Abstract
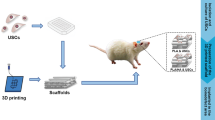
The bone regenerative scaffold with the tailored degradation rate matching with the growth rate of the new bone is essential for adolescent bone repair. To satisfy these requirement, we proposed bone tissue scaffolds with controlled degradation rate using osteoinductive materials (Ca-P bioceramics), which is expected to present a controllable biodegradation rate for patients who need bone regeneration. Physicochemical properties, porosity, compressive strength and degradation properties of the scaffolds were studied. 3D printed Ca-P scaffold (3DS), gas foaming Ca-P scaffold (FS) and autogenous bone (AB) were used in vivo for personalized beagle skull defect repair. Histological results indicated that the 3DS was highly vascularized and well combined with surrounding tissues. FS showed obvious newly formed bone tissues. AB showed the best repair effect, but it was found that AB scaffolds were partially absorbed and degraded. This study indicated that the 3D printed Ca-P bioceramics with tailored biodegradation rate is a promising candidate for personalized skull bone tissue reconstruction.







Similar content being viewed by others
References
Song P, Hu C, Pei X, Sun J et al (2019) Dual modulation on crystallinity and macro/micro structures of 3D printed porous titanium implants to enhance the stability and osseointegration. J Mater Chem B. 7:2865–2877
Ahadian S, Khademhosseini A (2018) A perspective on 3D bioprinting in tissue regeneration. Bio-Des Manuf 1(3):157–160
Pei X, Ma L, Zhang B, Sun J, Sun Y, Fan Y et al (2017) Creating hierarchical porosity hydroxyapatite scaffolds with osteoinduction by three-dimensional printing and microwave sintering. Biofabrication 9(4):045008
Miao X, Hu Y, Liu J, Wong AP (2004) Porous calcium phosphate ceramics prepared by coating polyurethane foams with calcium phosphate cements. Mater Lett 58(3–4):397–402
Samavedi S, Whittington AR, Goldstein AS (2013) Calcium phosphate ceramics in bone tissue engineering: a review of properties and their influence on cell behavior. Acta Biomater 9(9):8037–8045
Zhang B, Pei X, Zhou C, Fan Y, Jiang Q, Ronca R et al (2018) The biomimetic design and 3D printing of customized mechanical properties porous Ti6Al4V scaffold for load-bearing bone reconstruction. Mater Des 152(15):30–39
Yuan HYZ, Li Y et al (1998) Osteoinduction by calcium phosphate biomaterials. J Mater Sci Mater Med 72:3–6
Woodard JR, Hilldore AJ, Lan SK, Park CJ, Morgan AW, Eurell JA et al (2007) The mechanical properties and osteoconductivity of hydroxyapatite bone scaffolds with multi-scale porosity. Biomaterials 28(1):45–54
Ripamonti U, Van den Heever B, van Wyk J (1993) Expression of the osteogenic phenotype in porous hydroxyapatite implanted extraskeletally in baboons. Biomaterials 17:491–502
Carrodeguas RG, De Aza S (2011) Alpha-tricalcium phosphate: synthesis, properties and biomedical applications. Acta Biomater 7(10):3536–3546
Hirota M, Hayakawa T, Shima T, Ametani A, Tohnai I (2015) High porous titanium scaffolds showed higher compatibility than lower porous beta-tricalcium phosphate scaffolds for regulating human osteoblast and osteoclast differentiation. Mater Sci Eng C Mater Biol Appl. 49:623–631
Mohammadi Z, Sheikh-Mehdi Mesgar A, Rasouli-Disfani F (2016) Preparation and characterization of single phase, biphasic and triphasic calcium phosphate whisker-like fibers by homogenous precipitation using urea. Ceram Int 42(6):6955–6961
