Abstract
The research was designed to ascertain the potential of bambara groundnut inclusion in wheat bread to improve antioxidant activity, modulate carbohydrate hydrolyzing enzyme activities, and lower glyceamic index/ load. Protein (g/100 g) (11.2—11.73) and energy value (kcal/100 g) (421.5—435.5) of the bread were significantly higher than commercial wheat flour bread (CWF—10.45; 388.7). However, developed experimental bread samples exhibited higher growth performance in rats, free radical scavenging potentials, inhibitory activities against carbohydrate hydrolyzing enzymes and low glycemic index than other bread samples. Nevertheless, experimental bread samples were rated lower compared with the controls samples as regards organoleptic properties. The study authenticates that WBO3—25% wheat, and 75% bamabara groundnut WBO3 exhibits higher potentials as regards nutritional composition, growth indices, free radical scavenging potentials, ability to modulate carbohydrate hydrolyzing enzyme and lower glycemic index/ load. Hence, WBO3 may be recommended as functional bread for hyperglycemia prevention/ management.
Similar content being viewed by others
1 Background
Consumers attitude are tilted toward consumption of foods/ food products known as functional foods which are capable of providing health benefits beyond basic nutritional needs due to the presences of bioactive compounds [1]. Li et al. [2] further established that these foods possess the ability to reduce cardiovascular diseases, and maintain human wellness. Evidence has shown that there is a strong correlation between regular intakes of healthy diets and prevention of several degenerative diseases such as diabetes, hypertension etc. [3]. This has recently necessitated production of varieties of wheat-based functional foods like biscuits and bread enriched with indigenous agricultural crops to boost the functionality, nutritional content and antioxidative compounds.
Bread is a wheat-based baked product, rich in calories and widely consumed across all ages globally [4, 5]. However, it is limiting in protein and micronutrients [6]. Recent times, efforts have been shifted towards production of nutritious and healthy bread by supplementing with legumes [7, 8]. According to Adewale et al. [9] these plant-based food materials contain large amount of bioactive phytochemicals, protein and minerals, and possess medicinal properties. Studies have also shown that to reduce the glyceamic index of wheat-based bread it involves incorporation of dietary fiber either from the whole grains or through the inclusion of other plant-based food materials such as bambara groundnut into the formulation [10, 11]. Dietary fibres helps in reduction of hyperglyceamia and exhibites good therapeutical potential as regards insulin resistance syndrome and lipid oxidation [12, 13]. Hence, help in weight control and diabetic treatment [14].
Bambara groundnut (Vigna subterrenea, L.) is a known plant-based protein source, cultivated for the purpose of animal feeding [15, 16], and production of various local diets, beverage (“akara, moin-moin, okpa”, milk, "kunnu, tuwo”), and for management of diseases [17,18,19]. Recently, there is a shift towards production and consumption of functional foods in order to prevent oxidative stress and associated degenerative diseases like diabetes, hypertension, etc. [20]. In view of this, studies have formulated verities of functional foods from wheat enriched with local food materials [21,22,23]. However, there is scanty information on enriching wheat bread with inclusion of bambara groundnut. Bambara groundnut is included in the present study in bread production to boast the protein and bioactive compounds in experimental bread samples. Therefore, present research aimed at evaluating the potential of bambara groundnut inclusion in wheat bread to improve antioxidant activity, modulate carbohydrate hydrolyzing enzyme activities, and lower glyceamic index/ load.
2 Methods
2.1 Sources of food samples
Wheat, bambara groundnut and orange fruits were sourced from Kings Market, Akure, Ondo state, while Albino Wistar rats were obtained from Colony unit, Animal House, University of Ibadan, Ibadan, Nigeria.
2.2 Production of flour samples and food formulation
2.2.1 Processing of wheat and bambara groundnut flour samples
The raw wheat and bambara groundnut were processed into flour in which the food materials were thoroughly cleaned, dried at 55 °C in an automated electrical oven (Sunshine scientific equipment, 07AFHPN3371D1ZL, New Delhi, India) for 48 h, milled using Waring Commercial Laboratory Blender (Model WF2211210; Chicago, USA) and sieved through no 200 wire mesh sieve (British standard). The flour samples were then store at room temperature (27 °C) until further use for analysis.
2.3 Formulation of flour samples
The wheat and bamabara groundnut flour were blended in different proportions as follows: 75% wheat, and 25% bamabara groundnut (WBO1), 50% wheat, and 50% bamabara groundnut (WBO2), 25% wheat, and 75% bamabara groundnut (WBO3), while commercial wheat flour (CWF) and laboratory wheat flour (LWF) were used as control samples. Each of the wheat-bambara groundnut flour blends was mixed with other ingredients such as cocoa butter (4%), orange peel flour (3%), aspartame (0.5%), egg white (albumin—0.5%), skimmed milk (0.5%) and bread improver (ADA—0.05%).
2.4 Production of wheat-bambara groundnut bread
Each of the flour samples was mixed with water to form dough. The dough was properly prepared according to the method described by Famuwagun et al. [22] with slight modification as regards baking temperature. Dough proofing under controlled temperature (38 °C), for 55 min at 40% relative humidity was monitored for proper baking of dough using a mechanized oven at 220 °C for 15 min and baked bread samples were allowed to cool prior to storage at room temperature (27 °C) until further use for analysis.
2.5 Preparation of aqueous extracts of wheat-bamabara groundnut bread
The bread samples flour (500 g) was extracted exhaustively via maceration for 48 h, with 2.5 L of distilled water with continuous stirring for 48 h [24]. The mixture was centrifuged at 3,500 × g for 20 min. Supernant concentration was done using a Rotary evaporator (Model 349/ 2) Corning Limited at 35 °C for 24 h and thereafter, freeze-dried. The dried extract was stored (27 °C) until required for use.
2.6 Determination of proximate composition and energy values of wheat-bambara groundnut bread
The proximate composition, that is, moisture (AOAC 929.02), protein (AOAC 975.17), fat (AOAC 973.22), crude fibre (AOAC 962.09), and ash (922.02) of wheat-bambara groundnut bread samples were evaluated using the scientific method of AOAC [25]. Meanwhile, carbohydrate content was determined by subtracting values obtained above from 100.
Calories value were calculated using Atwater conversion factors [26].
2.7 Determination of mineral composition and mineral molar ratios of wheat-bambara groundnut bread samples
Selected minerals [Potassium (K), sodium (Na), phosphorus (P), zinc (Zn), copper (Cu), calcium (Ca), and magnesium (Mg)] were evaluated using AOAC [25] methods. While mineral ratios were calculated as earlier stated by Jacob et al. [27].
2.8 Determination of amino acid composition of wheat-bambara groundnut bread samples
Amino acid profile of the bread samples was evaluated scientific method of AOAC [25]. And results were calculated in gram per 100 g of crude protein.
