Abstract
A plethora of cyclotron options have been developed to fulfil the demands of nuclear medicine industries in PET and SPECT radioisotopes. As a remote site, the difficulties of transporting fluorine 18 radiopharmaceuticals for PET examinations were overcome by the installation of a 7.5 MeV cyclotron for in-house production. The addition of a third-party synthesis module enabled the synthesis of 7 additional radiotracers according to a ‘’dose on demand’’ principle. Radiochemical yield is considered the primary factor in producing sufficient activity for a single patient dose, since low energy cyclotrons can only offer low initial activities. We hereby report the average radiochemical yields, synthesis times and doses per production for [18F]FDG, [18F]PSMA-1007, [18F]DOPA, [18F]FET, [18F]FLT, [18F]FMISO, [18F]Choline and [18F]FES using a BG75 cyclotron and a Neptis Mosaic-RS. Additionally, the presence of radionuclidic impurities in the final product was examined.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Positron emission tomography with computed tomography (PET/CT) is gaining acceptance and is becoming the leading modality in diagnosis among the non-invasive diagnostic techniques in nuclear medicine [1]. Positron emitting isotopes have been used to radiolabel several compounds with different biodistributions, leading to an arsenal of radiopharmaceuticals that can be used to image certain reactions, metabolic routes and processes at the molecular level [1]. Due to its properties, fluorine-18 (F-18) is the radioisotope of choice for many radiopharmaceuticals. Among other characteristics, it has a half-life of 110 min, which is enough for production and administration or dispatching. However, its availability is challenging for remote sites where transport logistics are more complicated and time consuming. This has a direct impact on the examination costs and leads to the need for in-house production of F-18 using accelerators [2].
Nowadays there is a variety on available cyclotrons, differing on several properties and characteristics [3,4,5,6]. From the economical point of view, the cyclotron of choice depends primarily on the estimated number of PET examinations per year for the covering area of the institution [7]. Due to increasing demands of F-18 radiotracers remote sites could overcome their proximity ‘’disadvantage” using a ‘’dose on demand’’ small cyclotron. As a result, several companies have manufactured and offered lower energy cyclotrons in order to achieve multiple runs of bombardment and fluorine-18 activity yields during a single day, according to the needs of the site [4, 6].
Such an example is the BG-75 Biomarker Generator (Best ABT Molecular Imaging Inc., Knoxville, TN, USA), which consists of the mini cyclotron and the microchemistry unit. The cyclotron is a 7.5 MeV, self-shielded, proton beam, positive ion cyclotron, initially designed for Fluorodeoxyglucose ([18F]FDG) production only, using “a dose on demand” principle.
However, the growing need for further non-FDG PET examinations led us to the introduction of an additional radiosynthesis module which, to the best of our knowledge, has never before been used together with a 7.5 MeV cyclotron.
We herein report the feasibility on production of several other fluorine-18 radiotracers using a BG75 cyclotron and a third-party synthesis module (NEPTIS mosaic-RS automated system, ORA, Philippeville, Belgium) by reporting our experience after more than 2 years.
2 Materials and methods
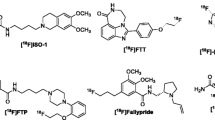
All chemicals, reagents and solvents needed for synthesis of 2-Deoxy-2-[18F]Fluoro-d-glucose ([18F]FDG), [18F]PSMA-1007, [18F]Fluoro-l-dihydroxyphenylalanine ([18F]DOPA), [18F]Fluoro-l-dihydroxyphenylalanine ([18F]FET), [18F]Fluorothymidine ([18F]FLT), [18F]Fluoromisonidazole ([18F]FMISO), [18F]Fluorocholine ([18F]Choline), [18F]Fluoroestradiol ([18F]FES) were purchased from ABX GmbH (Radeberg, Germany). Pre-conditioned Sep-Pak® Light Waters Accell™Plus QMA cartridges were used for 18F−/18O water separation. Additionally, the synthesis cassettes equipped with the cartridges required for each radiopharmaceutical and the reference compounds used for the quality control were also purchased from ABX GmbH. No modifications were made to the purchased Single-use synthesis cassettes and reagent kits. Methods and standard operating procedures for synthesis were provided by ORA NEPTIS manufacturer. Minor modifications on the sequences were made to avoid activity losses and ensure success on productions. For the better handling of low initial activity, a 6.2 cm line for introducing the activity to the synthesizer was used. The syntheses were hosted in a MecMurphil PET shielded isolator (MecMurphil S.r.l, Bologna, Italy) equipped with HEPA filters. HPLC solvents (HPLC grade) were purchased from Sigma-Aldrich.
