uk/research/climate/maps-and-data/uk-actual-and-anomaly-maps (Accessed 13 August 2021)
Pike JG (1971) Rainfall and evaporation in Botswana. Technical Document No. 1, FAO/UNDP/SP Project 359. FAO, Rome
Sim J, Wright CC (2005) The Kappa statistic in reliability studies: use, interpretation, and sample size requirements. Phys Ther 85:257–268
Starfield A, Bleloch AL (1986) Building Models for Conservation and Wildlife Management. Macmillan, p 253
Tucker MR (1997) Satellite-derived rainstorm distribution as an aid to forecasting African armyworm outbreaks. Weather 52:204–212
De Wit PV, Bekker RP (1990) Explanatory note on the land systems map of Botswana. Gaborone: FAO UN Development Programme/Government of Botswana. 43 pp. http://www.fao.org/docrep/field/009/ar782e/ar782e.pdf. [Accessed 23/11/2017]
Wohlleber B (1996) First results of research on the armoured bush cricket (Acanthoplus discoidalis) on pearl millet in Namibia: population dynamcis, biology and control. In: Proceedings of the SADC/ICRISAT Regional Sorghum and Pearl Millet Workshop: Drought tolerant crops in southern Africa, 25–29 July 1994, K Leuschner and CS Manthe (Eds) ICRISAT Patancheru, Gaberone
Wohlleber B (2000) Research on the armoured bush cricket (Acanthoplus discoidalis) on pearl millet in Namibia. In: Proceedings of the workshop on the management of sorghum and pearl millet pests in the SADC region, 10–13 February 1998, EM Minja and J van den Berg (Eds.) Matopos Research Station and ICRISAT Zimbabwe
Yen AL (2009) Edible insects: traditional knowledge or western phobia? Entomol Res 39:289–298
Acknowledgements
We thank the Plant Protection Research Institute, Pretoria, RSA and Sebele Research Station, Gaborone, Botswana for their technical support. J. Mitchell of the Plant Protection Institute and P. Mosupi of Sebele Research Station provided much appreciated assistance and comments and Mark Robertson, University of Pretoria reviewed and commented on an earlier draft. This study was part of Project No. R7428 funded by the UK Department for International Development; any views expressed are not necessarily those of DfID. The authors declare that they have no conflict of interest. His co-authors would like to dedicate this paper to the memory of Patrick Mviha.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mviha, P.J.Z., Holt, J. & Green, S.V. Armoured bush cricket, Acanthoplus discoidalis (Walker) (Orthoptera: tettigoniidae) outbreak prediction using rainfall patterns in three zones of Botswana. Int J Trop Insect Sci 42, 1113–1121 (2022). https://doi.org/10.1007/s42690-021-00627-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-021-00627-z