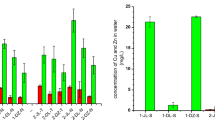
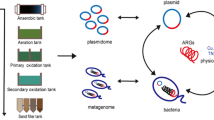
Abstract
An investigation was conducted in the vicinity of a sustainable pig farm to assess the presence of antibiotics through SPE-HPLC/MS/MS, microbial communities via Illumina high-throughput sequencing, and antibiotic resistance genes using SmartChip technology. The study revealed that tetracyclines were the predominant antibiotics detected in the soil and sediment surrounding the pig farm, with residual concentrations ranging from 33.3 to 1244.2 μg∙kg−1. The most prevalent phyla identified at various sampling sites were Firmicutes, Chloroflexi, Proteobacteria, Actinobacteria, and Acidobacteria. A total of 188 antibiotic resistance genes (ARGs) and 9 mobile genetic elements were found in the sediment, with aminoglycoside (particularly aadA2-03), sulfonamide (specifically sul2), and tetracycline (particularly tetX) resistance genes being the most frequently observed. The presence of tetracycline residue was observed to influence the composition of the microbial community, whereas no significant association was found between antibiotics and ARGs. Examination of the correlation between ARGs and bacteria at the phylum level demonstrated that Cyanobacteria, Acidobacteria, Planctomycetota, and Gemmatimonadota were the predominant phyla associated with ARG presence near an intensive pig farm. Notably, Cyanobacteria may function as a continual reservoir and/or shelter for ARGs, thereby potentially contributing to the dissemination of ARGs in the sediment environment in close proximity to a pig farm. This study presents evidence of the ecological risks posed by antibiotics in a pig farm-cropland system, highlighting the connection between microbial community structure and ARGs. Therefore, the issue of antibiotic residues must be factored into the sustainability of animal husbandry practices.
Article Highlights
-
An evaluation of the use of antibiotics was conducted in the area surrounding a sustainable pig farm.
-
Tetracycline residue had a considerable effect on the microbial community composition.
-
No meaningful connection was observed between antibiotics and their resistance genes.
-
Cyanobacteria may serve as a sustained reservoir for ARGs and thus contribute to the distribution of ARGs.






Similar content being viewed by others
Availability of Data and Materials
The data sets used and/or analyzed during the current study are available from the corresponding author on request.
References
Acosta-González A, Marqués S (2016) Bacterial diversity in oil-polluted marine coastal sediments. Curr Opin Biotechnol 38:24–32. https://doi.org/10.1016/j.copbio.2015.12.010
Arun J, Gopinath KP, Vigneshwar SS, Swetha A (2020) Sustainable and eco-friendly approach for phosphorus recovery from wastewater by hydrothermally carbonized microalgae: Study on spent biochar as fertilizer. J Water Process Eng 38:101567. https://doi.org/10.1016/j.jwpe.2020.101567
Bengtsson-Palme J, Kristiansson E, Larsson DGJ (2018) Environmental factors influencing the development and spread of antibiotic resistance. FEMS Microbiol Rev 42(1):fux053. https://doi.org/10.1093/femsre/fux053
Biswal BK, Balasubramanian R (2022) Adsorptive removal of sulfonamides, tetracyclines and quinolones from wastewater and water using carbon-based materials: recent developments and future directions. J Clean Prod 349:131421. https://doi.org/10.1016/j.jclepro.2022.131421
