Abstract
There have been speculations regarding rise in antimicrobial resistance (AMR) globally owing to indiscriminate antibiotic usage during the COVID-19 pandemic. To curb the menace through decisive policies, it is essential to assess the antibiotics, particularly the antibacterial agents. This systematic review and meta-analysis were performed to assess antibiotic use in COVID-19 patients. A thorough systematic search was undertaken in databases like PubMed, Cochrane library, Google Scholar, World Health Organization (WHO) database and clinicaltrials.gov by two independent reviewers for articles in English published from January 1, 2019 to October 31, 2020. Studies were included if they assessed confirmed COVID-19 cases and mentioned the use of antibiotics. The primary outcome was the proportion of COVID-19 patients subjected to specific antibacterial agents. An attempt to stratify the data based on study settings and disease severity was also performed. Of the total 6012 studies screened, 40 were eligible for qualitative review and 19 for meta-analysis. Specific antibacterial agents were mentioned in 23 studies (57.5%). In the random effect meta-analysis, pooled prevalence of azithromycin use was 24.5% (95% CI 22.9–26.2%) followed by cephalosporins as 26.6% (95% CI 24.9–28.4). None of the studies clearly specified indications for antibiotic use. Ten studies (25%) mentioned empirical use of antibiotics. Bacterial co-infections/secondary infections were documented in four studies with mean prevalence of infection of 1.9% (95% CI 1.2–2.8%). There is lack of data on use of specific antibacterial agents, indications for their use based on severity of infections and microbiological evidence of bacterial co-infections.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The global unpreparedness for tackling a pandemic of such magnitude like the coronavirus disease 2019 (COVID-19) has led to several direct and indirect challenges. Many of these challenges might not seem impending at the current stage of the pandemic but could have grave consequences even after the pandemic is contained. In this regard, there have been speculations that indiscriminate use of antibacterial agents or antibiotics during the pandemic without proper indications will impact antimicrobial resistance (AMR) particularly in bacteria (Getahun et al. 2020; Rossato et al. 2020). There are multiple threats of this pandemic towards a probable rise in AMR. On one side, increased hospital admissions due to the pandemic might lead to increase in healthcare associated infections and antibiotic use as a pre-emptive measure both in the community and hospitals. On the other side, widespread use of biocides against the virus in the community might result in an upsurge of drug resistant organisms, thus forming a vicious cycle (Caselli 2020). Evidence have suggested that the pandemic is ‘exacerbating’ the existing burden of AMR and antibiotics have been used beyond their indications (Strathdee et al. 2020). While appropriate management protocols for severe infections in COVID-19 and for bacterial coinfections remained largely unknown, there were sufficient gaps in expertise for differentiating COVID pneumonia and bacterial pneumonia, which in turn initiated unnecessary use of antibiotics (Nature Microbiology 2020). In order to tackle these problems, it is very essential to formulate effective and appropriate policies for judicious use of antibiotics. To decide on the policies, preliminary data on the trends in use of antibiotics in COVID-19 after almost 10–11 months of the ongoing pandemic is essential. To meet this end, we planned a systematic review to assess the proportions of various antibiotics, specifically the antibacterial agents used in COVID-19 patients.
Methods
Search strategy
Electronic databases particularly PubMed, Cochrane library, Google Scholar, World Health Organization (WHO) database and clinicaltrials.gov. were searched for articles that have been published between January 1, 2019 to October 31, 2020. The search terms and keywords used included “COVID-19”, “Covid-19”, “covid-19”, “SARS-CoV-2”, “antibiotics”, “antimicrobials” as per the search criteria of the individual databases.
Inclusion and exclusion criteria
All articles on COVID-19 patients across hospitals, in community or any long-term care facilities mentioning antibiotics were included. Studies only in English language within the said time period were considered. Studies with unreliable or overlapping data were excluded. Abstract only papers were also excluded. Besides, case reports, systematic or narrative reviews, opinions, editorials were not considered.