Yuan H, Li Y, Feng Z, Yang K, de Groot K, Zhang X (2001) Bone formation induced by calcium phosphate ceramics in soft tissue of dogs: a comparative study between porous a-TCP and b-TCP. J Mater Sci Mater Med 12:7–13
Zhu Y, Zhang K, Zhao R, Ye X, Chen X, Xiao Z et al (2017) Bone regeneration with micro/nano hybrid-structured biphasic calcium phosphate bioceramics at segmental bone defect and the induced immunoregulation of MSCs. Biomaterials 147:133–144
Marques CF, Perera FH, Marote A, Ferreira S, Vieira SI, Olhero S et al (2017) Biphasic calcium phosphate scaffolds fabricated by direct write assembly: mechanical, anti-microbial and osteoblastic properties. J Euro Cera Soci 37(1):359–368
Wang Z, Tang Z, Qing F, Hong Y, Zhang X (2012) Applications of calcium phosphate nanoparticles in porous hard tissue engineering scaffolds. NANO 07(04):1230004–1230018
Kim HJ, Park IK, Kim JH, Chong SC, Kim MS (2012) Erratum to: gas foaming fabrication of porous biphasic calcium phosphate for bone regeneration. Tissue Eng Regen Med 9(4):232
Tang Z, Li X, Tan Y, Fan H, Zhang X (2018) The material and biological characteristics of osteoinductive calcium phosphate ceramics. Regen Biomater 5(1):43–59
Perez RA, Mestres G (2016) Role of pore size and morphology in musculo-skeletal tissue regeneration. Mater Sci Eng C Mater Biol Appl 61:922–939
Phadke A, Hwang Y, Hee Kim S, Hyun Kim S, Yamaguchi T, Masuda K et al (2013) Effect of scaffold microarchitecture on osteogenic differentiation of human mesenchymal stem cells. Euro Cells Mater 25:114–129
Penk A, Forster Y, Scheidt HA, Nimptsch A, Hacker MC, Schulz-Siegmund M et al (2013) The pore size of PLGA bone implants determines the de novo formation of bone tissue in tibial head defects in rats. Magn Reson Med 70(4):925–935
Costa JB, Silva-Correia J, Reis RL, Oliveira JM (2018) Current advances in solid free-form techniques for osteochondral tissue engineering. Bio-Des Manuf 1(3):171–181
Jeong CG, Hollister SJ (2010) Mechanical and biochemical assessments of three-dimensional poly(1,8-octanediol-co-citrate) scaffold pore shape and permeability effects on in vitro chondrogenesis using primary chondrocytes. Tissue Eng A 16(12):3759–3768
Kujala S, Ryhänen J, Danilov A, Tuukkanen J (2003) Effect of porosity on the osteointegration and bone ingrowth of a weight-bearing nickel–titanium bone graft substitute. Biomaterials 24(25):4691–4697
Meza LR, Zelhofer AJ, Clarke N, Mateos AJ, Kochmann DM, Greer JR (2015) Resilient 3D hierarchical architected metamaterials. Proc Natl Acad Sci USA 112(37):11502–11507
Fukasawa T, Ando M, Ohji T, Kanzaki S (2001) Synthesis of porous ceramics with complex pore structure by freeze-dry processing. J Am Ceram Soc 84(1):230–232
Liu R, Xu T, Wang CA (2016) A review of fabrication strategies and applications of porous ceramics prepared by freeze-casting method. Ceram Int 42(2):2907–2925
Wu SC, Hsu HC, Hsiao SH, Ho WF (2009) Preparation of porous 45S5 Bioglass® -derived glass–ceramic scaffolds by using rice husk as a porogen additive. J Mater Sci Mater Med 20(6):1229
Ray AML, Gautier H, Bouler JM, Weiss P, Merle C (2010) A new technological procedure using sucrose as porogen compound to manufacture porous biphasic calcium phosphate ceramics of appropriate micro- and macrostructure. Ceram Int 36(1):93–101
