Abstract
The application of pre-treatment on oilseeds prior to extraction process may exert undesirable impact towards the quality of oils as well as microstructures of seed. The objectives of this study were to evaluate the effects of three drying methods on the microstructures of rambutan seeds and its effects on physicochemical properties of rambutan seed fat (RSF). The fats that being pre-treated with three different drying methods showed shrinkage or alteration of porous structure in terms of size, shape, and diameter. The differences between the RSF pre-treated with oven-, freeze-, and cabinet drying RSF were in fatty acids (oleic and arachidic acids), and free fatty acid (1.56–1.80 mg KOH/g fat). From the results obtained, the useful information regarding to the effects of pre-treatment on RSF, which is a potent ingredient to be used as a cocoa butter substitute in the formulation of chocolate in the confectionery industries. Moreover, the outcomes of this work able to provide information for better grasp about the correlation of drying methods and quality of RSFs, as well as its applications in other food industries.
Similar content being viewed by others
1 Introduction
Rambutan (Nephelium lappaceum Linn), a family of Sapindaceae, is a seasonal medium-sized tropical fruit tree that has been widely cultivated in Southeast Asia region including Malaysia, Philippines, Thailand, Vietnam, and even in Zaire in Central Africa, Madagascar in South Africa and Australia [1,2,3,4,5]. Furthermore, it can be adapted to temperate, moist and low evaporation rate environment with high amount of rainfall [5]. It is composed of hairy body, translucent white flesh and light-brown oblong seed [6, 7]. Basically, rambutan flesh is consumed either fresh, processed or canned. Besides, it is also being industrially processed into commercial food products, for instance, cans, syrups, juice, jams, jellies, and marmalades [6, 8,9,10,11]. Apart from that, the underutilized parts of rambutan (seed and rind) are often being considered as a major waste residue of the fruit and seldom gain interest by the manufacturers [5, 8, 9]. Yet, the light brown rambutan seed is actually safe for human consumption as it is non-toxic [12]. Most importantly, rambutan seed contained high amount of fats ranges from 33.4 to 37.35% [8, 13, 45]. The major fatty acids in RSFs are oleic and arachidic acids which ranging from 33.1 to 43.09% and 31.53 to 42.5%, respectively [8, 13,14,15,16,17]. The physicochemical properties make RSF capable for utilizing not only for cooking but for soaps [9], biodiesels [3, 18, 19] cosmetics [3, 18], medicinal purposes [20], and substitutes for cocoa butter [21].
In order to extract the oil from the seed of rambutan, a series of pre-treatment with the purpose to facilitate the drying process, maintain quality and ameliorate food safety are required [22]. The pre-treatment applied also help in improving extraction yield by rupturing the cell wall of oilseeds, thus, increasing the oil release from the cellular bodies [23]. However, the pre-treatment that being subjected to the oilseeds prior to the extraction process would probably possess impact towards the total yield of oil as well as its quality [24,25,26,27]. Food drying methods are the most ancient food preservation practice that still being widely utilized [28]. Nowadays, there are several methods have been employed to dry food, for instance, oven drying, cabinet drying, and freeze drying. According to Zhang et al. [26], the demucilaged flaxseed was being oven- and freeze- dried to assess its effect on extraction rate and quality of its oil. The mango seed was tray-dried for identifying the changes of seed properties associated with different temperatures by Divekar et al. [29]. In another study, the pomegranate seeds were being dried using freeze-dryer to evaluate its impact on colour attributes, phytochemicals, and antioxidant capacity [30].
The physicochemical properties such as iodine value, peroxide value, saponification value, slip melting point, and morphology of RSFs were reported in our previous study [31]. Nevertheless, the evaluation of the effects of varies drying methods on the microstructures of the rambutan seeds and the fatty acids of RSFs are limited. Therefore, the results obtained from this work will allow better comprehension regarding to the relationship between drying methods and quality of RSFs; to give the information on their application in all-around usages. Thus, the aim of this study was to evaluate the effects of different drying methods (i.e., oven-, cabinet-, and freeze- drying) on the physicochemical properties (i.e., fatty acids, free fatty acid, and refractive index) of RSFs. We also studied the microstructures of the rambutan seed using scanning electron microscope before and after the heat treatment.
