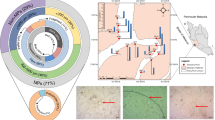
Abstract
Microplastics research in South Africa has focused on marine and coastal areas, with limited studies reporting microplastics in freshwaters. To address this gap, we conducted this study to investigate the presence, abundance, physical characteristics, and chemical composition of microplastics in the surface water of the Vaal River, one of the most important freshwater systems in South Africa. Twenty-two surface water samples were collected along 60 km of the middle part of the river using a 55 μm plankton net. Samples were treated by acid digestion, and MPs were separated by density separation. Physical identification was performed using a Nikon stereomicroscope, and chemical composition was identified using Raman spectroscopy. MPs were observed in all samples with 0.13 to 2.52 particles/m3 concentrations. Microplastics ranged in size from 0.06 – 4.95 mm, with more than 89% less than 2 mm, and 81% were fragments (39%) and fibers (42%). Microplastics were observed in numerous colours, of which green, black, and blue were the most dominant, representing 22%, 19%, and 18%, respectively. Chemical analysis revealed the polymer types as high-density polyethylene (HDPE), polypropylene (PP), and low-density polyethylene (LDPE). In addition, two colourants, pigment yellow 83 and carbon black, were detected. This study provides new data on microplastic occurrence and characteristics in the Vaal River system and offers a baseline for future monitoring. Our findings confirm that the Vaal River is under a real threat of microplastic contamination, which could impact its role as a significant freshwater system in South Africa.
Highlights
• Microplastics were prevalent in the surface water of the Vaal River.
• Fibrous, small-sized, and coloured microplastics were most abundant.
• Polymers were identified as HDPE, LDPE, and PP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Plastics were commercialised in the 1950s, and by the 1970s, they became widely used in countless applications (Amaral-Zettler et al. 2020). Due to their favourable properties like durability, mechanical strength, and lightweight, plastics are extensively used for various industrial and commercial purposes (Fred-Ahmadu et al. 2020). The global increase in the production and consumption of face masks during the COVID–19 pandemic has also increased plastic waste in the environment (Aragaw 2020). Plastic materials offer many benefits to society; nevertheless, their negative environmental impacts have become of great concern. Despite significant advances in waste management strategies and recycling technologies, only 18% of plastic waste is recycled (Chamas et al. 2020).
Plastics were considered a challenging environmental threat until the term “microplastics (MPs)” arose (Kutralam-Muniasamy et al. 2022). MPs are small plastic debris (< 5 mm). Based on how they originate, MPs are classified into primary or secondary MPs. Primary MPs are plastic particles manufactured in a microscopic size. They are commonly used in cosmetics, personal care products, and different industries. Secondary MPs are formed by the breaking down of large plastics (Onoja et al. 2022; Park and Park 2021; Samandra et al. 2021). MPs enter aquatic environment through different pathways such as direct disposal from the public, sewage sludge, beach littering, wastewater effluent, atmospheric fallout, and deposition, wear and tear of tyres, as well as runoff from urban areas, industrial, agricultural, and recreational activities (Nikiema et al. 2020a,b; Ramaremisa et al. 2022).
MPs are ubiquitous and have been extensively documented in various environments around the globe. However, MP research in Africa is a fledgling field mainly due to a lack of funding and resources available at research institutions. Most studies have thus been conducted in South Africa, possibly due to the availability of funding and resources in South African institutions compared to other African institutions (Aragaw 2021; Saad and Alamin 2024). Studies conducted have reported MP pollution in the Orange-Vaal River system (Weideman et al. 2020); surface water and freshwater fish (Cyprinus carpio) and sediments from the Vaal River, Gauteng province (Saad et al. 2022a, 2022b, 2024), urban estuaries and four species (i.e., Ambassis dussumieri, Terapon jarbua, Oreochromis mossambicus, Mugil sp.) in KwaZulu Natal (Naidoo et al. 2015, 2019), beach sediment and surf-zone water from the south-eastern coastline, Eastern Cape (Nel and Froneman 2015), wastewater treatment plants and the Rietvlei Dam in Gauteng province (Vilakati et al. 2021, 2022), marine water from the Atlantic Ocean, Western Cape province (Vilakati et al. 2020), and water and sediment from the Plankenburg River in the Western Cape (Apetogbor et al. 2023). However, most of these studies focused on the monitoring and quantification of MPs. The quantification of MPs in different environmental compartments is essential, but their ecological and health impacts can only be understood by studying their physical and chemical properties. Compared to other contaminants, and due to the variety in their physical characteristics, including the wide range of sizes, colours, and shapes. MP behaviour in aquatic environments is complex. Studying MP characteristics is essential to understanding their deposition, transport, distribution, and fate in different environmental compartments (Cheung et al. 2019; Guo et al. 2023; Hu et al. 2023; Shamskhany et al. 2021; Summers et al. 2023). For instance, MPs with different densities and shapes are distributed differently, which influences their bioavailability and fate and, thus, their ecological impact. In addition, the uptake/ingestion of MPs by plants and animals is influenced to a great extent by their colour, shape, and size (Li et al. 2023a; Saad et al. 2022a; Saad 2023). The toxicity of MPs is also influenced by their physiochemical properties (Beiras and Schönemann 2020; Caputo et al. 2021; Krishnan et al. 2023). This work, therefore, aimed to fill this gap and study the physical and chemical characteristics of MPs in the surface water of the Vaal River for better assessing and understanding of MP pollution in the Vaal River system. The Vaal River is primed for such study, considering its socioeconomic importance as the primary water source to various municipalities, industries, and agricultural sectors in South Africa’s economic hub (Gauteng province) and other surrounding provinces.
