Abstract
Background
Patient self-managed anticoagulant treatment with warfarin (PSM) has been proposed as an alternative to direct oral anticoagulants (DOACs) in patients with non-valvular atrial fibrillation (NVAF); however, direct evidence on the cost effectiveness of PSM compared with DOACs is lacking. We aimed to evaluate the cost effectiveness of PSM versus DOACs for NVAF patients in the Danish healthcare setting using a model-based cost-utility analysis.
Methods
A cost-utility analysis was performed using a decision-analytic model including two treatment alternatives: continuous PSM and DOACs. The analysis was performed from an extended Danish healthcare sector perspective, including patient-paid costs of medication related to the anticoagulant treatment, with a lifetime horizon. Inputs for the model comprised of probabilities of events, costs in Danish estimates, when possible, and effect in utilities. The probabilities of events are primarily based on real-life data from a direct comparison of PSM and DOACs. The results are presented as the incremental cost-effectiveness ratio (ICER) with an assumed cost-effectiveness threshold of £20,000/quality-adjusted life-year (QALY). Both deterministic and probabilistic sensitivity analyses were performed to investigate the robustness of the results.
Results
The base-analysis showed that PSM was dominant, with a decreased cost of £8495 and an increased QALY accumulation of 0.23 per patient (ICER = −£36,935/QALY). All deterministic sensitivity analyses indicated that PSM was dominant or at least cost effective. The probabilistic sensitivity analysis showed that 95% of the iterations were cost effective.
Conclusions
The present study found that PSM is dominant (i.e., both more effective and cost saving) compared with DOACs, adding to the scarce evidence of the comparative cost effectiveness of PSM and DOACs in NVAF.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This study found that PSM is dominant (i.e., more effective and cost-saving) compared to DOAC. Even when including the applied uncertainty of the input parameters in the sensitivity analyses, PSM seems to be at least as cost-effective. |
This study is based primarily on real-life data from a direct comparison of PSM and DOAC, which is expected to strengthen the results, and as a result of this, it presents a more realistic result of what to expect in real-life practice. |
1 Introduction
Individuals with non-valvular atrial fibrillation (NVAF) experience a fivefold increased risk of stroke [2,3,4,5,6]. NVAF-related strokes are associated with an increase in morbidity and mortality, decreased health-related quality of life, and increased healthcare utilization [6, 7]. To reduce the risk of stroke, NVAF patients are recommended anticoagulant treatment. Current guidelines include treatment with vitamin K antagonists (VKAs), such as warfarin, and direct oral anticoagulants (DOACs) [8]. DOACs have been shown to be superior to VKAs in decreasing the risk of thrombosis and avoiding adverse events, which is why guidelines recommend DOACs over VKAs [8,9,10,11,12,13]. However, the daily price of DOACs is currently approximately 10 times the daily price of warfarin [14]. To enable efficient utilization of the scarce resources of the health care system, it is important to identify cost-effective treatments that generate the most value for money for society.
A meta-analysis from 2020 [15] found that the comparative benefit of DOACs is decreased compared with patient self-managed anticoagulant treatment with warfarin (PSM). Studies have found PSM to be equal to [16,17,18], and even more effective than [19], various DOACs in preventing thrombotic events, and some evidence [18, 20] suggests PSM is cost saving compared with DOACs; however, several of these studies are based on mixed patient populations [16, 18, 19] and indirect comparisons of PSM with DOACs [15, 17,18,19,20,21], which is why the applicability of the results to NVAF is debatable [1, 15].
In a recent registry-based study of PSM versus DOACs in a Danish NVAF population [1], Grove et al. found that PSM is associated with a lower risk of thrombotic events and no difference in the occurrence of major bleeding and mortality when used in real-life practice.
The present study evaluated the cost effectiveness of PSM versus DOACs for NVAF patients in the Danish healthcare setting using a model-based cost-utility analysis (CUA). Evidence on the clinical effectiveness of PSM and DOACs is mainly based on the study by Grove et al. [1].