2 Material and methods
2.1 Sample preparation
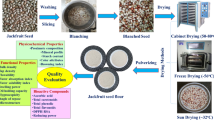
Matured and fresh red rambutan fruits were purchased from a market that located in segamat, Johor. The rambutans were carefully chosen and the skin was manually peeled off by using sterile kitchen knife. Then, the seeds were removed and cleaned under running tap water. Around 150 g of seeds were oven-dried at 60 °C for 48 h [14, 16]. While, a total of 300 g of seeds were also freeze-dried and cabinet-dried at − 40 °C for 48 h and 45 °C for 48 h, respectively. All of the dried seeds were kept in an airtight container under cool dry storage. With the purpose of extraction, the dried seeds were ground finely by using a mixer grinder (Panasonic MX-AC210S, India) before use. The rambutan seed powder was being passed through a sieve to obtain uniform particle size (< 200 μm).
2.2 Scanning electron micrographs
Microstructures observation for the surface of native and dried rambutan seeds dried by different methods were carried out by using scanning electron microscopy (EVO® MA 10, Zeiss, Germany) as described by Xie et al. [32] and Wang et al. [33]. The native and dried rambutan seeds were cut with sharp razor. The samples were sputter coated with gold using Emitech sputter coater (K550K, Emitech, France). The samples were then observed using SEM with an accelerating voltage of 15.0 kV.
2.3 Determination of free fatty acid value and refractive index
The free fatty acid value (FFA), and refractive index (RI) of RSFs were determined using AOCS Ca 5a-40 and Cc 7–25 [34]. For FFA, around 1.0 g fat samples were mixed with 25 mL diethyl ether and then add with 25 mL ethanol and 1 mL phenolphthalein indicator. The mixture was shaken vigorously to obtain homogenized solution. It was then titrated with 0.5 M sodium hydroxide (NaOH) (colorless changed to pink). The percentage of FFA was expressed as followed equation;
The refractive index for the RSF was determined using refractometer (ATAGO, Japan) at 40 °C. Melted samples (60–70 °C) were drops on the lower prism of refractometer and tighten firmly with the screwhead. The samples were allowed to stand for 1–2 min until the samples comes to the temperature of the instruments. The instrument and light were adjusted to obtain the most distinct reading possible and then determined the refractive index.
2.4 Determination of fatty acid by gas chromatography-flame ionization detector
The fatty acids of RSFs were determined by gas chromatography (Agilent 6890 N) equipped with flame ionization detector (HP EL-980) using BPX70 column (30 m × 0.25 μm × ID 0.25) SGE, France. The fatty acid methyl esters of RSF samples (50 mg) were prepared using 0.95 mL of n-hexane. The mixture was shaken to dissolve the RSF and then around 0.05 mL sodium methoxide was added into the mixture (shaken vigorously for 5–8 s). Approximately, 1 μL clear upper layer of FAMEs was injected into GC for fatty acids analysis. Detail procedure is described in our previous study [35].
2.5 Statistical analysis
All the analysis were performed in triplicates. The results were reported as mean ± standard deviation. The differences among means were evaluated by one-way analysis of variance (ANOVA), followed by Tukey’s test, using SPSS version 26.0. The statistical significance differences were considered at the level of p < 0.05.