HPLC analysis was performed on a Shimadzu 20A/AT system with a Gabi detector (Elysia-Raytest; Straubenhardt, Germany) for the determination of the radiochemical and chemical purity as well as the chemical identity of each compound. An ACE 3 C18 150 × 1.5 mm (ACE), Rezex ROA-Organic Acid H+ (8%) 250 × 4.6 mm and Luna® 5 µm Scx 100 Å 250 × 4.6 mm column was used. Prior the quality control, a reference standard of each compound was used for the determination of the retention time. GC (Shimadzu 17A, Lab solution 2.1 software; Kyoto, Japan) was used for the determination of residual solvents using a RESTEK RTX-624 (id 0.32 mm, length 1200 mm; Bellefonte, PA, USA) column. Radionuclidic identity was checked by half-life measurement via a dose calibrator (M.E.D. -Medizintechnik Dresden GmbH, Dresden, Germany). Radionuclidic purity was determined using a multi-channel analyser (Mucha star, Elysia -Raytest, Straubenhardt, Germany) with Gina star software (version 6.0, Elysia -Raytest, Straubenhardt, Germany). A spot test was used for the determination of both Tetrabutylammonium bicarbonate solution (TBA-HCO3) and Kryptofix levels in the final product, TLC (mini Gina detector and Gina star software, Elysia-Raytest; Straubenhardt, Germany) was performed using TLC Silica Gel 60 F254 Alu sheets (25 × 70 mm) and a mixture of MeOH:Ammonia (9:1 v/v ratio) as stationary and mobile phase respectively for the development of the plate. Reference standards of 111 ppm TBA-HCO3 solution and 600 ppm Kryptofix solution were used.
[18F]Fluoride was produced by irradiation of Oxygen-18 enriched water (97%, ABX, GmbH; Radeberg, Germany) with a 7.5-MeV cyclotron according to 18O(p,n)18F nuclear reaction. A 316-Stainless steel target of 0.28 mL maximum volume capacity and a window made of havar alloy, was used. The cyclotron’s target current was maintained at 5.5 μA. A total of 0.25 mL Oxygen-18 enriched water was used. Elution and purge of the transfer line was carried out in each production run, using the same volume of water (0.25 mL). Average time of bombardment for all syntheses was 87.3 ± 9.2 min. Irradiations were performed in the facility with the BG75; 7.5 MeV proton beam, positive-ion cyclotron. All syntheses were carried out using Neptis Mosaic-RS.
In order to investigate the existence and the identity of long-lived radioisotopes that might be present in the [O-18] water coming from the target, a further analysis was carried out. To determine those radioisotopes, a gamma spectroscopy of the recovered [O-18] water of all productions carried out with Neptis, since the first day of operation, was made (Fig. 1). Additionally, gamma spectroscopy for the recovered [O-18] water of single FDG run was performed, in order to determine the consistency of the output of radioisotopes (Fig. 2). The gamma spectroscopy data of a decayed [18F] FDG dose is also shown in Fig. 3.
3 Results
During the last 2 years of production, 1213 irradiations were carried out using both ABT’s microchemistry unit and Neptis Mosaic-RS. The average activity of F-18 transferred to synthesis modules was 4.08 ± 0.24 GBq after an average bombardment time of 87.3 ± 9.2 min. The data shown here were obtained using only Neptis Mosaic-RS. All radiochemical yields presented below are in the non-decay corrected form.
3.1 Radiopharmaceuticals
3.1.1 2-Deoxy-2-[18F]Fluoro-d-glucose
After 82 productions of FDG using the Neptis mosaic-RS radiosynthesis module, with 92.4 min of average bombardment time and a synthesis time of 23 min, the average production yield was 53 ± 8%. This equates to an average activity of 2.56 ± 0.84 GBq. The activity was considered enough for four to five patients to be administered, depending on their weight (target activity of 3.5 MBq/kg(BW)) and considering that only one PET/CT scanner is available.
3.1.2 [18F]-PSMA 1007
After 93 productions of 18F-PSMA-1007 with a synthesis time of 41 min, the average activity of 18F-PSMA-1007 was 1.97 ± 0.57 GBq, which translates to a radiochemical yield of 57 ± 8.1%. Based on the scan time (3 h post injection; target activity 4 MBq/kg(BW)) the average activity translates to the administration of up to three patients with one production.
3.1.3 [18F]Fluoro-l-dihydroxyphenylalanine
After 10 productions of 18F-L-DOPA with a synthesis time of 86 min, the average activity was 258 ± 78 MBq and the radiochemical yield was 7 ± 1%. This activity corresponds to a single patient (target activity 4 MBq/kg(BW)) for each production.