2 Methodology
2.1 Study Area and Sampling
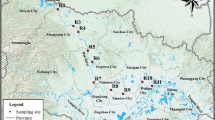
The Vaal River is a significant riverine system in South Africa with considerable economic value. It flows through five provinces and provides water needs for drinking, industrial use, and irrigation (Bouwman et al. 2018; Weideman et al. 2020). Sampling covered around 60 km long of the middle part of the river, between the Vaal barrage and Lethabo power station. This part of the river receives effluents from domestic, industrial, and WWTP discharges. It is also a hub for a variety of recreational activities (Weideman et al. 2020).
Samples were collected from twenty-two locations using a plankton net (0.25 m diameter, 55 μm mesh size) over two days in June 2021 (sampling locations are depicted in the map of Fig. 1, and a detailed description of each location is provided in Table SM1). The plankton net was towed along the river’s surface for 15 min using a rope, traveling downstream at 12 km/h. During sampling, the sampled materials were collected at the cod-end of the plankton net, which contains a small net bucket with a knob that can be opened to transfer the sample into glass jars (the manual of the plankton net, including description and image can be found using the link: https://www.hydrobios.de/images/datasheets/438%20001_Apstein.pdf, Accessed 10 Feb 2024). The samples were transferred to the laboratory in a cooler box and stored in a fridge at 4 °C for subsequent analysis. GPS and flow meter readings at the beginning and the end of each tow were used to calculate the distance travelled and the volume filtered. The distance covered was determined by converting the number of flowmeter rotations obtained from the difference between the initial and final flowmeter readings to metres using the following formula:
where: 26,873 is the Rotor constant; 999,999 is the 2ix-digit full-scale count.
The volume sampled was calculated by multiplying the distance trawled by the area of the net opening (Lam et al. 2020; Tamminga et al. 2019). The filtered volume was calculated as follows:
The flowmeter readings and calculated distances and volumes for each location are listed in Table SM2, and the GPS coordinates and calculated area sampled are listed in Table SM3.
2.2 Treatment of Samples
The preparation of samples was done with reference to the National Oceanic and Atmospheric Administration (NOAA) laboratory methods (Marine Debris Program 2015). Firstly, samples were digested using Fenton’s reagent (0.07 M FeSO4.7H2O catalyst and 30% (v/v) H2O2) to remove organic matter. Samples were left in the digestion solution for 24 to 48 h at room temperature to achieve complete digestion (Ramaremisa et al. 2022). Following this step, the two-step density separation protocol was followed to separate MP particles based on their density (Nuelle et al. 2014). The two-step density separation process was initiated by adding 45 \({{\text{cm}}}^{3}\) saturated NaCl (1.2 g cm−3) to the samples, left for 24 h to density separate before filtration. A second step was followed by adding 45 \({{\text{cm}}}^{3}\) NaI (1.8 g cm−3) to separate the high-density MPs. Mixtures were left for another 24 h to settle before final filtration. GF/A filter papers Whatman® (47 mm diameter, pore size: 1.6 μm) were used to extract MPs. Air-dried filter papers were stored in covered glass Petri dishes till analysis.
2.3 Quality Control
Extreme caution was taken during sample collection, processing, and analysis to prevent cross-contamination. Except for the plankton net, no plastic apparatus or containers were used during sampling and sample preparation, and experimental equipment and containers were washed with filtered Milli-Q® Type 1 Ultrapure water and rinsed with filtered ethanol. All laboratory experiments were conducted in a laminar flow cabin in a lab dedicated to MPs, and cotton lab coats were worn during all stages to prevent contamination risk from clothing fibers. To mitigate against the potential contamination of samples by MPs in chemicals, all solutions were filtered through a Whatman® GF/F glass microfiber filter (Cytiva Danaher Group, Buckinghamshire, United Kingdom) before use. In addition, blank experiments were conducted with filtered Milli-Q® Type 1 Ultrapure water to count for possible procedural contamination. Blank experiments were conducted for every five samples and processed along with the samples. No particles were detected in any blank sample.
2.4 Physical and Chemical Identification of MPs
The morphological examination was performed under a stereomicroscope (Nikon MET SMZ745T, Japan) with a detection limit of 0.04 mm. Potential MPs were counted, measured, and photographed.
The size of MP particles was measured using Nikon’s NIS Elements-D imaging software Version 5.30. The size of the pellets was determined by measuring their diameter because of their spherical nature. However, the size of fibers, fragments, and films was determined by measuring the separation between a particle’s two most distant points (Pivokonsky et al. 2018).