3 Results and discussion
3.1 Changes in microstructure of rambutan seed during drying
The scanning electron microscope was being employed in order to observe the structures of rambutan seed before and after pre-treated with oven-, freeze-, and cabinet- drying methods. Figure 1 shows the scanning electron micrographs of native (a), oven- (b), freeze- (c) and cabinet-dried (d) rambutan seeds, respectively. It can be seen from Fig. 1 that the pre-treatment leads to significant structural alterations on the cells of rambutan seed. As evidenced, all dried seed samples exhibited porous structure. Majority of the cells seem to be rectangular in shape in the tissue of native and oven-dried seeds. On the other hand, for freeze- and cabinet-dried seeds exhibited slightly circular and elongated cells shape, respectively. Besides, the diameter and perimeter of native seed cells were the largest among the other three seed samples that being pre-treated with different drying methods. The hot-air drying can cause the breakage and rupture of cell walls, thus, increase the cavities and form intercellular spaces [36]. The changes of cell shape for freeze-dried seed was probably resulted from the ice crystal expansion during pre-freezing, whereas the balance between the cell fluid turgor pressure forces and tension forces of cell walls may cause the cell structure of cabinet-dried seed [37]. The cell wall of cabinet-dried seed was ruptured more thoroughly which may be owing to the rapid moisture evaporation from the seed. This is due to the exposed of the samples to the higher temperature which resulting in the tissue collapse and cell damage thus increasing the rehydration capability (larger spaces) [38]. The destruction of cell structures is basically beneficial to the mass transfer of active compounds during oil extraction process [26].
3.2 Changes in physicochemical properties of RSF during drying
3.2.1 Fatty acids
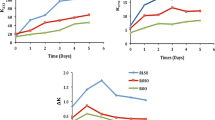
Table 1 shows the fatty acids of pre-dried RSF. There were no significant (p < 0.05) differences observed in the fatty acids of RSF when comparing the three drying methods. Similar findings were observed by Choo et al. and Latha et al. [39, 40] for rice bran and flaxseed oils during drying. The major fatty acids found in the RSF were oleic (32.02 to 33.75%), arachidic (37.57 to 39.80%), and stearic acids (6.78 to 7.94%). Figure 2 showed the gas chromatography-flame ionization detector chromatogram for fatty acid methyl ester of Freeze-dried (a), Cabinet-dried (b) and Oven-dried (c) RSFs. Among the fatty acids, oleic and arachidic acids were the dominant (up to 71%) with a significant presence of palmitic (4.68–5.04%), gondoic (5.72–5.95%) and behenic (3.78–3.97%) acids. Our results are in agreement with the stove dried RSF reported by Fuentes et al. [13]. All three drying methods showed around 50% of saturated fatty acids including the long chain fatty acid that has a relatively high melting point. Similar values were recently reported for RSF by Hernández-Hernández et al. and Mahisanunt et al. [17, 41], whereas Lourith et al. [9] reported low content of oleic (31.08%) and arachidic acids (28.65%), and high proportion of palmitic acid (5.84%) in rongrien rambutan variety. Oven-dried RSF exhibited lower oleic and higher arachidic acids compare to the two drying methods. Meanwhile, freeze-dried RSF showed lower arachidic (37.57%) and higher palmitic and stearic acids (5.04 and 7.94%) compared to the cabinet and oven dried RSF. It indicated that the freeze-dried RSF exhibited high-melting symmetrical triacylglycerols. RSF extracted from dried rambutan seed contained more palmitic and stearic acids, whereas less linolenic acid than that extracted from native rambutan seed. Fresh rambutan seed fat contain a relatively high arachidic ranges from 26 to 38% and relatively high saturated fatty acids [10, 16]. In addition, the fatty acid profiles obtained from three different drying methods showed different trends from the fermented RSF reported by Chai et al., [11, 42]. Fermented RSF showed higher oleic (41.02 to 42.38%) and linoleic acids (27.91 to 33.10%) compared to the dried RSF. The fatty acids of the fermented RSF gradually changed after seven and ten days of fermentation due to the selective hydrolysis of arachidic acid from the triacylglyerols containing arachidic acid and then replaced by the linoleic acid [42]. This indicating that the different drying methods showed less significant effect on the fatty acids composition of RSF compared to the nature of the extracted rambutan seed.
3.2.2 Free fatty acid (FFA)
The FFA values of RSF pre-treated with oven-, freeze- and cabinet- drying were 1.56, 1.78, and 1.80 mg KOH/g fat, respectively. The FFA values of RSF pre-treated with cabinet drying was the highest, manifesting that there is high rate of enzymatic rancidity that induced by lipase, if compared to those pre-treated by oven- and freeze- drying. The FFA values in this study were also significantly different (p < 0.05) among the RSFs being pre-treated with three different drying methods. Thus, it can be concluded that the FFA values of RSF are influenced by the drying methods applied. The identical results were also revealed by Taiwo et al. [43], who evaluated the consequences of drying methods on the properties of seed oils. The high FFA of cabinet-dried rambutan seed is probably resulted from the activity of lipase which catalysed the breakdown of RSF, thus, releasing the FFA [23]. Besides, the FFA value of freeze drying treated RSF is also significantly higher than those pre-treated by oven drying. Based on the literature, the type of chemical compounds exists and sample used determine the preservation effect of freeze drying, even though this drying method is capable in retaining raw quality and bioactive components of the seeds [44].