Bouwman L, Goldewijk KK, Van Der Hoek KW, Beusen AH, Van Vuuren DP, Willem J, Rufino MC, Stehfest E (2013) Exploring global changes in nitrogen and phosphorus cycles in agriculture induced by livestock production over the 1900–2050 period. Proc Natl Acad Sci 110(52):20882–20887. https://doi.org/10.1073/pnas.1012878108
Chen YS, Zhang HB, Luo YM, Song J (2012a) Occurrence and dissipation of veterinary antibiotics in two typical swine wastewater treatment systems in east China. Environ Monit Assess 184:2205–2217. https://doi.org/10.1007/s10661-011-2110-y
Chen YS, Zhang HB, Luo YM, Song J (2012b) Occurrence and assessment of veterinary antibiotics in swine manures: a case study in East China. Chin Sci Bull 57:606–614. https://doi.org/10.1007/s11434-011-4830-3
Chen Y, Xu J, Lv Z, Xie R, Huang L, Jiang J (2018) Impacts of biochar and oyster shells waste on the immobilization of arsenic in highly contaminated soils. J Environ Manage 217:646–653. https://doi.org/10.1016/j.jenvman.2018.04.007
Chen YS, Xi XP, Xu JH, Xie RJ, Jiang JP (2019) Distribution patterns of antibiotic residues in an urban river catchment. Water Environ J 33:31–39. https://doi.org/10.1111/wej.12366
Cheng D, Ngo HH, Guo W, Chang SW, Nguyen DD, Liu Y, Wei Q, Wei D (2019a) A critical review on antibiotics and hormones in swine wastewater: water pollution problems and control approaches. J Hazard Mater 387:121682. https://doi.org/10.1016/j.jhazmat.2019.121682
Cheng J, Tang X, Liu C (2019b) Distribution characteristics of antibiotic resistance genes in soil of livestock and poultry farms in purple soil hilly areas. Environ Sci 40:6–7. https://doi.org/10.13227/j.hjkx.2018.11.086. (in Chinese)
Cui H, Zhu D, Ding L, Wang Y, Su J, Duan G, Zhu Y (2023) Co-occurrence of genes for antibiotic resistance and arsenic biotransformation in paddy soils. J Environ Sci China 125:701–711. https://doi.org/10.1016/j.jes.2022.02.027
Di Cesare A, Eckert EM, Teruggi A, Fontaneto D, Bertoni R, Callieri C, Corno G (2015) Constitutive presence of antibiotic resistance genes within the bacterial community of a large subalpine lake. Mol Ecol 24:3888–3900. https://doi.org/10.1111/mec.2015.13293
Doyeni MO, Barcauskaite K, Buneviciene K, Venslauskas K, Navickas K, Rubezius M, Baksinskaite A, Suproniene S, Tilvikiene V (2023) Nitrogen flow in livestock waste system towards an efficient circular economy in agriculture. Waste Manag Res 41(3):701–712. https://doi.org/10.1177/0734242X221123484
Eisler MC, Lee MR, Tarlton JF, Martin GB, Beddington J, Dungait JA, Greathead H, Liu J, Mathew S, Miller H, Misselbrook T, Murray P, Vinod VK, Van Saun R, Winter M (2014) Agriculture: steps to sustainable livestock. Nature 507(7490):32–34. https://doi.org/10.1038/507032a
Elokil AA, Magdy M, Melak S, Ishfaq H, Bhuiyan A, Cui L, Jamil M, Zhao S, Li S (2020) Faecal microbiome sequences in relation to the egg-laying performance of hens using amplicon-based metagenomic association analysis. Animal 14(4):706–715. https://doi.org/10.1017/S1751731119002428
Feng Y, Lu Y, Chen Y, Xu J, Jiang J (2023) Microbial community structure and antibiotic resistance profiles in sediments with long-term aquaculture history. J Environ Manag 341:118052. https://doi.org/10.1016/j.jenvman.2023
Ghirardini A, Grillini V, Verlicchi P (2020) A review of the occurrence of selected micropollutants and microorganisms in different raw and treated manure—environmental risk due to antibiotics after application to soil. Sci Total Environ 707:136118. https://doi.org/10.1016/j.scitotenv.2019.136118