Study selection
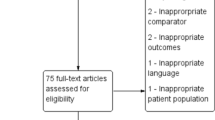
All published studies in English comprising of randomised and non-randomised controlled trials, case control studies, cohort studies, cross-sectional studies with sufficient data on antibiotic use in COVID-19 patients were included. Letter to Editor with results of original observational studies or randomised control trials (RCT) were also included. Pre-prints and ‘not yet peer-reviewed’ articles on MedRix-V were included if they contained relevant data. All duplicate studies were removed. The literature search as well as the screening of all the titles and abstracts of the included articles was independently done by two reviewers (SS and AS). All the full text articles were then reviewed by 2 independent reviewers (TB and SS/AS) for final inclusion. Disagreements were resolved by consensus, failing which the decision of the third reviewer was accepted. The reference section as well as the articles citing the selected articles were manually searched and checked for eligibility by the two independent reviewers. Authors were contacted by mail for any missing data in the selected articles. Reporting was done based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines for systematic review (Moher et al. 2009).
The study protocol was registered in PROSPERO register (CRD42020220042).
Data extraction
From the included studies, data was extracted by two independent reviewers (TB and AS) using a Microsoft Xcel spreadsheet and doubly cross checked for accuracy. Data was extracted under the following heads: first author, year of study, article type, study design, study setting, country of study, sample size, gender distribution, age of the study population, severity of COVID-19 in affected patients, number of patients on antibacterial therapy, antibacterial agents used, and indications of antibacterial therapy, presence of bacterial coinfections.
Data synthesis and statistical analysis
The primary outcome that was assessed was the overall proportions of antibacterial agents used in COVID-19 patients. We also tried to stratify the data based on the study settings and severity of COVID-19 in terms of mild, moderate or severe disease. Proportion data across all studies were pooled using a random effects meta-analysis with DerSimonian-Laird method. Results were displayed using forest plots for demonstration of the study effects along with the confidence intervals (CI). Heterogeneity was determined by I2 statistic. Heterogeneity was graded as considerably high at 75–100%, substantial at 50–90%, moderate at 30–60% and low at below 40% (Higgins et al. 2019). Publication bias was evaluated using the funnel plots. All categorical variables were expressed as relative frequencies and proportions. Continuous variables were expressed as mean or median with dispersion. All statistical analyses were performed using software STATA version 14 (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX: StataCorp LP.) and R package version 4.0 [R Core Team (2013). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria].
Results
Screening of studies
A total of 6012 search articles were screened of which 40 articles finally qualified for the systematic review (Table 1). Studies that were included consisted of confirmed COVID-19 cases by reverse transcriptase polymerase chain reaction (RT-PCR) and excluded suspected cases based on clinical presentations. Of these, 19 articles were considered for the meta-analysis. The study flow diagram has been shown in Fig. 1. The 40 articles for review comprised of 16 observational studies, 10 RCTs, 9 cohorts, 3 case series, 1 case control and 1 point-prevalence study. In this 1 article in ‘Letter to Editor’ and 1 in ‘Review” article format was also included due to relevance. This collection of 40 articles comprised of 35 published and 5 preprints ‘yet to be peer reviewed’ articles. The geographical distribution of the study locations has been shown in Fig. 2. Majority of the studies were from China, the epicentre of the pandemic. The summary of the articles has been shown in Table 1. All the studies were hospital based with only 1 specifically being an intensive care unit (ICU) based study. Sample size of the studied population varied from 20 to 136,855.
Meta-analysis of antibacterial agents used The names of specific antibacterial agents were mentioned in 23 of the 40 studies (57.5%) while the remaining studies mentioned only the use of ‘antibiotics’ without specifying the category of the antibacterial agents. In the random effect meta-analysis, pooled prevalence of azithromycin use was 24.5% (95% CI 22.9–26.2%), cephalosporins was 26.6% (95% CI 24.9–28.4) as shown in Table 2. There was considerable heterogeneity across all studies for all the antibacterial agents (I2 = 96–99%) with moderate heterogeneity in proportions of carbapenems use. The summary estimate of pooled prevalence of individual antibacterial agents has been shown in Fig. 3. Only two of the studies quantified antibiotic usage in terms of daily defined doses (DDD).
Indications for use of antibacterial agents None of the studies clearly specified indications for antibiotic use. While 10 studies (25%) mentioned empirical use of antibiotics, remaining studies did not specify the reason for use. Bacterial co-infections/secondary infections were documented in four studies only comprising of a sample size of 1323 cases where co-infections were seen in 26 (1.9%, 95% CI 1.2–2.8%, I2 = 44.2, moderate heterogeneity) of the cases (Fig. 4).