2.9 Determination of anti-nutritional factor of wheat-bambara groundnut bread samples
Tannin content was determined according to the method of Fagbemi et al. [28]. Flavonoid was evaluated using the method described by Boham and Kocipai [29]. Oxalate was determined as described by the modified method Adeniyi et al. [30]. Total alkaloids were evaluated using the method of Harborne [31]. Cardiac glycoside content in the sample was evaluated using Buljet’s reagent as described by El-Olemy et al. [32]. The phenol in the food samples was determined as described by the method of Georgé et al. [33] with minor modifications. The amount of phenolic compounds was expressed as mg of gallic acid per g of extract (mgGAE/g). Steroid content was determined with the method described by Okeke and Elekwa [34].
2.10 Molar ratio of oxalate and phytate to minerals
The moles of oxalate/phytate and minerals were determined using the methods earlier stated by Gemede [35].
2.11 Determination of antioxidative potential of wheat-bambara groundnut bread samples
The free radicals scavenging activity of aqueous extract of the bread samples on 2, 2- Diphenyl-1-picryhydrazyl (DPPH) was determined as described by Girgih et al. [36]. The free radical scavenging activity of the bread samples against 2,2-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid (ABTS) was determined using modified method of Re et al. [37]. The metal chelating activity of bread aqueous extract samples were determined according to the method of Girgih et al. [36]. The hydroxyl radical scavenging activity of the bread aqueous extract samples was determined as described by Girgih et al. [36]. The Ferric-reducing antioxidant power (FRAP) activity of the bread aqueous extract samples was determined using method of Mau et al. [38].
2.12 Carbohydrate hydrolyzing enzymes inhibitory ability of wheat-bambara groundnut bread samples
In vitro α – amylase and α – glucosidase inhibition assay of bread samples was determined using spectrophotometric method described by Worthington [39] and Oboh et al. [40] respectively.
2.13 Nutritional quality and glyceamic index of wheat-bambara groundnut bread samples
Statement of animal rights: The experiment on the animals were carried out in line with the rules and regulations guiding the use of animals as reported by Canadian Council on Animal Care Guidelines and Protocol Review [41].
2.14 Determination of glycemic index and glycemic load of flour blends:
Twenty-four Wistar Albino rats (male and female, body weights = 120 -150 g) obtained from Central Animal House University of Ibadan, Ibadan, Nigeria were grouped (4 rats/group), and housed individually in metabolic cages with free access to feed and water ad libitum. The rats were acclimatized under standard laboratory conditions (22 °C ± 3 °C; 12 h light and dark periods, respectively and humidity- 40–45%) [42] for 7 days. After 7 days acclimatization, the experimental food samples (WBO1, WBO2 and WBO3), control samples (CWF and LWF) and glucose in a portion that was calculated to contain 2.0 g of available carbohydrate were dissolved in warm distilled water (40 °C, 5 mL) and administered to the rats through oral gavage. Immediately after the oral feeding, the initial blood glucose concentration of the rat was measured via the tip tail, while the subsequent readings were taken at the interval of 30 min for 120 min. using an automatic glucose analyzer (‘Accu-Chek Active’ Diabetes monitoring kit; Roche Diagnostic, Indianapolis, USA). The glycaemic Index (GI) (%) for each food sample was calculated as described by Wolever et al. [43]. The Glycemic Load (GL) for each of the food samples was determined as described by Salmerón et al. [44] and categorized as follows:
-
Low-GI = < 55%, Medium-GI = 56—69%, and High-GI = > 70% [45].
-
Low-GL = < 10, Medium-GL = 11—19 and High-GL = > 20 [45].
2.15 Growth performance and biochemical activity in rats of wheat-bambara groundnut bread samples
Nutritional status of rats fed on Wheat-Bambara groundnut bread samples: The anthropometric measurements, i.e., weight and length, of the rats were measured at three days’ interval for 28 days. Length-For-Age (Stunting) and Weight-For-Age (Underweight) of Albino Wistar rats fed on experimental bread and control samples for 28 days were measured.
Biochemical activity of rats fed on wheat-bambara groundnut bread samples: On 28 day of the experimental period, all the rats were starved for about 3 h and weighed. Each rat was anaesthetized with chloroform before been euthanized. Blood was collected into Bijour bottles containing a speck of dried tetracetic ethylenediamine acid powder. The biochemical parameters were analysed using methods described by Jasper et al. [46]. After the experiment, the animals’ carcass was hygienically buried below the soil level as detailed by the study ethical protocol committee. The blood sample was first centrifuged at 1,500 × g for 10 min at ambient temperature. The serum was then separated and used for liver function assessment employing measurements of the enzymes aspartate aminotransferase (AST), alanine aminotransferase (ALT) and Alkaline Phosphate (ALP). Renal function was evaluated using serum concentrations of urea and creatinine. These tests were performed using disposable kits obtained from Labtest Diagnostica S.A. (Lagoa Santa, Minas Gerais, Brazil).
2.16 Determination of sensory attributes of wheat-bambara groundnut bread samples
Sensory attributes of developed bread samples were carried out under standard sensory conditions with respect to lighting and environmental odour using 30 semi-trained personnel. Bread samples were coded using three digits and randomly distributed to personnel for assessment based on product aroma, appearance, texture, and overall acceptability. Product ranking was done using 9-point hedonic scale ranging from 1 = dislike extremely and 9 = like extremely [47].
2.17 Statistical analysis
Data were obtained in triplicate and subjected to analysis using statistical package for social sciences (SPSS) (version 21), expressed as mean ± standard error of mean (SEM) using New Duncan Multiple Range Test (NDMRT) and Graphs were plotted using GraphPad Prism 8. Results were considered to be significant at p ≤ 0.05.
3 Results
3.1 Proximate composition and energy values of wheat-bambara groundnut bread samples
The proximate (g/100 g), minerals (mg/100 g) and calorie constituent (kcal/100 g) of wheat-Bambara groundnut bread (Table 1) shows that the moisture value of the wheat-bambara groundnut bread samples ranged from 20.61 in WBO3—33.22 in WBO1, which are significantly (p < 0.05) lower compared with (35.81) CWF and (38.33) LWF, respectively. The ash content of WBO3 (3.81 g/100 g) was significantly (p < 0.05) higher than WBO1 (2.34 g/100 g) and WBO2 (3.11 g/100 g), respectively. The protein content and energy values of WBO3 (11.73 g/100 g and 435.5 kcal/100 g) were significantly higher than WBO1 (11.12 and 421.5 kcal/100 g) WBO2 (11.6 g/100 g and 431.5 kcal/100 g), respectively; and were significantly (p < 0.05) higher than control samples, that is, CWF (10.45 g/100 g and 388.7 kcal/100 g) and LWF (10.87 g/100 g and 378.1 kcal/100 g).
3.2 Mineral composition and molar ratios of wheat-bambara groundnut bread samples
The mineral composition (mg/100 g) of wheat-bambara groundnut- bread samples ranged as follows: P (1.51—1.69), K (2.88—412), Na (280—361) and Ca (0.30—0.31), while Fe, Zn, Mn and Mg were 0.08—0.1, 0.05—0.06, 0.01—0.02 and 0.99—1.12, respectively (Table 1). These minerals were significantly (p < 0.05) higher in wheat-bambara groundnut bread than in control samples (CWF and LWF). The range values of sodium–potassium (Na:K) and calcium–phosphorous (Ca:P) ratios of the wheat-Bambara groundnut-orange peel based bread samples were 0.88—0.97 and 0.18—0.21, respectively.
