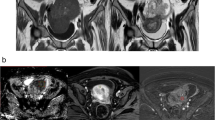
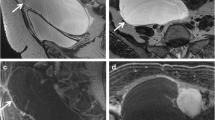
Abstract
Background
Characterization of adnexal masses calls for multidisciplinary involvement. Though a definitive pathological diagnosis mandates surgical intervention, a pre-interventional near-accurate characterization helps in treatment planning. Imaging is a mainstay in this regard.
Aim
This study aimed at analysing ADC values of ovarian lesions observed on a DW–MRI, with an intent to arrive at a probable demarcating value differentiating benign and malignant tumours. The significance of contrast enhancement pattern of lesions was also analysed.
Materials and Methods
All patients with histopathologically proven ovarian tumour (benign and malignant) who had pelvic MRI with DW sequence with b value of 0 and 1000 were included.
Results
Total patients were 62. Mean age was 44 ± 16.8 years. 33.88% were malignant. DWI and ADC values were plotted in 48 of the 62. Diffusion restriction was seen in 81.25% of malignant and 56.25% of benign lesions. The sensitivity and specificity of DWI in differentiating malignancies were 81.25% and 43.75%, respectively. The probable cut-off value of ADC differentiating a malignant and a benign lesion was found to be ≥ 1.13 × 10−3 mm2/s, statistically, using the receiver operating characteristics. 35.48% had early contrast enhancement, and 64.51% delayed enhancement.
Conclusion
DWI has an optimal sensitivity of 81.25% and a low specificity of 43.75% in differentiating malignant from benign lesions. In our study, we found an ADC cut-off of ≥ 1.13 × 10−3 mm2/s, to differentiate between malignant and benign tumours with a sensitivity of 84.4% and specificity of 87.5%. In differentiating between benign and malignant lesions, early enhancement on dynamic contrast study had a good sensitivity.





Similar content being viewed by others
References
Oktem O, Oktay K. The ovary anatomy and function throughout human life. Ann N Y Acad Sci. 2008;1127(1):1–9.
Shahrzad Ehdaivand. WHO classification of ovarian neoplasms. [Internet]. PathologyOutlines.com. 2019 [cited 2019 Mar 8]. http://www.pathologyoutlines.com/topic/ovarytumorwhoclassif.html.
Stewart SL. Ovarian cancer incidence: current and comprehensive statistics division of cancer prevention and control. In: Farghaly DS, editor. Ovarian cancer—clinical and therapeutic perspectives. 1st ed ed. Rijeka: InTech; 2012. p. 1–15.
Noone A, Howlader N, Krapcho M, Miller D, Brest A, Yu M, et al. SEER cancer statistics review, 1975–2015. Bethesda: National Cancer Institute; 2018.
Sohaib S, Sahdev A, Van Trappen P, Jacobs I, Reznek R. Characterization of adnexal mass lesions on MR imaging. Am J Roentgenol. 2003;180(5):1297–304.
Zhang P, Cui Y, Li W, Ren G, Chu C, Wu X. Diagnostic accuracy of diffusion-weighted imaging with conventional MR imaging for differentiating complex solid and cystic ovarian tumors at 1.5T. World J Surg Oncol. 2012;10(1):237.
Lemeshow S, Hosmer DW, Klar J, Lwanga SK. Adequacy of sample size in health studies. 1st ed. West Sussex: Wiley; 1990.
Foti PV, Attina G, Spadola S, Caltabiano R, Farina R, Palmucci S, et al. MR imaging of ovarian masses: classification and differential diagnosis. Insights Imaging. 2016;7(1):21–41.
Menon U, Hallett R, Ryan A, Burnell M. Sensitivity and specificity of multimodal and ultrasound screening for ovarian cancer, and stage distribution of detected cancers: results of the prevalence screen of the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS). Lancet Oncol. 2009;10(April):327–40.
Iyer VR, Lee SI. MRI, CT, and PET/CT for ovarian cancer detection and adnexal lesion characterization. Am J Roentgenol. 2010;194(2):311–21.
