Abstract
Purpose of Review
Urinary tract infection (UTI) in children is a major source of office visits and healthcare expenditure. Research into the diagnosis, treatment, and prophylaxis of UTI has evolved over the past 10 years. The development of new imaging techniques and UTI screening tools has improved our diagnostic accuracy tremendously. Identifying who to treat is imperative as the increase in multi-drug-resistant organisms has emphasized the need for antibiotic stewardship. This review covers the contemporary management of children with UTI and the data-driven paradigm shifts that have been implemented into clinical practice.
Recent Findings
With recent data illustrating the self-limiting nature and low prevalence of clinically significant vesicoureteral reflux (VUR), investigational imaging in children has become increasingly less frequent. Contrast-enhanced voiding urosonogram (CEVUS) has emerged as a useful diagnostic tool, as it can provide accurate detection of VUR without the need of radiation. The urinary and intestinal microbiomes are being investigated as potential therapeutic drug targets, as children with recurrent UTIs have significant alterations in bacterial proliferation. Use of adjunctive corticosteroids in children with pyelonephritis may decrease the risk of renal scarring and progressive renal insufficiency. The development of a vaccine against an antigen present on Escherichia coli may change the way we treat children with recurrent UTIs.
Summary
The American Academy of Pediatrics defines a UTI as the presence of at least 50,000 CFU/mL of a single uropathogen obtained by bladder catheterization with a dipstick urinalysis positive for leukocyte esterase (LE) or WBC present on urine microscopy. UTIs are more common in females, with uncircumcised males having the highest risk in the first year of life. E. coli is the most frequently cultured organism in UTI diagnoses and multi-drug-resistant strains are becoming more common. Diagnosis should be confirmed with an uncontaminated urine specimen, obtained from mid-stream collection, bladder catheterization, or suprapubic aspiration. Patients meeting criteria for imaging should undergo a renal and bladder ultrasound, with further investigational imaging based on results of ultrasound or clinical history. Continuous antibiotic prophylaxis is controversial; however, evidence shows patients with high-grade VUR and bladder and bowel dysfunction retain the most benefit. Open surgical repair of reflux is the gold standard for patients who fail medical management with endoscopic approaches available for select populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urinary tract infection (UTI) is one of the most common diagnoses affecting children and infants, accounting for 0.7% of physician office visits and 5–14% of emergency room visits per year [65]. Over the last 10 years, there has been a shift in the management of UTI, particularly regarding diagnosis and antibiotic prophylaxis for recurrent infections. In this review article, we cover the diagnosis, workup, and treatment of children with suspected UTI. We also explore the role of antibiotic prophylaxis, investigational imaging, and approaches to unique populations. Our goal is to help familiarize clinicians with the current management of pediatric UTI as new data becomes implemented in clinical practice.
Risk factors for UTI
UTI in children is strongly associated with white race, age, bowel and bladder dysfunction (BBD), circumcision status, and congenital anomalies of the kidney and urogenital tract (CAKUT) [1, 2]. Other risks include systemic disease (diabetes, HIV, immunocompromise, sickle cell disease), spinal dysraphism, sexual activity, kidney stones, and urinary tract instrumentation [4].
Uncircumcised neonates have a particularly high incidence of UTI (2.15%) when compared to circumcised males (0.22%) and females of the same age (2.05%) [1, 2]. Furthermore, uncircumcised males have been found to have a higher resistance to narrow-spectrum antibiotics when compared to females in the same age group (2–71 months) [26]. Data has shown a higher prevalence of UTIs in white children when compared to African American cohorts; however, Latinx populations have also been found to have increased risk [1, 2]. It is possible that this is due to the higher uncircumcised population in the Latinx community [2].
A recurrent UTI is defined as an individual who is diagnosed with 2 or more UTIs in a 6-month period, or 3 within a 1-year period. The two risk factors shown to have the highest 2-year recurrence rate for UTI are vesicoureteral reflux (VUR) and bladder and bowel dysfunction (BBD), with the combination of both producing an even higher risk (56%) [1].
Risk factors for renal scarring after febrile UTI include children with high-grade reflux, delayed initiation of antibiotics past 72 h, recurrent UTIs, infectious organisms other than E. coli, and older age [5,6,7,8, 17]. Laboratory findings linked to renal scarring are fever (> 39 °C), polymorphonuclear count > 60%, CRP > 40 mg/L, and an abnormal finding on renal and bladder ultrasound (RBUS) [9].
Diagnosis
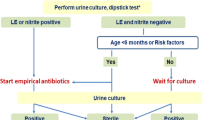
Pre-verbal children present a diagnostic challenge, as they cannot communicate lower urinary tract signs. The most clinically proven symptom in this age group is fever, but other common symptoms that should raise suspicion include irritability, vomiting, diarrhea, prolonged jaundice, poor feeding, and failure to thrive [14]. Recent advent of UTI risk calculators for febrile infants has improved detection in this cohort. The University of Pittsburgh used their risk calculator to reduce rates of UTI testing (8.1%), increase accuracy of testing, and reduce treatment delays (10.6%) when compared to the AAP algorithm and dipstick urinalysis [63]. At our institution, we have employed a UTI clinical pathway to streamline and simplify diagnosis and treatment (Fig. 1). The goal of the Connecticut Children’s Clinical Pathway program is to standardize best practice, improve patient outcomes, and reduce cost. This is accomplished by decreasing unnecessary variation while promoting safe, effective, and consistent patient care. The pathway for UTI was designed by physicians from the Department of Pediatrics with input from the urology and nephrology divisions. Quality metrics including appropriate use of antibiotics and imaging are tracked. Antibiotic recommendations are routinely reviewed to ensure that they are appropriate based on hospital antibiograms.
Since 5% of children between 2 and 24 months with a fever of unknown origin have a UTI [10], a urinalysis is an important diagnostic tool. In toilet-trained children, the collection method of choice is a mid-stream, clean-catch urine specimen after appropriate genital cleansing. Recent studies have shown a significant rate of urine culture contamination in children who did not perform perineal washings (23.9%) compared to those that did (7.8%) prior to mid-stream urine collection [11, 12].
In diapered children, three options exist: perineal bag collection, bladder catheterization, and suprapubic aspiration. Up to 85% of bag-collected specimens with a positive culture will be false positives [10]. Bag collection should be treated as an initial screening measure only: a negative result confirms the absence of a UTI [18]. Bladder catheterization is 95% sensitive and 99% specific, and is less technically difficult in comparison to suprapubic aspiration [10]. While some studies claim suprapubic aspiration is more painful and has a higher rate of collection failure, it has also been shown to have a lower rate of false positives [13,14,15]. In our experience, with proper technique and selective use of bedside ultrasound, suprapubic aspiration can be quick and well-tolerated when bladder catheterization is not a good option.
Urinalysis, urine culture, common organisms
The American Academy of Pediatrics defines a UTI as the presence of at least 50,000 CFU/mL of a single uropathogen obtained by bladder catheterization with a dipstick urinalysis positive for leukocyte esterase (LE) or WBC present on urine microscopy [16]. Due to the high sensitivity of LE, it is commonly used to rule out UTI in its absence. Nitrites have a higher specificity, and are thus used to rule in disease [88]. The presence of bacteria, WBC, and RBC on urine microscopy can add confirmation to the diagnosis. Table 1 shows the sensitivity and specificity of these clinical indicators alone and in combination for diagnosis of UTI.
Culture diagnosis is dependent on collection method, with > 100,000 CFU/mL required for mid-stream specimen, > 50,000 CFU/mL for catheterization, and > 1000 CFU/mL for suprapubic aspiration [16]. Emerging data has suggested that in infants with fever and pyuria, lowering the colony count for catheterized specimens to 10,000 CFU/mL will accurately diagnose UTI without sacrificing specificity [89]. The most common organism responsible for UTI in children is Escherichia coli, with Klebsiella pneumoniae, Enterococcus faecalis, and Proteus mirabilis also frequently cultured [16, 66, 82]. Table 2 shows a breakdown of common organisms in the community and nosocomial setting.