Gillings MR (2014) Integrons: past, present, and future. Microbiol Mol Biol Rev 78(2):257–277. https://doi.org/10.1128/MMBR.00056-13
Gu D, Feng Q, Guo C, Hou S, Lv J, Zhang Y, Yuan S, Zhao X (2019) Occurrence and risk assessment of antibiotics in manure, soil, wastewater, groundwater from livestock and poultry farms in Xuzhou, China. Bull Environ Contamin Toxicol 103(4):590–596. https://doi.org/10.1007/s00128-019-02692-0
Gu YR, Han BJ, Huang JY, Yang FX, Zhang KQ (2020) Occurrence characteristics and risk assessment of drug resistance genes in farmed feces from family farms in Tianjin. J Agro-Environ Sci 39(02):394–402 (in Chinese)
Guo XP, Yang Y, Lu DP, Niu ZS, Feng JN, Chen YR, Tou FY, Garner E, Xu J, Liu M, Hochella JMF (2018) Biofilms as a sink for antibiotic resistance genes (ARGs) in the Yangtze Estuary. Water Res 129:277–286. https://doi.org/10.1016/j.watres.2017.11.029
He LY, Liu YS, Su HC, Zhao JL, Liu SS, Chen J, Liu WR, Ying GG (2014) Dissemination of antibiotic resistance genes in representative broiler feedlots environments: identification of indicator ARGs and correlations with environmental variables. Environ Sci Technol 48(22):13120–13129. https://doi.org/10.1021/es5041267
He Y, Yuan Q, Mathieu J, Stadler L, Senehi N, Sun R, Alvarez PJJ (2020) Antibiotic resistance genes from livestock waste: occurrence, dissemination, and treatment. Npj Clean Water 3:4. https://doi.org/10.1038/s41545-020-0051-0
Ibekwe AM, Bhattacharjee AS, Phan D, Ashworth D, Schmidt MP, Murinda SE, Obayiuwana A, Murry MA, Schwartz G, Lundquist T, Ma J, Karathia H, Fanelli B, Hasan NA, Yang CH (2023) Potential reservoirs of antimicrobial resistance in livestock waste and treated wastewater that can be disseminated to agricultural land. Sci Total Environ 872:162194. https://doi.org/10.1016/j.scitotenv.2023.162194
Ji X, Shen Q, Liu F, Ma J, Xu G, Wang Y, Wu M (2012) Antibiotic resistance gene abundances associated with antibiotics and heavy metals in animal manures and agricultural soils adjacent to feedlots in Shanghai; China. J Hazard Mater 235–236:178–185. https://doi.org/10.1016/j.jhazmat.2012.07.040
Jin SQ, Zhang B, Wu B, Han DM, Hu Y, Ren CC, Zhang CZ, Wei X, Wu Y, Mol APJ, Reis S, Gu BJ, Chen J (2021) Decoupling livestock and crop production at the household level in China. Nat Sustain 4:48–55. https://doi.org/10.1038/s41893-020-00596-0
Ju F, Li B, Ma L, Wang Y, Huang D, Zhang T (2016) Antibiotic resistance genes and human bacterial pathogens: co-occurrence, removal, and enrichment in municipal sewage sludge digesters. Water Res 91:1–10. https://doi.org/10.1016/j.watres.2015.11.071
Karci A, Balcioğlu IA (2009) Investigation of the tetracycline, sulfonamide, and fluoroquinolone antimicrobial compounds in animal manure and agricultural soils in Turkey. Sci Total Environ 407(16):4652–4664. https://doi.org/10.1016/j.scitotenv.2009.04.047
Karkman A, Pärnänen K, Larsson DGJ (2019) Fecal pollution can explain antibiotic resistance gene abundances in anthropogenically impacted environments. Nat Commun 10(1):80. https://doi.org/10.1038/s41467-018-07992-3
Kielak AM, Barreto CC, Kowalchuk GA, van Veen JA, Kuramae EE (2016) The ecology of acidobacteria: moving beyond genes and genomes. Front Microbiol 7:744. https://doi.org/10.3389/fmicb.2016.00744
Kvesić M, Kalinić H, Dželalija M, Šamanić I, Andričević R, Maravić A (2022) Microbiome and antibiotic resistance profiling in submarine effluent-receiving coastal waters in Croatia. Environ Pollut 292(Pt A):118282. https://doi.org/10.1016/j.envpol.2021.118282
