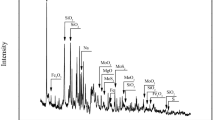
Abstract
MoO3 is a widely accessible foundation material and the primary compound used to produce numerous molybdenum products. The ore was first roasted, and the leaching behavior of concentrated molybdenite mineral was assessed. The optimal conditions for the leaching process were identified in the second step, and in a subsequent stage, the process' kinetics were assessed. Ammonia and ammonium nitrate concentration were used as independent variables in the trials, and the central composite design approach was used to analyze the results. At the conclusion of the tests, the optimum concentrations of ammonia and ammonium nitrate were found to be 2.42 mol/L and 1.86 mol/L, respectively. On the rate at which molybdenum leached from MoO3, the influences of temperature, stirring speed, solid-to-liquid ratio, and particle size were investigated. It was found that the leaching rate increased when temperature and stirring speed increased, along with decreasing the solid-to-liquid ratio and particle size. Leaching efficiency of molybdenum (93%) such as temperature 20 °C, 1.859 mol/L ammonium nitrate concentration, 2.419 mol/L ammonia concentration, solid/liquid ratio 6/100 g/mL, stirring speed 400 rpm, and average particle size 0.115 mm optimum conditions have been achieved. The kinetic study proposed a mathematical model for the leaching process, which revealed that the leaching kinetics matched the Avrami model. This process' activation energy was determined to be 22.66 kJ/mol. Calcium molybdate is used in various fields, such as construction, transportation, anti-corrosion pigments, and flame retardants.
Graphical Abstract


















Similar content being viewed by others
References
Saji VS, Lee C (2012) Molybdenum, molybdenum oxides, and their electrochemistry. ChemSusChem 5:1146–1161. https://doi.org/10.1002/cssc.201100660
McGill I, Habashi F (1997) Handbook of extractive metallurgy, vol III. Wiley-VCH, Weinheim
Cao Z, Zhong H, Qiu Z et al (2009) A novel technology for molybdenum extraction from molybdenite concentrate. Hydrometallurgy 99:2–6. https://doi.org/10.1016/j.hydromet.2009.05.001
Vosough M, Khayati GR, Sharafi S (2022) Ammonia leaching of MoO3 concentrate: finding the reaction mechanism and kinetics analysis. Chem Pap 76:3227–3237. https://doi.org/10.1007/s11696-022-02098-z
Shalchian H, Ferella F, Birloaga I et al (2019) Recovery of molybdenum from leach solution using polyelectrolyte extraction. Hydrometallurgy. https://doi.org/10.1016/j.hydromet.2019.105167
Kim BS, Jha MK, Jeong J, Lee J (2008) Leaching of impurities for the up-gradation of molybdenum oxide and cementation of copper by scrap iron. Int J Miner Process 88:7–12. https://doi.org/10.1016/j.minpro.2008.04.002
Habbache N, Alane N, Djerad S, Tifouti L (2009) Leaching of copper oxide with different acid solutions. Chem Eng J 152:503–508. https://doi.org/10.1016/j.cej.2009.05.020
Ekmekyapar A, Aktaş E, Künkül A, Demirkiran N (2012) Investigation of leaching kinetics of copper from malachite ore in ammonium nitrate solutions. Metall Mater Trans B 43:764–772. https://doi.org/10.1007/s11663-012-9670-2
Künkül A, Gülezgin A, Demirkiran N (2013) Investigation of the use of ammonium acetate as an alternative lixiviant in the leaching of malachite ore. Chem Ind Chem Eng Q 19:25–34. https://doi.org/10.2298/CICEQ120113039K
Rudnik E, Pierzynka M, Handzlik P (2016) Ammoniacal leaching and recovery of copper from alloyed low-grade e-waste. J Mater Cycles Waste Manage 18:318–328. https://doi.org/10.1007/s10163-014-0335-x