Cultures are susceptible to contamination from feces and skin flora. Signs of contamination include > 10 squamous epithelial cells/HPF, presence of > 2 bacteria, low bacterial colony count, and growth of non-uropathogens (Lactobacillus, Corynebacterium, Viridans streptococci) [18]. When asymptomatic bacteriuria (AB) is encountered on culture analysis, treatment should be avoided as it can promote symptomatic infections, antibiotic-resistant organisms, and side effects. While contamination is common, true AB is an extremely rare event, with prevalence in infant boys and girls found to be < 0.5%, and in boys after infancy, < 0.1% [91]. Higher rates exist in children requiring use of foreign body instrumentation in the GU tract, such as those requiring daily CIC [90].
Treatment
Once the clinician has reached a suspected diagnosis of UTI, the child should be treated with antibiotics within the first 48 h to prevent likelihood of renal scarring [6, 7, 25]. There is likely a linear relationship between the prevalence of renal scar formation and delayed time to treatment [7].
Antimicrobial stewardship is more important than ever with the rise of multi-drug-resistant pathogens. This includes extended spectrum beta-lactamase (ESBL) producing organisms, which have been found increasingly in community acquired UTIs [27,28,29]. One study found ESBL-producing E. coli composed nearly half of the bacterial strains in their culture-proven cohort [66]. The AAP recommends Bactrim, Augmentin, or cephalosporins (cefixime, cefpodoxime, cephalexin) as first-line agents for uncomplicated pediatric UTI; our institution’s clinical pathway recommends Ceftriaxone IV or Cephalexin PO. Unfortunately, there is data suggesting the most common empiric antibiotics (Bactrim, Augmentin, ampicillin, and first-generation cephalosporins) have the highest growth of antibiotic resistance among UTI-producing bacteria [27,28,29]. Regardless of the first-line agent typically chosen, clinicians must also review patient-specific culture results and follow local antibiograms and antimicrobial sensitivity patterns available at their institution.
Multiple studies show a 2- to 4-day course of oral antibiotics is just as effective as a 7- to 14-day course in children with cystitis [18, 30, 31]. Those with acute pyelonephritis can also be treated with oral antibiotics for 10–14 days or IV antibiotics for 2–4 days followed by oral therapy [18, 32].
Imaging
The purpose of imaging is to identify genitourinary anomalies that increase the risk of recurrent UTI and injury from repeat infections (renal scarring, renal insufficiency). A renal and bladder ultrasound (RBUS) should be obtained in all infants aged 2–24 months after first febrile UTI [18]. Children who are older than 2–24 months who experience recurrent febrile UTIs should also be evaluated, though RBUS has a poor sensitivity for detecting mild-moderate VUR [18].
Voiding cystourethrogram (VCUG) is indicated if an abnormality is found on RBUS or for recurrent febrile UTI [18]. VCUG is used to identify children with high-grade (IV–V) reflux who are at risk for renal deterioration but should be used selectively given the need for catheterization and the radiation exposure [18]. Multiple studies have found that low-grade VUR does not increase risk of renal damage and is frequently self-limiting; therefore, the use of VCUG has become increasingly selective. Data has shown less than 40% of children are found to have VUR after their first febrile UTI and among this group, less than 10% have high-grade VUR (IV, V) [16, 61, 62]
Contrast-enhanced voiding urosonogram (CEVUS) has emerged as a radiation-free method of evaluating children for VUR. The method works by using contrast-containing echogenic microbubbles that are easily detected using low-mechanical index ultrasound. The diagnostic accuracy of CEVUS for VUR is excellent, with recent studies quoting a sensitivity of 90.4–92% and a specificity of 92.8–98% [20, 21•]. Concordance with VCUG for detection and grading of VUR is 84.3% and 81.8%, respectfully [21•], with a multitude of data confirming non-inferiority of CEVUS when compared to VCUG [20, 21•, 22,23,24].
Dimercaptosuccinate (DMSA) scans can be used to identify patients with renal scarring, however are also sensitive for detecting renal anomalies such as small or absent kidneys, ectopic kidneys, and duplex systems. The AUA recommends DMSA scans when initial RBUS is abnormal or when there is a concern for renal scarring (breakthrough UTIs) [19]. A DMSA scan has a higher sensitivity for detecting renal parenchymal injury when compared to RBUS; however, inflammatory changes to the kidney can cause false-positive scans if performed in the first 4–6 months following an infection [16, 19]. Similar to VCUG, the pitfall of DMSA is radiation exposure, with one study proving the average exposure per scan is 2.84 mSv, equal to 28 chest X-rays per year [60]. DMSA can also be difficult for radiologists to obtain.
Antibiotic prophylaxis
Continuous antibiotic prophylaxis (CAP) can be prescribed to prevent renal injury in children who experience recurrent febrile UTIs. Recommendations on prophylaxis can be controversial. Proponents for CAP reference the RIVUR trial, a randomized study that investigated use of CAP in preventing recurrent febrile UTIs in patients with dilated and non-dilated VUR. Bactrim decreased the rate of recurrent UTIs by 50% in all patients with VUR; dilated VUR found the most benefit [33•]. However, the study found no difference in the rate of renal scarring between treatment groups, the outcome of interest to most physicians. This was confirmed by the CUTIE trial, which showed no difference in renal scarring between children with or without VUR who did not receive CAP after febrile UTI [3]. To date, there is no level I data to show CAP has any effect on renal scarring [3, 33•, 44].
A meta-analysis sought to validate the benefit of CAP in children with VUR and found that with the addition of the RIVUR trial data, statistical significance was achieved [34•]. However, a recent publication using advanced analysis has doubted the statistical power of these results [34•, 35]. Adding to the skepticism is the multitude of publications noting CAP has no significant effect on preventing recurrent UTI in kids with VUR [39,40,41,42, 45, 46], not to mention that there is a three-fold higher risk of antibiotic-resistant organisms present in infected patients with low-grade VUR on CAP therapy [48]. Nonetheless, children with high-grade VUR and BBD have consistently been shown to derive benefit from CAP [33•, 34•, 36,37,38, 43]. The work comparing time to febrile UTI after discontinuing CAP has identified BBD and high-grade VUR as independent risk factors for early recurrence [37, 38].
Given this evidence, the American Urologic Association and our own practice are to recommend continued antibiotic prophylaxis (CAP) for a child < 1 year of age with VUR and a history of febrile urinary tract infection [19]. Prophylaxis should be considered for all patients with high-grade VUR (III–V). CAP is also recommended for children > 1 year of age if they have BBD and VUR [19]. For children with VUR or renal cortical anomalies without BBD, CAP is optional and should be discussed as part of a shared decision-making process.
Treatment of bladder and bowel dysfunction
Bladder and bowel dysfunction is defined by a constellation of lower urinary tract symptoms (LUTS) in conjunction with bowel dysfunction, most notably constipation or encopresis [82, 96, 97]. LUTS include, but are not limited to, dysuria, frequency, incomplete emptying, incontinence, nocturia, and retention. It is thought that the retention of stool in the rectal vault mechanically compresses the bladder, leading to decreased compliance and frequent urination. In addition, there seems to be a neurologic component, with chronic retention of urine and stool leading to a decreased afferent response from the bladder and pelvic floor. This results in an attenuated sensation to urinate and poor relaxation of the pelvic floor during voiding [82, 96, 97]. Children with BBD have increased rates of reflux, UTI, and renal sequelae [96, 97]. Compounding the issue, those with BBD treated medically or surgically for VUR have increased rates of recurrence and poorer outcomes [64, 82].
About 50% of children with BBD will improve with behavioral intervention alone [98]. Treatment starts with adequate hydration to improve bladder cycling, timed voiding every 2–3 h, appropriate voiding posture, and pelvic floor muscle training (PFMT) with or without biofeedback therapy [82, 96]. In biofeedback, monitors placed on the skin electronically record pelvic floor musculature in real time allowing for improved voluntary control during voiding. Studies have shown a UTI prevention rate of 68–86% in children treated with behavioral therapy and PFMT alone [96, 99,100,101,102,103]. Medications such as alpha agonists and muscarinic antagonists are pursued if symptoms persist in spite of behavioral treatment and adequate management of constipation for 6 months. If these treatments fail, OnabotulinumtoxinA and sacral neuromodulation are among last resort options [96].