2 Methods
This study focuses on a cohort of NVAF patients considered eligible for DOAC for whom PSM could be an alternative treatment option. In PSM, patients are educated in monitoring and adjusting their VKA treatment based on international normalized ratio (INR) measurements. The patients are in continuous contact with both a nurse and chief physician at a specialized outpatient clinic, and attend regular check-ups with their general practitioner (besides medication, DOAC treatment consists of regular check-ups with a general practitioner). The cohort was assumed to resemble the population reported by Grove et al. [1], while baseline characteristics of the study population have been described elsewhere [1]. The approach of the study was from an extended Danish health care sector perspective, which consists of the perspective of the Danish healthcare sector but where patient-paid costs of medication related to anticoagulant treatment are also included. The choice of perspective was based on factors where the alternatives were expected to be different, i.e., the risk of acute events and medication costs. The results are expressed as the incremental cost-effectiveness ratio (ICER) of PSM compared with DOACs, based on the effect measure quality-adjusted life-years (QALYs). Since no threshold for cost effectiveness exists in Denmark, the British threshold of £20,000/QALY was used [22]. It was assumed that the British threshold was relevant, due to the similarities between the financing of the Danish and British health care systems [23]. An annual discount rate of 3.5% was applied to both costs and effects [24, 25]. This study was conducted in accordance with the Consolidated Health Economic Evaluations Reporting Standards (CHEERS) checklist [26]. (For elaboration of the literature search, please see the electronic supplementary material [ESM].)
2.1 Model Structure
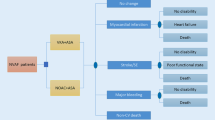
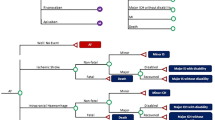
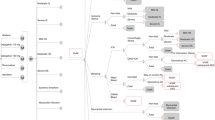
The CUA was performed using a Markov model with a lifetime horizon. The included alternatives in the Markov model, representing PSM and DOAC treatment, were structurally similar but included different probabilities of the events. Events included ischemic stroke (IS), myocardial infarction (MI), pulmonary embolism (PE), systemic embolism (SY), deep venous thromboembolism (DVT), transient ischemic attack (TIA), intracranial bleeding (IB), gastrointestinal bleeding (GB), other major bleedings (MB), and death (Fig. 1).
Model structure. The Markov model for the two alternative treatment options (patient self-managed anticoagulant treatment with warfarin and direct oral anticoagulants) is structurally similar. A The events: No major clinical events, pulmonary embolism, systemic embolism, deep venous thromboembolism, transient ischemic attack, major bleeding, and gastrointestinal bleeding lead to continuation in the current health state (well, post intracranial bleeding, post ischemic stroke, or post myocardial infarction). B Surviving an ischemic stroke leads to transition to the post ischemic stroke health state, unless the current health state is post intracranial bleeding (in which case it will lead to continuation in the post intracranial bleeding health state). C Surviving a myocardial infarction leads to continuation in the post myocardial infarction health state, unless the current health state is post ischemic stroke (in which case it will lead to continuation in the post ischemic stroke health state) or post intracranial bleeding (in which case it will lead to continuation in the post intracranial bleeding health state)
A 3-month cycle length was chosen, reflecting the assumption that more adverse events were unlikely to occur per quarter [27]. IB, IS, and MI lead to ‘post-event health states’, which were created to reflect the assumption that the initiating events affect cost accumulation and/or patients’ health-related quality of life. Health states representing a combination of events were not included in the model. The transition to worse post-event health states was always applied and followed the hierarchy of MI, IS, and IB, with MI being considered the least severe event. For example, a patient with prior IB who experienced an IS would transition back to the post-IB health state, as living with previous IB was considered worse than the permanent impact that an IS would have on patients’ health-related quality of life and the need for healthcare over time. Input parameters for the model were based on estimates reflecting current Danish practice.
To account for events occurring at discrete time points and not continuously over time, a half-cycle correction was performed for all cost and effects, counteracting potential overestimation of costs and effects due to rewards accumulating at the start of cycles, and transitioning occurring at the end of cycles [28]. One of the authors (TBL) reviewed the model structure and possible transitions to ensure consistency with what is observed in clinical practice. To evaluate whether the modeled risk of thrombotic events, bleeding events and mortality can be compared with the risks expected in real-life practice, the modeled 10-year risk of thrombotic events, bleeding events and mortality was compared between PSM and DOACs. The model was constructed using TreeAge Pro 2022 R1.2 (Healthcare version; TreeAge Software, Williamstown, MA, USA; the software is available at http://www.treeage.com).