Il NY, Seo SY, Kang YC et al (2011) Purification of molybdenum trioxide calcine by selective leaching of copper with HCl-NH4Cl. Hydrometallurgy 109:9–17. https://doi.org/10.1016/j.hydromet.2011.05.001
Shariat MH, Setoodeh N, Dehghan RA (2001) Optimizing conditions for hydrometallurgical production of purified molybdenum trioxide from roasted molybdenite of sarcheshmeh. Miner Eng 14:815–820. https://doi.org/10.1016/S0892-6875(99)00000-X
Çalban T, Çolak S, Yeşilyurt M (2005) Optimization of leaching of copper from oxidized copper ore in NH3-(NH4)2SO4 medium. Chem Eng Commun 192:1515–1524. https://doi.org/10.1080/009864490896106
Hinkelmann K (2012) Design and analysis of experiments. Wiley, Hoboken
Tanaydın MK, Bakıcı Tanaydın Z, Demirkıran N (2021) Determination of optimum process conditions by central composite design method and examination of leaching kinetics of smithsonite ore using nitric acid solution. J Sustain Metall 7:178–191. https://doi.org/10.1007/s40831-020-00333-z
Tanaydin MK, Goksu A (2021) Optimization of the adsorption of methyl green dye on almond shells using central composite design. Desalin Water Treat 227:425–439. https://doi.org/10.5004/dwt.2021.27297
Tanaydin MK, Tanaydin ZB, Demirkiran N (2022) Optimization of process parameters and kinetic modelling for leaching of copper from oxidized copper ore in nitric acid solutions. Trans Nonferrous Met Soc China 32:1301–1313. https://doi.org/10.1016/S1003-6326(22)65875-8
Fan X, Deng Q, Gan M, Chen X (2019) Roasting oxidation behaviors of ReS2 and MoS2 in powdery rhenium-bearing, low-grade molybdenum concentrate. Trans Nonferrous Met Soc China (English Ed) 29:840–848. https://doi.org/10.1016/S1003-6326(19)64994-0
Kan S, Benzeşik K, Odabaş ÖC, Yücel O (2021) Investigation of molybdenite concentrate roasting in chamber and rotary furnaces. Min Metall Explor 38:1597–1608. https://doi.org/10.1007/s42461-021-00429-4
Lee J-Y, Kumar JR, Jeon H-S, Kim J-S (2013) Up-gradation of MoO3 and separation of copper, iron, zinc from roasted molybdenum ore by a leaching process. Braz J Chem Eng 30:391–397. https://doi.org/10.1590/S0104-66322013000200016
Zhang M, Liu C, Zhu X et al (2021) Preparation of ammonium molybdate by oxidation roasting of molybdenum concentrate: a comparison of microwave roasting and conventional roasting. Chem Eng Process—Process Intensif 167:108550. https://doi.org/10.1016/j.cep.2021.108550
Levenspiel O (1972) Chemical reaction engineering, 3rd edn. Wiley, New York
Wen CY (1968) Noncatalytic heterogeneous solid-fluid reaction models. Ind Eng Chem 60:34–54. https://doi.org/10.1021/ie50705a007
Zheng YJ, Chen KK (2014) Leaching kinetics of selenium from selenium-tellurium-rich materials in sodium sulfite solutions. Trans Nonferrous Met Soc China (English Ed) 24:536–543. https://doi.org/10.1016/S1003-6326(14)63093-4
Sohn HY (1976) Kinetics of non-catalytic fluid-solid reactions in chemical and extractive metallurgical processes: part I. Reaction of a single nonporous particle. Hwahak Konghak 14:3–16
Demirkiran N, Özdemir GDT (2019) A kinetic model for dissolution of zinc oxide powder obtained from waste alkaline batteries in sodium hydroxide solutions. Metall Mater Trans B 50:491–501. https://doi.org/10.1007/s11663-018-1469-3
Gu K, Li W, Han J et al (2019) Arsenic removal from lead-zinc smelter ash by NaOH-H2O2 leaching. Sep Purif Technol 209:128–135. https://doi.org/10.1016/j.seppur.2018.07.023
Shi M, Min X, Shen C et al (2021) Separation and recovery of copper in Cu−As-bearing copper electrorefining black slime by oxidation acid leaching and sulfide precipitation. Trans Nonferrous Met Soc China 31:1103–1112. https://doi.org/10.1016/S1003-6326(21)65564-4