The initial treatment for constipation in children includes increased fiber intake, proper hydration, and stool softeners. Polyethylene glycol is the most commonly prescribed stool softener in children [96, 98]. Children are often encouraged to sit on the toilet for 10 min after meals to take advantage of the gastrocolic reflex. If these treatments fail to resolve constipation, retrograde enemas, dis-impaction, and anterograde continence enema (ACE) procedures can be employed [96, 98]. There is some data showing the use of neuromodulation can simultaneously improve overactive bladder and constipation in children with BBD; however, this should be selectively utilized given the invasive nature of the treatment [104, 105].
Surgical treatment
The index patient who may benefit from surgery is one who has proven VUR and experiences breakthrough UTIs while receiving CAP therapy [19]. Other reasons include non-compliance with antibiotics or new renal cortical lesions found on surveillance screening [16, 19].
The most common approaches for ureteral reimplantation are the Politano-Leadbetter, Lich-Gregoir, modified Lich-Gregoir, and Cohen cross-trigonal reimplant. Historically, open surgery remained the gold standard; in recent years, robotic-assisted laparoscopic approaches have good data supporting their efficacy, with lower rates of post-operative pain, less opioid use, shorter hospital stay, and improved cosmesis [50, 51, 53,54,55]. Furthermore, there is data proving non-inferiority in VUR downgrading and radiographic resolution when compared to open techniques [50, 52, 54]. In non-experienced centers, robotic-assisted approaches have seen a higher 90-day complication rate and a significantly higher cost to the patient’s family [56].
Endoscopic treatment (ET) involves an injection of a subureteral or intraureteral bulking agent. Dextranomer and hyaluronic acid (Deflux) is the most common anti-reflux agent. There is data showing no difference in prevention of UTI recurrence or renal scarring in patients with VUR undergoing ET when compared to CAP alone [47, 49]. However, endoscopic therapy is still used commonly in clinical practice with the index patient being one with low-grade VUR or an older patient in which reimplantation would be more technically difficult.
It is important to note that children with lower urinary tract dysfunction have an increased surgical failure rate for reflux resolution in both open and endoscopic series. Thus, treating the underlying urinary symptoms should preclude any attempt at surgical intervention for VUR [64].
Unique populations
The majority of children born with spina bifida (SB) suffer chronically from increased bladder storage pressures, difficulty emptying, and dependency on catheter drainage, which put them at risk for recurrent UTI and renal deterioration [67]. One paper demonstrated 50% of children with spina bifida will experience a UTI by 15 months and 44% will have > 5 UTIs by 15 years old [68]. Based on the National Spina Bifida Patient Registry, > 70% of spina bifida patients perform CIC. This population presents a diagnostic challenge to discern asymptomatic colonization versus symptomatic infection. There is no uniform consensus on symptomatic UTIs in SB patients; however, in a systematic review, fever, positive culture, and symptoms were the most reported criteria [70]. Symptoms included flank pain, abdominal pain, change in continence patterns, increased urine odor, and dysuria [70, 71]. The primary goal in identifying SB patients with symptomatic UTIs is to prevent renal damage; however, multiple studies have shown that renal function is largely preserved in patients with asymptomatic bacteriuria in the absence of VUR and hostile bladder parameters on urodynamics [69]. Therefore, complete evaluation including urodynamics and ultrasound is key to identify patients at risk for infectious sequelae. Preventing high detrusor pressures, urinary stasis, and constipation will also attenuate risk for infection. While the research does not provide uniform guidelines, patients with fever, culture-positive urine, and new-onset symptoms should be prioritized for antibiotic treatment.
Bladder exstrophy (BE) is a rare condition with an exposed bladder plate, epispadias, and pelvic diastasis [82]. After initial bladder closure, ureteral insertion is commonly at a 90° angle with a very limited intramural tunnel. Thus, a significant number of patients will have VUR and may be at risk of recurrent febrile UTI if ureteral reimplantation is not performed during the operation [81]. Ureteral reimplantation during bladder exstrophy repair has been shown to decrease rates of infection and renal scarring [72, 73, 81]. If reimplantation is not performed primarily, emphasis should be placed in identifying those with VUR and use of prophylactic antibiotics should be considered. Continence remains a challenging issue in this population, with a high rate of CIC dependence or augmentation cystoplasty, both of which are associated with UTI [107]. In addition, those found to have continence after repair tend to have higher rates of VUR and recurrent febrile UTI [81, 108]. One unique option for UTI in patients with urologic reconstructions who already perform CIC is intravesical antibiotic instillation, either as prophylaxis or treatment. In one study that evaluated intravesical gentamycin, serum levels were indetectable and development of antibiotic resistance low (4.16%) [109]. Since patients with BE report high rates of recurrent UTI and renal scarring in adulthood, this population requires close urologic care in childhood and beyond [107].
Future directions
Emerging research has identified a relationship between vaginal, intestinal, and urinary bacteria and the development of recurrent UTI. Intestinal bacteria have historically been a focus of research as introital and periurethral colonization with these organisms remains a common etiology for adult female UTIs [74]. In a recent publication, children with recurrent UTIs were found to have higher proportions of fecal bacteria from the Enterobacter family as compared to the Peptostreptococcaceae family found in those without UTI [77]. It is an important distinction, as a 1% relative abundance of Escherichia or Enterococcus species acts as independent risk factor for UTI [75]. In another study, patients with recurrent UTI and Clostridium difficile colitis underwent a fecal transplant, leading to a significant reduction in their UTI recurrence [76]. While intestinal bacteria can act as a nidus for genitourinary infections, the vaginal flora has a number of mechanisms to protect against bacterial invasion and proliferation. In Stapleton et al.’s randomized phase II trial of adult women, intravaginal instillation of Lactobacillus crispatus led to a significant reduction in recurrent UTIs [78]. Studies on vaginal microbiome transplant have shown preliminary evidence for remission of recurrent bacterial vaginosis, further validating this area as a potential therapeutic target [79]. While research on the pediatric urinary microbiome is still growing, there is data suggesting bacterial dysbiosis in children with recurrent UTI. Kinneman et al. found differences in both the alpha diversity (number of different species at a single site) and the beta diversity (differences in species at multiple sites) in children presenting with UTI when compared to those without UTI [80], highlighting an investigational difference in bacterial proliferation between these two cohorts.
Clinicians have long sought to prevent renal parenchymal scarring secondary to urinary infection which can lead to hypertension, proteinuria, and chronic renal failure. In recent years, it has been proposed that adjuvant use of corticosteroids in children with pyelonephritis can limit the development of inflammatory cytokines that lead to renal scarring. The data has been mixed, with the largest known clinical trial failing to reach accrual but claiming a non-statistically significant trend towards favorability of adjunctive use of steroids [58]. Smaller investigations have also boasted a significant decrease in DMSA-proven renal scarring [57, 59]. All three studies were underpowered, but two clinical trials are underway (NCT04654507, NCT02034851) which hope to shed more light on this controversial topic.
With the advent of the HPV vaccine in the mid-2000s and now the multitude of vaccines targeting the novel coronavirus, primary prevention has become an intense topic of research, and UTIs are no exception. Scientists from Sequoia Sciences Inc. have recently published a phase I, dose escalation trial of a vaccine targeted against a preserved antigen present in E. coli bacteria [110•]. The vaccine consists of a protein called FimCH adjuvanted with a TLR4 agonist. FimCH is a naturally occurring antigen present on type I pili in E. coli. Type I pili are bacterial appendages that help bacteria bind to oligomannose-containing glycoproteins in human bladders [110•]. The researchers theorize that patients inoculated with their vaccine will build a IgG-mediated antibody response to FimCH proteins that will penetrate the mucosal surface of the bladder and prevent recurrent infections from occurring. The results of the study proved the vaccine to be extremely well-tolerated, with adverse events limited to mild localized skin reactions, headaches, nausea, and fatigue. All subjects receiving the four-dose vaccination schedule were seropositive after 30 days. The cohort of women with recurrent UTI demonstrated a 150-fold increase in antibodies against FimH proteins [110•]. While this is the first study of a FimH vaccine in humans, prior research has proven its efficacy with a 99% reduction of in vivo E. coli colonization using murine models [111]. A randomized, phase II, placebo-controlled trial has been approved.