2.2 Transition Probabilities
To fit the 3-month cycle length of the model, all included transition probabilities were scaled to 3 months, assuming a constant rate in accordance with the methods described by Briggs et al. [29]. Rates were converted into probabilities under the assumption of a constant rate model [31, 32]. Input parameters limited within the range of 0–1, as for probabilities, were assumed to be outcomes of beta-distributed derivates [30], whereas gamma distributions were assumed for non-negative parameters as rates. The probability of adverse events was assumed to be associated with the treatment (either PSM or DOAC), whereas the short-term mortality associated with adverse events was assumed to be associated with the adverse event. The probability of adverse events was based on unpublished data from the study by Grove et al. [1], while short-term mortality associated with IS, MI, IB, GB, and MB was based on the literature [33,34,35,36,37,38]. DVT, PE, SY, and TIA were not assumed to cause death within the same cycle. Other-cause (non-cardiovascular) mortality was included based on Danish national mortality charts, dependent on age [33]. When possible, estimates of standard error (SE) and 95% confidence interval (CI) were retrieved directly from the data or the literature, or otherwise they were estimated based on either the relevant 95% CI or SE, respectively. When neither the SE nor the 95% CI was available, an assumed SE of 15% of the mean was used and the 95% CI was thereby calculated. Uncertainty in the input of other cause (non-cardiovascular) mortality was not included in the sensitivity analysis (Table 1).
2.3 Costs
All costs are presented in 2019 values and, when necessary, adjusted using the consumer price index of hospital services [33]. Costs are presented in British pounds sterling (£) using exchange rates from the Bank of England as of 18 June 2021 [39]. Relevant costs included costs related to the treatment and derived incident and long-term costs related to the occurrence of adverse events (Table 2).
PSM was associated with an initial cost in relation to patient education and an operational cost in relation to INR monitoring. In addition to contact with the specialized outpatient clinic, patients receiving warfarin treatment are advised to have 2-yearly check-ups with their general practitioner [3]. Patient education consists of contact with a specialized outpatient clinic and written material/handouts. The costs associated with INR monitoring comprise equipment, medication, and feedback on measurements. Unit prices for the equipment, i.e., CoaguChek INRange monitor, CoaguChek Softclix lancets, and CoaguChek XS [PT(PST)] test strips, were estimated with help from Roche Diagnostics A/S, Denmark. A discount on PSM equipment was included based on negotiation under the auspices of the North Denmark region, while estimation of labor costs associated with patient education and feedback was based on effective hourly wages. On average, PSM medication included warfarin 5 mg daily (2 × 2.5 mg tablets).
DOAC use was associated with an operational cost in relation to medication use. In addition, 4-yearly check-ups with a general practitioner were recommended in the first year of treatment, with 2-yearly check-ups in subsequent years [3]. In the study by Grove et al. [1], 68% of NVAF patients were treated with dabigatran etexilate, 23% were treated with rivaroxaban, and 9% were treated with apixaban. In the present analysis, the same distribution of DOAC use was assumed. Daily doses of dabigatran etexilate were 110 or 150 mg, rivaroxaban 15 or 20 mg, and apixaban 2.5 or 5 mg [1]. (For elaboration of the estimation of costs associated with PSM and DOAC treatment, please see the ESM.)
It was assumed that the costs of adverse events were referable to the 3-month cycle in which the adverse event occurred; however, IS and IB were also expected to entail long-term economic consequences. MI was assumed to not consist of long-term economic consequences, hence the main costs of MI are related to inpatient care [40], which are reflected in the diagnosis-related group (DRG) tariff. The costs of IS, IB, GB, and MB were based on Danish cost-of-illness studies [41, 42]. The incident costs consisted of healthcare costs, including inpatient and outpatient hospital care, and contact with private practicing health professionals, such as general practitioners and physiotherapists. The long-term costs of IS and IB included costs of social care and contacts within the healthcare system that could be ascribed to the initial event. Incident costs of MI, SY, DVT, PE, and TIA were based on the Danish DRG system [43] from 2019.