Lacroix MZ, Ramon-Portugal F, Huesca A, Angastiniotis K, Simitopoulou M, Kefalas G, Ferrari P, Levallois P, Fourichon C, Wolthuis-Fillerup M, De Roest K, Bousquet-Mélou A (2023) Residues of veterinary antibiotics in manures from pig and chicken farms in a context of antimicrobial use reduction by implementation of health and welfare plans. Environ Res 238(Pt 2):117242. https://doi.org/10.1016/j.envres.2023.117242
Larsson DGJ, Flach CF (2022) Antibiotic resistance in the environment. Nat Rev Microbiol 20(5):257–269. https://doi.org/10.1038/s41579-021-00649-x
Lekagul A, Tangcharoensathien V, Yeung S (2019) Patterns of antibiotic use in global pig production: a systematic review. Vet Anim Sci 7:100058. https://doi.org/10.1016/j.vas.2019.100058
Li C, Chen J, Wang J, Ma Z, Han P, Luan Y, Lu A (2015) Occurrence of antibiotics in soils and manures from greenhouse vegetable production bases of Beijing, China and an associated risk assessment. Sci Total Environ 521–522:101–107. https://doi.org/10.1016/j.scitotenv.2015.03.070
Li J, Xin Z, Zhang Y, Chen J, Yan J, Li H, Hu H (2017) Long-term manure application increased the levels of antibiotics and antibiotic resistance genes in a greenhouse soil. Appl Soil Ecol Sect Agric Ecosyst Environ 121:193–200. https://doi.org/10.1016/j.apsoil.2017.10.007
Li X, Chen L, Mei Q, Dong B, Dai X, Ding G, Zeng EY (2018) Microplastics in sewage sludge from the wastewater treatment plants in China. Water Res 142:75–85. https://doi.org/10.1016/j.watres.2018.05.034
Li N, Li H, Su G, Chen J (2022) Heavy metal distribution profiles in soil and groundwater near pig farms in China. Chemosphere 294:133721. https://doi.org/10.1016/j.chemosphere.2022.133721
Liu F, Liao D, Li K, Liu L, Liu C, Huang X (2013) Pollution characteristics of sulfa quinolones and macrolides antibiotics in livestock and poultry breeding bases. J Agro-Environ Sci 32(04):847–853 (in Chinese)
Liu S, Xu Q, Lou S, Tu J, Yin W, Li X, Jin Y, Radnaeva LD, Nikitina E, Makhinov AN, Araruna JT, Fedorova IV (2023) Spatiotemporal distributions of sulfonamide and tetracycline resistance genes and microbial communities in the coastal areas of the Yangtze River Estuary. Ecotoxicol Environ Saf 259:115025. https://doi.org/10.1016/j.ecoenv.2023.115025
Luo Y, Mao D, Rysz M, Zhou Q, Zhang H, Xu L, Alvarez JJP (2010) Trends in antibiotic resistance genes occurrence in the Haihe River, China. Environ Sci Technol 44(19):7220–7225. https://doi.org/10.1021/es100233w
Ma L, Li AD, Yin XL, Zhang T (2017) The prevalence of integrons as the carrier of antibiotic resistance genes in natural and man-made environments. Environ Sci Technol 51(10):5721–5728. https://doi.org/10.1021/acs.est.6b05887
Malolan R, Jayaraman RS, Adithya S, Arun J, Gopinath KP, SundarRajan P, Nasif O, Kim W, Govarthanan M (2021) Anaerobic digestate water for Chlorella pyrenoidosa cultivation and employed as co-substrate with cow dung and chicken manure for methane and hydrogen production: A closed loop approach. Chemosphere 266:128963. https://doi.org/10.1016/j.chemosphere.2020.128963. (Epub 2020 Nov 12)
Marutescu LG, Jaga M, Postolache C, Barbuceanu F, Milita NM, Romascu LM, Schmitt H, de Roda Husman AM, Sefeedpari P, Glaeser S, Kämpfer P, Boerlin P, Topp E, Gradisteanu Pircalabioru G, Chifiriuc MC, Popa M (2022) Insights into the impact of manure on the environmental antibiotic residues and resistance pool. Front Microbiol 13:965132. https://doi.org/10.3389/fmicb.2022.965132