3.3 Amino acid composition of wheat-bambara groundnut bread
The amino acid profiles of wheat-bambara groundnut bread samples are presented in Table 2. Glutamic acid (18.48—31.03 mg/100 g protein) was present in abundant concentration, while tryptophan (1.2—1.29 mg/100 g protein) had the lowest concentration in the experimental bread samples, and these values were comparable to that of CWF (32.17 and 1.37 mg/100 g protein) and LWF (30.98 and 1.78 mg/100 g protein). Likewise, a large increase in the content of lysine was observed, which certainly affects the color of the bread and the level of the non-enzymatic browning reaction compounds.
3.4 Anti-nutritional factors and phytate/mineral molar ratios of wheat-bambara groundnut bread samples
The antinutrients in wheat-bambara groundnut bread samples are presented in Table 3. The antinutrients in the formulated bread samples were oxalate (0.59—0.77 mg/g), phytate (11.12—16.89 mg/g), tannin (1.8—3.24 mg/g), glycosides (15.24—22.22 mg/g), flavonoid (1.77—9.63 mg/g), alkaloid (22.55—27.60%) steroid (9.79—12.60 mg/g) and phenol (1.32—2.08 mg/g).
The phytate/calcium, Phytate/Zinc, Phytate/iron and Phytat*Ca/Zn molar ratios ranged from 0.001—0.155, 0.001—0.066, 0.001—0.094 and 0.002 – 0.113, respectively, while that of Oxalate/Calcium ratio ranged from 0.002—0.113.
3.5 In vitro antioxidant activities of wheat-bambara groundnut bread samples
The in vitro antioxidant activities of wheat-bambara groundnut bread samples are shown in Table 4. Antioxidant activity of the formulated bread samples against ABTS, DPPH and OH- free radicals ranged from 7.59—8.05 mmolTEAC/100 g, 1.37 – 2.56 mg/mL and 059—0.77 mg/mL, respectively, while that of FRAP and Fe2+ chelation varied from 25.75—81.93 mgAAE/100 g and 0.54—0.88 mg/mL. Antioxidant activity of the formulated bread samples was significantly (p < 0.05) higher against ABTS, DPPH, OH free radicals, iron chelation and FRAP than control samples compared to experimental bread samples.
3.6 Carbohydrate hydrolyzing enzyme inhibitory activities of wheat-bambara groundnut bread samples
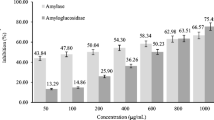
The α-amylase and α-glucosidase enzyme inhibitory activity of the wheat-bambara groundnut bread samples are shown in Fig. 1. The result showed that WBO3 (83.7% and 81%) had highest inhibitory activity against α-glucosidase and α-amylase enzyme, followed by WBO2 (81.9% and 78.4%) and WBO1 (76.8% and 60.6%), respectively.
3.7 Glycaemic index and glycaemic load of wheat-bambara groundnut bread samples
The glycaemic index (GI) and glycaemic load (GL) of the experimental bread samples are presented in Fig. 2. Glycaemic index of the formulated bread samples varied from 35.0 in WBO3 to 55% in WBO1, and the values were significantly (p < 0.05) lower than in the control samples, that is, CWF (60.8%) and LWF (57.9%), respectively. For the GL, the values varied from 17% in WBO3 to 27% in WBO1, and were significantly lower than that of CWF (28.8%) and LWF (28.3%), respectively.
3.8 Growth performance and biochemical activities of rats fed on wheat-bambara groundnut bread samples
Growth performance: The effect of formulated bread samples on the growth performance of rats is presented in Fig. 3. The rats fed on WBO3 sample had highest growth performance when compared with the rats fed on WBO2, WBO1 and control samples (CWF and LWF).
Biochemical properties: The effects of formulated bread samples on the biochemical activities of the liver and kidney with reference to aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), creatinine and urea are presented in Table 5. The ALP, AST, ALT and AST/ALT ratios are useful biomarkers of liver injury and their values in this study ranged from 21.04—33.12 U/L, 45.75—56.13 U/L, 39.17—48.83 U/L and 1.11—1.35, respectively. The bilirubin, creatinine, urea and urea/creatinine ratio are useful biomarkers of heamoglobin breakdown and kidney injury, and the values varied from 0.79—0.88 mg/dL, 0.95—1.38 mg/dL, 0.71—0.85 mg/dL and 0.52—1.02, respectively.
3.9 Sensory attributes of wheat-bambara groundnut bread samples
The sensory attributes of bread formulated from wheat-bambara groundnut flour blends and control samples are presented in Table 6. The results of aroma, appearance and taste ranged from 4.35—7.8, 4.3—8.0 and 2.95—8.1, respectively, while that of texture and overall acceptability were 2.65—7.9 and 3.9—8.0, respectively.
4 Discussion
The moisture content of bread produced in this present study was in line with the wheat-leafy vegetable-based bread [23], wheat- Bambara Groundnut- based bread [7] and wheat-bambara groundnut- yellow cassava-based bread [48]. Food products with high moisture content tends to have rapid reproduction rate of spoilage microorganism, hence reduced storage life. Experimental bread moisture content in this study indicates that the food products can only be stored for a short time.
The ash content indicates that WBO3 might have higher mineral content when compare to other experimental bread samples including controls (CWF, 0.85 g/100 g; LWF, 1.03 g/100 g). The ash contents of wheat-bambara groundnut-orange peel-based bread in this study agreed with values reported for wheat-legume bran-based bread (3.2 -5.4 g/100 g) [21]; but, higher than what reported for wheat-bambara groundnut-based bread (1.71–2.44 g/100 g) [7] and that of wheat-leafy vegetable-based bread (1.1 – 2.4 g/100 g) [23]. The variation in ash contents of these products mighty be attributed to differences in food materials, climatic condition and processing techniques.
The protein content and energy value of the bread samples were observed to be increasing as the level of bambara groundnut added increased. This finding could be as a result of inclusion effect between protein, fat and carbohydrate content of the wheat and Bambara groundnut. For instance, study has shown that bambara groundnut contains between 18–24% of protein, while wheat contain 11–12% protein [49]. The protein content and energy value of bread samples in this present study were similar to the reports of Yusufu and Ejeh [7] and that of Odunlade et al. [23]. The high protein and energy of the bread samples in this study mighty be useful to prevent protein-energy malnutrition in all age groups, particularly among low-income people who cannot afford expensive quality diets [49].
The findings observed in minerals contents of experimental bread samples may be associated with inclusion effects of mineral composition of the blended raw materials, i.e., wheat and bambara groundnut [50]. One major nutrients required for growth in children and healthy well-being in adults are minerals. Hence, these formulated bread samples in this study are rich in minerals that could promote such good health for both children and adults [51].