Severity of COVID-19 infections in the studies In 14 of the 40 studies, severity of COVID-19 infections as mild, moderate and severe was not mentioned. In the remaining studies, use of antibiotics based on severity of infections was mentioned only in two studies. However, in both the studies specific antibacterial agents for each category were not mentioned.
Assessment of bias
As detected by the funnel plots in Fig. 5, there was no bias in selection of studies on cephalosporin, carbapenem, amoxycillin-clavulanate and doxycycline while asymmetrical funnel plots were seen for azithromycin and macrolides, fluoroquinolones, piperacillin-tazobactam. There was no bias in article selection for antibacterial agents used in bacterial coinfections vis-a-vis empirical therapy.
Discussion
This systematic review on the antibacterial agents used in COVID-19 patients tried to identify the specific agents that have been used and their indications to generate a baseline data on antibiotic use in COVID-19. As speculations on inappropriate and excessive usage of antibiotics is remarkably high, this review paves the way for early recognition of several problems related to antibiotic consumption, research studies documenting them and prevailing antibiotic policies and stewardship programmes during the pandemic. Systematic reviews on antibacterial agents used in the pandemic are not previously available in literature.
The review clearly showed that azithromycin was the most frequently used antibacterial agent during the pandemic. While the role of azithromycin in treatment of COVID-19 has been debatable throughout, there has been evidence of increase in its consumption in the community against a background of decrease in consumption of other antibiotics (Gagliotti et al. 2020). Efficacy of azithromycin is well established in cases of community acquired pneumonia (CAP), but it is effects on viruses lacks evidence (Sultana et al. 2020). Besides azithromycin, clarithromycin has also been used in COVID-19 patients (Calderón et al. 2020). WHO has also reported the widespread use of azithromycin especially with hydroxychloroquine even beyond recommendations (WHO 2020, NICE 2020).
Empirical use of cephalosporins, especially third generation cephalosporins, in ICUs has been common entity even before the pandemic (Hariri et al. 2017; Banerjee et al. 2013, 2018). While the pandemic has mostly concentrated on repurposing and repositioning of the drugs, consumption of antibiotics for presumptive co-infections might have already changed the existing AMR scenario. In this regard, among the cephalosporins, cefuroxime has also been proposed for repurposing (Durojaiye et al. 2020). This drug has also been used in 220 patients out of the 556 cases of empirical therapy in one of the studies (Karami et al. 2020). Another repurposed drug in this aspect is doxycycline which has been used with the reason that tetracyclines possess both antiviral and anti-inflammatory properties that might help to fight COVID-19 (Yates et al. 2020). Broad spectrum antibiotics in form of cephalosporins and fluoroquinolones have been used empirically in nearly 74% of the patients as suggested by another meta-analysis on bacterial coinfection in COVID-19 (Langford et al. 2020). The same analysis has shown that against this high antibiotic prescribing attitude, co-infections and secondary bacterial infections were demonstrated only in 3.5% and 14.3% of the COVID-19 cases respectively, despite recommendations for antibiotic use specifically in cases of documented infections (WHO 2020). In this systematic review too, of the included studies only four studies discussed the prevalence of lower rate of documented bacterial infections against high empirical antibiotic use.
The major strength of this study is that it is the first documentation of the specific antibacterial agents used in COVID-19, a preliminary step for necessary actions to curb the menace of AMR. However, the study was not without limitations. Though 19 studies were identified for meta-analysis, use of antibiotics might have been under-represented as majority of the studies focussed on other aspects of treatment of COVID-19, not specifying antibiotic therapy. Besides, antibacterial agents have also been used in clinically suspected patients of COVID-19 which were not included in this review. The classes of antibiotics used had not been mentioned in most of the studies and data on use of antibacterial agents based on disease severity was lacking. Unequal distribution of the studies throughout the globe might have affected the results owing to variations in local policies prevailing in the regions, besides differences in patient population (Huttner et al. 2020). The considerable heterogeneity in the study effects could have resulted from the wide differences in the study designs of the selected articles among other factors affecting heterogeneity (Glasziou and Sanders 2002).
