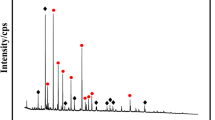
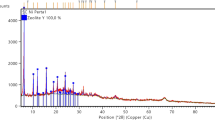
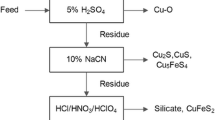
Abstract
This research took the roasted products obtained by leaching copper (Cu) refining slag using waste acids and roasting leaching residues with calcium oxide (CaO) as raw materials and used oxidative leaching to extract Cu from roasted products using waste acids. Influences of the leaching temperature, liquid–solid ratio, and sulfuric acid (H2SO4) concentration in waste acids on the leaching rate of Cu in the oxidative leaching process of roasted products using waste acids were investigated. The unreacted-core shrinking model of heterogeneous reactions was adopted to reveal the leaching kinetics of Cu in the oxidative leaching process of roasted products using waste acids. Results show that the leaching rate of Cu reaches 91.68% when the leaching temperature, liquid–solid ratio, H2SO4 concentration in waste acids, and injected airflow are 80 ℃, 8:1, 1.5 mol/L, and 200 mL/min, respectively. If the leaching temperature is below 80 ℃, the leaching process of Cu is controlled by the solid-film diffusion, and the apparent activation energy Ea is 11.0835 kJ/mol; at a leaching temperature above 80 ℃, the leaching process of Cu is governed by the interfacial chemical reaction, with an apparent activation energy Ea of 42.3183 kJ/mol. The liquid–solid ratio can positively influence the reaction rate constant. The apparent order of the leaching reaction of Cu is 0.632.
















Similar content being viewed by others
Data Availability
All data reported in this study are available upon request by contact with the corresponding author.
References
Roy S, Datta A, Rehani S (2015) Flotation of copper sulphide from copper smelter slag using multiple collectors and their mixtures. Int J Miner Process 143:43–49. https://doi.org/10.1016/j.minpro.2015.08.008
Turan MD, Sari ZA, Miller JD (2017) Leaching of blended copper slag in microwave oven. Trans Nonferrous Met Soc China 27(6):1404–1410. https://doi.org/10.1016/S1003-6326(17)60161-4
Li S, Pan J, Zhu D, Guo Z, Xu J, Chou J (2019) A novel process to upgrade the copper slag by direct reduction-magnetic separation with the addition of Na2CO3 and CaO. Powder Technol 347:159. https://doi.org/10.1016/j.powtec.2019.02.046
Banza AN, Gock E, Kongolo K (2002) Base metals recovery from copper smelter slag by oxidising leaching and solvent extraction. Hydrometal 67(1–3):63–69. https://doi.org/10.1016/S0304-386X(02)00138-X
Khalid MK, Hamuyuni J, Agarwal V, Pihlasalo J, Haapalainen M, Lundström M (2019) Sulfuric acid leaching for capturing value from copper rich converter slag. J Cleaner Prod 215:1005–1013. https://doi.org/10.1016/j.jclepro.2019.01.083
Tian H, Guo Z, Pan J, Zhu D, Yang C, Xue Y, Wang D (2021) Comprehensive review on metallurgical recycling and cleaning of copper slag. Resour Conserv Recycl 168:105366. https://doi.org/10.1016/j.resconrec.2020.105366
Lye CQ, Koh SK, Mangabhai R, Dhir RK (2015) Use of copper slag and washed copper slag as sand in concrete: a state-of-the-art review. Mag Concr Res 67(12):665–679. https://doi.org/10.1680/macr.14.00214
Phiri TC, Singh P, Nikoloski AN (2022) The potential for copper slag waste as a resource for a circular economy: A review–Part I. Miner Eng 180:107474. https://doi.org/10.1016/j.mineng.2022.107474
CaO ZL, Wang ZY (1996) Handbook of inorganic chemical reaction equations. Hunan Science and Technology Press, Changsha
Demirkıran N (2009) Dissolution kinetics of ulexite in ammonium nitrate solutions. Hydrometall 95(3–4):198–202. https://doi.org/10.1016/j.hydromet.2008.05.041
Raza N, Zafar ZI, Kumar RV (2015) Leaching of natural magnesite ore in succinic acid solutions. Int J Miner Process 139:25–30. https://doi.org/10.1016/j.minpro.2015.04.008
RaoS YT, Zhang D, Liu W, Chen L, Hao Z, Wen J (2015) Leaching of low grade zinc oxide ores in NH4Cl–NH3 solutions with nitrilotriacetic acid as complexing agents. Hydrometall 158:101–106. https://doi.org/10.1016/j.hydromet.2015.10.013
Ma A, Zheng X, Li S, Wang Y, Zhu S (2018) Zinc recovery from metallurgical slag and dust by coordination leaching in NH3–CH3COONH4–H2O system. Soc Open Sci 5(7):180660. https://doi.org/10.1098/rsos.180660
Jemal M (2008) Effect of temperature on the attack of fluorapatite by a phosphoric acid solution. Sci Res Essays 3(1):035–039. https://doi.org/10.5897/SRE.9000411
Li H, Zhao L, Wang L, Liang J, Yan H, Liu J (2021) Leaching kinetics of secondary zinc oxide in a NH3–NH4HCO3–H2O System. Crystals 11(5):496. https://doi.org/10.3390/cryst11050496
Funding
This study was supported by Graduate Research and Innovation Projects of Jiangsu Province (CN) (KYCX22_3085).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tang, C.F., Zhang, R.L., Zhang, W. et al. Kinetics Study on the Leaching of Copper from Calcification Roasting Copper Refining Slag Using Waste Acid. Mining, Metallurgy & Exploration 40, 171–179 (2023). https://doi.org/10.1007/s42461-022-00727-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42461-022-00727-5