Abstract
An innovative and sustainable way for production of vanadium (V) is to co-process V titanomagnetite ores with V-bearing steel slag, via reduction. V-slag produced from highly enriched Fe–V melt contains as high as 20 mass% V. The phase relationship in FeO–SiO2 based slag with V fixed at 20.4 mass% was investigated experimentally at 1673 K. The compositions covered the normal production range, viz. Al2O3 from 0 to 6 mass%, CaO from 1 to 5 mass% and SiO2 from 10 to 17 mass%. The content of MnO was fixed at 5.5 mass%. Samples quenched at 1673 K were found to be solid–liquid mixtures, either in a 2-phase region consisting of liquid and a solid spinel phase, or in 3-phase region consisting of liquid, spinel phase and silica phase. Free silica was only found in samples with a composition of 0 mass% Al2O3, 1 mass% CaO and 17 mass% SiO2. Formation of free silica was prohibited in samples with 1 mass% CaO and up to 17 mass% SiO2 by adding 5 mass% Al2O3. Addition of 4 mass% Al2O3 was also sufficient to prevent the presence of free silica in samples with 3 mass% CaO and 13.5 mass% SiO2. Most of the V was kept in the spinel phase, while the solubility of V in the liquid was generally less than 1 mass% as V2O3. Fe, Mn and Al coexisted in both liquid phase and spinel phase. On the other hand, Si and Ca only distributed in the liquid phase. The fraction of solid phase, viz. spinel was found to decrease with increasing SiO2 content.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
There are several vanadium (V) titanomagnetite ore deposits in the world, particularly in Scandinavia. Due to the small size of these iron mines, the feasibility for an economic exploitation is dependent on the possibility to valorize the V as a co-product to iron. V titanomagnetite ores globally constitute the main source for production of V containing commodities, most importantly vanadium pentoxide (V2O5) and ferrovanadium (FeV) [1, 2]. FeV is produced from vanadium (III) oxide (V2O3) or V2O5 [1, 2]. Extraction of V as a co-product to iron is usually done by concentrating the V into a V-slag. The V slag is then converted to V2O5 by a salt roast and leach process [1]. V-slag is commonly oxidized by oxygen and transformed into water soluble sodium vanadates in the presence of sodium salts (Na2CO3, NaCl, NaOH and/or Na2SO4) [1, 3–5]. Thereafter, V2O3 or V2O5 is obtained from the leachate by precipitation and calcination [1, 5]. Production of V-slag involves two main pyrometallurgical steps. At first, the ore concentrate or DRI (Direct Reduced Iron) is reduced to a hot metal with a V-content in the range of 0.4–1.3 mass% [1, 6]. In the second step, the V in the hot metal is oxidized to the V slag at around 1673 K [1, 6]. The V slag is an acid FeO–SiO2 based slag with normally 9–15 mass% V [1, 6–8]. High FeO activity in the slag results in high V distribution ratio between slag and hot metal [9–12]. Low P content is essential in order to satisfy the requirements on the final products [1].