Nevertheless, the study clearly reveals that azithromycin and cephalosporins have been the most frequently used antibiotics in the pandemic despite evidence of low bacterial co-infection rates. To meet this increase in antibiotic consumption in healthcare set ups, initiation of ‘One Health’ approach is very essential along with a concomitant curb on antibiotic use in agribusiness (Strathdee et al. 2020). Speculating a rise in AMR based on these preliminary findings, we need to prioritize antimicrobial stewardship at double the existing pace to combat the impending crisis in the post-COVID era. Surge in AMR is likely to affect low- and middle-income countries (LMIC) drastically as some of them are also the major producers of antibiotics (Nature Microbiology 2020). Epidemiological studies with more and more evidence of actual quantity and indication of antibiotic use should be encouraged along with justifications for use based on severity of COVID-19.
It should be mentioned that AMR is a worldwide threat that incurs huge expenditures on the world economy and global health. The major concern is the fact that LMIC which are the major facilitators for easy dissemination of AMR are also the ones most affected by the burden of infections. Therefore, we suggest that utmost importance should be given to the implementation of antimicrobial stewardship in hospitals even in low resource countries based on our understanding of pathogenesis of the SARS-CoV-2 virus. Prudent and judicious use of antibacterial agents are necessary to curb the already existing menace of AMR. Lastly, it is always advisable to follow policies based on evidence and therefore, documentation of the real need for antibacterial therapy in cases of bacterial co-infections should be prioritized.
Conclusion
This study showed that among the COVID-19 patients, azithromycin and cephalosporins have been used mostly on empirical basis in hospitals. There is lack of data on the use of specific antibacterial agents as most of the studies have not mentioned the name of the antimicrobial agents administered. Though presumptive antibacterial therapy for suspected bacterial co-infections was the commonest indication for their use, documented microbiological evidence of bacterial co-infections was not mentioned in majority of the studies. The data from the available studies had also not specified the administration or indication of antibacterial therapy based on the severity of the COVID-19. Though it was evident that azithromycin and cephalosporins were the commonest antibacterial agents used during the pandemic, dearth of details on several aspects could not predict their exact role in AMR. Therefore, future studies should consider these lacking details for better analysis of the situation.
References
Abd-Elsalam S, Esmail ES, Khalaf M, Abdo EF, Medhat MA, Abd El Ghafar MS, Ahmed OA, Soliman S et al (2020) Hydroxychloroquine in the treatment of COVID-19: a multicenter randomized controlled study. Am J Trop Med Hyg 103:1635–1639
An MH, Kim MS, Kim BO, Kang SH, Kimn WJ, Park SK, Park HW, Yang W et al (2020) Treatment response to hydroxychloroquine and antibiotics for mild to moderate COVID-19: a retrospective cohort study from South Korea. medRxiv. https://doi.org/10.1101/2020.07.04.20146548
Angus DC, Berry S, Lewis RJ, Al-Beidh F, Arabi Y, van Bentum-Puijk W, Bhimani Z, Bonten M et al (2020) The REMAP-CAP (randomized embedded multifactorial adaptive platform for community-acquired pneumonia) study. Rationale and design. Ann Am Thorac Soc 17:879–891
Banerjee T, Anupurba S, Singh DK (2013) Poor compliance with the antibiotic policy in the intensive care unit (ICU) of a tertiary care hospital in India. J Infect Dev Ctries 15:7
Banerjee T, Mishra A, Das A, Sharma S, Barman H, Yadav G (2018) High prevalence and endemicity of multidrug resistant Acinetobacter spp. in intensive care unit of a Tertiary Care Hospital, Varanasi, India. J Pathog 2018:9129083