Conclusion
Pediatric UTI remains a significant source of morbidity in children, especially uncircumcised males in the first year and females throughout life. UTI risk calculators, algorithms, and optimized collection techniques will decrease the rate of false positives and unnecessary antibiotic treatment. Antibiotic-resistant organisms are increasing rapidly; therefore, antibiotic stewardship and use of local antibiograms are more important than ever. Young children with febrile UTI and older children recurrent UTI should undergo investigational imaging. VCUG is invasive and requires radiation; however, newer imaging techniques such as CEVUS provide excellent detection of upper tract disease without the use of radiation. CAP in the setting of VUR is controversial; children with BBD and high-grade VUR derive the most benefit. Patients with complex urologic conditions such as SB and BE require unique approaches to urinary infections. Research into corticosteroids, urinary microbiomes, and UTI vaccinations could change how we treat UTIs and prevent chronic renal insufficiency in high-risk patients.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Schoen EJ, Colby CJ, Ray GT. Newborn circumcision decreases incidence and costs of urinary tract infections during the first year of life. Pediatrics. 2000;105(4 Pt 1):789–93.
Shaikh N, Morone NE, Bost JE, Farrell MH. Prevalence of urinary tract infection in childhood: a meta-analysis. Pediatr Infect Dis J. 2008;27(4):302–8. https://doi.org/10.1097/INF.0b013e31815e4122 (PMID: 18316994).
Keren R, Shaikh N, Pohl H, Gravens-Mueller L, Ivanova A, Zaoutis L, Patel M, deBerardinis R, Parker A, Bhatnagar S, Haralam MA, Pope M, Kearney D, Sprague B, Barrera R, Viteri B, Egigueron M, Shah N, Hoberman A. Risk Factors for Recurrent Urinary Tract Infection and Renal Scarring. Pediatrics. 2015;136(1):e13-21. https://doi.org/10.1542/peds.2015-0409 (Epub 2015 Jun 8. PMI).
Becknell B, Schober M, Korbel L, Spencer JD. The diagnosis, evaluation and treatment of acute and recurrent pediatric urinary tract infections. Expert Rev Anti Infect Ther. 2015;13(1):81–90.
Snodgrass WT, Shah A, Yang M, Kwon J, Villanueva C, Traylor J, Pritzker K, Nakonezny PA, Haley RW, Bush NC. Prevalence and risk factors for renal scars in children with febrile UTI and/or VUR: a cross-sectional observational study of 565 consecutive patients. J Pediatr Urol. 2013;9(6 Pt A):856–63. https://doi.org/10.1016/j.jpurol.2012.11.019 (Epub 2013 Mar 7. PMID: 23465483; PMCID: PMC3770743.).
Karavanaki KA, Soldatou A, Koufadaki AM, Tsentidis C, Haliotis FA, Stefanidis CJ. Delayed treatment of the first febrile urinary tract infection in early childhood increased the risk of renal scarring. Acta Paediatr. 2017;106(1):149–54. https://doi.org/10.1111/apa.13636 (Epub 2016 Nov 17 PMID: 27748543).
Oh MM, Kim JW, Park MG, Kim JJ, Yoo KH, du Moon G. The impact of therapeutic delay time on acute scintigraphic lesion and ultimate scar formation in children with first febrile UTI. Eur J Pediatr. 2012;171(3):565–70. https://doi.org/10.1007/s00431-011-1614-3 (Epub 2011 Nov 3 PMID: 22048628).
Shaikh N, Haralam MA, Kurs-Lasky M, Hoberman A. Association of Renal Scarring With Number of Febrile Urinary Tract Infections in Children. JAMA Pediatr. 2019;173(10):949–52. https://doi.org/10.1001/jamapediatrics.2019.2504 (PMID:31381021;PMCID:PMC6686976).
Shaikh N, Craig JC, Rovers MM, Da Dalt L, Gardikis S, Hoberman A, Montini G, Rodrigo C, Taskinen S, Tuerlinckx D, Shope T. Identification of children and adolescents at risk for renal scarring after a first urinary tract infection: a meta-analysis with individual patient data. JAMA Pediatr. 2014;168(10):893–900. https://doi.org/10.1001/jamapediatrics.2014.637 (PMID: 25089634).
Finnell SM, Carroll AE, Downs SM. Subcommittee on Urinary Tract Infection: Diagnosis and management of an initial UTI in febrile infants and young children. Pediatrics. 2011;128:749–70.
Vaillancourt S, McGillivray D, Zhang X, Kramer MS. To clean or not to clean: effect on contamination rates in midstream urine collections in toilet-trained children. Pediatrics. 2007;119(6):e1288–93. https://doi.org/10.1542/peds.2006-2392 (Epub 2007 May 14 PMID: 17502345).
Marzuillo P, Guarino S, Furlan D, Pecoraro A, Pedullà M, Miraglia Del Giudice E, La Manna A. Cleaning the genitalia with plain water improves accuracy of urine dipstick in childhood. Eur J Pediatr. 2018;177(10):1573–9. https://doi.org/10.1007/s00431-018-3215-x (Epub 2018 Jul 27 PMID: 30054720).
Kozer E, Rosenbloom E, Goldman D, Lavy G, Rosenfeld N, Goldman M. Pain in infants who are younger than 2 months during suprapubic aspiration and transurethral bladder catheterization: a randomized, controlled study. Pediatrics. 2006;118(1):e51–6. https://doi.org/10.1542/peds.2005-2326 (PMID: 16818537).
Eliacik K, Kanik A, Yavascan O, Alparslan C, Kocyigit C, Aksu N, Bakiler AR. A Comparison of Bladder Catheterization and Suprapubic Aspiration Methods for Urine Sample Collection From Infants With a Suspected Urinary Tract Infection. Clin Pediatr (Phila). 2016;55(9):819–24. https://doi.org/10.1177/0009922815608278 (Epub 2015 Sep 29 PMID: 26423890).
El-Naggar W, Yiu A, Mohamed A, Shah V, Manley J, McNamara P, Taddio A. Comparison of pain during two methods of urine collection in preterm infants. Pediatrics. 2010;125(6):1224–9. https://doi.org/10.1542/peds.2009-3284 (Epub 2010 May 17 PMID: 20478939).
Mattoo TK, Shaikh N, Nelson CP. Contemporary Management of Urinary Tract Infection in Children. Pediatrics. 2021;147(2): e2020012138. https://doi.org/10.1542/peds.2020-012138 (PMID: 33479164).
Peters CA, Skoog SJ, Arant BS Jr, Copp HL, Elder JS, Hudson RG, Khoury AE, Lorenzo AJ, Pohl HG, Shapiro E, Snodgrass WT, Diaz M. Summary of the AUA Guideline on Management of Primary Vesicoureteral Reflux in Children. J Urol. 2010;184(3):1134–44. https://doi.org/10.1016/j.juro.2010.05.065 (Epub 2010 Jul 21 PMID: 20650499).
SUBCOMMITTEE ON URINARY TRACT INFECTION. Reaffirmation of AAP Clinical Practice Guideline: The Diagnosis and Management of the Initial Urinary Tract Infection in Febrile Infants and Young Children 2–24 Months of Age. Pediatrics. 2016;138(6): e20163026. https://doi.org/10.1542/peds.2016-3026 (PMID: 27940735).
Management and Screening of Primary Vesicoureteral Reflux in Children (2010, amended 2017). Vesicoureteral reflux guideline - american urological association. (2010). Retrieved January 4, 2022, from https://www.auanet.org/guidelines/guidelines/vesicoureteral-reflux-guideline
Chua ME, Mendoza JS, Ming JM, Dy JS, Gomez O. Diagnostic accuracy of contrast-enhanced voiding urosonogram using second-generation contrast with harmonic imaging (CEVUS-HI) study for assessment of vesicoureteral reflux in children: a meta-analysis. World J Urol. 2019;37(10):2245–55. https://doi.org/10.1007/s00345-018-2587-x (Epub 2018 Dec 12 PMID: 30542962).