An innovative and sustainable way for production of V is to co-process V titanomagnetite ores with V-bearing steel slag (BOF-slag). Some BOF-slags in Europe, in particular in Scandinavia, contains as high as 3 mass% as V. The presence of heavy metals like V and chromium (Cr) limits the external use of the slag. In many cases the slags are disposed. In addition to V, iron (Fe) is recovered and the lime (CaO) in the BOF-slag is reused. In a parallel work, an ore concentrate from the Mustavaara deposit in northern Finland containing about 0.8 mass% V was co-processed with a V-bearing steel slag with around 2 mass% V [13]. The hot metal was composed of about 1.7 mass% V, 0.1 mass% P, 0.3 mass% Si and 0.3 mass% Mn. The V-slag produced from the highly enriched Fe–V melt contained as high as 20 mass% V [13]. Typical V slags also contains up to 6 mass% Al2O3, up to 5 mass% CaO and SiO2 in the range of 10–17 mass% [1, 6–8]. These components originate from carry-over slag, gangue in cooling agents and by oxidation of silicon in the hot metal. However the concentration of these oxides becomes diluted by the increasing V content in the slag. Very little information is available regarding the impact of these components on the phase relationship. Fang et al. studied the influence of CaO at 1673 K with Al2O3, SiO2 and V fixed at 3–4, 16–17 and 7–9 mass%, respectively [14]. It was reported that the slag was composed of a spinel phase surrounded by silicate phases. At 2.81 mass% CaO the identified silicate phase was olivine (Me2SiO4) with 5.27 mass% CaO, the olivine had to some extent been transformed to augite (Ca(Mg, Fe, Mn)(Si, Al)2O6). The V concentration in the spinel was also reported to decrease with increasing CaO-content in the slag. The melting point of the FeO·V2O3 spinel in the FeO–V2O3-system is over 1973 K [15]. Both the FeO·V2O3 spinel and V2O3 is formed when the Fe to V molar ratio is larger than 2 (>67.6 mass% V2O3) [15]. V2O3 (karelianite) can also form solid solutions with Al and Fe [16–20]. CaO decrease the melting point of V2O3 and the spinel [21, 22]. Diao et al. reported that the V, Cr and Ti in V-slag were concentrated in the spinel phase, to some extent together with Mg and Ca [7, 8]. The Si was found to be concentrate in the silicate phases while Fe, Mn and Al coexist in both the spinel and silicate phase with uniform distribution [7]. Ge et al. reported that free silica is not generated in the production of V slag [23]. This work aims at providing a fundamental understanding of the phase relationship in the FeO–SiO2 based V slags at 1673 K. The results are expected to provide important knowledge in the production of V slags and its impact on the following extraction processes. The studied compositions will cover the normal production range of Al2O3 from 0 to 6 mass%, CaO from 1 to 5 mass% and SiO2 from 10 to 17 mass%, with V and MnO fixed at 20.4 and 5.5 mass%, respectively.
Experimental Setup and Procedure
V slags were prepared synthetically from Al2O3, CaO, FeO, MnO, SiO2 and V2O3 powders. The suppliers and purities of the powders are listed in Table 1. The Al2O3, CaO and SiO2 powders were kept at 1173 K for 10 h to remove moisture and volatiles. The Fe2O3 and MnO were dried at 373 K for 12 h. First, the FeO was prepared from a Fe and Fe2O3 mixture with a total composition of 51 mol % oxygen. The Fe and Fe2O3 powders were mixed thoroughly in an agate mortar. The mixture was packed tightly into an iron crucible and sealed with a conical lid. A vertical resistance furnace equipped with MoSi2 heating elements and Al2O3 working tube, sealed with rubber stoppers, was used. The mixture was heated up with a rate of 2 K/min to 1163 K in argon atmosphere (supplied by Aga Linde, H2O ≤ 0.5 ppm, O2 ≤ 0.5 ppm). The Ar gas flow was kept at about 0.1 l/min at standard ambient temperature (298 K) and pressure (1 bar) (hereinafter referred to as ln/min). The mixture was kept at 1163 K for 60 h before cooling to room temperature at a rate of 2 K/min. The V slag samples were then prepared by mixing appropriate ratios of the oxides in an agate mortar. The mixtures were pressed at 10 MPa into pellets with a diameter of 11 mm and a weight of about 5 g. The MnO and V2O3 contents were targeted to 5.5 and 30 mass% (20.4 mass% V) in all samples. The slag compositions were balanced with FeO. The ratios of the mixed powders for all 24 samples are shown in Table 2. In order to examine the reliability of the present technique with respect to both the equilibrium and quenching, experiments no. 23 and no. 24 were carried out with the same initial composition.