The mineral ratios are nutritional indices, which provides information on the interrelationships of dietary minerals regarding their status and disease states [27]. Na:K ratio of the experimental bread samples was less than recommended value (< 1) [27]. Nutritionally, it could be deduced that the bread samples contained higher amount of potassium relatively to low sodium, hence, regular consumption of these bread samples may be suitable for management of hypertension [52, 53]. Ca:P of experimental bread were less than recommended values (< 0.5 poor; > 1 good) [52]. This indicates that the formulated bread samples were low in phosphorous relatively to calcium. Higher calcium-phosphorous content is recommended in diets for effective calcium metabolism and utilization in the body [54], likewise, diet containing protein and phosphorus may enhance calcium wastage via urine [55]. The low Ca/P obtained in experimental bread samples has implications nutritionally, as regards the growing-aged infants and adults, in need of high intake of calcium and phosphorus for strong bone and teeth formation and prevention of osteoporosis. Hence, consumer of experimental bread samples may need to source for calcium supplements to avoid deformation of bones.
The glutamic acid concentration in the bread samples was observed to be statistically (P < 0.05) higher compared with other amino acids evaluated, and this observation is similar to Ndungu et al. [56] and Wiedemair et al. [57], reports that glutamic acid is abundant in plant-based food products. The aromatic, hydrophobic, branched chain, essential amino acids and Arginine/lysine ratios of the formulated bread samples ranged from 9.32—10.31, 39.09—42.93, 14.79—15.77, 31.11—40.53 mg/100 g protein and 0.95—1.46, respectively, and these values were statistically (P < 0.05) higher in the formulated bread samples compared with control samples, except for Arginine/lysine ratio. Constant consumption of these amino acids may prevent chronic diseases like type-2 diabetes and high blood pressure [58, 59]. For instance, arginine intakes help in secretion of nitric oxide, which relax arteries for easy transfer of blood, hence prevent the occurrence of hypertension [58]. Comparatively, total essential amino acids in experimental bread samples were higher compared with wheat-based bread enriched with Oyster Mushroom reported by Ndungu et al. [56]. This may be associated to high essential amino acids in legume (bambara groundnut) than Oyster Mushroom.
The antinutrients in experimental bread samples were either lower or higher than in control samples. However, the contents of antinutrients in experimental bread samples are within the tolerable levels. Antinutrients are generally toxic at high concentration, but have some health benefits at lower concentration. For instance, phytate has ability to chelate with divalent cations resulting into formation of insoluble complexes with proteins at elevated pH [60, 61], thereby impair digestibility and bioavailability of these vital nutrients in humans and/or animals. It is well established that some of these antinutrients have antioxidant activities against free radicals’ formation in humans [62]; they help scavenge free radicals and prevent oxidative stress hence, preventing chronic diseases like cancer, hypertension and diabetes [63].
The phytate and divalent minerals molar ratios (an index of mineral bioavailability) of the formulated bread samples were calculated.
The phyatate/ mineral molar ratios of the experimental bread samples were significantly lower than LWF, but higher than in CWF, except for Phytate/Calcium and Phytate/iron molar ratios. However, these values were comparably lower than the critical molar ratio values (Phyt/Ca, < 0.24; Phyt/Zn, < 10; Phyt/Fe, > 0.15; Phytate*Ca/Zn, 0.5 and Oxalate/Calcium, < 1), indicating high absorption and bioavailability of calcium, zinc and iron in the formulated bread samples [64,65,66,67]. The Oxalate/Calcium molar ratio was less than one in this study; this is of significance since the amount of oxalate in the bread sample may not interfere with the absorption of calcium. Oxalate in foods may possess negative effects on human health, by reducing the rate of calcium metabolism as well as facilitating kidney stones occurrence [64].
Antioxidants, are chemical substance that acts as oxidation reaction inhibitors and as such prevents the production of free radicals, and plays major roles in preventing oxidative stress and associated chronic diseases like hypertension, diabetes, obesity and cancers [68, 69]. The antioxidant activity of these formulated bread samples indicates that regular intakes of the food products could inhibit free radicals’ formation and thereby preventing oxidative stress occurrence as well as hypertension, diabetes etc.
The enzyme inhibitory activities of the formulated bread samples were observed to be increasing with inclusion of bambara groundnut, which may be attributed to the additive effects of bioactive components in Bambara groundnut, wheat and orange peel. Comparatively, the enzyme inhibitory power of experimental bread samples was statistically (p < 0.05) higher compared with CWF (50.7% and 51.7%) and LWF (71.5% and 49.9%). The difference between the enzyme inhibitory activities of the formulated bread samples and the control could be due to inhibitory activities of bambara groundnut flour included in the production of experimental bread samples which is absent in control samples. The carbohydrate hydrolyzing enzyme inhibitory activities of the formulated bread samples may be of health benefits, particularly to control overweight/obesity and high blood glucose in diabetes. It is evident that α-amylase-glucosidase enzyme inhibition from hydrolyzing carbohydrate are the main strategy to prevent obesity and diabetes [70]. α-amylase is the enzyme that hydrolyse α-(1,4)-D-glycosidic linkages of starch to disaccharides and oligosaccharides, while α-glucosidase hydrolyse oligosaccharides to monosaccharides (glucose) [71]. Inhibition of these enzymes usually leads to prevention of starch from breakdown and thereby lowering blood glucose levels [70, 71].
In this study, it was recorded that the GI of experimental bread samples reduce as the proportion of bambara groundnut inclusion increase. This finding may be associated to difference in carbohydrate properties of the blending food materials. It is well established that mixing two or more carbohydrates such as cereals (wheat) of higher GI value and legumes (bambara groundnut) of lower GI value may resulted into modification of the physical property and chemical composition of the resulting foods [72]. Hence, reducing the GI of the food blends, which may be of nutritional benefits to disorders like diabetes. The GI values of experimental bread samples in this study were lower than what obtained for whole grain based multigrain Indian bread samples (Rotis) (63.2—66.2%) reported by Nagaraju et al. [73], and also, lower than recommended value for low GI foods (< 55%). This indicates that the formulated bread samples, particularly WBO3, may be recommended as functional bread for the prevention of weight gain and hyperglycaemia in diabetes. Foods with GI values in the range of < 55, 55—69, and > 70 are classified as low, medium, and high GI foods, respectively [73]. Low GI and GL foods play vital roles in the management of diabetes [74] and overweight/obese [75].
The high growth performance of rats that were fed on WBO3 could be attributed to additive effects of protein, essential amino acids and energy value of the bread products. It is well established that a food that deficient in quality protein, energy value and micronutrient may lead to development of malnutrition, hence, hindering growth and development in humans or animals. Epidemiological studies have implicated deficient of protein and energy in foods as a major contributory factor to high prevalence of protein-energy malnutrition in developing countries [76,77,78]. Nutritionally, the growth performance of rats fed on the formulated bread samples, particularly WBO3, is an indication that the bread products are of nutritional quality, and that regular intakes of the bread products could prevent protein related diseases in children and adults.