All costs were included as gamma distributions [30] based on means and an assumed SE of 15% of the mean value of the parameter.
2.4 Quality-Adjusted Life-Years
Utility and disutility weights were ascribed to health states and events to enable estimation of QALYs gained with PSM and DOACs. Because no utility and disutility weights have been estimated for the Danish population, these were based on the literature, with estimates primarily from a British population and one estimate from an American population [44, 45].
The cohort was ascribed a baseline utility reflecting a starting age of 64 years [1]. Disutility weights were applied for NVAF, treatment, adverse events, and increasing age. The disutility associated with adverse events was assumed to be temporary, lasting for the cycle in which they occurred, although IB, IS, and MI was assumed to also cause permanent disutility. Death was ascribed a utility weight of zero. Disutility related to age was included, with a constant linear increase of 0.00029 per year. Uncertainty in the input disutility related to age was not included in the sensitivity analysis. The estimates of baseline utility and disutility of other events were included as beta distributions [30]. Weights and parameters for estimation of QALYs are given in Table 3.
2.5 Sensitivity Analyses
Sensitivity analyses were performed to investigate the robustness of the results. The deterministic sensitivity analyses included one-way sensitivity analyses. For these, input parameters were ascribed a range based on their 95% CI. When a 95% CI was not available, a symmetric range was applied based on the mean value of the parameter ± 1.96 × SE. When an SE of 15% of the mean value was assumed, this was used.
Scenario analyses were also performed as follows.
-
Scenario 1: The average daily price of a DOAC was reduced by 80% to reflect the DOAC patent expiry [46].
-
Scenario 2: Changing the time horizon to 1 year.
-
Scenario 3: Discount rate of 0% was applied for both costs and effects.
-
Scenario 4: The discount on PSM equipment included in the base-case analysis was removed.
-
Scenario 5: The IS risk associated with DOACs was applied for both DOACs and PSM.
The probabilistic sensitivity analysis (PSA) consisted of a second-order Monte Carlo simulation with 1000 iterations based on random draws from the input parameter distributions.
3 Results
The base-case analysis showed that PSM, with a cost accumulation of £10,364 and QALY accumulation of 9.78 over the lifetime horizon, dominated DOACs, which accumulated £18,860 and 9.55 QALYs, rendering PSM both cost saving and more effective compared with DOACs (ICER = −£36,935/QALY). The modeled 10-year risks of bleeding events and mortality in PSM and DOACs were, in general, similar (18% vs. 13%, and 14% vs. 15%, respectively), whereas the risk of thrombotic events was increased with DOACs by more than a factor of 2 (27% vs. 12%). This is consistent with the findings of Grove et al., that PSM is associated with a lower risk of thrombotic events and no difference in bleeding events and death [1].
All sensitivity analyses indicated that the result was robust. The one-way sensitivity analyses showed that PSM remains cost effective compared with DOACs within the investigated input parameter ranges (see Fig. S1 in the ESM). The PSA was visualized in an incremental cost-effectiveness scatterplot and a cost-effectiveness acceptability curve (Figs. 2 and 3). The cost-effectiveness acceptability curve showed that 95% of the iterations were cost effective at a threshold of £20,000/QALY (Fig. 3). Table 4 shows the results of the base-case analysis and the scenario analyses. None of the investigated scenarios indicated that PSM would be cost ineffective compared with DOACs when applying the threshold for cost effectiveness of £20,000/QALY. Given the changes investigated, all sensitivity analyses indicated that PSM is either dominant or cost effective compared with DOACs (Table 4).
Incremental cost-effectiveness scatter plot (PSM vs. DOACs). The dotted line illustrates the assumed threshold for cost effectiveness of £20,000/QALY. DOACs direct oral anticoagulants, PSM patient self-managed anticoagulant treatment with warfarin, QALY quality-adjusted life-year, WTP willingness to pay
4 Discussion
This CUA found that PSM is both cost saving and more effective than DOACs. The results were robust, although some scenario analyses indicated that, under specific conditions, the total cost of PSM may be higher than the total cost of DOACs, but given the assumed threshold for cost effectiveness of £20,000/QALY, PSM would remain cost effective.