For every sample, four particles representing all shapes detected were selected at random and analysed using Scanning Electron Microscopy (SEM) to examine their surface morphology; particles were extracted using tweezers from the filters and mounted on the sample holder for analysis. Chemical identification of MPs was performed using a Raman spectrometer (Horiba LabRAM HR). On average, fifteen particles were analysed per sample. SLOPP Library of MPs and polymer databases of KnowItAll software (Bio-Rad Laboratories, Inc.) were used as references to compare the acquired Raman spectra and to identify polymer types.
3 Results and Discussion
3.1 MP Abundance
In total, 848 particles were identified as MPs, ranging from as low as 8 particles in L3 to as high as 159 particles in L7. An average of 38.5 ± 35.5 particles were detected per tow. MP abundance per sample is reported in Table 1.
It is worth mentioning that the sampling locations were near urban areas (e.g., Parys, Vereeniging, Sasolburg, and Vanderbijlpark); informal settlements (e.g., several informal settlements under the R59 road); industrial hubs (e.g., Arcelor Mittal Vaal works and Sasol); wastewater treatment plants (e.g., Rietspruit, Midvaal, Sebokeng, and Leeuwkuil); and agricultural activities (e.g., the land around the Vaal River is widely used for agriculture). This is in addition to several anthropogenic activities and tourism (Ramaremisa et al. 2022; Wepener et al. 2011). These are reported as primary sources of plastic waste entering water bodies (Qiu et al. 2023; Shu et al. 2023; Yin 2022; Zhou et al. 2023). High abundance values were detected at locations L5 to L10 (0.73, 1.2, 2.5, 0.74, 1.6, and 1.3 particles/m3, respectively). These sampling spots were located within the region from the Taaibosspruit-Vaal River confluence to the confluences of the Vaal River with the Klip and Suikerbosrand Rivers. Pollution input from tributary streams is known to contribute to the pollution profile of main rivers (Zhang et al. 2021). This explains the high abundance of MPs in this region of the Vaal.
Several recent studies reported MPs in surface waters of various freshwater systems across the globe (Kieu-Le et al. 2023; Liu et al. 2024; Rodrigues dos Santos et al. 2023; Tran-Nguyen et al. 2024). However, the lack of standard analytical protocols for sampling, preparation, and identification makes comparisons between studies unattainable. In addition, these studies report MP abundance in different units, which makes the comparison inaccurate. Nonetheless, most studies revealed higher abundances at urban centres, industrial and residential areas, and recreational activities (Manbohi et al. 2023; Zhao et al. 2024).
3.2 Physical Characteristics
3.2.1 Distribution of Sizes of MPs
Overall MPs ranged in size from 0.06 to 4.95 mm, with an average of 0.96 ± 0.88 mm. Figure 2 shows the size distribution of MPs detected in all samples. Small MPs were more abundant (MPs < 2 mm accounted for 89%, the remaining 11% in the size range of 2 – 5 mm). Among the small size range (< 2 mm), sizes less than 0.5 mm were found to be the most abundant (38%), followed by 0.5 – 1 mm (29%), and less (22%), in the range of 1 – 2 mm.
The prevalence of small-sized MPs has been reported in several studies. Zhao et al. (2024) studied MPs in the surface water of the XJ River in China; the authors reported 60% of MPs in the size range of 0.05 to 0.1 mm. Similarly, Imbulana et al. (2024) observed that 50% of MPs detected in a primary river network in Japan were smaller than 0.04 mm (Imbulana et al. 2024; Zhao et al. 2024).
Spearman correlation test was used to assess the relationship, if any, between MP abundance and their sizes (Fig. 3). The statistical analysis revealed a weak correlation (p-value = 0.60). The greater abundance of small-sized MPs could be explained by the frequent degradation of MPs, possibly due to weathering conditions in the Vaal River, which results in a continuous decrease in MPs size (Koelmans and Kalčíková 2019). Thus, unlike other pollutants, MPs become more abundant with time (Saad et al. 2022b).
MP size affects their transport, distribution, pathways, and fate. For instance, size affects the buoyancy and the drag force. Consequently, it determines their dispersal and distribution in water and accumulation in sediment. For example, small MPs are frequently reported in near-surface water, whereas larger particles are found in deep waters at different depths depending on their density (Shamskhany et al. 2021). This explains the higher abundance of small-sized MPs in this study. However, with time, MPs tend to sink in sediments due to density change, which results in biofouling, adsorption of co-existing contaminants from the surroundings, and other environmental conditions (Tekman et al. 2020). MP particle size also influences their mobility and long-distance transport (Zhang et al. 2022). Further, MP size is tied to hydrodynamic parameters such as relaxation time, settling, and rise velocity; ultimately, this correlation defines the mobility and transport of MPs. For instance, compared to larger MPs, small-sized MPs are reported to have low relaxation time and low settling velocity but faster sinking due to biofouling, implying that small MPs are more mobile and have a fast transport rate compared to larger MPs (Kumar et al. 2021; Shamskhany et al. 2021).
3.2.2 Distribution of Shapes of MPs
Particles were categorised into fibers (slender shape); films (very thin particles); fragments (irregular shape); and pellets (spherical shape) (Su et al. 2016). Typical MP shapes observed in this study are shown in Fig. SM1. Proportionally, fibers and fragments were dominantly present, accounting for 81% of the total particles (Fig. 4).The remaining particles were identified as films and pellets (19%).