Ntoulia A, Back SJ, Shellikeri S, Poznick L, Morgan T, Kerwood J, Christopher Edgar J, Bellah RD, Reid JR, Jaramillo D, Canning DA, Darge K. Contrast-enhanced voiding urosonography (ceVUS) with the intravesical administration of the ultrasound contrast agent Optison™ for vesicoureteral reflux detection in children: a prospective clinical trial. Pediatr Radiol. 2018 Feb;48(2):216–226. doi: https://doi.org/10.1007/s00247-017-4026-3. Epub 2017 Nov 27. PMID: 29181582. This prospective clinical trial helped validate the use of contrast enhanced voiding urosonography (CeVUS) in the diagnosis of children with vesicoureteral reflux. Highlighted in the paper was the sensitivity (91.7%) and specificty (98%) of CeVUS. Further, the concordance with voiding cystourethrography was found to be greater than 80% on average. Thus supplanting CeVUS as a trusted diagnostic entity in the setting of pediatric VUR.
Woźniak MM, Wieczorek AP, Pawelec A, Brodzisz A, Zajączkowska MM, Borzęcka H, Nachulewicz P. Two-dimensional (2D), three-dimensional static (3D) and real-time (4D) contrast enhanced voiding urosonography (ceVUS) versus voiding cystourethrography (VCUG) in children with vesicoureteral reflux. Eur J Radiol. 2016;85(6):1238–45. https://doi.org/10.1016/j.ejrad.2015.11.006 (Epub 2015 Nov 5 PMID: 26597418).
Mane N, Sharma A, Patil A, Gadekar C, Andankar M, Pathak H. Comparison of contrast-enhanced voiding urosonography with voiding cystourethrography in pediatric vesicoureteral reflux. Turk J Urol. 2018;44(3):261–7. https://doi.org/10.5152/tud.2018.76702 (Epub 2018 Mar 6. PMID: 29733800; PMCID: PMC5937646).
Wong LS, et al. Voiding urosonography with second-generation ultrasound contrast versus micturating cystourethrography in the diagnosis of vesicoureteric reflux. Eur J Pediatr. 2014;173(8):1095–101.
Karavanaki K, Koufadaki AM, Soldatou A, Tsentidis C, Sourani M, Gougourelas D, Haliotis FA, Stefanidis CJ. Fever duration during treated urinary tract infections and development of permanent renal lesions. Arch Dis Child. 2019;104(5):466–70. https://doi.org/10.1136/archdischild-2017-314576 (Epub 2018 Nov 2 PMID: 30389675).
Shaikh N, Hoberman A, Keren R, Ivanova A, Gotman N, Chesney RW, Carpenter MA, Moxey-Mims M, Wald ER. Predictors of Antimicrobial Resistance among Pathogens Causing Urinary Tract Infection in Children. J Pediatr. 2016;171:116–21. https://doi.org/10.1016/j.jpeds.2015.12.044 (Epub 2016 Jan 13. PMID: 26794472; PMCID: PMC4808618).
Erol B, Culpan M, Caskurlu H, Sari U, Cag Y, Vahaboglu H, Özumut SH, Karaman MI, Caskurlu T. Changes in antimicrobial resistance and demographics of UTIs in pediatric patients in a single institution over a 6-year period. J Pediatr Urol. 2018;14(2):176.e1-176.e5. https://doi.org/10.1016/j.jpurol.2017.12.002 (Epub 2018 Jan 9 PMID: 29428362).
Patwardhan V, Kumar D, Goel V, Singh S. Changing prevalence and antibiotic drug resistance pattern of pathogens seen in community-acquired pediatric urinary tract infections at a tertiary care hospital of North India. J Lab Physicians. 2017;9(4):264–8. https://doi.org/10.4103/JLP.JLP_149_16 (PMID: 28966488; PMCID: PMC5607755).
Konca C, Tekin M, Uckardes F, Akgun S, Almis H, Bucak IH, Genc Y, Turgut M. Antibacterial resistance patterns of pediatric community-acquired urinary infection: Overview. Pediatr Int. 2017;59(3):309–15. https://doi.org/10.1111/ped.13139 (Epub 2016 Nov 6 PMID: 27542568).
Tran D, Muchant DG, Aronoff SC. Short-course versus conventional length antimicrobial therapy for uncomplicated lower urinary tract infections in children: a meta-analysis of 1279 patients. J Pediatr. 2001;139(1):93–9. https://doi.org/10.1067/mpd.2001.114698 (PMID: 11445800).
Michael M, Hodson EM, Craig JC, Martin S, Moyer VA. Short versus standard duration oral antibiotic therapy for acute urinary tract infection in children. Cochrane Database Syst Rev. 2003;1:CD003966. https://doi.org/10.1002/14651858.CD003966 (PMID: 12535494).
Strohmeier Y, Hodson EM, Willis NS, Webster AC, Craig JC. Antibiotics for acute pyelonephritis in children. Cochrane Database Syst Rev. 2014;28(7):CD003772. https://doi.org/10.1002/14651858.CD003772.pub4.
RIVUR Trial Investigators. 2014 Antimicrobial prophylaxis for children with vesicoureteral reflux. NEJM 370: 2367. A multisite, randomized, placebo-controlled trial published in 2014 that had a significant impact on pediatric medicine. The most important finding being that children with VUR and recurrent UTI’s who received antibiotic prophylaxis reduced the risk of recurrence by 50%. Further, the effect on renal scarring was found to be non-significantly different between the placebo and the intervention group. The cohort receiving the most benefit after statistical analysis were those with bladder and bowel dysfunction and high grade VUR.
de Bessa J Jr, de Carvalho Mrad FC, Mendes EF, Bessa MC, Paschoalin VP, Tiraboschi RB, Sammour ZM, Gomes CM, Braga LH, Bastos Netto JM. Antibiotic prophylaxis for prevention of febrile urinary tract infections in children with vesicoureteral reflux: a meta-analysis of randomized, controlled trials comparing dilated to nondilated vesicoureteral reflux. J Urol. 2015 May;193(5 Suppl):1772–7. doi: https://doi.org/10.1016/j.juro.2014.10.092. Epub 2015 Mar 25. PMID: 25817142. This study was published after the major impact of the RIVUR trial. The authors set to determine a consensus on antibiotic prophylaxis in children with VUR and recurrent UTI’s. With the addition of the RIVUR trial data, the paradigm was changed from favorability in children with high grade VUR to children with all grades of VUR.
Weng H, Zeng XT. Antibiotic prophylaxis for prevention of febrile urinary tract infections in children with vesicoureteral reflux: a meta-analysis of randomized, controlled trials comparing dilated to nondilated vesicoureteral reflux. World J Urol. 2017;35(5):847–8. https://doi.org/10.1007/s00345-016-1935-y (Epub 2016 Sep 12 PMID: 27620895).
Drzewiecki BA, Thomas JC, Pope JC 4th, Adams MC, Brock JW 3rd, Tanaka ST. Observation of patients with vesicoureteral reflux off antibiotic prophylaxis: physician bias on patient selection and risk factors for recurrent febrile urinary tract infection. J Urol. 2012;188(4):1480–4. https://doi.org/10.1016/j.juro.2012.02.033 (Epub 2012 Aug 17. PMID: 22906676; PMCID: PMC3459350).
Leslie B, Moore K, Salle JL, Khoury AE, Cook A, Braga LH, Bägli DJ, Lorenzo AJ. Outcome of antibiotic prophylaxis discontinuation in patients with persistent vesicoureteral reflux initially presenting with febrile urinary tract infection: time to event analysis. J Urol. 2010;184(3):1093–8. https://doi.org/10.1016/j.juro.2010.05.013 (Epub 2010 Jul 21 PMID: 20650501).
Moriya K, Mitsui T, Kitta T, Nakamura M, Kanno Y, Kon M, Nishimura Y, Shinohara N, Nonomura K. Early discontinuation of antibiotic prophylaxis in patients with persistent primary vesicoureteral reflux initially detected during infancy: outcome analysis and risk factors for febrile urinary tract infection. J Urol. 2015;193(2):637–42. https://doi.org/10.1016/j.juro.2014.08.007 (Epub 2014 Aug 9 PMID: 25111912).