Borba MGS, Val FFA, Sampaio VS, Alexandre MAA, Melo GC, Brito M, Mourão MPG, Brito-Sousa JD et al (2020) Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial. JAMA Netw Open 3:e208857
Buetti N, Mazzuchelli T, Priore EL, Balmelli C, Llamas M, Pallanza M, Elzi L, Consonni V et al (2020) Early administered antibiotics do not impact mortality in critically ill patients with COVID-19. J Infect 81:e148–e149
Calderón JM, Flores MD, Coria LP, Garduño JC, Figueroa JM, Contretas MJ, De la Cruz ÁL, Meza SD et al (2020) Nitazoxanide against COVID-19 in three explorative scenarios. J Infect Dev Ctries 14:982–986
Caselli E (2020) Hygiene: microbial strategies to reduce pathogens and drug resistance in clinical settings. Microb Biotechnol 10:1079–1083
Chen J, Bai H, Liu J, Chen G, Liao Q, Yang J, Wu P, Wei J et al (2020) Distinct clinical characteristics and risk factors for mortality in female COVID-19 inpatients: a sex-stratified large-scale cohort study in Wuhan, China. Clin Infect Dis 8:ciaa920
National Institute for Health and Care Excellence (NICE) (2020) COVID-19 rapid guideline: antibiotics for pneumonia in adults in hospital [Internet]. https://www.nice.org.uk/guidance/ng173. Accessed 1 Oct 2020
Davoudi-Monfared E, Rahmani H, Khalili H, Hajiabdolbaghi M, Salehi M, Abbasian L, Kazemzadeh H, Yekaninejad MS (2020) A randomized clinical trial of the efficacy and safety of interferon β-1a in treatment of severe COVID-19. Antimicrob Agents Chemother 64:e01061-e1120
de Melo AC, Thuler LC, da Silva JL, de Albuquerque LZ, Pecego AC, Rodrigues LD, da Conceicao MS, Garrido MM et al (2020) Cancer inpatients with COVID-19: a report from the Brazilian National Cancer Institute. PLoS One 15:e0241261
Durojaiye AB, Clarke JR, Stamatiades GA, Wang C (2020) Repurposing cefuroxime for treatment of COVID-19: a scoping review of in silico studies. J Biomol Struct Dyn 13:1–8
Editorial (2020) Antimicrobial resistance in the age of COVID-19 Nat Microbiol 5:779
Feng Y, Ling Y, Bai T, Xie Y, Huang J, Li J, Xiong W, Yang D et al (2020) A Multicenter Study of Clinical Features. Am J Respir Crit Care Med 201:1380–1388
Furtado RHM, Berwanger O, Fonseca HA, Corrêa TD, Ferraz LR, Lapa MG, Zampieri FG, Veiga VC et al (2020) Azithromycin in addition to standard of care versus standard of care alone in the treatment of patients admitted to the hospital with severe COVID-19 in Brazil (COALITION II): a randomised clinical trial. Lancet 396:959–967
Gagliotti C, Buttazzi R, Ricchizzi E, Di Mario S, Tedeschi S, Moro ML (2020) Community use of antibiotics during the COVID-19 lockdown. Infect Dis (lond) 19:1–3
Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, Doudier B, Courjon J et al (2020) Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents 56:105949
Getahun H, Smith I, Trivedi K, Paulin S, Balkhy HH (2020) Tackling antimicrobial resistance in the COVID-19 pandemic. Bull World Health Organ 98:442-442A
Glasziou PP, Sanders SL (2002) Investigating causes of heterogeneity in systematic reviews. Stat Med 21:1503–1511
Hariri G, Tankovic J, Boëlle PY, Dubée V, Leblanc G, Pichereau C, Bourcier S, Bigé N et al (2017) Are third-generation cephalosporins unavoidable for empirical therapy of community-acquired pneumonia in adult patients who require ICU admission? A retrospective study. Ann Intensive Care 7:35
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (eds) (2019) Cochrane handbook for systematic reviews of interventions, 2nd edn. Wiley, Chichester
Hu W, Lv X, Li C, Xu Y, Qi Y, Zhang Z, Li M, Cai F et al (2020) Disorders of sodium balance and its clinical implications in COVID-19 patients: a multicenter retrospective study. Intern Emerg Med 16:1–10
Huang Q, Deng X, Li Y, Sun X, Chen Q, Xie M, Liu S, Qu H, Liu S, Wang L, He G (2020) Clinical characteristics and drug therapies in patients with the common-type coronavirus disease 2019 in Hunan, China. Int J Clin Pharm 42:837–845
Hung IF, Lung KC, Tso EY, Liu R, Chung TW, Chu MY, Ng YY, Lo J et al (2020) Triple combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet 395:1695–1704