Wang XX, Li W, Kumar V (2006) A method for solvent-free fabrication of porous polymer using solid-state foaming and ultrasound for tissue engineering applications. Biomaterials 27(9):1924–1929
Moghadam MZ, Hassanajili S, Esmaeilzadeh F, Ayatollahi M, Ahmadi M (2017) Formation of porous HPCL/LPCL/HA scaffolds with supercritical CO2 gas foaming method. J Mech Behav Biomed Mater 69:115–127
Jing X, Mi HY, Turng LS (2017) Comparison between PCL/hydroxyapatite (HA) and PCL/halloysite nanotube (HNT) composite scaffolds prepared by co-extrusion and gas foaming. Mater Sci Eng C Mater Biol Appl 72:53–61
Jakus AE, Rutz AL, Jordan SW, Kannan A, Mitchell SM, Yun C et al (2016) Hyperelastic “bone”: a highly versatile, growth factor-free, osteoregenerative, scalable, and surgically friendly biomaterial. Sci Trans Med 8(358):15
Wang X, Ao Q, Tian X, Fan J, Wei Y, Hou W et al (2016) 3D Bioprinting technologies for hard tissue and organ engineering. Materials 9(10):23
Chen Z, Li Z, Li J, Liu C, Lao C, Fu Y et al (2019) 3D printing of ceramics: a review. J Euro Ceram Soc 39(4):661–687
Bose S, Vahabzadeh S, Bandyopadhyay A (2013) Bone tissue engineering using 3D printing. Mater Today 16(12):496–504
Hwa LC, Rajoo S, Noor AM, Ahmad N, Uday MB (2017) Recent advances in 3D printing of porous ceramics: a review. Curr Opin Solid State Mater Sci 21(6):323–347
Zhang B, Pei X, Song P, Sun H, Li H, Fan Y et al (2018) Porous bioceramics produced by inkjet 3D printing: effect of printing ink formulation on the ceramic macro and micro porous architectures control. Compos B Eng 155:112–121
Wang J, Chen Y, Zhu X, Yuan T, Tan Y, Fan Y et al (2015) Effect of phase composition on protein adsorption and osteoinduction of porous calcium phosphate ceramics in mice. J Biomed Mater Res A 102(12):4234–4243
Ji K, Wang Y, Wei Q, Zhang K, Jiang A, Rao Y et al (2018) Application of 3D printing technology in bone tissue engineering. Bio-Des Manuf 1(3):203–210
Shao H, Yang X, He Y, Fu J, Liu L, Ma L, Zhang L, Yang G, Gao C, Gou Z (2015) Bioactive glass-reinforced bioceramic ink writing scaffolds: sintering, microstructure and mechanical behavior. Biofabrication 7(3):035010
Wang J, Chen Y, Zhu X, Yuan T, Tan Y, Fan Y et al (2014) Effect of phase composition on protein adsorption and osteoinduction of porous calcium phosphate ceramics in mice. J Biomed Mater Res A 102(12):4234–4243
Ripamonti U (1991) Bone induction in nonhuman primates. An experimental study on the baboon. Clin Orthop Relat Res 269:284–294
Acknowledgements
This work was supported by the National Key Research and Development Program of China (No. 18YFB1105600, 2018YFC1106800), National Natural Science Foundation of China (51875518), Sichuan Province Science & Technology Department Projects (2016CZYD0004, 2017SZ0001, 2018GZ0142, 2019YFH0079), Research Foundation for Young Teachers of Sichuan University (2018SCUH0017) and The “111” Project (No. B16033).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
The animal experiments were approved by the Animal Care and Use Committee of Sichuan University. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Zhang, B., Sun, H., Wu, L. et al. 3D printing of calcium phosphate bioceramic with tailored biodegradation rate for skull bone tissue reconstruction. Bio-des. Manuf. 2, 161–171 (2019). https://doi.org/10.1007/s42242-019-00046-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42242-019-00046-7