Pennesi M, Travan L, Peratoner L, Bordugo A, Cattaneo A, Ronfani L, Minisini S, Ventura A, North East Italy Prophylaxis in VUR study group. Is antibiotic prophylaxis in children with vesicoureteral reflux effective in preventing pyelonephritis and renal scars? A randomized, controlled trial. Pediatrics. 2008;121(6):e1489-94. https://doi.org/10.1542/peds.2007-2652 (Epub 2008 May 19. PMID: 18490378).
Montini G, Rigon L, Zucchetta P, Fregonese F, Toffolo A, Gobber D, Cecchin D, Pavanello L, Molinari PP, Maschio F, Zanchetta S, Cassar W, Casadio L, Crivellaro C, Fortunati P, Corsini A, Calderan A, Comacchio S, Tommasi L, Hewitt IK, Da Dalt L, Zacchello G, IRIS Group. Prophylaxis after first febrile urinary tract infection in children? A multicenter, randomized, controlled, noninferiority trial. Pediatrics. 2008;122(5):1064–71. https://doi.org/10.1542/peds.2007-3770 (PMID: 18977988).
Roussey-Kesler G, Gadjos V, Idres N, Horen B, Ichay L, Leclair MD, Raymond F, Grellier A, Hazart I, de Parscau L, Salomon R, Champion G, Leroy V, Guigonis V, Siret D, Palcoux JB, Taque S, Lemoigne A, Nguyen JM, Guyot C. Antibiotic prophylaxis for the prevention of recurrent urinary tract infection in children with low grade vesicoureteral reflux: results from a prospective randomized study. J Urol. 2008;179(2):674-9 discussion 679. https://doi.org/10.1016/j.juro.2007.09.090 (Epub 2007 Dec 20. PMID: 18082208).
Garin EH, Olavarria F, Garcia Nieto V, Valenciano B, Campos A, Young L. Clinical significance of primary vesicoureteral reflux and urinary antibiotic prophylaxis after acute pyelonephritis: a multicenter, randomized, controlled study. Pediatrics. 2006;117(3):626–32. https://doi.org/10.1542/peds.2005-1362 (PMID: 16510640).
Wang ZT, Wehbi E, Alam Y, Khoury A. A Reanalysis of the RIVUR Trial Using a Risk Classification System. J Urol. 2018;199(6):1608–14. https://doi.org/10.1016/j.juro.2017.11.080 (Epub 2017 Dec 2 PMID: 29198997).
Wang HH, Gbadegesin RA, Foreman JW, Nagaraj SK, Wigfall DR, Wiener JS, Routh JC. Efficacy of antibiotic prophylaxis in children with vesicoureteral reflux: systematic review and meta-analysis. J Urol. 2015;193(3):963–9. https://doi.org/10.1016/j.juro.2014.08.112 (Epub 2014 Sep 6. PMID: 25196653; PMCID: PMC4380260).
Williams G, Hodson EM, Craig JC. Interventions for primary vesicoureteric reflux. Cochrane Database Syst Rev. 2019;2:CD001532.
Williams G, Craig JC. Long-term antibiotics for preventing recurrent urinary tract infection in children. Cochrane Database Syst Rev. 2019;4:CD001534.
Wheeler D, Vimalachandra D, Hodson EM, et al. Antibiotics and surgery for vesicoureteric reflux: a meta-analysis of randomised controlled trials. Arch Dis Childhood. 2003;88:688–94.
Williams G, Hodson EM, Craig JC. Interventions for primary vesicoureteric reflux. Cochrane Database Syst Rev. 2019;2(2):CD001532. https://doi.org/10.1002/14651858.CD001532.pub5 (PMID: 30784039; PMCID: PMC6380991).
Nordenström J, Sjöström S, Sillén U, Sixt R, Brandström P. The Swedish infant high-grade reflux trial: UTI and renal damage. J Pediatr Urol. 2017;13(2):146–54. https://doi.org/10.1016/j.jpurol.2016.12.023 (Epub 2017 Feb 2 PMID: 28215835).
Boysen WR, Akhavan A, Ko J, Ellison JS, Lendvay TS, Huang J, Garcia-Roig M, Kirsch A, Koh CJ, Schulte M, Noh P, Monn MF, Whittam B, Kawal T, Shukla A, Srinivasan A, Gundeti MS. Prospective multicenter study on robot-assisted laparoscopic extravesical ureteral reimplantation (RALUR-EV): Outcomes and complications. J Pediatr Urol. 2018;14(3):262.e1-262.e6. https://doi.org/10.1016/j.jpurol.2018.01.020 (Epub 2018 Feb 20 PMID: 29503220).
Arlen AM, Broderick KM, Travers C, Smith EA, Elmore JM, Kirsch AJ. Outcomes of complex robot-assisted extravesical ureteral reimplantation in the pediatric population. J Pediatr Urol. 2016;12(3):169.e1-6. https://doi.org/10.1016/j.jpurol.2015.11.007 (Epub 2015 Dec 17 PMID: 26747012).
Schomburg JL, Haberman K, Willihnganz-Lawson KH, Shukla AR. Robot-assisted laparoscopic ureteral reimplantation: a single surgeon comparison to open surgery. J Pediatr Urol. 2014;10(5):875–9. https://doi.org/10.1016/j.jpurol.2014.02.013 (Epub 2014 Apr 4 PMID: 24766855).
Harel M, Herbst KW, Silvis R, Makari JH, Ferrer FA, Kim C. Objective pain assessment after ureteral reimplantation: comparison of open versus robotic approach. J Pediatr Urol. 2015;11(2):82.e1-8. https://doi.org/10.1016/j.jpurol.2014.12.007 (Epub 2015 Feb 26 PMID: 25864615).
Smith RP, Oliver JL, Peters CA. Pediatric robotic extravesical ureteral reimplantation: comparison with open surgery. J Urol. 2011;185(5):1876–81. https://doi.org/10.1016/j.juro.2010.12.072 (Epub 2011 Mar 21 PMID: 21421231).
Deng T, Liu B, Luo L, Duan X, Cai C, Zhao Z, Zhu W, Wu W, Zeng G. Robot-assisted laparoscopic versus open ureteral reimplantation for pediatric vesicoureteral reflux: a systematic review and meta-analysis. World J Urol. 2018;36(5):819–28. https://doi.org/10.1007/s00345-018-2194-x (Epub 2018 Jan 27 PMID: 29374841).
Kurtz MP, Leow JJ, Varda BK, Logvinenko T, Yu RN, Nelson CP, Chung BI, Chang SL. Robotic versus open pediatric ureteral reimplantation: Costs and complications from a nationwide sample. J Pediatr Urol. 2016;12(6):408.e1-408.e6. https://doi.org/10.1016/j.jpurol.2016.06.016 (Epub 2016 Aug 23 PMID: 27593917).
Huang YY, Chen MJ, Chiu NT, Chou HH, Lin KY, Chiou YY. Adjunctive oral methylprednisolone in pediatric acute pyelonephritis alleviates renal scarring. Pediatrics. 2011;128(3):e496-504. https://doi.org/10.1542/peds.2010-0297 (Epub 2011 Aug 15 PMID: 21844061).
Shaikh N, Shope TR, Hoberman A, Muniz GB, Bhatnagar S, Nowalk A, Hickey RW, Michaels MG, Kearney D, Rockette HE, Charron M, Lim R, Majd M, Shalaby-Rana E, Kurs-Lasky M, Cohen DM, Wald ER, Lockhart G, Pohl HG, Martin JM. Corticosteroids to prevent kidney scarring in children with a febrile urinary tract infection: a randomized trial. Pediatr Nephrol. 2020;35(11):2113–20. https://doi.org/10.1007/s00467-020-04622-3 (Epub 2020 Jun 15. PMID: 32556960; PMCID: PMC7529851).
Da Dalt L, Bressan S, Scozzola F, Vidal E, Gennari M, La Scola C, Anselmi M, Miorin E, Zucchetta P, Azzolina D, Gregori D, Montini G. Oral steroids for reducing kidney scarring in young children with febrile urinary tract infections: the contribution of Bayesian analysis to a randomized trial not reaching its intended sample size. Pediatr Nephrol. 2021;36(11):3681–92. https://doi.org/10.1007/s00467-021-05117-5 (Epub 2021 May 25. PMID: 34032923; PMCID: PMC8497283).