The dominance of fibrous MPs over other shapes is reported in the Mekong River Delta in Vietnam, ten urban and rural freshwater sites in Australia, the Ergene River in Turkey, and the West River in China (Akdogan et al. 2023; Huang et al. 2021; Kieu-Le et al. 2023; Nan et al. 2020).
The shape of MPs is another essential characteristic that impacts their behaviour. For example, it influences the biofouling rate and, thus, affects the sinking/rising behaviour of MPs (Godoy et al. 2019; Murphy et al. 2022). The shape of MPs can also indicate their sources; for instance, potential sources of fibers may include domestic sewage from residential areas around the Vaal, namely, Sedibeng and Metsimaholo districts. It is important to note that this region of the Vaal receives copious amounts of wastewater effluent from several WWTPs, namely, Leeuwkuil, Sebokeng, Midvaal, and Rietspruit WWTPs (Saad et al. 2022a; Weideman et al. 2020). Fragments could be attributed to recreational activities; the Vaal River is a renowned tourism destination in the country.
Pellets are primary MPs produced by ultrafine grinding in mills and are extensively used in cosmetics and other personal-care products, primarily in small sizes (Fahrenfeld et al. 2019; Hartmann et al. 2017; Koelmans and Kalčíková 2019). These can enter the Vaal as people engage in different water activities. The average size of pellets in personal care products such as sunscreens is between 0.06 and 0.8 mm (Cheung and Fok 2017; Sun et al. 2020). This agrees with the findings of this study, noting that 86% of pellets were < 0.5 mm. The remaining 14% were in the 0.5–1 mm (13%) and 1–2 mm (1%), with an average size of 0.32 ± 0.19 mm (Fig. SM2). The average sizes (mean value ± stdev) of fibers, fragments, and films were 1.2 ± 0.86 mm, 0.64 ± 0.59 mm, and 1.7 ± 1.3 mm, respectively.
The distribution of shapes across different size ranges showed that in all samples, the highest proportion (> 80%) of fibers were in the gauges of < 0.5 mm (20%), between 0.5 and 1 mm (33%), and between 1 and 2 mm (32%). Lower quantities were in higher size ranges of 2 to 3 mm (11%), 3 to 4 mm (2%), and 4 to 5 mm (2%). Similarly, 54% of fragments were less than 0.5 mm, 28% were between 0.5 to 1 mm, and 13% were between 1 to 2 mm. Smaller quantities were detected in the gauge of 2 to 3 mm (3%) and 3 to 4 mm (1%). Films were distributed as follows: 15% were less than 0.5 mm, 25% were between 0.5 and 1 mm, 31% between 1 and 2 mm, 11% were in the range of 2 to 3 mm, 9% were in the range of 3 to 4 mm, and only 8% were in the range of 4 to 5 mm. The size distribution across fibers, fragments, and films indicates that large plastic debris may undergo successive fragmentation when exposed to different environmental processes/conditions in the Vaal River. Considering the gradual disintegration of plastics into smaller particles over time, their size distribution in rivers and other water systems is more likely to be skewed towards lower-size classes (Mason et al. 2016; Onoja et al. 2022).
3.2.3 Distribution of Colours of MPs
Numerous colours were observed, and green, black, and blue were the most dominant, accounting for 22%, 19%, and 18%, respectively (Fig. 5). MPs with white and red colour were less prevalent, accounting for 11% and 6%, respectively. Other minor colours (pink, purple, yellow, and brown) that were least prevalent were grouped as “others”; accounting for 5%. Transparent MPs were detected in a smaller portion of 17% of the total particles.
The distribution of colours across different shapes is depicted in Fig. SM3. The wide variety in colours suggests that MPs in the Vaal River are from various sources. For instance, the presence of fragments in relatively equal proportions of different colours (blue (19%), green and white (18%) each, black (15%), red (7%), and other colours (8%)), reflects their multiple sources (fragmentation of a wide range of plastic items). The dominance of coloured fibers over transparent, and the absence of white fibers, suggest that wastewater discharge is the primary source of these fibrous MPs. Coloured fibers are known to originate from synthetic textiles, whereas white fibers come from fishing nets and lines. The prevalence of transparent films (66%) is also consistent with their potential sources (packaging materials and wrapping films) (Dris et al. 2018; Lahiri et al. 2023; Sadan and de Kock 2020; Zhang et al. 2021).
3.2.4 Surface Texture of MPs
SEM images of selected MPs are depicted in Fig. 6. The images clearly show cracks, scratches, and pores on the surfaces of the particles, which are signs of degradation. Such signs include the rough surface of the microplastic fragment (Fig. 6b), the cracked and porous surface of the pellet (Fig. 6d), and fine particles pitting on the surface of the fiber and film particles (Fig. 6a and Fig. 6c). In natural environments like the Vaal River, plastics/MPs are exposed to different weathering processes. Additionally, the degradation increases the surface area of MPs, resulting in an increased sorption capacity/sites (Saad 2022, 2023; Zhang et al. 2021). This explains the presence of small particles on the surface of MPs (Fig. 6a and Fig. 6c). Therefore, degraded MPs are of great concern due to their higher adsorption capacity of co-existing pollutants (Guo et al. 2023; Hu et al. 2023; Zhong et al. 2023).