Mu M, Yang F, Han B, Tian X, Zhang K (2022) Manure application: a trigger for vertical accumulation of antibiotic resistance genes in cropland soils. Ecotoxicol Environ Saf 237:113555. https://doi.org/10.1016/j.ecoenv.2022.113555
Peng FJ, Zhou LJ, Ying GG, Liu YS, Zhao JL (2014) Antibacterial activity of the soil-bound antimicrobials oxytetracycline and ofloxacin. Environ Toxicol Chem 33(4):776–783. https://doi.org/10.1002/etc.2513
Peng FJ, Ying GG, Liu YS, Su HC, He LY (2015) Joint antibacterial activity of soil-adsorbed antibiotics trimethoprim and sulfamethazine. Sci Total Environ 506–507:58–65. https://doi.org/10.1016/j.scitotenv.2014.10.117
Pironti C, Ricciardi M, Motta O, Miele Y, Proto A, Montano L (2021) Microplastics in the environment: intake through the food web, human exposure and toxicological effects. Toxics 9(9):224. https://doi.org/10.3390/toxics9090224
Qiu Z, Yu Y, Chen Z, Jin M, Yang D, Zhao Z, Wang J, Shen Z, Wang X, Qian D, Huang A, Zhang B, Li JW (2012) Nanoalumina promotes the horizontal transfer of multiresistance genes mediated by plasmids across genera. Proc Natl Acad Sci USA 109(13):4944–4949. https://doi.org/10.1073/pnas.1107254109
Qiu JC, Chen YS, Feng Y, Li XF, Xu JH, Jiang JP (2023) Adaptation of rhizosphere microbial communities to continuous exposure to multiple residual antibiotics in vegetable farms. Int J Environ Res Public Health 20:3137. https://doi.org/10.3390/ijerph20043137
Ren J, Shi H, Liu J, Zheng C, Lu G, Hao S, Jin Y, He C (2023) Occurrence, source apportionment and ecological risk assessment of thirty antibiotics in farmland system. J Environ Manag 335:117546. https://doi.org/10.1016/j.jenvman.2023
Roose-Amsaleg C, Laverman AM (2016) Do antibiotics have environmental side-effects? Impact of synthetic antibiotics on biogeochemical processes. Environ Sci Pollut Res 23(5):4000–4012. https://doi.org/10.1007/s11356-015-4943-3
Shi B, Wang S, Jiao J, Li G, Yin C (2022) Recognition on characteristics and applicability of typical modes for manure & sewage management in pig farming: a case study in Hebei, China. Waste Manag 148:83–97. https://doi.org/10.1016/j.wasman.2022.05.018
Szekeres E, Baricz A, Chiriac CM, Farkas A, Opris O, Soran ML, Andrei AS, Rudi K, Balcázar JL, Dragos N, Coman C (2017) Abundance of antibiotics, antibiotic resistance genes and bacterial community composition in wastewater effluents from different Romanian hospitals. Environ Pollut 225:304–315. https://doi.org/10.1016/j.envpol.2017.01.054
Tiwari A, Gomez-Alvarez V, Siponen S, Sarekoski A, Hokajärvi AM, Kauppinen A, Torvinen E, Miettinen IT, Pitkänen T (2022) Bacterial genes encoding resistance against antibiotics and metals in well-maintained drinking water distribution systems in Finland. Front Microbiol 12:803094. https://doi.org/10.3389/fmicb.2021.803094
Wang J, Ben W, Yang M, Zhang Y, Qiang Z (2016) Dissemination of veterinary antibiotics and corresponding resistance genes from a concentrated swine feedlot along the waste treatment paths. Environ Int 92–93:317–323. https://doi.org/10.1016/j.envint.2016.04.020
Wang L, Wang J, Wang J, Zhu L, Yang L, Yang R (2019) Distribution characteristics of antibiotic resistant bacteria and genes in fresh and composted manures of livestock farms. Sci Total Environ 695:133781. https://doi.org/10.1016/j.scitotenv.2019.133781
Wang Z, Han M, Li E, Liu X, Ning K (2020) Distribution of antibiotic resistance genes in an agriculturally disturbed lake in China: their links with microbial communities, antibiotics, and water quality. J Hazard Mater 393:122426. https://doi.org/10.1016/j.jhazmat.2020.122426