Michaud JE, Gupta N, Baumgartner TS, Kim B, Bosemani T, Wang MH. Cost and radiation exposure in the workup of febrile pediatric urinary tract infections. J Surg Res. 2016;203:313–8.
Hoberman A, Charron M, Hickey RW, Baskin M, Kearney DH, Wald ER. Imaging studies after a first febrile urinary tract infection in young children. N Engl J Med. 2003;348(3):195–202. https://doi.org/10.1056/NEJMoa021698 (PMID: 12529459).
Juliano TM, Stephany HA, Clayton DB, Thomas JC, Pope JC 4th, Adams MC, Brock JW 3rd, Tanaka ST. Incidence of abnormal imaging and recurrent pyelonephritis after first febrile urinary tract infection in children 2 to 24 months old. J Urol. 2013;190(Suppl 4):1505–10. https://doi.org/10.1016/j.juro.2013.01.049 (Epub 2013 Jan 23. PMID: 23353046; PMCID: PMC3778088).
Shaikh N, Hoberman A, Hum SW, Alberty A, Muniz G, Kurs-Lasky M, Landsittel D, Shope T. Development and Validation of a Calculator for Estimating the Probability of Urinary Tract Infection in Young Febrile Children. JAMA Pediatr. 2018;172(6):550–6. https://doi.org/10.1001/jamapediatrics.2018.0217 (PMID:29710324;PMCID:PMC6137527).
Austin PF, Vricella GJ: Functional disorders of the lower urinary tract in children, in Wein AJ, Kavoussi LR, Partin AW, Peters CA (eds): CAMPBELL-WALSH UROLOGY, ed 11. Philadelphia, Elsevier, 2015, vol 4, chap 143, pp 3298–3299.
Kaufman J, Temple-Smith M, Sanci L. Urinary tract infections in children: an overview of diagnosis and management. BMJ Paediatr Open. 2019;3(1):e000487. https://doi.org/10.1136/bmjpo-2019-000487 (Published 2019 Sep 24).
Koçak M, Büyükkaragöz B, Çelebi Tayfur A, Çaltik A, Köksoy AY, Çizmeci Z, Günbey S. Causative pathogens and antibiotic resistance in children hospitalized for urinary tract infection. Pediatr Int. 2016;58(6):467–71. https://doi.org/10.1111/ped.12842 (Epub 2016 Feb 3 PMID: 26513232).
Campbell MF, Wein AJ, Kavoussi LR. Campbell-Walsh Urology. 9th ed. Philadelphia, PA: W.B. Saunders; 2007.
Filler G, Gharib M, Casier S, Lödige P, Ehrich JH, Dave S. Prevention of chronic kidney disease in spina bifida. Int Urol Nephrol. 2012;44(3):817–27. https://doi.org/10.1007/s11255-010-9894-5 (Epub 2011 Jan 13 PMID: 21229390).
Ottolini MC, Shaer CM, Rushton HG, Majd M, Gonzales EC, Patel KM. Relationship of asymptomatic bacteriuria and renal scarring in children with neuropathic bladders who are practicing clean intermittent catheterization. J Pediatr. 1995;127(3):368–72. https://doi.org/10.1016/s0022-3476(95)70065-x (PMID: 7658264).
Madden-Fuentes RJ, McNamara ER, Lloyd JC, Wiener JS, Routh JC, Seed PC, Ross SS. Variation in definitions of urinary tract infections in spina bifida patients: a systematic review. Pediatrics. 2013;132(1):132–9. https://doi.org/10.1542/peds.2013-0557 (PMID: 23796735).
Elliott SP, Villar R, Duncan B. Bacteriuria management and urological evaluation of patients with spina bifida and neurogenic bladder: a multicenter survey. J Urol. 2005;173(1):217–20. https://doi.org/10.1097/01.ju.0000146551.87110.f4 (PMID: 15592079).
Braga LHP, Lorenzo AJ, Jrearz R, Bagli DJ, Salle JLP. Bilateral ureteral reimplantation at primary bladder exstrophy closure. J Urol. 2010;183:2337–41.
Dickson AP. The management of bladder exstrophy: the manchester experience. J Pediatr Surg. 2014;49:244–50.
Curley T, Forster CS. Recurrent UTIs in Girls: What Is the Role of the Microbiome? Urology. 2021;151:94–7. https://doi.org/10.1016/j.urology.2020.04.091 (Epub 2020 May 7 PMID: 32389817).
Magruder M, Sholi AN, Gong C, et al. Gut uropathogen abundance is a risk factor for development of bacteriuria and urinary tract infection. Nat Commun. 2019;10:5521. https://doi.org/10.1038/s41467-019-13467-w.
Tariq R, Pardi DS, Tosh PK, Walker RC, Razonable RR. Fecal microbiota transplantation for recurrent clostridium difficile infection reduces recurrent urinary tract infection frequency. Clin Infect Dis. 2017;65:1745–7. https://doi.org/10.1093/cid/cix618.
Paalanne N, Husso A, Salo J, et al. Intestinal microbiome as a risk factor for urinary tract infections in children. Eur J Clin Microbiol Infect Dis. 2018;37:1881–91. https://doi.org/10.1007/s10096-018-3322-7.
AE Stapleton, M Au-Yeung, TM Hooton, et al. 2011 Randomized, placebo-controlled phase 2 trial of a lactobacillus crispatus probiotic given intravaginally for prevention of recurrent urinary tract infection Clin Infect Dis 52(2011):1212–1217. Doi: https://doi.org/10.1093/cid/cir183
Lev-Sagie A, Goldman-Wohl D, Cohen Y, et al. Vaginal microbiome transplantation in women with intractable bacterial vaginosis. Nat Med. 2019;25:1500–4. https://doi.org/10.1038/s41591-019-0600-6.
L Kinneman, W Zhu, SW Wendy, et al. 2020 Assessment of the urinary microbiome in children younger than 48 months Pediatr Infect Dis J (2020) https://doi.org/10.1097/INF.0000000000002622
Ramji J, Weiss DA, Romao RLP, Eftekharzadeh S, Shah J, Frazier JR, Reddy PP, Merguerian PA, Pippi Salle JL, Canning DA, Joshi RS, Shukla AR. Impact of bilateral ureteral reimplantation at the time of complete primary repair of bladder exstrophy on reflux rates, renogram abnormalities and bladder capacity. J Pediatr Urol. 2021;17(3):393.e1-393.e7. https://doi.org/10.1016/j.jpurol.2021.01.020 (Epub 2021 Jan 21 PMID: 33583747).
Baskin, L. S., Kogan, B. A., & Stock, J. A. (2019). Handbook of Pediatric Urology. Wolters Kluwer.
Langley JM, Hanakowski M, Leblanc JC. Unique epidemiology of nosocomial urinary tract infection in children. Am J Infect Control. 2001;29(2):94–8. https://doi.org/10.1067/mic.2001.111537 (PMID: 11287876).
Marcus N, Ashkenazi S, Samra Z, Cohen A, Livni G. Community-acquired enterococcal urinary tract infections in hospitalized children. Pediatr Nephrol. 2012;27(1):109–14. https://doi.org/10.1007/s00467-011-1951-5 (Epub 2011 Aug 7 PMID: 21822790).
Seifi Z, Azish M, Salehi Z, Zarei Mahmoudabadi A, Shamsizadeh A. Candiduria in children and susceptibility patterns of recovered Candida species to antifungal drugs in Ahvaz. J Nephropathol. 2013;2(2):122–8. https://doi.org/10.12860/JNP.2013.20.
Devillé WL, Yzermans JC, van Duijn NP, Bezemer PD, van der Windt DA, Bouter LM. 2004 The urine dipstick test useful to rule out infections. A meta-analysis of the accuracy. BMC Urol. 4:4. Published doi:https://doi.org/10.1186/1471-2490-4-4
Marques AG, Doi AM, Pasternak J, Damascena MDS, França CN, Martino MDV. Performance of the dipstick screening test as a predictor of negative urine culture. Einstein (Sao Paulo). 2017;15(1):34–9. https://doi.org/10.1590/S1679-45082017AO3936.