3.3 Chemical Composition
3.3.1 Polymer Types
The chemical composition of MPs was identified with reference to their Raman spectra (Fig. SM4). The Raman-Forensic-Horiba database is abbreviated as RAX, RHX for Raman-Forensic-Horiba, and WSARX for the Raman-Sigma-Aldrich Library of Raman Spectra-Wiley database. The polymer identity number and names in the databases are also given. Three polymers were identified, namely, high-density polyethylene (HDPE), low-density polyethylene (LDPE), and polypropylene (PP).
The Raman spectra osf LDPE (Fig. SM4a) and HDPE (Fig. SM4b) have noticeable differences. In the CH2 bending and CH2 twisting regions, the intensity of the CH2 bending mode was higher for HDPE (1414.40 cm−1 and 1440.51 cm−1) compared to that of LDPE (1418.34 cm−1 and 1439.55 cm−1). Due to its crystalline state, PE has a more pronounced doublet of narrow bands in the region 1400–1500 cm−1 (Ibrahim and He 2017; Jumeau et al. 2013). The differences between HDPE and LDPE are also pronounced in the C-H stretching region. The intensity of the symmetric CH2 stretching mode at 2849.50 cm−1 (relative to the asymmetric CH2 stretching mode at 2882.29 cm−1) is higher for LDPE compared to HDPE with symmetric and asymmetric CH2 stretching modes at 2881.10 cm−1 and 2848.32 cm−1, respectively. The stretching of C-H (2800–2900 cm−1) and the bending of CH2 (1400–1500 cm−1) are sensitive to the density of PE; thus, these regions were used to differentiate between the different Crystalline and amorphous phases of the PE (Ibrahim and He 2017).
The Raman spectrum of the sample in Fig. SM4c was superimposable to a PP homopolymer reference spectrum. The sample and reference spectra had a 98% HQI and shared major peaks at 399.13 cm−1, 807–842.68 cm−1, 1151.24 cm−1, and 1459.80 cm−1, all assigned to PP (Nielsen et al. 2002).
In South Africa, the largest plastic production companies (Sasol Polymers and Safripol) polymerise ethylene and propylene into polyolefins (PE or PP) (Sadan and de Kock 2020). Polyolefins are the most ubiquitous synthetic commercial polymers, and PE is the most mass-produced polymer worldwide. Numerous studies have reported PE and PP in different aquatic compartments of various water systems (Lee and Kim 2023; Li et al. 2023a; Yang et al. 2023; Yu et al. 2023).
PE plastics are classified according to their molecular structure, melting point, and density as either LDPE or HDPE. LDPE has a highly branched chain structure and is a product of a non-catalyzed high-pressure polymerization. Its applications include packaging, bottles, plastic bags, clothes, water tanks, plastic furniture, and films. On the other hand, HDPE is a highly crystalline polymer with a linear chain produced by low-pressure polymerization. Its applications include bottles, large containers, drums, fuel tanks, pipes, wrapping films, and crates. Although mostly inert, PE can slowly degrade in the environment due to structural defects or under the influence of various forces in nature (Chamas et al. 2020).
PP is harder than PE, is translucent, semi-rigid, tough, and has good heat and chemical resistance. It is available in three commercial grades and used in several applications (automotive, patio furniture, bottles, films, tapes, webbing straws, and appliances). PP products can undergo similar degradation processes to PE and generate MPs (Chamas et al. 2020).
3.3.2 Colourants
The sample and reference spectra in Fig. SM4d had a 91.56% HQI and major peaks at 1259.71–1293.73 cm−1 (stretching of CC moieties), 1406 cm−1 (stretching of NN moieties), and 1601.04 cm−1 (stretching of CC and bending of CH moieties), all assigned to Pigment Yellow 83 (Yakes et al. 2017). The Raman spectrum in Fig. SM4e had carbon bands of Vine black (Pigment Black 6) at 1337.14 cm−1 and 1534.30 cm−1 (Coccato et al. 2015; Tomasini et al. 2012). The reference spectrum is superimposable to other carbon blacks like Ivory black and Van Dyck brown with HQI values greater than 97%. Carbon blacks are carbon-based pigments used in polymers/polymer blends. In terms of the volume of all pigments used by the plastic industry, carbon blacks are second only to TiO2 (Pigment White 6). Polymers containing carbon blacks can exhibit a carbon-like Raman spectrum, and for this reason, black particles are sparsely confirmed via Raman spectroscopy (Huang 2002; Coccato et al. 2015; Käppler et al. 2016).
4 Implications of the Physical Characteristics of MPs
The size, shape, and colour of MPs have significant impacts on their behaviour, transport, and distribution in aquatic environments, thus, significant consequences on their fate, bioavailability, and potential health risks (Gad and Midway 2022; Gray and Weinstein 2017; Shamskhany et al. 2021; Saad 2023; Zhang et al. 2022).