The values of bilirubin, creatinine, urea and urea/creatinine enzymes in this study are either lower or within the normal recommended values. Evidences have shown that values above the normal reference of ALP, AST, ALT, urea and creatinine indicate haemoglobin, liver and kidney breakdown and dysfunction, respectively [79,80,81]. It is well established increased serum AST and ALT are associated with internal tissue damaged. Hence, AST/ALT ratio (> 1.5) are termed injured viral hepatitis [82]. Creatinine are kidney waste products in which elevated serum concentration is associated with poor kidney efficiency. The low concentration of these enzymes in experimental animal fed on the form with experimental bread samples implies safety for consumption.
The scoring of sensory attributes of experimental bread samples with respects to assessed parameters decrease from WBO1 to WBO3, and this observation could be associated to inclusion of bambara groundnut in bread samples. Sensory attributes of experimental bread samples were statically (p < 0.05) rated lower compared to LWF and CWF, respectively. This finding could be attributed to the familiarity of the panelists to the commercial bread (i.e., wheat-based bread), bread composition or processing techniques. Rating WBO1 by the panelists above other formulated bread samples in sensory attributes indicates that inclusion of 25% Bambara groundnut in wheat bread samples could be acceptable. This finding agreed with the report of wheat-based biscuit and bread enriched with Bambara groundnut-orange peel and waxy rice- wheat respectively [83, 84]. However, the texture of the experimental bread samples was poorly rated lower compared to the control samples which may be attributed to the inclusion of bambara groundnut flour.
5 Conclusion
The study developed and evaluated nutritional quality, antioxidant, carbohydrate enzyme inhibitory activity and glycaemic index of bread products from wheat, and bambara groundnut flour blends. The finding established that the formulated bread samples, particularly WBO3 (75% wheat, and 25% bambara groundnut) exhibited high nutritional quality, free radical scavenging potentials, carbohydrate enzyme inhibitory activity and low glyceamic index. Hence, this bread sample may be suitable to prevent hyperglycaemia and oxidative stress. However, there is a need for clinical study to substantiate the therapeutical potentials of the bread products.
Data availability
Data are available upon request by contacting the authors.
Abbreviations
- WBO1 :
-
75% Wheat, and 25% bambara groundnut
- WBO2 :
-
50% Wheat, and 50% bambara groundnut
- WBO3 :
-
75% Wheat, and 25% bambara groundnut
- CWF:
-
Commercial wheat flour
- LWF:
-
Laboratory wheat flour
- AOAC:
-
Association of Official Analytical Chemist
- DPPH:
-
2, 2- Diphenyl-1-picryhydrazyl
- ABTS:
-
2,2-Azinobis-(3-ethylbenzothiazoline-6-sulfonic acid
- OH:
-
Hydroxyl radical
- FRAP:
-
Ferric-reducing antioxidant power
- CCAC:
-
Canadian Council on Animal Care Guidelines and Protocol Review
- GL:
-
Glycemic Load
- GI:
-
Glycemic Index
- PCV:
-
Packed cell volume
- RBC:
-
Red blood cell count
- Hb:
-
Hemoglobin
- MCHC:
-
Mean corpuscular hemoglobin concentration
- MCH:
-
Mean corpuscular hemoglobin
- MCV:
-
Mean corpuscular volume
- AST:
-
Aspartate aminotransferase
- ALT:
-
Alanine aminotransferase
- ALP:
-
Alkaline Phosphate
- WFA:
-
Weight-for-age
- LFA:
-
Length-for-age
References
Meléndez-Martínez AJ, Böhm V, Borge GIA, Cano MP, Fikselová M, Gruskiene R, O’Brien NM (2021) Carotenoids: Considerations for Their Use in Functional Foods, Nutraceuticals, Nutricosmetics, Supplements, Botanicals, and Novel Foods in the Context of Sustainability, Circular Economy, and Climate Change. Ann RevFood Sci Technol 12:433–460
Li Y, He D, Li B, Lund MN, Xing Y, Wang Y, Li L (2021) Engineering polyphenols with biological functions via polyphenol-protein interactions as additives for functional foods. Trends Food Sci Technol
Oluwajuyitan TD, Ijarotimi OS, Fagbemi TN, Oboh G (2021) Blood glucose lowering, glycaemic index, carbohydrate-hydrolysing enzyme inhibitory activities of potential functional food from plantain, soy-cake, rice-bran and oat-bran flour blends. J Food Measurem Characterizat. 1–9
Bolarinwa IF, Tawakalitu EA, Akeem OR (2017) Nutritive value and acceptability of bread fortified with moringa seed powder. J Saudi Soc Agric Sci 18:195–200. https://doi.org/10.1016/j.jssas.2017.05.002
Mollakhalili-Meybodi N, Arab M, Nematollahi A, Khaneghah AM (2021) Prebiotic Wheat Bread: Technological, Sensorial and Nutritional perspectives and Challenges. LWT, 111823
Ameh MO, Dick IG, Bibiana DI (2013) Physico-chemical and sensory evaluation of wheat bread supplemented with stabilized undefatted rice bran. Food Nutri Sci 4:43. https://doi.org/10.4236/fns.2013.49A2007
Yusufu MI, Ejeh DD (2018) Production of bambara groundnut substituted whole wheat bread: Functional properties and quality characteristics. J Nutri Food Sci 8:731
Bajka BH, Pinto AM, Ahn-Jarvis J, Ryden P, Perez-Moral N, Van Der Schoot A, Edwards CH (2021) The impact of replacing wheat flour with cellular legume powder on starch bioaccessibility, glycaemic response and bread roll quality: A double-blind randomised controlled trial in healthy participants. Food Hydrocol 114:106565
Adewale OO, Brimson JM, Odunola OA, Gbadegesin MA, Owumi SE, Isidoro C, Tencomnao T (2015) The potential for plant derivatives against acrylamide neurotoxicity. Phytoth Resear 29:978–985. https://doi.org/10.1002/ptr.5353
Marangoni F, Poli A (2008) The glycemic index of bread and biscuits is markedly reduced by the addition of a proprietary fiber mixture to the ingredients. Nutri Metabol Cardiovasc Dis 2008:602–605. https://doi.org/10.1016/j.numecd.2007.11.003
Prabhakar PK (2021) Nutritional composition, anti-nutritional factors, pretreatments-cum-processing impact and food formulation pote. LWT-Food Sci Technol 138:110796
Livesey G, Taylor R, Hulshof T, Howlett J (2008) Glycemic response and health—a systematic review and meta-analysis: relations between dietary glycemic properties and health outcomes. Am J Clin Nutri 87:258S-268S. https://doi.org/10.1093/ajcn/87.1.258S