Ilhan S, Akgün D (2021) Leaching kinetics of Mo, Ni, and Al oxides from spent nickel-molybdenum hydrodesulfurization catalyst in H2SO4 solution. J Sustain Metall 7:470–480. https://doi.org/10.1007/s40831-021-00351-5
Aracena A, Azocar A, Ibáñez JP, Jerez O (2019) Mechanism and leaching kinetics of molybdenite concentrate in a hydrogen peroxide-acid system. Physicochem Probl Miner Process 55:140–152. https://doi.org/10.5277/ppmp18139
Zhao Z, Guo M, Zhang M (2015) Extraction of molybdenum and vanadium from the spent diesel exhaust catalyst by ammonia leaching method. J Hazard Mater 286:402–409. https://doi.org/10.1016/j.jhazmat.2014.12.063
Alguacil FJ, Cobo A (1998) Separation of copper and nickel from ammoniacal/ammonium carbonate solutions using ACORGA PT5050. Sep Sci Technol 33:2257–2264. https://doi.org/10.1080/01496399808545726
Yang R, Wang S, Duan H et al (2016) Efficient separation of copper and nickel from ammonium chloride solutions through the antagonistic effect of TRPO on Acorga M5640. Hydrometallurgy 163:18–23. https://doi.org/10.1016/j.hydromet.2016.03.006
Alguacil FJ (1999) Recovery of copper from ammoniacal/ammonium carbonate medium by LIX 973N. Hydrometallurgy 52:55–61. https://doi.org/10.1016/S0304-386X(99)00012-2
Oishi T, Koyama K, Alam S et al (2007) Recovery of high purity copper cathode from printed circuit boards using ammoniacal sulfate or chloride solutions. Hydrometallurgy 89:82–88. https://doi.org/10.1016/j.hydromet.2007.05.010
Gameiro MLF, Ismael MRC, Reis MTA, Carvalho JMR (2008) Recovery of copper from ammoniacal medium using liquid membranes with LIX 54. Sep Purif Technol 63:287–296. https://doi.org/10.1016/j.seppur.2008.05.009
Sengupta B, Bhakhar MS, Sengupta R (2007) Extraction of copper from ammoniacal solutions into emulsion liquid membranes using LIX 84 I®. Hydrometallurgy 89:311–318. https://doi.org/10.1016/j.hydromet.2007.08.001
Gameiro MLF, Machado RM, Ismael MRC et al (2010) Copper extraction from ammoniacal medium in a pulsed sieve-plate column with LIX 84-I. J Hazard Mater 183:165–175. https://doi.org/10.1016/j.jhazmat.2010.07.006
Vidya S, Solomon S, Thomas JK (2012) Synthesis, sintering and optical properties of CaMoO4: a promising scheelite LTCC and photoluminescent material. Phys Status Solidi Appl Mater Sci 209:1067–1074. https://doi.org/10.1002/pssa.201127620
Oliveira FKF, Oliveira MC, Gracia L et al (2018) Experimental and theoretical study to explain the morphology of CaMoO4 crystals. J Phys Chem Solids 114:141–152. https://doi.org/10.1016/j.jpcs.2017.11.019
Xiang Y, Song J, Hu G, Liu Y (2015) Synthesis of CaMoO4 hierarchical structures via a simple slow-release co-precipitation method. Appl Surf Sci 349:374–379. https://doi.org/10.1016/j.apsusc.2015.04.228
Reuss G, Disteldorf W, Gamer AO, Hilt A (2000) Ullmann’s encyclopedia of industrial chemistry. Wiley, Hoboken
Acknowledgements
We thank Mineral Research and Exploration Institute (MTA), Ankara, Türkiye. for providing us with free samples. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of ınterest
All authors have declared no conflict of interest.
Additional information
The contributing editor for this article was Atsushi Shibayama.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karacahan, M.K. Investigation of Leaching Kinetics of Molybdenum from MoO3 Concentrate in Ammonia–Ammonium Nitrate Solutions. J. Sustain. Metall. 9, 1564–1577 (2023). https://doi.org/10.1007/s40831-023-00748-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40831-023-00748-4