The dominance of small-sized, fibrous, and coloured MPs in the Vaal River is of great concern. The small size of MPs makes them available to organisms throughout aquatic food webs. Further, smaller-sized MPs may interact with microbial biofilms such as fungi and bacteria and cause a higher probability of being mistakenly ingested by organisms. Additionally, small-sized MPs have larger surfaces available for the attachment of other contaminants, thus, they present more hazards to aquatic organisms (Li et al. 2023b; Saad et al. 2023; Saad 2023). In short, the effect of MPs size on their mobility, distribution, fate, and environmental behaviour, ultimately defines their potential ecological impacts and health risks. Smaller-sized MPs pose higher risks; thus, the high prevalence of small-sized MPs in the Vaal River is of great concern. Moreover, it is also noted that the efficiency of water and wastewater treatment plants in removing MPs is determined, among others, by particle size (Hu et al. 2019; Krishnan et al. 2023; Leslie et al. 2017; Sun et al. 2020).
Fibers, the most abundant shape in this study, are reported to cause more harm to aquatic organisms than other MP shapes (Liang et al. 2023). For instance, they had higher mortality in amphipods (Hyalella azteca), stronger acute toxicity in grass shrimp (Palaemonetes pugio), and caused intestinal epithelial cell necrosis in zebrafish (Danio rerio) (Blarer and Burkhardt-Holm 2016; Gray and Weinstein 2017; Qiao et al. 2019). MPs were detected in all colon tissue samples, with an average of 331 MPs per individual, with fibers accounting for 96.1% of particles (Ibrahim et al. 2021; Schwabl et al. 2019). The high prevalence of fibrous MPs in the surface water of the Vaal River, therefore, alarms great potential health risks.
Lastly, the high abundance of coloured MPs in the Vaal is also worrying; colour may have significant implications on exposure and potential health risks of MPs. Several studies have reported that aquatic organisms may selectively prey on coloured MPs due to colour similarity to their natural prey (Saad et al. 2022a). The preferential uptake of MPs with specific colours by different aquatic biota is well documented (de Sá et al. 2015; Ory et al. 2018; Sequeira et al. 2020). The ingestion of coloured MPs by organisms is of great concern. In addition to their possible health risk, coloured MPs contain colourant agents such as pigments and dyes, which are toxic. The toxicity of coloured MPs comes from the plastic and the chemicals added to plastics during manufacturing (Onoja et al. 2022; Rochman 2013).
5 Environmental Consequences and Mitigation
Our findings demonstrate the prevalence of MPs in the Vaal River, which may affect a large population. The impacts are not limited to the population around the Vaal triangle but also to towns and farming communities downstream of the river. The Vaal River has considerable economic and social value; it is the largest tributary of the Orange River, which flows through five out of the nine provinces of South Africa. It provides water supply to industries, agricultural activities, breeding livestock, recreation, and drinking water ( (Iloms et al. 2020; Weideman et al. 2020). Further, the pollution profile concerning the physical and chemical properties of the detected MPs indicates serious threats and health risks. Small-sized, coloured, and fibrous MPs that were abundantly detected are easily transported and ingested by aquatic organisms, which put the aquatic species in the Vaal in real danger. The Vaal River is home to various species, e.g., yellowfish and carp fish, both widely consumed in South Africa. Notably, the impacts of MP pollution are not limited to the bio-physical elements of the environment. There are also implications for the social and economic systems of society. For example, the productivity, viability, profitability, and safety of the sectors that the Vaal River supports are highly vulnerable to plastic pollution.
The government of South Africa should pay enough attention to address MP pollution; the country is listed within the top 20 countries with the highest mass of mismanaged plastic waste (Verster et al. 2017; Verster and Bouwman 2020). Yet, the South African National Waste Management Strategy and the Annual Performance Plan (APP) for the Department of Environmental Affairs do not address MP or plastic pollution. Therefore, to contribute to the global initiative of addressing Sustainable Development Goals, the South African government is yet to recognise MP pollution within its environmental management frameworks. The United Nations has already requested countries to join the UN treaty, which addresses the urgent need to amplify efforts and catalyse comprehensive global efforts toward plastic pollution (Islam et al. 2023).
To effectively address and mitigate plastic and MP pollution in South Africa, collective efforts are needed. Authorities, the scientific community, and stakeholders, including communities, should all play their role. Effective waste management strategies need to be developed; these include source segregation and improved transportation to promote waste recycling, effective legislation for plastic and microplastic waste management, enforcement of policies to reduce illegal dumping, and supportive policies to reduce landfilling and promote recycling. It is also essential to educate the public about MP pollution and the associated health, environmental, social, and economic impacts of MPs. Awareness of MP pollution consequences is key to generating public interest that would lead to a longstanding behavioural change in which people use plastics. However, these efforts must be supported by legislation and economic instruments if a significant change is ultimately to be achieved (Nikiema et al. 2020b). Legislative interventions include aspects such as reduction of the usage of plastic through a tax on plastic bags and banning microplastic beads in cosmetics and personal care products; taxes on plastic bags have already been introduced in South Africa in 2003 (Dikgang et al. 2012). However, a lot of ground remains to be covered in formulating and enforcing guidelines on contaminant levels of MPs in aquatic and other environments.