Hajar et al., [7] reported that the FFA value of RSF was 1.22 mg KOH/g fat, which is obviously lower if compared to the FFA values of RSF pre-treated with three different drying methods. The varied in FFA values could be because of the inappropriateness of the duration as well as the conditions of storage of oilseeds that enabling lipase activity [31]. In this study, the high FFA values could be due to the prolonged storage period of RSF after extraction. According to Afoakwa et al. [45], the FFA value of CB is much lower than the FFA of RSF. Thus, the refining process of RSF is necessary if wished to use as a CBA as this process can stabilize the oil, and hence, enhance its quality prior to the applications in the confectionery industries [31].
3.2.3 Refractive index (RI)
The RI is generally applied for the measurement of the purity of fats and oils [46]. The RSF with oven- and freeze- drying pre-treatment had a RI of 1.48, respectively, whereas cabinet drying treated RSF had a RI of 1.47, which are in line with those reported in the literature [7, 8]. A non-significant difference (p > 0.05) were observed in the RI of RSF when comparing the three different drying methods. This indicated that RI of RSF were not affected by drying methods. These similarities also being demonstrated by Ajala and Adeleke [47], who evaluated the effects of drying methods on the yields and qualities of seed oils. The RI indicates the intermolecular interaction of the fat system [7]. Besides, the RI has a strong relationship with density, since the molar refractivity is directly proportional to the molecular weight of the hydrocarbon molecule [7]. Based on the study carried out by George and Singh [48], the RI will increase along with the increase in temperature, increment in length of carbon chain and number of double bond [7].
4 Conclusion
The present study enables the improvement regarding to the understanding of the relationship between the effects of drying methods on the quality of RSF in terms of physicochemical properties and renders advantageous information about the application of RSF in the food industries, especially in the confectionery industries. The impacts of different drying methods (oven-, freeze-, and cabinet-drying) on the quality of RSF were evaluated. Based on the results, the physicochemical properties, for instance, fatty acids, FFA (1.56 to 1.80 mg KOH/g fat), and RI (1.47 to 1.48) were obtained in RSF pre-treated with oven-, freeze-, and cabinet-drying methods. The results obtained manifested that the RSF is a potential alternative source of ingredient that can be utilized as a blending component for cocoa butter equivalent formulation. In a nutshell, the most preferred drying method for producing the desirable quality of RSF is freeze drying based on the results obtained in this study.