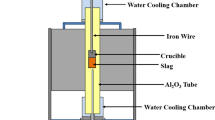
A vertical resistance furnace, equipped with MoSi2 heating elements and Al2O3 working tube (OD: 80 mm, ID: 70 mm, H:1000 mm), was used for the investigations (see Fig. 1). In order to quench the samples rapidly without withdrawal from the furnace, the working tube was interconnected to a water cooled Al-chamber. The pellet was placed in a molybdenum (Mo) crucible (OD 14 mm, ID 13 mm, H 40 mm). For each run, 3 crucibles were placed in a Mo sample holder (OD 54 mm, ID 48 mm, H 120 mm). In view that the slag in the reactor is exposed to different oxygen partial pressures through the thickness of the slag phase, to study the phases of the slag, the slag composition must be maintained. Because of the presence of vanadium oxide in the slag and its multi-valence nature at different oxygen potentials, it was necessary to use a closed system for all samples. A Mo lid fixed with a weight was placed on the top of each sample crucible. In addition, the sample holder was sealed with a Mo cap. This arrangement would ensure that the slag would not react with the gas phase in the reaction chamber. The holder was connected to the lifting unit via a steel rod with a Mo extension. A B-type thermocouple was placed just below the samples inside the reaction chamber. After the reaction chamber was sealed, the whole system was evacuated with a vacuum pump and then filled with the high quality argon. This procedure was repeated at least 3 times. The furnace was heated up to 1873 K at a rate of 2 K/min. The samples were then lowered down slowly to the reaction chamber. The samples were kept at this temperature for 6 h before being cooled down with a rate of 2 K/min to the targeted temperature of 1673 K, and kept there for 48 h. Finally the samples were either cooled to room temperature at a rate of 2 K/min or quenched in the cooling chamber. Throughout the tests, Ar was flushed through the chamber with a flow rate of 0.1 ln/min. The weights of the sample before and after experiment were carefully weighed. The weight change was negligible, indicating thereby that no reaction between the sample and gas phase took place (e.g. decomposition).
Slag samples were prepared for SEM–EDS analysis (Hitachi S-3700N with a Bruker EDS analyzer) to identify the phases and their compositions. For this purpose, the samples were mounted into conductive embedding material and polished in ethanol. Gold coating was made using a JEOL JFC-1300. The phase fractions were estimated using a Leica DM4500 P instrument equipped with Leica LAS and QWin image analysis software.
Results
In total, 4 main phases are identified in the quenched samples, namely (1) spinel phase, (2) free silica and a matrix consisting of two silicate phases. The multiphase matrix has dendritic structure and the coarseness varied between the samples. Samples slow cooled at a rate of 2 K/min had the coarsest matrix structure. This indicates that these phases have precipitated from a liquid phase during cooling. This aspect will be discussed in detail later. The quenched samples consisted either of two phases (spinel and matrix) or of 3-phases (spinel, free silica and matrix) at 1673 K. As examples, Fig. 2 presents a SEM microphotograph of the 2-phase equilibrium and Fig. 3 of the 3-phase equilibrium. Figure 4 presents a SEM microphotograph for a slow cooled sample. Due to the tiny sizes of the dendrites in sample no. 2, the matrix in Fig. 2 is marked solely as M. Note that certain amount of Mo was detected in the samples. The total amount of dissolved Mo in the samples ranges from about 1–3 mass% as MoO2. The Mo was expected to originate from the sample crucible. This aspect will be discussed in detail later. The phases found in the Al2O3–CaO–FeO–MnO–SiO2–V2O3 system as well as the average compositions (over four measured values) of the matrix are presented in Table 3 for the quenched samples. The matrix was analyzed with area scan at 600 times magnification. Normalization was done by taking the sum of the six components, Mo excluded, as 100 mass%. The average compositions (over four measured values) of the spinel phase are presented for the quenched samples in Table 4. It is well documented that the spinel phase is a solid solution, the composition of which depends on the weighed-in composition [24, 25].
As mentioned earlier, sample 23 and 24 were prepared with the same weighed-in composition in order to examine the reliability of the present technique. Both samples were found to be in the 3-phase equilibrium region. The good agreement between the samples indicates the reproducibility of the experimental technique. Some minor content of Al2O3 was found in the matrix and the spinel phase in sample 23 and 24, despite a weighed-in composition of 0 mass% Al2O3. Some Al2O3 could originate as a trace element in the used chemicals. However, it should be pointed out that this concentration range (<0.4 mass%) is below the limit of the measurement technique. The average relative weight loss of all samples (including the whole crucible) was 0.38 %. This indicates that the impact from the surroundings was limited. V has been reported to concentrate in the spinel phase; Ca to some extent distributed to the spinel phase, the Si to concentrate in the silicate phases while Fe, Mn and Al coexist in the spinel phase and the silicate phases [7]. While the present results are mostly in line with these findings, no Ca is detected in the spinel phase.