Wang Z, Chen Q, Zhang J, Guan T, Chen Y, Shi W (2020b) Critical roles of cyanobacteria as reservoir and source for antibiotic resistance genes. Environ Int 144:106034. https://doi.org/10.1016/j.envint.2020.106034
Wang J, Zhang Q, Chu H, Shi Y, Wang Q (2022) Distribution and co-occurrence patterns of antibiotic resistance genes in black soils in Northeast China. J Environ Manag 319:115640. https://doi.org/10.1016/j.jenvman.2022.115640
Wang B, Song L, Li W, Hou L, Li J, Xu X, Sheng G (2023a) Distribution and migration of antibiotic resistance genes, as well as their correlation with microbial communities in swine farm and its surrounding environments. Environ Pollut 316(Pt 2):120618. https://doi.org/10.1016/j.envpol.2022.120618
Wang F, Fu Y, Lin Z, Zhang B, Se J, Guo X, Fan J, Jia Y, Xu X, Jiang Y, Shen C (2023b) Neglected drivers of antibiotic resistance: survival of extended-spectrum β-lactamase-producing pathogenic escherichia coli from livestock waste through dormancy and release of transformable extracellular antibiotic resistance genes under heat treatment. Environ Sci Technol 57(27):9955–9964. https://doi.org/10.1021/acs.est.3c02377
Wu N, Qiao M, Zhang B, Cheng WD, Zhu YG (2010) Abundance and diversity of tetracycline resistance genes in soils adjacent to representative swine feedlots in China. Environ Sci Technol 44(18):6933–6939. https://doi.org/10.1021/es1007802
Wu J, Wang JY, Li ZT, Guo SM, Li KJ, Xu PS, Ok YS, Jones DL, Zou JW (2023) Antibiotics and antibiotic resistance genes in agricultural soils: a systematic analysis. Crit Rev Environ Sci Technol 53(7):847–864. https://doi.org/10.1080/10643389.2022.2094693
Xi X, Wang M, Chen Y, Yu S, Hong Y, Ma J, Wu Q, Lin Q, Xu X (2015) Adaption of the microbial community to continuous exposures of multiple residual antibiotics in sediments from a salt-water aquacultural farm. J Hazard Mater 290:96–105. https://doi.org/10.1016/j.jhazmat.2015.02.059
Xing G, Lu J, Xuan L, Chen J, Xiong J (2022) Sediment prokaryotic assembly, methane cycling, and ammonia oxidation potentials in response to increasing antibiotic pollution at shrimp aquafarm. J Hazard Mater 434:128885. https://doi.org/10.1016/j.jhazmat.2022.128885
Xiong S, Wang K, Yan H, Hou D, Wang Y, Li M, Zhang D (2022) Geographic patterns and determinants of antibiotic resistomes in coastal sediments across complex ecological gradients. Front Microbiol 13:922580. https://doi.org/10.3389/fmicb.2022.922580
Xu QH, Liu SG, Lou S, Radnaeva LD, Nikitina E, Nikolavich MA, Tavares AJ, Jin YC, Li X (2023a) Distributions of antibiotic resistance genes and microbial communities in the nearshore area of the Yangtze River Estuary. Environ Sci 44(01):158–168. https://doi.org/10.13227/j.hjkx.202203160. (in Chinese)
Xu M, Jiang J, Feng Y, Li X, Ye L, Chen Y (2023b) Resistance profiles of microbial communities in maize rhizospheres to the introduction of exogenous antibiotics to agricultural systems with a high arsenic geological background. Environ Sci Process Impacts 25(7):1224–1237. https://doi.org/10.1039/d3em00149k
Yan Z, Wang C, Liu T (2020) An analysis of the environmental efficiency of pig farms and its determinants-a field study from China. Environ Sci Pollut Res 27(30):38084–38093. https://doi.org/10.1007/s11356-020-09922-7
Yang Q, Tian T, Niu T, Wang P (2017) Molecular characterization of antibiotic resistance in cultivable multidrug-resistant bacteria from livestock manure. Environ Pollut 229:188–198. https://doi.org/10.1016/j.envpol.2017.05.073