Lopes da Silva M, de Càssia Gonçalves Alfenas R (2011) Efecto del índice glucémico sobre la oxidación lipídica y la composición corporal. Nutri Hospit 26:48–55. http://scielo.isciii.es/pdf/nh/v26n1/revision_2.pdf
Esfahani A, Wong JM, Mirrahimi A, Srichaikul K, Jenkins DJ, Kendall CW (2009) The glycemic index: physiological significance. J Am Coll Nutri 28:439S-445S. https://doi.org/10.1080/07315724.2009.10718109
Adeleke OR, Adiamo OQ, Fawale OS (2018) Nutritional, physicochemical, and functional properties of protein concentrate and isolate of newly-developed Bambara groundnut (Vigna subterrenea L.) cultivars. Food Sci Nutri 6:229–242. https://doi.org/10.1002/fsn3.552
Chinma CE, Abu JO, Asikwe BN, Sunday T, Adebo OA (2021) Effect of germination on the physicochemical, nutritional, functional, thermal properties and in vitro digestibility of Bambara groundnut flours. LWT 140:110749
Atiku S, Mohammed K (2004) Cultivation of Bambara groundnut in the Nigeria. Ahmedu Bello University, Zaria: Report of a field study, 2004
Adebowale YA, Adeyemi IA, Oshodi AA (2005) Functional and physicochemical properties of flours of six mucuna species. Afri J Biotechnol 4: 416– 468. http://www.academicjournals.org/AJB
Piyarat S (2008) Structure and functional properties of starch and flour from Bambara groundnut. Songklanak J Sci Technol 30, 51– 56. http://rdo.psu.ac.th/sjst/journal/30-Suppl-1/0125-3395-30-S1-51-56.pdf
Krawęcka A, Sobota A, Sykut-Domańska E (2019) Functional Cereal Products in the Diet for Type 2 Diabetes Patients. Intern J Food Sci 2019. https://doi.org/10.1155/2019/4012450
Elawad RMO, Yang TA, Ahmed AHR, Ishag KEA, Mudawi HA, Abdelrahim SMK (2016) Chemical composition and functional properties of wheat bread containing wheat and legumes bran. Intern J Food Sci Nutri 1: 10–15. http://www.foodsciencejournal.com/
Famuwagun AA, Taiwo KA, Gbadamosi SO, Oyedele DJ (2016) Optimization of production of bread enriched with leafy vegetable powder. J Food Process Technol 7: 1–7. http://hdl.handle.net/10625/55661
Odunlade TV, Famuwagun AA, Taiwo KA, Gbadamosi SO, Oyedele DJ, Adebooye OC (2017) Chemical composition and quality characteristics of wheat bread supplemented with leafy vegetable powders. J Food Qual 2017:1–8. https://doi.org/10.1155/2017/9536716
Malik VS, Li Y, Tobias DK, Pan A, Hu FB (2015) Dietary protein intake and risk of type 2 diabetes in US men and women. Diab 64:A424–A424
AOAC (2012) Association of Official Analytical Chemist. Official Methods Analysis Analytical Chemist Intern 18th ed. Gathersburg, MD USA
Nguyen TTT, Loiseau G, Icard-Vernière C, Rochette I, Trèche S, Guyot JP (2007) Effect of fermentation by amylolytic lactic acid bacteria, in process combinations, on characteristics of rice/soybean slurries: A new method for preparing high energy density complementary foods for young children. Food Chem 100:623–631. https://doi.org/10.1016/j.foodchem.2005.09.080
Jacob AG, Etong DI, Tijani A (2015) Proximate, mineral and anti-nutritional compositions of melon (Citrullus lanatus) seeds. Bri J Res 2:142–151
Fagbemi TN, Oshodi AA, Ipinmoroti KO (2005) Processing effects on some antinutritional factors and in vitro multienzyme protein digestibility (IVPD) of three tropical seeds: breadnut (Artocarpus altilis), cashewnut (Anacardium occidentale) and fluted pumpkin (Telfairia occidentalis). Pak J Nutri 4:250–256. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.567.6498andrep=rep1andtype=pdf
Boham BA, Kocipai-Abyazan R (1994) Medicinal and Poisonous Plant of Southern and Eastern Africa, 2nd edn. Livingstone, Edinburgh
Adeniyi SA, Orjiekwe CL, Ehiagbonare JE, Arimah BD (2010) Preliminary phytochemical analysis and insecticidal activity of ethanolic extracts of four tropical plants (Vernonia amygdalina, Sida acuta, Ocimum gratissimum and Telfaria occidentalis) against beans weevil (Acanthscelides obtectus). Inter J Phys Sci 5(6):753–762. https://doi.org/10.5897/IJPS.9000459
Harborne JB (1973) Phenolic compounds. In Phytochemical methods (pp. 33–88). Springer, Dordrecht
El-Olemy MM, Al-Muhtadi FJ, Afifi AFA (1994) Experimental phytochemistry. A laboratory manual. King Saud University Press
Georgé S, Brat P, Alter P, Amiot MJ (2005) Rapid determination of polyphenols and vitamin C in plant-derived products. J Agric Food Chem 53:1370–1373. https://doi.org/10.1021/jf048396b
Okeke CU, Elekwa I (2003) Phytochemical study of the extract of Gongronema latifolium Benth. J Health Visual Sci 5:47–55. https://www.ajol.info/index.php/jhvs/article/view/53091
Gemede HF (2020) Nutritional and antinutritional evaluation of complementary foods formulated from maize, pea, and anchote flours. Food Sci Nutri 8:2156–2164. https://doi.org/10.1002/fsn3.1516
Girgih AT, Udenigwe CC, Aluko RE (2011) In vitro antioxidant properties of hemp seed (Cannabis sativa L.) protein hydrolysate fractions. J Am Oil Chem Soc 88:381–389. https://doi.org/10.1007/s11746-010-1686-7
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Rad Biol Med 26:1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Mau JL, Lin HC, Song SF (2002) Antioxidant properties of several speciality mushrooms. Food Res Inter 35:519–526. https://doi.org/10.1016/S0963-9969(01)00150-8
Worthington K (1993) Alpha amylase: In Worthington enzyme manual. Freehold, NJ: Worthington Biochemical Corp, 36–41
Oboh G, Ademiluyi AO, Akinyemi AJ, Henle T, Saliu JA, Schwarzenbolz U (2012) Inhibitory effect of polyphenol-rich extracts of jute leaf (Corchorus olitorius) on key enzyme linked to type 2 diabetes (α-amylase and α-glucosidase) and hypertension (angiotensin I converting) in vitro. J Funct Foods 4:450–458. https://doi.org/10.1016/j.jff.2012.02.003
CCAC (1993). Canadian Council on Animal Care. In: Olfert, E.D., Cross, B.M., McWilliam, A.A. (Eds.), Guide to the Care and Use of Experimental Animals. second ed., 1 CCAC, Ontario. https://ci.nii.ac.jp/naid/10005104578/
Oluwajuyitan TD, Ijarotimi OS, Fagbemi TN (2020) Nutritional, biochemical and organoleptic properties of high protein-fibre functional foods developed from plantain, defatted soybean, rice-bran and oat-bran flour. Nutri Food Sci. https://doi.org/10.1108/NFS-06-2020-0225