Discussion
The Closed System
As mentioned in the experiment part, a closed system is used for each sample in the experiment. It implies that the chemical potentials in the closed crucible are determined by the sample compositions. This arrangement is necessary, since the slag in the reactor is exposed to different oxygen partial pressures through the thickness of the slag phase. The closed system would ensure that the sample having different compositions being studied under well-defined equilibrium conditions, with respect to the chemical potentials and temperature.
Precipitation of the Silicate Phases During Cooling
While the matrix was found to consist of mainly five metal oxides, e.g. Al, Ca, Fe, Mn and Si oxide, the two major components in the matrix are FeO x and SiO2. There is no phase diagram available for this complex system. Therefore, the three ternary systems, Al2O3–FeO n –SiO2, CaO–FeO n –SiO2 and FeO n –MnO x –SiO2 are selected in order to estimate the liquidus temperature of the matrix [26]. Normalization is carried out by excluding all other components than FeO n , SiO2 and the third component. The concentrations of the three components are normalized in proportion to their original fractions with their sum being 100 mass%. The normalized compositions of the matrix phase in the quenched samples are marked in the ternary systems in Fig. 5a–c.
According to these ternary systems, it is very likely that the matrix phase is liquid at 1673 K. It is commonly known that a mixing of three liquids would not increase the liquidus temperature. It is also indicated that the liquidus temperature decreases with increasing Al2O3 and CaO content in the investigated concentration range. The dendritic structure of the matrix suggests that the two silicate phases have precipitated during cooling, due to insufficient quenching. This dendritic structure would further support the argument that the matrix is originally liquid at the experimental temperature. Samples 17–22 were slow-cooled. The compositional range of the silicate phases in samples 17–22 is presented in Table 5. The molar ratio of Fe to Si is about 2 in the light grey matrix phase shown in Fig. 4 (sample no. 21). Therefore, this phase could be referred to as olivine (Fe2SiO4) [7, 14]. The molar ratio of Ca to (Mn + Fe) and Ca to (Si + Al) is about 1 and 2, respectively in the dark grey matrix phase shown in Fig. 4 (sample no. 21). Hence, this phase is very likely the augite phase. (Ca(Fe, Mn)(Si, Al)2O6) [7, 14]. Note that the melting temperatures of both olivine and augite are lower than the experimental temperature; the presence of the two phases again strengthens the conclusion that the matrix is liquid at the experimental temperature.
Presence of a Solid Spinel Phase at 1673 K
To verify the phase equilibrium of a solid spinel phase at 1673 K, line analyses of spinel crystals are made. An example of a line analysis is presented in Fig. 6 (sample no. 1), where the position of the line analysis is shown in Fig. 6a and the concentration of Al, Fe, Mn, V and Si as function of the distance along the line in Fig. 6b. The concentrations of the components are relatively constant over the spinel crystal and no overall gradients can be observed. An abrupt change in the composition is seen at the interface between the spinel crystal and the matrix. The V2O3 content in the spinel phase is in average about 52 mass%. According to the phase diagram of the 2-phase system FeO–V2O3 [15], the melting point for the spinel phase with a FeO to V2O3 ratio of about 1 is over 1973 K. The mean size of spinel crystals in industrial samples has been reported to be in the range of 17–33 µm [8]. The crystal size has been found to increase with increasing holding time and decreasing cooling rate [7, 8, 27]. Most of the spinel grains in the quenched samples are larger than 30 µm. Agglomerates of spinel larger than 100 µm are also observed, shown in Fig. 2. The large size of the crystals and the absence of concentration gradients in the spinel grains strongly suggest that the growth of the spinel has occurred during the long holding time at 1673 K.