Bafna P, Deepanjali S, Mandal J, Balamurugan N, Swaminathan RP, Kadhiravan T. Reevaluating the true diagnostic accuracy of dipstick tests to diagnose urinary tract infection using Bayesian latent class analysis. PLoS ONE. 2020;15(12): e0244870. https://doi.org/10.1371/journal.pone.0244870 (PMID:33382863;PMCID:PMC7774958).
Tzimenatos, Leah, et al. Accuracy of the urinalysis for urinary tract infections in febrile infants 60 days and younger. Pediatrics. (2018);141(2).
Shekarriz B, Upadhyay J, Freedman AL, Fleming P, Barthold JS, González R. Lack of morbidity from urodynamic studies in children with asymptomatic bacteriuria. Urology. 1999;54(2):359–61; discussion 362. https://doi.org/10.1016/s0090-4295(99)00214-9 (PMID: 10443739).
Shaikh N, Osio VA, Wessel CB, Jeong JH. Prevalence of Asymptomatic Bacteriuria in Children: A Meta-Analysis. J Pediatr. 2020;217:110-117.e4. https://doi.org/10.1016/j.jpeds.2019.10.019 (Epub 2019 Nov 28. Erratum in: J Pediatr. 2020 Jan 31; PMID: 31787323).
Marschner CA, Schwarze V, Stredele R, Froelich MF, Rübenthaler J, Geyer T, Clevert DA. Evaluation of the Diagnostic Value of Contrast-Enhanced Voiding Urosonography with Regard to the Further Therapy Regime and Patient Outcome-A Single-Center Experience in an Interdisciplinary Uroradiological Setting. Medicina (Kaunas). 2021;57(1):56. https://doi.org/10.3390/medicina57010056 (PMID:33435420;PMCID:PMC7826578).
Colleran GC, et al. Residual intravesical iodinated contrast: a potential cause of false-negative reflux study at contrast-enhanced voiding urosonography. Pediatr Radiol. 2016;46(11):1614–7.
Ferrara F, Dickson AP, Fishwick J, Vashisht R, Khan T, Cervellione RM. Delayed exstrophy repair (DER) does not compromise initial bladder development. J Pediatr Urol. 2014;10(3):506–10. https://doi.org/10.1016/j.jpurol.2013.10.026 (Epub 2013 Nov 28 PMID: 24331166).
Baradaran N, Cervellione RM, Stec AA, Gearhart JP. Delayed primary repair of bladder exstrophy: ultimate effect on growth. J Urol. 2012 Dec;188(6):2336–41. https://doi.org/10.1016/j.juro.2012.08.037 (Epub 2012 Oct 22 PMID: 23088977).
Santos JD, Lopes RI, Koyle MA. Bladder and bowel dysfunction in children: An update on the diagnosis and treatment of a common, but underdiagnosed pediatric problem. Can Urol Assoc J. 2017;11(1–2):S64–72. https://doi.org/10.5489/cuaj.4411.
Chase JW, Homsy Y, Siggaard C, Sit F, Bower WF. Functional constipation in children. J Urol. 2004;171(6 Pt 2):2641–3. https://doi.org/10.1097/01.ju.0000109743.12526.42 (PMID: 15118440).
Santos et al 2014 PediatTherapeut 4 1 https://doi.org/10.4172/2161-0665.1000191
Amira P, Dušan P, Gordana M. Bladder control training in girls with lower urinary tract dysfunction. Int Braz J Urol. 2013;39:118–27. https://doi.org/10.1590/S1677-5538.IBJU.2013.01.15.
Zivkovic V, Lazovic M, Vlajkovic M. Diaphragmatic breathing exercises and pelvic floor retraining in children with dysfunctional voiding. Eur J Phys Rehabil Med. 2012;48:413–21.
Kajbafzadeh A, Sharifi-Rad L, Ghahestani S. Animated biofeedback: An ideal treatment for children with dysfunctional elimination syndrome. J Urol. 2011;186:2379–85.
Vesna Z, Milica L, Marina V. Correlation between uroflowmetry parameters and treatment outcome in children with dysfunctional voiding. J Pediatr Urol. 2010;6:396–402.
Vasconcelos M, Lima E, Caiafa L. Voiding dysfunction in children. Pelvic-floor exercises or biofeedback therapy: A randomized study. Pediatr Nephrol. 2006;21:1858–64.
Barroso U, Lordêlo P. Electrical nerve stimulation for overactive bladder in children. Nat Rev Urol. 2011;8:402–7.
Veiga ML, Lordêlo P, Farias T, Barroso U Jr. Evaluation of constipation after parasacral transcutaneous electrical nerve stimulation in children with lower urinary tract dysfunction–a pilot study. J Pediatr Urol. 2013;9(5):622–6. https://doi.org/10.1016/j.jpurol.2012.06.006 (Epub 2012 Aug 1 PMID: 22857873).
Taylor AS, Cabo JJ, Lauderdale C, Maskan N, Thomas JC, Tanaka ST, Pope JC, Adams MC, Brock JW, Shannon CN, Clayton DB. Pelvic floor biofeedback therapy in children: Assessment of symptom scores in responders and non-responders. Neurourol Urodyn. 2019;38(1):254–60. https://doi.org/10.1002/nau.23842 (Epub 2018 Oct 23 PMID: 30350888).
Eyer de Jesus L, Dekermacher S, Pippi-Salle JL. Bladder exstrophy: We need to improve A lot. J Pediatr Urol. 2021;S1477–5131(21):00530–1. https://doi.org/10.1016/j.jpurol.2021.11.007 (Epub ahead of print. PMID: 34876380).
Sinatti C, Spinoit AF, Raes A, Van Laecke E, Hoebeke P. Long-Term fate of the upper urinary tract and ITS association with continence in exstrophy patients. J Pediatr Urol. 2021;17(5):655.e1-655.e7. https://doi.org/10.1016/j.jpurol.2021.06.009 (Epub 2021 Jun 11 PMID: 34172386).
Marei MM, Jackson R, Keene DJB. Intravesical gentamicin instillation for the treatment and prevention of urinary tract infections in complex paediatric urology patients: evidence for safety and efficacy. J Pediatr Urol. 2021;17(1):65.e1-65.e11. https://doi.org/10.1016/j.jpurol.2020.08.007 (Epub 2020 Aug 19 PMID: 33309610).
Eldridge GR, Hughey H, Rosenberger L, Martin SM, Shapiro AM, D'Antonio E, Krejci KG, Shore N, Peterson J, Lukes AS, Starks CM. Safety and immunogenicity of an adjuvanted Escherichia coli adhesin vaccine in healthy women with and without histories of recurrent urinary tract infections: results from a first-in-human phase 1 study. Hum Vaccin Immunother. 2021 May 4;17(5):1262–1270. doi: https://doi.org/10.1080/21645515.2020.1834807. Epub 2020 Dec 16. PMID: 33325785; PMCID: PMC8078672. A first of its kind Phase I, dose escalation trial investigating the use of a vaccine against FimH protein present on gram negative bacteria responsible for recurrent UTI’s. The results showed that the vaccine was well-tolerated among all participants and induction of binding and functional antibodies was successful. A double blinded, randomized, placebo-controlled phase II study has been approved based on the paper’s results.
Langermann S, Palaszynski S, Barnhart M, Auguste G, Pinkner JS, Burlein J, Barren P, Koenig S, Leath S, Jones CH, Hultgren SJ. Prevention of mucosal Escherichia coli infection by FimH-adhesin-based systemic vaccination. Science. 1997;276(5312):607–11. https://doi.org/10.1126/science.276.5312.607 (PMID: 9110982).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Philip Olson, Anne G. Dudley, and Courtney K. Rowe declare that they have no conflict of interest.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Olson, P., Dudley, A.G. & Rowe, C.K. Contemporary Management of Urinary Tract Infections in Children. Curr Treat Options Peds 8, 192–210 (2022). https://doi.org/10.1007/s40746-022-00242-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40746-022-00242-1