Wolever TM, Jenkins DJ, Jenkins AL, Josse RG (1991) The glycemic index: methodology and clinical implications. Am J Clin Nutri 54:846–854. https://doi.org/10.1093/ajcn/54.5.846
Salmerón J, Ascherio A, Rimm EB, Colditz GA, Spiegelman D, Jenkins DJ, Willett WC (1997) Dietary fiber, glycemic load, and risk of NIDDM in men. Diab care 20:545–550. https://doi.org/10.2337/diacare.20.4.545
Dona AC, Guilhem P, Robert GG, Philip WK (2010) Digestion of starch: in vivo and in vitro kinetic models used to characterize oligosaccharide or glucose release. Carbohyd Polymer 80:599–617. https://doi.org/10.1016/j.carbpol.2010.01.002
Jasper R, Locatelli OG, Pilati C, Locatelli C (2012) Evaluation of biochemical, haematological and oxidative parameters in mice exposed to the herbicide glyphosate-Roundup. Interdisciplin Toxicol 5:133–140. https://doi.org/10.2478/v10102-012-0022-5
Olapade AA (2014) Chemical and sensory evaluation of African Breadfruit (Treculia africana) seeds processed with alum and trona. Niger Food J 32:80–88. https://doi.org/10.1016/S0189-7241(15)30099-0
Okoye EC, Ezeugwu EH (2019) Production, Quality Evaluation and Acceptability of Bread from Wheat, Bambara Groundnut and Yellow Root Cassava Flours. Inter J Food Biosci 2: 11–17. https://www.innovationinfo.org/articles/IJFB/IJFB-2-112.pdf
Basman A, Köksel H, Ng PKW (2003) Utilization of transglutaminase to increase the level of barley and soy flour incorporation in wheat flour breads. J Food Sci 68:2453–2460. https://doi.org/10.1111/j.1365-2621.2003.tb07045.x
Mi Y, Ejeh DD (2018) Production of Bambara Groundnut Substituted Whole Wheat Bread: Functional Properties and Quality Characteristics. J Nutri 8:1000731. https://doi.org/10.4172/2155-9600.1000731
Amoakoah TL, Kottoh ID, Asare IK, Torby-Tetteh W, Buckman ES, Adu‐Gyamfi A (2015) Physicochemical and elemental analyses of bananas composite flour for infants. Bri J Appl Sci Technol 6(3), 277–284. http://www.sciencedomain.org/review-history.php?iid=766andid=5andaid=7345
Alinnor IJ, Oze R (2011) Chemical evaluation of the nutritive value of Pentaclethra macrophylla benth (African Oil Bean) seeds. Pak J Nutri 10: 355–359. http://docsdrive.com/pdfs/ansinet/pjn/2011/355-359.pdf
Perez V, Chang ET (2014) Sodium-to-potassium ratio and blood pressure, hypertension, and related factors. Adv Nutri Inter Review J 5:712–741. https://doi.org/10.3945/an.114.006783
Adeyeye EI, Orisakeye OT, Oyarekua MA (2012) Composition, mineral safety index, calcium, zinc and phytate interrelationships in four fast-foods consumed in Nigeria. Bull Chem Soci Ethiop 26:233–241. https://doi.org/10.4314/bcse.v26i1.5
Adeoti OA, Elutilo OO, Babalola JO, Jimoh KO, Azeez LA, Rafiu KA (2013) Proximate, mineral, amino acid and fatty acid compositions of maize tuwo-cirina forda flour blends. Greener J Biol Sci 3:165–171
Ndungu SW, Christina AO, Calvin O, Fredrick M (2015) Nutritional Composition, Physical Qualities and Sensory Evaluation of Wheat Bread Supplemented with Oyster Mushroom. Am J Food Technol 10:279–288
Wiedemair V, Scholl-Bürgi S, Karall D, Huck CW (2020) Amino Acid Profiles and Compositions of Different Cultivars of Panicum miliaceum L. Chromatograp 83:829–837. https://doi.org/10.1007/s10337-020-03899-8
Tejero J, Biswas A, Wang ZQ, Page RC, Haque MM, Hemann C, Stuehr DJ (2008) Stabilization and characterization of a heme-oxy reaction intermediate in inducible nitric-oxide synthase. J Biol Chem 283: 33498–33507. https://www.jbc.org/content/283/48/33498.short
Floegel A, Stefan N, Yu Z, Mühlenbruch K, Drogan D, Joost HG, Fritsche A, Häring HU, de Angelis MA, Peters A, Roden M, Prehn C, Wang-Sattler R, Illig T, Schulze MB, Adamski J, Boeing H, Pischon T (2013) Identification of serum metabolites associated with risk of type 2 diabetes using a targeted metabolomic approach. Diab 62:639–648
Carnovale E, Lugaro E, Lombardi-Boccia G (1988) Phytic acid in faba bean and pea: effect on protein availability. Cereal Chem 65:114–117
Tavajjoh M, Yasrebi J, Karimian N, Olama V (2011) Phytic acid concentration and phytic acid: Zinc molar ratio in wheat cultivars and bread flours Fars province, Iran. J Agric Sci Technol 13: 743–755. https://www.sid.ir/en/journal/ViewPaper.aspx?ID=216794
Liu Z, Ren Z, Zhang J, Chuang CC, Kaswamy E, Zhou T, Zuo L (2018) Role of ROS and nutritional antioxidants in human diseases. Front Physiol 9:477
Janeb M, Thompson LU (2002) Role of Phytic Acid in Cancer and Other Diseases. In: Reddy NR, Sathe SK (eds) Food Phytates. CRC Press, Boca Raton, pp 225–248
Bhandari MR, Kawabata J (2004) Assessment of antinutritional factors and bioavailability of calcium and zinc in wild yam (Dioscorea spp.) tubers of Nepal. Food Chem 85:281–287. https://doi.org/10.1016/j.foodchem.2003.07.006
Frontela C, Ros G, Martínez C (2009) Effect of dephytinization on bioavailability of iron, calcium and from infant cereals assessed in the Caco-2 cell model. World J Gastroenterol 15:1977–1984. https://doi.org/10.3748/wjg.15.1977
Woldegiorgis AZ, Abate D, Haki GD, Ziegler GR (2015) Major, minor and toxic minerals and anti-nutrients composition in edible mushrooms collected from Ethiopia. J Food Process Technol 6:234–244. https://doi.org/10.4172/2157-7110.1000430
Gemede HF, Haki GD, Beyene F, Woldegiorgis AZ, Rakshit SK (2016) Proximate, mineral, and antinutrient compositions of indigenous Okra (Abelmoschus esculentus) pod accessions: implications for mineral bioavailability. Food Sci Nutri 4:223–233. https://doi.org/10.1002/fsn3.282
Lillioja S, Neal AL, Tapsell L, Jacobs DR Jr (2013) Whole grains, type2 diabetes, coronary heart disease, and hypertension: links to the aleurone preferred over indigestible fiber. Biofact 39:242–258. https://doi.org/10.1002/biof.1077
Sharma T, Kanwar SS (2018) Phytomolecules for Obesity and Body Weight Management”. J Biochem Cell Biol 1(1):1–8
Mahmood N (2016) A review of α-amylase inhibitors on weight loss and glycemic control in pathological state such as obesity and diabetes. Compreh Clin Pathol 25:1253–1264. https://doi.org/10.1007/s00580-014-1967-x