Precipitation of “Free” Silica
Free silica is found in the samples with a weighed-in composition of 0 mass% Al2O3, 1 mass% CaO and 17 mass% SiO2 (sample no. 23 and no. 24). Formation of free silica is prohibited in samples with 1 mass% CaO and up to 17 mass% SiO2 by adding 5 mass% Al2O3 (sample no. 1, 5 and 16). Addition of 4 mass% Al2O3 is sufficient to prevent the presence of free silica in samples with 3 mass% CaO and 13.5 mass% SiO2 (sample no. 6). It is shown in Fig. 5a–c that the liquid compositions are close to the tridymite composition triangle. It is also apparent according to these figures that the precipitation of tridymite is less likely to occur with increasing Al2O3 and CaO contents. These findings differ from the suggestion of Ge et al. based on a phase diagram analysis by considering the pseudo-ternary CaO–FeO–MgO–SiO2 system [23]. They report that precipitation of free silica is possible only if the SiO2-content is above 40 mass% in the liquid [23]. In the present work, free silica was found in samples (no. 23 and no. 24) with a SiO2-content of below 30 mass% in the matrix.
To adjust the composition of the slag in order to prohibit precipitation of free silica is important for the roasting performance. Silica is also formed during roasting when olivine is oxidized and decomposed to Fe2O3 and SiO2 [5, 28]. Detrimental effect of silica is associated with the formation of sodium silicates with low-melting point, which encapsulate unreacted spinel grains, consequently limiting oxygen diffusion towards the spinel core [5, 28]. V oxides are thus retained in water insoluble solid solutions. Another detrimental effect of silica is that sodium silicates can hydrolyze during water leaching to produce hydrated silica, which increases the viscosity of the leaching solution and slows down diffusion of vanadate ions in water [5]. It has been reported that Al2O3, if present, forms sodium aluminum silicates, which melt at higher temperature than those usually applied in roasting and prevents formation of sodium silicates with low-melting point [2, 5]. As a result, formation of water-insoluble sodium aluminum silicates such as NaAlSiO4 enhances V leaching [5]. Lime can cause difficulties by forming insoluble calcium vanadates during roasting [2, 28]. It can therefore be concluded that alumina is the preferred additive to prevent precipitation of silica in the V slag. However, to what extent an addition of 5 mass% Al2O3 in the V slag will prohibit the formation of sodium silicates with low-melting point, as olivine is oxidized and decomposed, needs investigation in the future.
The Effect of Al2O3 on the Phase Compositions
The effect of Al2O3 on the phase compositions is investigated in the range of 4–6 mass% with CaO fixed at 3 mass% and SiO2 at 13.5 mass%. The contents of the components in the matrix and the spinel phase as functions of the total initial Al2O3 content are shown in Fig. 7a and b, respectively. The bars represent the standard deviation (STD) for each sample (over four measured values). It is shown that Al2O3, in the matrix and the spinel phase, increases with increasing total initial Al2O3 content. FeO in the matrix and spinel decreases while the other components are unaffected by the increasing initial Al2O3 content. Al2O3 substitutes FeO in both the matrix and the spinel phase, in similar proportions, as the total initial FeO content decreases.
The Effect of CaO on the Phase Compositions
The effect of CaO on the phase compositions is investigated in the range of 1–5 mass% with Al2O3 fixed at 5 mass% and SiO2 at 13.5 mass%. The contents of the components and their STD in the matrix and the spinel phase (over four measured values) as a function of the initial CaO content are presented in Fig. 8a and b, respectively. The increasing CaO content in the matrix results in a correlative decrease of FeO in the matrix, without having any significant effect on the FeO content or any other component in the spinel phase. The solely substitution of FeO in the matrix shows that CaO distributes to the matrix. In an earlier work by Fang et al. [14], V in spinel was found to decrease with increasing CaO in the range of 2.81 and 14.94 mass%. In the present work, CaO is found to substitute FeO in the matrix; no decrease of V in the spinel is seen. In the work done by Fang et al., the increasing CaO content in the samples was not balanced by a correlative decrease of FeO [14]. CaO substitution of FeO in the matrix could be used to explain the decreasing V with increasing CaO content in the spinel phase, as reported by Fang et al., since the distribution of FeO to the spinel phase increases with increasing CaO.