Huttner BD, Catho G, Pano-Pardo JR, Pulcini C, Schouten J (2020) COVID-19: don’t neglect antimicrobial stewardship principles! Clin Microbiol Infect 26:808–810
Ji M, Yuan L, Shen W, Lv J, Li Y, Li M, Lu X, Hu L et al (2020) Characteristics of disease progress in patients with coronavirus disease 2019 in Wuhan. China. Epidemiol Infect 148:e94
Jin X, Lian JS, Hu JH, Gao J, Zheng L, Zhang YM, Hao SR, Jia HY et al (2020) Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms. Gut 69:1002–1009
Karami Z, Knoop BT, Dofferhoff ASM, Blaauw MJT, Janssen NA, van Apeldoorn M, Kerckhoffs APM, van de Maat JS (2020) Few bacterial co-infections but frequent empiric antibiotic use in the early phase of hospitalized patients with COVID-19: results from a multicentre retrospective cohort study in The Netherlands. Infect Dis (lond) 24:1–9
Khamis F, Al-Zakwani I, Al Naamani H, Al Lawati S, Pandak N, Omar MB, Al Bahrani M, Bulushi ZA et al (2020) Clinical Characteristics and outcomes of the first 63 adult patients hospitalized with COVID-19: an experience from Oman. J Infect Public Health 13:906–913
Langford BJ, So M, Raybardhan S, Leung V, Westwood D, MacFadden DR, Soucy JR, Daneman N (2020) Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis. Clin Microbiol Infect 22
Lehmann CJ, Pho MT, Pitrak D, Ridgway JP, Pettit NN (2020) Community acquired co-infection in COVID-19: a retrospective observational experience. Clin Infect Dis 1:ciaa902
Lian J, Jin X, Hao S, Jia H, Cai H, Zhang X, Hu J, Zheng L et al (2020) Epidemiological, clinical, and virological characteristics of 465 hospitalized cases of coronavirus disease 2019 (COVID-19) from Zhejiang province in China. Influenza Other Respir Viruses 14:564–574
Mancilla-Galindo J, Garcia-Mendez JO, Marquez-Sanchez J, Reyes-Casarrubias RE, Aguirre-Aguilar E, Rocha-Gonzalez HI, Kammar-Garcia A (2020) Use of antivirals and antibiotics for COVID-19 in Mexico City: a real-world Multicenter Cohort Study. medRxiv. https://doi.org/10.1101/2020.10.13.20211797
Mason CY, Kanitkar T, Richardson CJ, Lanzman M, Stone Z, Mahungu T, Mack D, Wey EQ et al (2020) Exclusion of bacterial co-infection in COVID-19 using baseline inflammatory markers and their response to antibiotics. medRxiv. https://doi.org/10.1101/2020.10.09.20199778
Mato AR, Roeker LE, Lamanna N, Allan JN, Leslie L, Pagel JM, Patel K, Osterborg A et al (2020) Outcomes of COVID-19 in patients with CLL: a multicenter international experience. Blood 136:1134–1143
Moher D, Liberati J, Tetzlaff DG, Altman P, Grp (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement (reprinted from Annals of Internal Medicine). Phys Ther 89:873–880
Piva S, Filippini M, Turla F, Cattaneo S, Margola A, De Fulviis S, Nardiello I, Beretta A et al (2020) Clinical presentation and initial management critically ill patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in Brescia, Italy. J Crit Care 58:29–33
Prata-Barbosa A, Lima-Setta F, Santos GRD, Lanziotti VS, de Castro REV, de Souza DC, Raymundo CE, de Oliveira et al (2020) Pediatric patients with COVID-19 admitted to intensive care units in Brazil: a prospective multicenter study. J Pediatr (Rio J) 96:582–592
Rossato L, Negrão FJ, Simionatto S (2020) Could the COVID-19 pandemic aggravate antimicrobial resistance? Am J Infect Control 48:1129–1130
Sadeghi A, Ali Asgari A, Norouzi A, Kheiri Z, Anushirvani A, Montazeri M, Hosamirudsai H, Afhami S et al (2020) Sofosbuvir and daclatasvir compared with standard of care in the treatment of patients admitted to hospital with moderate or severe coronavirus infection (COVID-19): a randomized controlled trial. J Antimicrob Chemother 75:3379–3385
Santoro F, Monitillo F, Raimondo P, Lopizzo A, Brindicci G, Gilio M, Musaico F, Mazzola M et al (2020) QTc interval prolongation and life-threatening arrhythmias during hospitalization in patients with COVID-19. Results from a multi-center prospective registry. Clin Infect Dis 24:ciaa1578