3.1.4 [18F]Fluoroethyl-l-tyrosine
After 24 productions of 18F-FET with a synthesis time of 49 min, the average activity of 1.31 ± 0.3 GBq, with a radiochemical yield of 30 ± 6% was achieved. This activity was considered to be enough for up to three patients to be administered (target activity of 200 MBq/patient).
3.1.5 [18F]Fluorothymidine
18F-FLT was produced once with an activity of 169 MBq, which results in a radiochemical yield of 5.6%. The low achievable activity of 18F-FLT (therefore high cost) have discouraged the use of 18F-FLT and favoured the use of 3.1.4 in the daily routine. Synthesis time 43 min.
3.1.6 [18F]Fluoromisonidazole
With two productions of 18F-FMISO, the activities of 629 MBq and 527 MBq can be reported. This translates to radiochemical yields of 17% and 11% respectively. The achieved activity was enough for a single patient (target activity 5.5 MBq/kg(BW)). The time needed for synthesis was 55 min.
3.1.7 [18F]Fluorocholine
After two syntheses we report doses of 973 MBq and 847 MBq, which translate to a radiochemical yield of 24% and 18%. The synthesis time was 49 min. A single patient was administered in each case (target activity 3 MBq/kg(BW)).
3.1.8 [18F]Fluoroestradiol
Two productions of 18F-FES were carried out. After 71 min of syntheses the final activities recorded were 800 MBq and 852 MBq and radiochemical yields of 23% and 28%. Those were administered to patients in a radioactive dose of 111–222 MBq as a slow intravenous bolus injection over 1–2 min [8]. Due to the uptake time of 80–100 min, the final activity is considered to be enough for two patients.
All radiopharmaceuticals passed the specifications for their quality control stated by European Pharmacopoeia.
3.2 Radionuclides generated using 7.5 MeV cyclotron
During normal operation, the BG-75 cyclotron activates certain materials, such as the target and its window. Since the target and the target window are made of stainless steel and havar alloy respectively, the generation of unwanted radionuclides cannot be avoided [9].
However, due to its low energy, the BG75 cyclotron only generates a very small amount of other, long-lived radioisotopes. Although most of the generated radionuclides expected [4, 10] are detectable in both cases (Figs. 1, 2) shown below, the activities recorded are very low. The sum of the integrated values of the peaks generated are 400 cps and 20 cps for all time and for a single day, respectively. An FDG dose was left to decay for five days. Despite that the data from Figs. 1 and 2 are comparable, there is no indication that any of these radioisotopes are present in the final product.
The data from Figs. 1 and 2 show that there are certainly 2 radioisotopes that can be considered activation products of Havar and stainless steel, Manganese 52 (t1/2 = 5.6 days) and Cobalt 56 (t1/2 = 77 days). One more isotope that is expected to be appeared cannot be identify but only hypotheses can be made due to lack of equipment and the low of activity. That isotope is Technetium 96× (t1/2 = 4.6 days) where its parent isotope (Molybdenum) has been used as addition to the stainless-steel alloy.
4 Discussion
The need for specialized examinations led to the installation of Neptis module due to the limitation of Best ABT’s microchemistry unit on producing radiopharmaceuticals other than FDG. The production of the new radiotracers has been shown to be feasible. All of them passed the quality control specifications and they can be considered as suitable for a routine nuclear medicine clinical practice. Eight radiopharmaceuticals were produced with comparable radiochemical yields to those reported by commercial cyclotron-synthesis modules [1, 11, 12]. Although the production of more FDG doses with one batch was not planned initially, the use of a third-party synthesis module enabled that option as well leading to the administration of even 5 patients with one hardware and reagents kit of FDG. Therefore, Best ABT’s 7.5 MeV cyclotron can be considered efficient enough for a small clinical site of 30–40 cases per week depending on the availability of synthesis modules.
Furthermore, the lower initial activity restricts the number of patients that can be administered. A Longer line was added on introducing the activity to the module through a 10 mL reservoir column in order to avoid spillage of droplets on its sides. And therefore, to minimize losses of activity.
Moreover, [18F]DOPA, [18F]FMISO, [18F]Choline, [18F]FES and [18F]FLT could only be administered to a single patient at a time, even though sufficient activity was generated for [18F]Choline and [18F]FES to administer up to two patients. Long synthesis times combined with low initial activities and patient availability results in single dose productions. The rest of the radiopharmaceuticals could be administered to multiple patients. Despite similarities in imaging, FET was preferred over FLT because of its better ability to cross the blood–brain barrier [11] as well as the better yields that were obtained.
Additionally, the unwanted production of other radionuclides did not deviate from what has been reported previously [13, 14] or suggested by cyclotron manufacturer. A similar study with different cyclotrons and target materials have shown similar results on the spectroscopic data [14, 15]. Although there is a production of other radionuclides, their low activity compared to commercial cyclotrons allows for better waste or equipment management in order to comply with national and local regulations. Additionally, there was no indication that any of those by-products were present in the final product even after 5 days of decay.