The Effect of SiO2 on the Phase Compositions
The effect of SiO2 on the phase compositions is investigated in the range of 10–17 mass% with Al2O3 fixed at 5 mass% and CaO fixed at 3 mass%. The contents of the components and their standard deviation in the matrix and the spinel phase (over four measured values) as a function of the total initial SiO2 content are shown in Fig. 9a and b, respectively. The increase of the SiO2 content in the matrix from about 25–30 mass% corresponds to the decrease of FeO and CaO together. The MnO and Al2O3 contents in the matrix seem unaffected. On the other hand, the increasing V2O3 content results in most significantly a correlative decrease of FeO in the spinel phase.
The Effect of Al2O3, CaO and SiO2 on the Phase Relationship
The effect of the total initial Al2O3, CaO and SiO2 contents on the phase fraction of the spinel phase is shown in Fig. 10. The phase fraction of the spinel phase is derived according to the following:
whereas Asp is the area of the spinel phase, Atot the total area and finally Ap is the area of the pores. No impact on the phase fraction can be observed with varying initial Al2O3 and CaO contents. This is explained by the fact that Al2O3 and CaO substitute FeO. As the samples are balanced with FeO, increased weighed-in contents of Al2O3 and CaO lead to the corresponding reduction of the FeO content. The phase fraction of the spinel is however decreasing with increasing SiO2. This is clearly in line with the impact of SiO2 on the phase compositions. The decreasing phase fraction of the spinel phase explains the increasing V2O3 content in the spinel phase, as V2O3 is fixed at 30 mass% and the V2O3 content in matrix is generally below 1 mass%. The Al2O3 and MnO contents in the matrix are not diluted with increasing initial SiO2 content due to the combined effect of decreasing spinel phase fraction and decreasing Al2O3 and MnO contents in the spinel phase. Increasing V content could be expected to increase the phase fraction of the spinel phase, since V is mainly distributed to this phase. Diao et al. have also reported a spinel phase fraction of 0.45 and 0.36 for V slags with 12.3 and 5.1 mass% V2O3, respectively [8]. Viscosity of liquid slag has been found to decrease with increasing V2O3 content if V is dissolved in liquid slag [18, 20]. Spinel and/or karelianite precipitate in V saturated slags [16–20] which results in a sharp increase of the slag viscosity [20]. The viscosity of V slags, i.e. the solid–liquid mixture, is therefore expected to increase with increasing V content and/or decreasing SiO2 content due to the higher solid fraction in the slag. The V dissolved in the liquid fraction is low and thus should have negligible impact on the slag viscosity. The viscosity of the solid–liquid mixture is an important topic to study, since it has great impact on both the production of V slags and on the following extraction processes. It is hoped that the present information about the phase relationship will throw some lights on the extraction of V, and therfore the sustainable use of the V in the BOF slag.
Uncertainties Due to Dissolution of Mo From the Crucible
Normalization was done by excluding Mo originating from the crucible. Mo was mainly detected in the spinel phase and was in the range of 2–6 mass% as MoO2. The spinel phase fraction was in average 0.55 and consequently the total amount of dissolved MoO2 in the samples ranges from about 1–3 mass%. As the Mo content in the matrix is small, its impact on the phase composition of the matrix and the precipitation of free silica is expected to be minor. Compositional variation of the spinel phase was only observed with variable total initial SiO2 content. The V2O3 content increases from 47.7 mass% to 54.1 mass% and the FeO decreases from 43.0 mass% to 38.3 mass% in the spinel phase with increasing total initial SiO2 content from 10.0 to 17.0 mass% (shown in Fig. 9b). In comparison, the compositional variation of MoO2 in the spinel phase is small and thus expected to have a minor effect on the observed relationships.