Seaton RA, Gibbons CL, Cooper L, Malcolm W, McKinney R, Dundas S, Griffith D, Jeffreys D et al (2020) Survey of antibiotic and antifungal prescribing in patients with suspected and confirmed COVID-19 in Scottish hospitals. J Infect 26:952–960
Sekhavati E, Jafari F, SeyedAlinaghi S, Jamalimoghadamsiahkali S, Sadr S, Tabarestani M, Pirhayati M, Zendehdel A et al (2020) Safety and effectiveness of azithromycin in patients with COVID-19: An open-label randomised trial. Int J Antimicrob Agents 56:106143
Spinner CD, Gottlieb RL, Criner GJ, López JR, Cattelan AM, Viladomiu AS, Ogbuagu O, Malhotra P et al (2020) Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: a randomized clinical trial. JAMA 324:1048–1057
Staub MB, Beaulieu RM, Graves J, Nelson GE (2020) Changes in antimicrobial utilization during the COVID-19 pandemic after implementation of a multispecialty clinical guidance team. Infect Control Hosp Epidemiol 26:1–28
Stevens RW, Jensen K, O’Horo JC, Shah A (2020) Antimicrobial prescribing practices at a tertiary-care center in patients diagnosed with COVID-19 across the continuum of care. Infect Control Hosp Epidemiol 24:1–4
Strathdee SA, Davies SC, Marcelin JR (2020) Confronting antimicrobial resistance beyond the COVID-19 pandemic and the 2020 US election. Lancet 396:1050–1053
Sultana J, Cutroneo PM, Crisafulli S, Puglisi G, Caramori G, Trifirò G (2020) Azithromycin in COVID-19 patients: pharmacological mechanism, clinical evidence and prescribing guidelines. Drug Saf 43:691–698
Sun L, Shen L, Fan J, Gu F, Hu M, An Y, Zhou Q, Fan H et al (2020) Clinical features of patients with coronavirus disease 2019 from a designated hospital in Beijing, China. J Med Virol 92:2055–2066
Tang W, Cao Z, Han M, Wang Z, Chen J, Sun W, Wu Y, Xiao W et al (2020) Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial. BMJ 369:1849
Tawfik GM, Dila KA, Mohamed MY, Tam DN, Kien ND, Ahmed AM, Huy NT (2019) A step by step guide for conducting a systematic review and meta-analysis with simulation data. Trop Med Health 47:46
Tian R, Wu W, Wang C, Pang H, Zhang Z, Xu H, Luo Q, Gao P et al (2020) Clinical characteristics and survival analysis in critical and non-critical patients with COVID-19 in Wuhan, China: a single-center retrospective case control study. Sci Rep 10:17524
Wei W, Ortwine JK, Mang NS, Joseph C, Hall BC, Prokesch BC (2020) Limited role for antibiotics in COVID-19: scarce evidence of bacterial coinfection (SSRN 3622388)
Williams EJ, Mair L, de Silva TI, Green DJ, House P, Cawthron K, Gillies C, Wigfull J et al (2020) Routine measurement of serum procalcitonin allows antibiotics to be safely withheld in patients admitted to hospital with SARS-CoV-2 infection. medRxiv. https://doi.org/10.1101/2020.06.29.20136572
World Health Organization (2020) Clinical management of COVID-19: interim guidance. World Health Organization
Yang Q, Xie L, Zhang W, Zhao L, Wu H, Jiang J, Zou J, Liu J et al (2020) Analysis of the clinical characteristics, drug treatments and prognoses of 136 patients with coronavirus disease 2019. J Clin Pharm Ther 45:609–616
Yin X, Liu L, Xu X, Huang L, Jing P, Li H, Jiang N, Wang J et al (2020) Evaluation of early antibiotics use in non-severe COVID-19 patients admitted with low risk of bacterial infection. medRxiv. https://doi.org/10.21203/rs.3.rs-39522/v1
Yu B, Li C, Chen P, Zhou N, Wang L, Li J, Jiang H, Wang DW (2020) Low dose of hydroxychloroquine reduces fatality of critically ill patients with COVID-19. Sci China Life Sci 15:1–7
Funding
None.
Author information
Authors and Affiliations
Contributions
SS and AS have performed literature search. SS, AS and TB have done final selection for articles. AS and TB have extracted data. SS and TB have synthesized data and performed statistical analysis. SS, AS and TB have finally drafted the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sharma, S., Singh, A. & Banerjee, T. Antibacterial agents used in COVID-19: A systematic review and meta-analysis. Environmental Sustainability 4, 503–513 (2021). https://doi.org/10.1007/s42398-021-00194-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42398-021-00194-6