This study does have some limitations. The applied models may not reflect the management that would be observed in real-life practice. For example, it is likely that adverse events trigger a clinical evaluation of whether to discontinue or switch treatment. In addition, the occurrence of adverse events is known to increase the risk of secondary events [36] but this has not been incorporated into the models. However, we do not assume that these limitations affect the overall results of the analysis. It should be noted that this analysis was conducted in the setting of the Danish health care sector and transferability to other settings could affect the results. Furthermore, this analysis was limited to a narrow perspective, i.e., the public Danish health care sector extended with costs related to medication. A societal perspective would include more costs, i.e., costs related to the consumption of time for patients and relatives, consumption of materials, and productivity loss, which could possibly affect the results. Furthermore, the study was based on British and American utility estimates, which consist of a limitation in the reflection of a Danish population; however, this is assumed to not reflect the incremental effectiveness of this study since the estimates are used in both alternatives.
This study is strengthened by the fact that events in the model are based on real-life data from a direct comparison of PSM and DOACs [1]. The application of effectiveness data rather than efficacy data, as would have been retrieved from trials, expectedly provides more realistic results for the expected comparative cost effectiveness of PSM and DOACs. To the best of our knowledge, this study is the first model-based CUA comparing PSM and DOACs using direct, real-life data.
The results of this study are consistent with the findings of other studies, which have also found that PSM is cost saving [18, 20] and equally [16,17,18], if not more, effective as DOACs [19].
Even though this study presents PSM as a viable alternative to DOACs, not all patients are eligible for PSM. The quality of PSM is highly dependent on patients’ ability to self-monitor and their acceptability of the treatment. A Danish Health Technology Assessment from 2009 [47] indicated that only 33% of patients are likely to accept PSM. This is comparable with a British study [48], which found that 24% of patients were likely to agree to self-monitor and only 14% of them would persist in the long-term. However, it is debatable whether these estimates are obsolete given the age of the publications. It is possible that a higher level of acceptability and adherence to self-monitoring and other digital healthcare solutions would be observed today due to a general increase in digitized everyday life. Thus, the proportion of patients who could be eligible for and/or prefer PSM could be higher.
5 Conclusions
With the application of an extended Danish healthcare sector perspective and a lifetime horizon, PSM appears to be both more effective and cost saving compared with DOACs in NVAF, producing an ICER of −£36,935/QALY. Sensitivity analyses indicated that the result was robust. Even in simulations of patent expiry of DOACs, PSM remained cost effective. The evidence base for the current study is highly specific to the Danish setting, and direct transferability of results to other settings must be addressed in that context. Nonetheless, this study adds to the scarce evidence on the cost effectiveness of PSM and DOACs in NVAF.
References
Grove EL, Skjøth F, Nielsen PB, Christensen TD, Larsen TB. Effectiveness and safety of self-managed oral anticoagulant therapy compared with direct oral anticoagulants in patients with atrial fibrillation. Sci Rep Engl. 2018;8:15805.
Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation as an independent risk factor for stroke: the Framingham study. Stroke. 1991;22:983–8.
Marini C, De Santis F, Sacco S, Russo T, Olivieri L, Totaro R, et al. Contribution of atrial fibrillation to incidence and outcome of ischemic stroke. Stroke [Internet]. 2005;36:1115–9. https://doi.org/10.1161/01.STR.0000166053.83476.4a.
Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation: a major contributor to stroke in the elderly. The Framingham Study. Arch Intern Med [Internet]. 1987;147:1561–4.
Savelieva I, Bajpai A, Camm AJ. Stroke in atrial fibrillation: update on pathophysiology, new antithrombotic therapies, and evolution of procedures and devices. Ann Med. 2007;39:371–91.
Camm AJ, Kirchhof P, Lip GYH, Schotten U, Savelieva I, Ernst S, et al. Guidelines for the management of atrial fibrillation: The Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J [Internet]. 2010;31:2369–429. https://doi.org/10.1093/eurheartj/ehq278.