Conclusions
The phase relationship in FeO–SiO2 based V slag was investigated experimentally at 1673 K. The compositions studied covered the normal production range, namely Al2O3 from 0 to 6 mass%, CaO from 1 to 5 mass% and SiO2 from 10 to 17 mass%. The V and MnO were fixed at 20.4 and 5.5 mass%, respectively. The main findings can be summarized as follows:
-
Samples quenched at 1673 K were found to be solid–liquid mixtures, either in 2-phase region consisting of liquid and a solid spinel phase, or in 3-phase region consisting of liquid, spinel phase and free silica.
-
Free silica was only found in samples with a weighed-in composition of 0 mass% Al2O3, 1 mass% CaO and 17 mass% SiO2. Formation of free silica was prohibited in samples with 1 mass% CaO and up to 17 mass% SiO2 by adding 5 mass% Al2O3. Addition of 4 mass% Al2O3 was sufficient to prevent the presence of free silica in samples with 3 mass% CaO and 13.5 mass% SiO2.
-
The V content in the liquid was generally below 1 mass% as V2O3, thereby indicating that almost all V was kept in the spinel phase.
-
Fe, Mn and Al coexisted in both the liquid and spinel phase. Si and Ca were only found in the liquid phase.
-
The fraction of solid phase, viz. spinel was found to decrease with increasing SiO2 content.
-
The SiO2 content in the solid–liquid mixture had a large impact on the phase relations.
References
Lindvall M (2006) Selective oxidation of vanadium prior to iron and phosphorus. E-publications Luleå University of Technology. http://epubl.ltu.se/1402-1617/2006/288/LTU-EX-06288-SE.pdf. Accessed 11 April 2016
Gupta CK, Krishnamurthy N (1992) Extractive metallurgy of vanadium. Elsevier Sci Publishers BV, Amsterdam, pp 278–307
Voglauer B, Geyrhofer W, Jörgl HP(2004) A model based control concept with knowledge based overhead control of a roasting process. In: Proc Am Control Conf, IEEE, New York, pp 3581–3586
Song WC, Li K, Zheng Q, Li H (2014) A novel process of vanadium extraction from molten vanadium bearing slag. Waste Biomass Valoris 5:327–332
Li HY, Fang HX, Wang K, Zhou W, Yang Z, Yan XM, Ge WS, Li QW, Xie B (2015) Asynchronous extraction of vanadium and chromium from vanadium slag by stepwise sodium roasting-water leaching. Hydrometall 156:124–135
Steinberg WS, Geyser W, Nell J (2011) The history and development of the pyrometallurgical processes at Evraz Highveld Steel & Vanadium. In: Proc of South Afr Pyrometall, Johannesburg, pp 63–76
Diao J, Xie B, Wang Y, Ji CQ (2009) Mineralogical characterization of vanadium slag under different treatment conditions. Ironmak Steelmak 36:476–480
Diao J, Xie B, Ji C, Guo X, Wang Y (2009) Growth of spinel crystals in vanadium slag and their characterization. Cryst Res Technol 44:707–712
Werme A (1988) Distribution of vanadium between SiO2 rich slags and carbon saturated liquid iron. Steel Res Int 59:6–15
Zhang YL, Zhao F, Wang YG (2011) Effects of influencing factors on distribution behaviors of vanadium between hot metal and FeO–SiO2–MnO (–TiO2) slag system. Steel Res Int 82:940–950
Inoue R, Suito H (1982) Distribution of vanadium between liquid iron and MgO saturated slags of the system CaO–MgO–FeOx–SiO2*. Trans Iron Steel Inst Jpn 22:705–714
Howard RL, Richards SR, Welch BJ, Moore JJ (1994) Vanadium distribution in melts intermediate to ferroalloy production from vanadiferous slag. Metall Mater Trans B 25B:27–32