References
Lasekan OO, Shabnam M (2013) Effect of traditional fermentation as a pre-treatment to decrease the antinutritional properties of rambutan seed (Nephelium lappaceum L.). Int Conf Food Agric Sci 55:67–72
Akhter S, McDonald MA, Marriott R (2016) Mangifera sylvatica (Wild Mango): a new cocoa butter alternative. Sci Rep 6:32050. https://doi.org/10.1038/srep32050
Nguyen HD, Nguyen MH, Nguyen TD, Nguyen PT (2016) Nephelium lappaceum oil: a low-cost alternative feedstock for sustainable biodiesel production using magnetic solid acids. Environ Prog Sustain Energy 35(2):603–610. https://doi.org/10.1002/ep.12254
Mahmood K, Kamilah H, Alias AK, Ariffin F (2018) Nutritional and therapeutic potentials of rambutan fruit (Nephelium lappaceum L.) and the by-products: a review. J Food Meas Charact 12(3):1556–1571. https://doi.org/10.1007/s11694-018-9771-y
Wahini M, Miranti MG, Lukitasari F, Novela L (2018) Rambutan seed (Nephelium lappaceum L.) optimization as raw material of high nutrition value processed food. IOP Conf Ser: Mater Sci Eng 306(1):012089. https://doi.org/10.1088/1757-899X/306/1/012089
Goenaga R (2011) Dry matter production and leaf elemental concentrations of rambutan grown on an acid Ultisol. J Plant Nutr 34:753–761. https://doi.org/10.1080/01904167.2011.540690
Hajar N, Mohamad N, Tokiman A, Munawar N, Shahrizan NS (2017) Physicochemical properties of rambutan seed fat. J Acad UiTM Negeri Sembilan 5:82–91
Sirisompong W, Jirapakkul W, Klinkesorn U (2011) Response surface optimization and characteristics of rambutan (Nephelium lappaceum L.) kernel fat by hexane production. LWT Food Sci Technol 44(9):1946–1951. https://doi.org/10.1016/j.lwt.2011.04.011
Lourith N, Kanlayavattanakul M, Mongkonpaibool K, Butsaratrakool T, Chinmuang T (2016) Rambutan seed as a new promising unconventional source of specialty fat for cosmetics. Ind Crops Prod 83:149–154. https://doi.org/10.1016/j.indcrop.2015.12.045
Chai KF, Adzahan NM, Karim R, Rukayadi Y, Ghazali HM (2018) Characteristics of fat, and saponin and tannin contents of 11 varieties of rambutan (Nephelium lappaceum L.) seed. Int J Food Prop 21(1):1091–1106. https://doi.org/10.1080/10942912.2018.1479857
Chai KF, Adzahan NM, Karim R, Rukayadi Y, Ghazali HM (2019) Characterization of rambutan (Nephelium lappaceum L.) seed fat and anti-nutrient content of the seed during the fruit fermentation: effect of turning intervals. LWT Food Sci Technol 103:199–204. https://doi.org/10.1016/j.lwt.2019.01.008
Polanditya P (2007) Biji rambutan sebagai alternative makanan baru. J. Ilmu Kimia FPMIPA Universitas Islam Indonesia, Yogyakarta, pp 1–4
Fuentes SJA, Camey-Ortiz G, Hernández-Medel MR, Pérez-Mendoza F, Durán-de-Bazúa C (2010) Composition, phase behaviour and thermal stability of natural edible fat from Rambutan (Nephelium lappaceum L.) seed. Bioresour Technol 101(2):799–803. https://doi.org/10.1016/j.biortech.2009.08.031
Harahap SN, Ramli N, Vafaei N, Said M (2012) Physicochemical and nutritional composition of rambutan anak sekolah (Nephelium lappaceum L.) seed and seed oil. Pak J Nutr 11(11):1073–1077. https://doi.org/10.3923/pjn.2012.1073.1077
Sonwai S, Ponprachanuvut P (2012) Characterization of physicochemical and thermal properties andcrystallization behavior of krabok (Irvingia Malayana) and rambutan seed fats. J Oleo Sci 61(12):671–679. https://doi.org/10.5650/jos.61.671
Manaf YNA, Marikkar JMN, Long K, Ghazali HM (2013) Physico-chemical characterization of the fat from the reed-skin rambutan (Nephellium lappaceum L.) seed. J Oleo Sci 62(6):335–343. https://doi.org/10.5650/jos.62.335