Watcharachaisoponsiri T, Sornchan P, Charoenkiatkul S, Suttisansanee U (2016) The α-glucosidase and α-amylase inhibitory activity from different chili pepper extracts. Inter Food Res J 23: 1439–1445. http://ifrj.upm.edu.my/23%20(04)%202016/(13).pdf
Abdullah MT, Adnan AMMA (2010) Blood glucose response and glycemic index of diets containing different sources of carbohydrate in healthy rats. Mesopot J Agric 38: 1–11. https://magrj.mosuljournals.com/article_27736_3dc1ef2fee464d096c22e0299e9ed6fc.pdf
Nagaraju R, Sobhana PP, Thappatla D, Epparapalli S, Kandlakunta B, Korrapati D (2020) Glycemic Index and Sensory Evaluation of Whole Grain Based Multigrain Indian Breads (Rotis). Prev Nutri Food Sci 25:194–202. https://doi.org/10.3746/pnf.2020.25.2.194
Rizkalla SW, Taghrid L, Laromiguiere M, Huet D, Boillot J, Rigoir A, Slama G (2004) Improved plasma glucose control, whole-body glucose utilization, and lipid profile on a low-glycemic index diet in type 2 diabetic men: a randomized controlled trial. Diab Care 27:1866–1872. https://doi.org/10.2337/diacare.27.8.1866
McMillan-Price J, Petocz P, Atkinson F, O’Neill K, Samman S, Steinbeck K, Brand-Miller J (2006) Comparison of 4 diets of varying glycemic load on weight loss and cardiovascular risk reduction in overweight and obese young adults: a randomized controlled trial. Arch Inter Med 166:1466–1475. https://doi.org/10.1001/archinte.166.14.1466
Daelmans B, Saadeh R (2003) Global initiatives to improve complementary feeding. In: Newsletter SCN (ed) Meeting the Challenge to Improve Complementary Feeding. Lavenham Press, UK, pp 10–70
Caballero B, Allen L, Prentice A (eds) (2005) Encyclopedia of Human Nutrition, 2nd edn. Elsevier Academic Press, Oxford
Rolfes SR, Pinna K, Whitney E (2008) Understanding Normal and Clinical Nutrition, 8th edn. Wadsworth Cengage Learning, Canada
Toya K, Babazono T, Hanai K, Uchigata Y (2014) Association of serum bilirubin levels with development and progression of albuminuria, and decline in estimated glomerular filtration rate in patients with type 2 diabetes mellitus. J Diab Invest 5:228–235. https://doi.org/10.1111/jdi.12134
Aliyu R, Adebayo AH, Gasting D, Garba IH (2007) The effects of ethanolic leaf extract of Commiphora Africana (Burseraceae) on rat liver and kidney functions. J Pharmacol Toxicol 2: 373–379. http://eprints.covenantuniversity.edu.ng/277/1/Publication_on_Commiphora_africana_JPT.pdf
Thammitiyagodage MG, de Silva NR, Rathnayake C, Karunakaran R, Kumara WGSS, Gunatillka MM, Thabrew MI (2020) Biochemical and histopathological changes in Wistar rats after consumption of boiled and un-boiled water from high and low disease prevalent areas for chronic kidney disease of unknown etiology (CKDu) in north Central Province (NCP) and its comparison with low disease prevalent Colombo. Sri Lanka BMC Nephrol 21:38
Botros M, Sikaris KA (2013) The de ritis ratio: the test of time. Clin Biochem Rev 34: 117–130. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3866949/
Nakamura S, Suzuki K, Ohtusbo K (2009) Characteristics of bread prepared from wheat flours blended with various kinds of newly developed rice flours. J Food Sci 74:121–130. https://doi.org/10.1111/j.1750-3841.2009.01088.x
Adefegha SA, Oboh G (2013) Sensory Qualities, Antioxidant Activities, and in vitro Inhibition of Enzymes Relevant to Type-2 Diabetes by Biscuits Produced from 5 Wheat-Bambara Groundnut Flour Blends. Inter J Food Engin 9:17–28. https://doi.org/10.1515/ijfe-2012-0182
CODEX CAC, GL 08, (1991) Codex Alimentarius: Guidelines on Formulated Supplementary Foods for Older Infants and Young Children. FAO/WHO Joint Publications 4:144
Morris ER, Ellis R (1985) Usefulness of the dietary phytic acid/zinc molar ratio as an index of zinc bioavailability to rats and humans. Biolog Trace Elem Res 19:107–117
Hurrell RF (2003) Influence of vegetable protein sources on trace elements and minerals bioavailability. J Nutri 133:2973S-2977S. https://doi.org/10.1093/jn/133.9.2973S“\t”_blank“10.1093/jn/133.9.2973S
Hemalatha S, Platel K, Srinivasan K (2007) Influence of germination and fermentation on bioaccessibility of zinc and iron from food grains. Europ J Clin Nutri 61(3):342–348. https://doi.org/10.1038/sj.ejcn.1602524“\t“_blank”10.1038/sj.ejcn.1602524
Giannini E, Botta F, Fasoli A, Ceppa P, Risso D, Lantieri PB, Celle G, Testa R (1999) Progressive liver functional impairment is associated with an increase in AST/ALT ratio. Digest Dis Sci 44:1249–1253
Diana NC (2007) Appendix: therapeutic drug monitoring and laboratory reference ranges. In: Stephen JM, Maxine AP (Eds), Current Medical Diagnosis and Treatment (46th ed)
Acknowledgements
The authors would like to acknowledge the laboratory staff of the Department of Food Science and Technology as well as Department of Biochemistry of the Federal University of Technology, Akure, Nigeria.
Author information
Authors and Affiliations
Contributions
OSO carried out the laboratory analysis and wrote the first draft of the manuscript while OTD carried out the laboratory and statistical analysis. ISO monitored the analysis, carried out statistical analysis, proof read the first draft and corrected the final manuscript draft. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author certify that there were no affiliations with or involvement in any organization or entity with any financial interest or nonfinancial interest in the subject matter or materials discussed in this manuscript.
Ethical approval and consent to participate
Ethical Committee of School of Agriculture and Agricultural Technology, Federal University of Technology Akure, Nigeria approved the study protocol with approval number FUTA/SAAT/2019/011.
Consent for publication
Not Applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oguntuase, S.O., Ijarotimi, O.S., Oluwajuyitan, T.D. et al. Nutritional, antioxidant, carbohydrate hydrolyzing enzyme inhibitory activities, and glyceamic index of wheat bread as influence by bambara groundnut substitution. SN Appl. Sci. 4, 121 (2022). https://doi.org/10.1007/s42452-022-05018-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-022-05018-8