Kinkel K, Lu Y, Mehdizade A, Pelte M-F, Hricak H. Indeterminate ovarian mass at US: incremental value of second imaging test for characterization—meta-analysis and bayesian analysis. Radiology. 2005;236:85–94.
Adusumilli S, Hussain HK, Caoili EM, Weadock WJ, Murray JP, Johnson TD, et al. MRI of sonographically indeterminate adnexal masses. Am J Roentgenol. 2006;187(3):732–40.
Komatsu T, Konishi I, Mandai M, Togash K, Kawakami S, Konishi J, et al. Adnexal masses: transvaginal US and gadolinium-enhanced MR imaging assessment of intratumoral structure. Radiology. 1996;198(1):109–15.
Yuan X, Guo L, Du W, Mo F, Liu M. Diagnostic accuracy of DWI in patients with ovarian cancer. Medicine. 2017;96(19):1–7.
Bakir B, Bakan S, Tunaci M, Bakir VL, Iyibozkurt AC, Berkman S, et al. Diffusion-weighted imaging of solid or predominantly solid gynaecological adnexial masses: Is it useful in the differential diagnosis? Br J Radiol. 2011;84(1003):600–11.
Thomassin-Naggara I, Daraï E, Cuenod C, Fournier L, Toussaint I, Marsault C, et al. Contribution of diffusion-weighted MR imaging for predicting benignity of complex adnexal masses. Eur Radiol. 2009;19(6):1544–52.
Thomassin-Naggara I, Toussaint I, Perrot N. Characterization of complex adnexal masses: value of adding perfusion- and diffusion-weighted MR imaging to conventional MR imaging. Radiology. 2011;258(3):793–803.
Takeuchi M, Matsuzaki K, Nishitani H. Diffusion-weighted magnetic resonance imaging of ovarian tumors: differentiation of benign and malignant solid components of ovarian masses. J Comput Assist Tomogr. 2010;34(2):173–6.
Kovač JD, Terzić M, Mirković M, Banko B, Đikić-Rom A, Maksimović R. Endometrioid adenocarcinoma of the ovary: MRI findings with emphasis on diffusion-weighted imaging for the differentiation of ovarian tumors. Acta Radiol. 2016;57(6):758–66.
Katayama M, Masui T, Kobayashi S, Ito T, Sakahara H, Nozaki A, et al. Diffusion-weighted echo planar imaging of ovarian tumors: is it useful to measure apparent diffusion coefficients? J Comput Assist Tomogr. 2002;26(2):250–6.
Cappabianca S, Iaselli F, Reginelli A, D’Andrea A, Urraro F, Grassi R, et al. Value of diffusion-weighted magnetic resonance imaging in the characterization of complex adnexal masses. Tumori. 2013;99(2):210–7.
Lee NK, Kim S, Kim KH, Suh DS, Kim TU, Han GJ, et al. Diffusion-weighted magnetic resonance imaging in the differentiation of endometriomas from hemorrhagic cysts in the ovary. Acta Radiol. 2016;57(8):998–1005.
Kim HJ, Lee SY, Shin YR, Park CS, Kim K. The value of diffusion-weighted imaging in the differential diagnosis of ovarian lesions: a meta-analysis. PLoS One. 2016;11(2):1–13.
Motekia T, Horikoshic H, Endo K. Relationship between apparent diffusion coefficient and signal intensity in endometrial and other pelvic cysts. Magn Reson Imaging. 2002;20(6):463–70.
Mastrolia SA, Alvarez-Arguelles H, Carballo J, Amaya F, de la Torre J. Steroid cell tumor (NOS) of the ovary associated to high levels of CA-125. Gynecol Surg. 2011;8(2):231–3.
Funding
The authors have not received any funding either from the institution or from an external source.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jayasankaran, S.C., Chelakkot, P.G., Prabhakaran, P. et al. Role of Diffusion-Weighted Magnetic Resonance Imaging in the Evaluation of Ovarian Tumours. Indian J Gynecol Oncolog 17, 64 (2019). https://doi.org/10.1007/s40944-019-0305-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40944-019-0305-2