6 Conclusions
This work was dedicated to investigating MP pollution in the Vaal River. The study was designed with a particular focus on morphological and chemical characteristics of MPs and factors driving distribution patterns. MP particles were prevalent in considerable concentrations, and the abundance of small-sized, fibrous, and coloured MPs is of additional concern due to the potential health risks associated with these properties. In addition to the three polymers (LDPE, HDPE, and PP), two pigments were also identified, which are believed to be colouring agents added to polymers during production.
Wastewater effluents and recreational activities are identified as primary potential sources of MP pollution in the Vaal River. The study also revealed that the proximity to urban centres and the presence of tributaries had significant input on MP abundance. Despite a lack of standardised sampling and analysis procedures, and the need for more monitoring studies in various environments to amass more data, the implications of MPs on biota, water quality, and human health are undeniable and detrimental. This study provided insights into understanding microplastic behaviour, fate, bioavailability, and toxicity concerning their physicochemical properties. It is worth mentioning that the characteristics reported and discussed in this study are not unique to the Vaal River or South Africa; such insights are beneficial to broaden our understanding of MP pollution as a global environmental concern.
Data Availability
Data are available on request.
References
Akdogan Z, Guven B, Kideys AE (2023) Microplastic distribution in the surface water and sediment of the Ergene River. Environ Res 234:116500. https://doi.org/10.1016/J.ENVRES.2023.116500
Amaral-Zettler LA, Zettler ER, Mincer TJ (2020) Ecology of the plastisphere. Nat Rev Microbiol 18(3):139–151. https://doi.org/10.1038/s41579-019-0308-0
Apetogbor K, Pereao O, Sparks C, Opeolu B (2023) Spatio-temporal distribution of microplastics in water and sediment samples of the Plankenburg River, Western Cape, South Africa. Environ Pollut 323. https://doi.org/10.1016/j.envpol.2023.121303
Aragaw TA (2020) Surgical face masks as a potential source for microplastic pollution in the COVID-19 scenario. Mar Pollut Bull 159:111517. https://doi.org/10.1016/j.marpolbul.2020.111517
Aragaw TA (2021) Microplastic pollution in African countries’ water systems: a review on findings, applied methods, characteristics, impacts, and managements. SN Appl Sci 3. https://pubmed.ncbi.nlm.nih.gov/34002166/
Beiras R, Schönemann AM (2020) Currently monitored microplastics pose negligible ecological risk to the global ocean. Sci Rep 10:22281. https://doi.org/10.1038/s41598-020-79304-z
Bin YuY, Choi JH, Choi CY et al (2023) Toxic effects of microplastic (polyethylene) exposure: Bioaccumulation, hematological parameters and antioxidant responses in crucian carp. Carassius Carassius Chemosphere 332:138801. https://doi.org/10.1016/J.CHEMOSPHERE.2023.138801
Blarer P, Burkhardt-Holm P (2016) Microplastics affect assimilation efficiency in the freshwater amphipod Gammarus fossarum. Environ Sci Pollut Res 23:23522–23532. https://doi.org/10.1007/S11356-016-7584-2/FIGURES/3
Bouwman H, Minnaar K, Bezuidenhout C, Verster C (2018) Microplastic in freshwater environments a scoping study report to the water research commission. Water Res Comission 85. https://www.wrc.org.za/wp-content/uploads/mdocs/2610-1-18.pdf
Caputo F, Vogel R, Savage J et al (2021) Measuring particle size distribution and mass concentration of nanoplastics and microplastics: addressing some analytical challenges in the sub-micron size range. J Colloid Interface Sci 588:401–417. https://doi.org/10.1016/j.jcis.2020.12.039
Chamas A, Moon H, Zheng J et al (2020) Degradation rates of plastics in the environment. ACS Sustain Chem Eng 8:3494–3511. https://doi.org/10.1021/ACSSUSCHEMENG.9B06635
Cheung PK, Fok L (2017) Characterisation of plastic microbeads in facial scrubs and their estimated emissions in Mainland China. Water Res 122:53–61. https://doi.org/10.1016/J.WATRES.2017.05.053
Cheung PK, Hung PL, Fok L (2019) River microplastic contamination and dynamics upon a rainfall event in Hong Kong, China. Environ Processes 6. https://doi.org/10.1007/s40710-018-0345-0
Coccato A, Jehlicka J, Moens L, Vandenabeele P (2015) Raman spectroscopy for the investigation of carbon-based black pigments. J Raman Spectrosc 46.https://doi.org/10.1002/jrs.4715
de Sá LC, Luís LG, Guilhermino L (2015) Effects of microplastics on juveniles of the common goby (Pomatoschistus microps): Confusion with prey, reduction of the predatory performance and efficiency, and possible influence of developmental conditions. Environ Pollut 196:359–362. https://doi.org/10.1016/J.ENVPOL.2014.10.026