Tikka J, Lindvall M (2013) Smelting Mustavaara Titanomagnetite concentrate using DC-furnace and selective oxidation of vanadium from the hot metal. In: Proceedings of 43rd ironmak and raw mater semin & 14th Brazilian symposium on iron ore & 1st Brazilian symposium on agglom of iron ore, ABM, Sao Paulo
Fang HX, Li HY, Zhang T, Liu BS, Xie B (2015) Influence of CaO on existence form of vanadium-containing phase in vanadium slag. ISIJ Int 55:200–206
Körber F, Oelsen W (1940) Die Schlackenkunde als Grundlage der Metallurgie der Eisenerzeugung. Stahl Eisen 60:948–949
Lindvall M, Gran J, Sichen D (2014) Determination of the vanadium solubility in the Al2O3–CaO (25 mass%)–SiO2 system. Calphad 47:50–55
Lindvall M, Sichen D (2015) Determination of vanadium solubility in the Al2O3–CaO(30 Mass Pct)–SiO2 and Al2O3–CaO(35 Mass Pct)–SiO2 system. Metall Mater Trans B 46B:733–740
Nakano J, Duchesne M, Bennett J, Kwong KS, Nakano A, Hughes R (2015) Thermodynamic effects of calcium and iron oxides on crystal phase formation in synthetic gasifier slags containing from 0 to 27 wt% V2O3. Fuel 161:364–375
Nakano J, Kwong KS, Bennett J, Lam T, Fernandez L, Komolwit P, Sridhar S (2011) Phase equilibria in synthetic coal-petcoke slags (Al2O3–CaO–FeO–SiO2–V2O3) under Simulated Gasification Conditions. Energy Fuels 25:3298–3306
Duchesne M, Ilyushechkin AY, Hughes RW, Lu DY, McCalden DJ, Macchi A, Anthony EJ (2012) Flow behavior of slags from coal and petroleum coke blends. Fuel 97:321–328
Muan A, Spring L, Mitri V, Najjar S, Falls W (1911) Compositions involving V2O3–CaO. US Pat 5077260. http://patentimages.storage.googleapis.com/pdfs/US5077260.pdf. Accessed 14 April 2016
Peimin G, Pei Z (2005) Activity model and its application of quarternary system CaO–FeO–SiO2–V2O3. Iron Steel Vanadium Titan 26:1–6
Ge W, Chen L, Li L, Li Q, Huang Z (2013) Study on the behavior of SiO2 related phases in the vanadium slag. Adv Mater Res 634–638:3098–3101
Richardson HM, Ball F, Rigby GR (1954) The preparation and properties of vanadium spinels*. Trans J Br Ceram Soc 53:376–387
Wakihara M, Shimizu Y, Katsura T (1971) Preparation and magnetic properties of the FeV2O4–Fe3O4 system. J Solid State Chem 3:478–483
Allibert M, Gaye H, Geiseler J, Janke D, Keene BJ, Kirner D, Kowalski M, Lehmann J, Mills KC, Neuschütz D, Parra R, Saint-Jours C, Spencer PJ, Susa M, Tmar M, Woermann E (1995) Slag atlas, 2nd edn. Verlag Stahleisen GmbH, Düsseldorf
Diao J, Qiao Y, Zhang X, Ji CQ, Xie B (2015) Growth mechanisms of spinel crystals in vanadium slag under different heat treatment conditions. CrystEngComm 17:7300–7305
Li XS, Xie B, Wang G, Li X (2011) Oxidation process of low-grade vanadium slag in presence of Na2CO3. Trans Nonferr Met Soc Ch 21:1860–1867
Acknowledgments
Funding from ERA-MIN, the ERA-NET program on the Industrial Handling of Raw Materials for European industries and supported by the European Commission’s 7th Framework Programme, via VINNOVA, Sweden’s innovation agency, are gratefully acknowledged. Special thanks to Dr. G Ye of Swerea MEFOS and Jukka Pitkäjärvi of Mustavaaran Kaivos Oy for your support.
Author information
Authors and Affiliations
Corresponding author
Additional information
The contributing editor for this article was Katsunori Yamaguchi.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lindvall, M., Berg, M. & Sichen, D. The Effect of Al2O3, CaO and SiO2 on the Phase Relationship in FeO–SiO2 Based Slag with 20 Mass% Vanadium. J. Sustain. Metall. 3, 289–299 (2017). https://doi.org/10.1007/s40831-016-0088-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40831-016-0088-y