Rajsic S, Gothe H, Borba HH, Sroczynski G, Vujicic J, Toell T, et al. Economic burden of stroke: a systematic review on post-stroke care. Eur J Heal Econ [Internet]. 2019;20:107–34. https://doi.org/10.1007/s10198-018-0984-0.
The Danish Council for the use of expensive hospital medicines (RADS). Guideline of oral anticoagulant treatment in non-valvular atrial fibrillation [In Danish: Behandlingsvejledning for oral antikoagulationsbehandling ved non-valvulær atrieflimren] [Internet]. 2016. https://rads.dk/media/3415/beh-atrieflimmer-juni-2016.pdf. Accessed 10 May 2022.
Giugliano RP, Ruff CT, Braunwald E, Murphy SA, Wiviott SD, Halperin JL, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013;369:2093–104.
Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883–91.
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med [Internet]. 2009;361:1139–51. https://doi.org/10.1056/NEJMoa0905561.
Granger CB, Alexander JH, McMurray JJV, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981–92.
Ruff CT, Giugliano RP, Braunwald E, Hoffman EB, Deenadayalu N, Ezekowitz MD, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383:955–62.
Danish Medicines Agency. List of medicinal products. [In Danish: Medicinpriser] [Internet]. https://medicinpriser.dk/?lng=1
Ng SS, Lai NM, Nathisuwan S, Jahan NK, Dilokthornsakul P, Kongpakwattana K, et al. Comparative efficacy and safety of warfarin care bundles and novel oral anticoagulants in patients with atrial fibrillation: a systematic review and network meta-analysis. Sci Rep Nat Res. 2020;10:662.
Nagler M, Bachmann LM, Schmid P, Raddatz Müller P, Wuillemin WA. Patient self-management of oral anticoagulation with vitamin K antagonists in everyday practice: efficacy and safety in a nationwide long-term prospective cohort study. PLoS ONE. 2014;9:e95761.
Alonso-Coello P, Zhou Q, Guyatt G. Home-monitoring of oral anticoagulation vs. dabigatran: An indirect comparison. Thromb Haemost. 2012;108:647–53.
Carles M, Brosa M, Souto JC, Garcia-Alamino JM, Guyatt G, Alonso-Coello P. Cost-effectiveness analysis of dabigatran and anticoagulation monitoring strategies of vitamin K antagonist. BMC Health Serv Res BioMed Central. 2015;15:289.
Heneghan CJ, Garcia-Alamino JM, Spencer EA, Ward AM, Perera R, Bankhead C, et al. Self-monitoring and self-management of oral anticoagulation. Cochrane Database Syst Rev. 2016;7:1–81. https://doi.org/10.1002/14651858.CD003839.pub3
Moloney E, Craig D, Holdsworth N, Smithson J. Optimising clinical effectiveness and quality along the atrial fibrillation anticoagulation pathway: an economic analysis. BMC Health Serv Res. 2019;19:1007.
Matchar DB, Jacobson A, Dolor R, Edson R, Uyeda L, Phibbs CS, et al. Effect of home testing of international normalized ratio on clinical events. N Engl J Med. 2010;363:1608–20.
National Institute for Health and Care Excellence (NICE). Guide to the methods of technology appraisal 2013—process and methods [Internet]. 2013. https://www.nice.org.uk/process/pmg9/chapter/the-reference-case. Accessed 10 May 2022.
OECD. Health at a Glance 2017. Chapter: financing of health care. [Internet]. OECD; 2017. https://www.oecd-ilibrary.org/social-issues-migration-health/health-at-a-glance-2017_health_glance-2017-en. Cited 17 Mar 2022.
Ministry of Finance. Documentation note - the socio-economic discount rate [In Danish: Dokumentationsnotat - den samfundsøkonomiske diskonteringsrente] [Internet]. 2021. https://fm.dk/media/18371/dokumentationsnotat-for-den-samfundsoekonomiske-diskonteringsrente_7-januar-2021.pdf. Accessed 10 May 2022.
Ehlers LH, Vestergaard AS. Costing in health economic evaluation—theory and practice. 1st ed. Aalborg: Aalborg University Press; 2019.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated health economic evaluation reporting standards (CHEERS) statement. Value Health. 2013;16:e1-5.