Hernández-Hernández C, Aguilar CN, Rodríguez-Herrera R, Flores-Gallegos AC, Morlett-Chávez J, Govea-Salas M, Ascacio-Valdés JA (2019) Rambutan (Nephelium lappaceum L.): nutritional and functional properties. Trends Food Sci Technol 85:201–210. https://doi.org/10.1016/j.tifs.2019.01.018
Kalayasiri P, Jeyashoke N, Krisnangkura K (1996) Survey of seed oils for use as diesel fuels. J Am Oil Chem Soc 73(4):471–474. https://doi.org/10.1007/BF02523921
Lim TK (2013) Nephelium lappaceum. In: Lim TK (ed) Edible medicinal and non-medicinal plants, volume 6: fruits. Springer, Amsterdam, pp 62–71
Soeng S, Evacuasiany E, Widowati W, Fauziah N, Manik V, Maesaroh M (2015) Inhibitory potential of rambutan seeds extract and fractions on adipogenesis in 3T3-L1 cell line. J Exp Integr Med 5(1):55–60. https://doi.org/10.5455/jeim.200115.or.120
Issara U, Zzaman W, Yang TA (2014) Rambutan seed fat as a potential source of cocoa butter substitute in confectionery product. Int Food Res J 21(1):25–31
da Silva GD, Barros ZMP, de Medeiros RAB, de Carvalho CBO, Rupert Brandão SC, Azoubel PM (2016) Pretreatments for melon drying implementing ultrasound and vacuum. LWT Food Sci Technol 74:114–119. https://doi.org/10.1016/j.lwt.2016.07.039
Qin X, Zhong J (2016) A review of extraction techniques for avocado oil. J Oleo Sci 65(11):881–888. https://doi.org/10.5650/jos.ess16063
Qu Q, Yang X, Fu M et al (2016) Effects of three conventional drying methods on the lipid oxidation, fatty acids composition, and antioxidant activities of walnut (Juglans regia L.). Dry Technol 34(7):822–829. https://doi.org/10.1080/07373937.2015.1081931
Wanyo P, Meeso N, Kaewseejan N, Siriamornpun S (2016) Effects of drying methods and enzyme aided on the fatty acid profiles and lipid oxidation of rice by-products. Dry Technol 34(8):953–961. https://doi.org/10.1080/07373937.2015.1087407
Zhang ZS, Liu YL, Che LM (2018) Effects of different drying methods on the extraction rate and qualities of oils from demucilaged flaxseed. Dry Technol 36(13):1642–1652. https://doi.org/10.1080/07373937.2017.1421220
Özcan MM, Aljuhaimi F, Uslu N, Ahmed IA, Osman MA, Gassem MA, Salih HA (2019) Effect of oven drying on antioxidant activity, phenolic compounds, fatty acid composition and tocopherol contents of pomegranate aril and oils. J Food Proc Preserv 43(3):e13885. https://doi.org/10.1111/jfpp.13885
Guiné R (2018) The drying of foods and its effect on the physical-chemical, sensorial and nutritional properties. Int J Food Eng 2(4):93–100. https://doi.org/10.18178/ijfe.4.2.93-100
Divekar SP, Bisen RD, Sonawane SP (2016) Drying Characteristics of Kesar Mango Stone. Int J Appl Pure Sci Agric 2(11):95–99
Al-Sanabani AS, Youssef KM, Shatta AA, El-Samahy SK (2016) Impact of freeze and freeze-drying processes on color, phytochemical contents and antioxidant capacity of pomegranate seeds. J Food Sci 3(1):27–34. https://doi.org/10.21608/scuj.2016.6659
Jahurul MHA, Ying LN, Amir HMS et al (2019) Effects of drying methods on the characteristics of rambutan (Nephelium lappaceum L.) seed fat: an optimisation approach. Eng Rep. https://doi.org/10.1002/eng2.12050
Xie L, Mujumdar AS, Fang XM, Wang J, Dai JW, Du ZL, Xiao HW, Liu Y, Gao ZJ (2017) Far-infrared radiation heating assisted pulsed vacuum drying (FIR-PVD) of Wolfberry: effects on drying kinetics and quality attributes. Food Bioprod Process 102:320–331. https://doi.org/10.1016/j.fbp.2017.01.012
Wang J, Bai TY, Wang D, Fang XM, Xue LY, Zheng ZA, Gao ZJ, Xiao HW (2019) Pulsed vacuum drying of Chinese ginger (Zingiber officinale Roscoe) slices: Effects on drying characteristics, rehydration ratio, water holding capacity, and microstructure. Dry Technol 37(3):301–311. https://doi.org/10.1080/07373937.2017.1423325