Zhang XX, Zhang T, Fang HH (2009) Antibiotic resistance genes in water environment. Appl Microbiol Biotechnol 82(3):397–414. https://doi.org/10.1007/s00253-008-1829-z
Zhang QQ, Ying GG, Pan CG, Liu YS, Zhao JL (2015) Comprehensive evaluation of antibiotics emission and fate in the river basins of China: source analysis, multimedia modeling, and linkage to bacterial resistance. Environ Sci Technol 4949(11):6772–6782. https://doi.org/10.1021/acs.est.5b00729
Zhang YJ, Hu HW, Gou M, Wang JT, Chen D, He JZ (2017) Temporal succession of soil antibiotic resistance genes following application of swine, cattle and poultry manures spiked with or without antibiotics. Environ Pollut 231(Pt 2):1621–1632. https://doi.org/10.1016/j.envpol.2017.09.074
Zhang YJ, Hu HW, Chen QL, Singh BK, Yan H, Chen D, He JZ (2019) Transfer of antibiotic resistance from manure-amended soils to vegetable microbiomes. Environ Int 130:104912. https://doi.org/10.1016/j.envint.2019.104912
Zhang Z, Zhang Q, Wang T, Xu N, Lu T, Hong W, Penuelas J, Gillings M, Wang M, Gao W, Qian H (2022) Assessment of global health risk of antibiotic resistance genes. Nat Commun 13(1):1553. https://doi.org/10.1038/s41467-022-29283-8
Zhao L, Dong YH, Wang H (2010) Residues of veterinary antibiotics in manures from feedlot livestock in eight provinces of China. Sci Total Environ 408(5):1069–1075. https://doi.org/10.1016/j.scitotenv.2009.11.014
Zhao J, Bi CJ, Chen ZL, Shi Q, Li LR, Yao CX (2017) Contents of fluoroquinolone-type antibiotics in the surroundings of livestock farms in chongming Island of Shanghai. J Ecol Rural Environ 33(2):120–126. https://doi.org/10.11934/j.issn.1673-4831.2017.02.004
Zhao Y, Lin H, Liu Y, Jiang Y, Zhang W (2022) Abundant bacteria shaped by deterministic processes have a high abundance of potential antibiotic resistance genes in a plateau river sediment. Front Microbiol 13:977037. https://doi.org/10.3389/fmicb.2022.977037
Zheng D, Yin G, Liu M, Chen C, Jiang Y, Hou L, Zheng Y (2021) A systematic review of antibiotics and antibiotic resistance genes in estuarine and coastal environments. Sci Total Environ 777:146009. https://doi.org/10.1016/j.scitotenv.2021.146009
Zhu YG, Johnson TA, Su JQ, Qiao M, Guo GX, Stedtfeld RD, Hashsham SA, Tiedje JM (2013) Diverse and abundant antibiotic resistance genes in Chinese swine farms. Proc Natl Acad Sci USA 110(9):3435–3440. https://doi.org/10.1073/pnas.1222743110
Zhu D, Xiang Q, Yang XR, Ke X, O’Connor P, Zhu YG (2019) Trophic transfer of antibiotic resistance genes in a soil detritus food chain. Environ Sci Technol 53(13):7770–7781. https://doi.org/10.1021/acs.est.9b00214
Acknowledgements
This work was supported by the National Natural Science Foundation of China (no. 41977358) and Environmental protection technology plan project of Fujian Province (2022R003).
Author information
Authors and Affiliations
Contributions
YC designed the work plan; MY, JX and XL: performed all experiments; MY, JX and YF: analyzed the data; MY and YC wrote manuscript; JJ and YC: helped in modifying this manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, M., Xu, J., Li, X. et al. Adaptation and Resistance of Microbial Communities to Antibiotic Contamination in the Surrounding Environment of a Sustainable Pig Farm. Int J Environ Res 18, 47 (2024). https://doi.org/10.1007/s41742-024-00601-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41742-024-00601-w