Vestergaard AS, Ehlers LH. A health economic evaluation of stroke prevention in atrial fibrillation: guideline adherence versus the observed treatment strategy prior to 2012 in Denmark. Pharmacoeconomics. 2015;33:967–79.
Half-Cycle Correction. York; York Health Economics Consortium. [Internet]. 2016. https://yhec.co.uk/glossary/half-cycle-correction/. Accessed 10 May 2022.
Briggs A, Claxton K, Sculpher M. Decison modelling for health economic evaluation. 1st ed. New York: Oxford University Press; 2006.
Edlin R, McCabe C, Hulme C, Hall P, Wright J. Cost effectiveness modelling for health technology assessment—a practical course. Cost Eff. Model. Heal. Technol. Assess. Springer International Publishing; 2015.
Li X, Tse VC, Lau WCY, Cheung BMY, Lip GYH, Wong ICK, et al. Cost-effectiveness of apixaban versus warfarin in chinese patients with non-valvular atrial fibrillation: a real-life and modelling analyses. PLoS ONE. 2016;11: e0157129.
Kongnakorn T, Lanitis T, Annemans L, Thijs V, Marbaix S. Cost effectiveness of apixaban versus aspirin for stroke prevention in patients with non-valvular atrial fibrillation in Belgium. Clin Drug Investig [Internet]. T. Kongnakorn, Evidera, 26-28 Hammersmith Grove, London, United Kingdom; 2014;34:709–21. https://doi.org/10.1007/s40261-014-0224-z.
Statistics Denmark. StatBank Denmark [In Danish: Statistikbanken] [Internet]. 2018. https://www.statistikbanken.dk/statbank5a/default.asp?w=1536. Accessed 10 May 2022.
Andersen KK, Andersen ZJ, Olsen TS. Predictors of early and late case-fatality in a nationwide danish study of 26 818 patients with first-ever ischemic stroke. Stroke. 2011;42:2806–12.
Sorensen S, Kansal A, Connolly S, Peng S, Linnehan J, Bradley-Kennedy C, et al. Cost-effectiveness of dabigatran etexilate for the prevention of stroke and systemic embolism in atrial fibrillation: a Canadian payer perspective. Thromb Haemost [Internet]. 2011;105:908–19. https://doi.org/10.1160/TH11-02-0089.
Nielsen PB, Larsen TB, Gorst-Rasmussen A, Skjøth F, Rasmussen LH, Lip GYH. Intracranial hemorrhage and subsequent ischemic stroke in patients with atrial fibrillation: a nationwide cohort study. Chest. 2015;147:1651–8.
Rasmussen S, Abildstrom SZ, Rosén M, Madsen M. Case-fatality rates for myocardial infarction declined in Denmark and Sweden during 1987–1999. J Clin Epidemiol. 2004;57:638–46.
Ottesen TP. Trends in intracerebral haemorrhage epidemiology in Denmark between 2004 and 2012: incidence, risk-profile and case-fatality. Research year report. [Internet]. 2014. https://kea.au.dk/fileadmin/KEA/filer/Research_year_reports/Tobias_Ottesen_Research_year.pdf. Accessed 10 May 2022.
Bank of England. Bank of England. Statistics. Exchange rates. GBP daily rates. [Internet]. 2020. https://www.bankofengland.co.uk/statistics/exchange-rates. Accessed 10 May 2022.
Borah BJ, Roger VL, Mills RM, Weston SA, Anderson SS, Chamberlain AM. Association between atrial fibrillation and costs after myocardial infarction: a community study. Clin Cardiol [Internet]. 2015;38:548–54. https://doi.org/10.1002/clc.22448.
Jakobsen M, Kolodziejczyk C, Fredslund EK, Poulsen PB, Dybro L, Johnsen SP. Societal costs of first-incident ischemic stroke in patients with atrial fibrillation—a Danish Nationwide Registry Study. Value Health [Internet]. 2016;19:413–8. https://linkinghub.elsevier.com/retrieve/pii/S1098301516000152
Jakobsen M, Kolodziejczyk C, KlausenFredslund E, Poulsen PB, Dybro L, Paaske JS. Costs of major intracranial, gastrointestinal and other bleeding events in patients with atrial fibrillation—a nationwide cohort study. BMC Health Serv Res [Internet]. 2017;17:398. https://doi.org/10.1186/s12913-017-2331-z.