AOCS (2003) Official methods and recommended practices of the American oil chemists' society (Part 1, A–C), 5th edn. AOCS, Champaign
Jahurul MHA, Zaidul ISM, Norulaini N, Sahena F, Rahman MM, Mohd Omar AK (2015) Optimization of supercritical carbon dioxide extraction parameters of cocoa butter analogy fat from mango seed kernel oil using response surface methodology. J Food Sci Technol 52(1):319–326. https://doi.org/10.1007/s13197-013-0979-x
Gutiérrez L, Ratti C, Belkacemi K (2008) Effects of drying method on the extraction yields and quality of oils from quebec sea buckthorn (Hippophaë rhamnoides L.) seeds and pulp. Food Chem 106(3):896–904. https://doi.org/10.1016/j.foodchem.2007.06.058
Karunasena HCP, Hesami P, Senadeera W, Gu Y, Brown RJ, Oloyede A (2014) Scanning electron microscopic study of microstructure of gala apples during hot air drying. Dry Technol 32(4):455–468. https://doi.org/10.1080/07373937.2013.837479
Wang J, Law CL, Nema PK, Zhao JH, Liu ZL, Deng LZ, Gao ZJ, Xiao HW (2018) Pulsed vacuum drying enhances drying kinetics and quality of lemon slices. J Food Eng 224:129–138. https://doi.org/10.1016/jfoodeng.2018.01.002
Choo WS, Birch EJ, Dufour JP (2007) Physicochemical and stability characteristics of flaxseed oils during pan-heating. J Am Oil Chem Soc 84(8):735–740. https://doi.org/10.1007/s11746-007-1096-7
Latha RB, Nasirullah DR (2014) Physico-Chemical changes in rice bran oil during heating at frying temperature. J Food Sci Technol 51(2):335–340. https://doi.org/10.1007/s13197-011-0495-9
Mahisanunt B, Jom KN, Matsukawa S, Klinkesorn U (2017) Solvent fractionation of rambutan (Nephelium lappaceum L.) kernel fat for production of non-hydrogenated solid fat: Influence of time and solvent type. J King Saud Univ Sci 29(1):32–46. https://doi.org/10.1016/j.jksus.2016.08.004
Chai KF, Adzahan NM, Karim R, Rukayadi Y, Ghazali HM (2019) Fat properties and antinutrient content of rambutan (Nephelium lappaceum L.) during solid-state fermentation of rambutan fruit. Food Chem 274:808–815. https://doi.org/10.1016/j.foodchem.2018.09.065
Taiwo AA, Agbotoba MO, Oyedepo JA, Shobo OA, Oluwadare I, Olawunmi MO (2008) Effects of drying methods on properties of watermelon (Citrullus lanatus) seed oil. Afr J Food Agric Nutr Dev 8(4):492–501. https://doi.org/10.4314/ajfand.v8i4.19208
Raja KS, Taip FS, Azmi MMZ, Shishir MRI (2017) Effect of pre-treatment and different drying methods on the physicochemical properties of Carica papaya L. leaf powder. J Saudi Soc Agric Sci 18(2):150–156. https://doi.org/10.1016/j.jssas.2017.04.001
Afoakwa EO, Ofosu-Ansah E, Takrama JF, Budu AS, Mensah-Brow H (2014) Changes in chemical quality of cocoa butter during roasting of pulp pre-conditioned and fermented cocoa (Theobroma cacao) beans. Int Food Res J 1(6):2221–2227
Essien EA, Eduok UM (2013) Chemical analysis of Citrullus lanatus seed oil obtained from southern Nigeria. Org Chem 54:12700–12703
Ajala AS, Adeleke SA (2014) Effect of drying temperatures on physicochemical properties and oil yield of African star apple (Chrysophyllum alibidum) seeds. Global J Eng Des Technol 3(3):12–16
George A, Singh R (2015) Correlation of refractive index and density of crude oil and liquid hydrocarbon. Int J Chem Environ Biol Sci 3(5):2320–4087
Acknowledgements
This research was supported by the Centre for Research and Innovation, Universiti Malaysia Sabah (UMS) (GUG0336-1/2019 and SDN0061-2019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jahurul, M.H.A., Nee, S.S., Norazlina, M.R. et al. Changes in microstructures of rambutan seed and the quality of its fat during drying. SN Appl. Sci. 2, 841 (2020). https://doi.org/10.1007/s42452-020-2649-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-020-2649-8