5 Conclusion
In conclusion, the addition of Neptis Mosaic-RS to the existing ABT’s 7.5 MeV cyclotron, made the production of additional radiopharmaceuticals feasible. Not only that, but also allowed the production of batches of radiopharmaceuticals needed for a certain number of patients. Most of the radiotracers have been produced to be administered to a single patient; however, the capabilities of the system in administering more patients with the same batch depends on the PET/CT scanners’ and patients’ availability. Due to the limitation of low energy cyclotrons in providing high initial activities of F-18, the radiochemical yield is considered the parameter that will define the capabilities of each batch.
References
Coenen HH, Elsinga PH, Iwata R, Kilbourn MR, Pillai MRA, Rajan MGR, Wagner HN, Zaknun JJ (2010) Fluorine-18 radiopharmaceuticals beyond [18F] FDG for use in oncology and neurosciences. Nucl Med Biol 37:727–740
Zaman MU, Fatima N, Mehdi SU, Sami M, Khan K, Khan G (2019) Personalized 18FDG dose synthesis using BG-75 generator: 1st year experience at JCI accredited tertiary care hospital in Pakistan. J Biomed Phys Eng 9:409–416
Tilbury RS, Laughlin JS (1974) Cyclotron production of radioactive isotopes for medical use. Semin Nucl Med 4:245–255
Awasthi V, Watson J, Gali H, Matlock G, McFarland A, Bailey J, Anzellotti A (2014) A “dose on demand” Biomarker Generator for automated production of [18F] F− and [18F] FDG. Appl Radiat Isot 89:167–175
Pashentsev VN (2015) Production of radionuclides for cyclotron positron-emission tomography. At Energy 118:405–409
Synowiecki MA, Perk LR, Nijsen JFW (2018) Production of novel diagnostic radionuclides in small medical cyclotrons. EJNMMI Radiopharm Chem 3:3
International Atomic Energy Agency (2009) Cyclotron produced radionuclides: guidelines for setting up a facility, Technical reports series no. 471, IAEA, Vienna
Chae SY, Ahn SH, Kim S, Han S, Lee SH, Oh SJ, Lee SJ, Kim HJ, Ko BS (2019) Diagnostic accuracy and safety of 16α-[18F] fluoro-17β-oestradiol PET-CT for the assessment of oestrogen receptor status in recurrent or metastatic lesions in patients with breast cancer: a prospective cohort study. Lancet Oncol 2045:1–10
Schmor P (2011) Review of cyclotrons used in the production of radioisotopes for biomedical applications. In: Proceedings of international conference of cyclotrons and their application, Lanzhou, China, pp 419–424, FRM2CIO01
International Atomic Energy Agency (2012) Cyclotron Produced Radionuclides: guidance on facility design and production of [18 F]Fluorodeoxyglucose (FDG). IAEA, Vienna
Kjoelhede M, Regner S, Perryman L, Erler J, Skovgaard H, Stockhausen M, Lassen U, Kjaer A (2016) Comparison of 18F-FET and 18F-FLT small animal PET for the assessment of anti-VEGF treatment response in an orthotopic model of glioblastoma. Nucl Med Biol 43:198–205
Diksic M, Toda Y (1983) Production of 18F-labelled molecular fluorine with a medical mini-cyclotron. Can J Chem 61:661–664
Martinez-Serrano JJ, de los Rios AD (2014) Predicting induced activity in the Havar foils of the 18F production targets of a PET cyclotron and derived radiological risk. Health Phys 107:103–110
Chaves JC, Vargas MJ, Sánchez RS (2016) Measurement of activation products generated in the [18F] FDG production by a 9.6 MeV cyclotron. Radiat Phys Chem 126:32–36
Manickam V, Brey RR, Jenkins PA, Christian PE (2009) Measurements of activation products associated with Havar foils from a GE PETtrace medical cyclotron using high resolution gamma spectroscopy. Health Phys 96:S37–S42
Acknowledgements
Not applicable.
Author information
Authors and Affiliations
Contributions
AF and MRP carried out the radiosyntheses, quality controls and collection of cyclotron’s data. AV conceived of the study and helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests. The authors alone are responsible for the content and writing of this article.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Not applicable.
Availability of data and material
Upon request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fesas, A., Pourkhessalian, M.R. & Vrachimis, A. Single center experience on the production of fluorine-18 radiopharmaceuticals using a 7.5 MeV cyclotron: capabilities and challenges. SN Appl. Sci. 2, 890 (2020). https://doi.org/10.1007/s42452-020-2695-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-020-2695-2