The Danish Health and Medicines Authority. Danish DRG-tariff system[In Danish: Takstsystem 2019]. 2019.
Sullivan PW, Slejko JF, Sculpher MJ, Ghushchyan V. Catalogue of EQ-5D scores for the United Kingdom. Med Decis Mak. 2011;31:800–4.
Gage BF, Cardinalli AB, Albers GW, Owens DK. Cost-effectiveness of warfarin and aspirin for prophylaxis of stroke in patients with nonvalvular atrial fibrillation. JAMA J Am Med Assoc. 1995;274:1839–45.
Drug Price Reduction After Patent Expires [Internet]. https://dyingforacure.org/key-facts/. Accessed 10 May 2022.
Hvilsted Rasmussen L, Jespersen J, Bloch Münster A, Godtfredsen J, Husted S, Lassen J, et al. Self-monitored blood-thinning treatment—an annotated foreign medical technology assessment [In Danish: Selvmonitoreret blodfortyndende behandling – en kommenteret udenlandsk medicinsk teknologivurdering] [Internet]. København; 2009. https://www.sst.dk/-/media/Udgivelser/2009/Publ2009/MTV/blodfortyndende_medicin/Selvmonitoreret_blodfortyndende_medicin_net_final,-d-,pdf.ashx. Accessed 10 May 2022.
Connock M, Stevens C, Fry-Smith A, Jowett S, Fitzmaurice D, Moore D, et al. Clinical effectiveness and cost-effectiveness of different models of managing long-term oral anticoagulation therapy: a systematic review and economic modelling. Health Technol Assess Engl. 2007;11:iii–66.
Danish Medical Association. Consultation fee table. [In Danish: Læger.dk. PLO. Overenskomser og aftaler. Honoarer og takster. Honoartabel. Gruppe-1-takster pr. 1. oktober 2019] [Internet]. https://www.laeger.dk/sites/default/files/honorartabel_2019_oktober-1.pdf. Accessed 10 May 2022.
Acknowledgements
The authors would like to thank Dorte Reffeldt Lund, sales leader at Roche Diagnostics Denmark, for help in estimating the unit costs concerning equipment used in PSM treatment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None for this study.
Conflict of interest
T.B. Larsen has served as an investigator for Janssen Scientific Affairs, LLC, and Boehringer Ingelheim; Larsen has also participated in speaker panels for Bayer, Bristol-Myers Squibb, Pfizer, Roche Diagnostics, and Boehringer Ingelheim; Larsen also received honoraria for consulting activities from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, and Pfizer. T.B. Larsen's institution has received unrestricted funds for investigator-initiated research activities from Bayer, Pfizer, and Daiichi Sankyo. All other authors declare no conflicts of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data Availability
Parts of this study are based on Danish registry data from the study by Grove et al. [1]. The data used in the current publication are reported where relevant. The model used in this study was provided to the journal's peer reviewers for their reference when reviewing the manuscript.
Code availability
All data generated or analysed during this study are included in this published article [and its supplementary information files]. The Markov model are available from the corresponding author on reasonable request.
Author contributions
All authors contributed to the study conception and design. Sabine Michelsen Raunbak performed the modeling in collaboration with Anne Sig Sørensen, Louise Hansen and Lars Holger Ehlers. Input parameters for the model was identified by Sabine Michelsen Raunbak in collaboration with Flemming Skjøth and Torben Bjerregaard Larsen. Torben Bjerregaard Larsen reviewed the model to reflect real clinical practice. All authors contributed to the analysis and interpretation of the results. The first draft of the manuscript was written by Sabine Michelsen Raunbak, and all authors commented on later versions of the manuscript. All authors read and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Raunbak, S.M., Sørensen, A.S., Hansen, L. et al. Cost Effectiveness of Patient Self-Managed Warfarin Compared with Direct Oral Anticoagulants in Atrial Fibrillation: An Economic Evaluation in a Danish Healthcare Sector Setting. PharmacoEconomics Open 6, 483–494 (2022). https://doi.org/10.1007/s41669-022-00337-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-022-00337-3