Dikgang J, Leiman A, Visser M (2012) Analysis of the plastic-bag levy in South Africa. Resour Conserv Recycl 66:59–65. https://doi.org/10.1016/j.resconrec.2012.06.009
Dris R, Gasperi J, Rocher V et al (2018) Standard practice for preparation of water samples with high, medium, or low suspended solids for identification and quantification of microplastic particles and fibers using Raman Spectroscopy, IR Spectroscopy, or Pyrolysis-GC/MS. Environ Pollut 12:4. https://webstore.ansi.org/standards/astm/astmd833320
Fahrenfeld NL, Arbuckle-Keil G, Naderi Beni N, Bartelt-Hunt SL (2019) Source tracking microplastics in the freshwater environment. TrAC - Trends Anal Chem 112:248–254. https://www.sciencedirect.com/science/article/pii/S0165993618301584
Fred-Ahmadu OH, Bhagwat G, Oluyoye I et al (2020) Interaction of chemical contaminants with microplastics: principles and perspectives. Sci Total Environ 706:135978. https://doi.org/10.1016/J.SCITOTENV.2019.135978
Gad AK, Midway SR (2022) Relationship of microplastics to Body Size for Two Estuarine Fishes. Microplastics 1:211–220. https://doi.org/10.3390/microplastics1010014
Godoy V, Martín-Lara MA, Calero M, Blázquez G (2019) Physical-chemical characterization of microplastics present in some exfoliating products from Spain. Mar Pollut Bull 139:91–99. https://doi.org/10.1016/J.MARPOLBUL.2018.12.026
Gray AD, Weinstein JE (2017) Size- and shape-dependent effects of microplastic particles on adult daggerblade grass shrimp (Palaemonetes pugio). Environ Toxicol Chem 36:3074–3080. https://doi.org/10.1002/ETC.3881
Guo C, Wang L, Lang D et al (2023) UV and chemical aging alter the adsorption behavior of microplastics for tetracycline. Environ Pollut 318:120859. https://doi.org/10.1016/J.ENVPOL.2022.120859
Hartmann NB, Rist S, Bodin J et al (2017) Microplastics as vectors for environmental contaminants: Exploring sorption, desorption, and transfer to biota. Integr Environ Assess Manag 13:488–493. https://doi.org/10.1002/IEAM.1904
Hu Y, Gong M, Wang J, Bassi A (2019) Current research trends on microplastic pollution from wastewater systems: a critical review. Rev Environ Sci Biotechnol 18:207–230. https://link.springer.com/article/10.1007/s11157-019-09498-w
Hu J, Lim FY, Hu J (2023) Characteristics and behaviors of microplastics undergoing photoaging and Advanced Oxidation Processes (AOPs) initiated aging. Water Res 232:119628. https://doi.org/10.1016/J.WATRES.2023.119628
Huang JC (2002) Carbon black filled conducting polymers and polymer blends. Adv Polym Technol 21. https://onlinelibrary.wiley.com/doi/epdf/10.1002/adv.10025
Huang D, Li X, Ouyang Z et al (2021) The occurrence and abundance of microplastics in surface water and sediment of the West River downstream, in the South of China. Sci Total Environ 756:143857. https://doi.org/10.1016/J.SCITOTENV.2020.143857
Ibrahim M, He H (2017) Classification of polyethylene by Raman spectroscopy application note. https://www.gammadata.se/assets/Uploads/Vibrational-Compendium-r18-05-10.pdf#page=48
Ibrahim YS, Tuan Anuar S, Azmi AA et al (2021) Detection of microplastics in human colectomy specimens. JGH Open 5:116. https://doi.org/10.1002/jgh3.12457
Iloms E, Ololade OO, Ogola HJO, Selvarajan R (2020) Investigating industrial effluent impact on municipal wastewater treatment plant in Vaal, South Africa. Int J Environ Res Public Health 17:1096. https://doi.org/10.3390/ijerph17031096
Imbulana S, Tanaka S, Yukioka S, Oluwoye I (2024) Occurrence and distribution of plastic particles (10–25,000 μm) and microfibers in the surface water of an urban river network in Japan. Environ Monit Assess 196:1–19. https://doi.org/10.1007/S10661-023-12221-6/FIGURES/7
Islam MS, Lee Z, Shaleh A, Sen SH (2023) The United Nations Environment Assembly resolution to end plastic pollution: challenges to effective policy interventions. Environ Dev Sustain. https://doi.org/10.1007/s10668-023-03639-6
Jumeau R, Bourson P, Ferriol M et al (2013) Identification of LDPE Grades Focusing on Specific CH 2 Raman Vibration Modes. Int J Spectrosc 2013. https://doi.org/10.1155/2013/720598
Käppler A, Fischer D, Oberbeckmann S et al (2016) Analysis of environmental microplastics by vibrational microspectroscopy: FTIR, Raman or both? Anal Bioanal Chem 408:8377. https://doi.org/10.1007/s00216-016-9956-3
Kieu-Le TC, Thuong QT, Truong TNS et al (2023) Baseline concentration of microplastics in surface water and sediment of the northern branches of the Mekong River Delta. Vietnam Mar Pollut Bull 187:114605. https://doi.org/10.1016/J.MARPOLBUL.2023.114605
Koelmans B, Kalčíková G (2019) A scientific perspective on microplastics in nature and society. SAPEA. file:///Users/daliasaad/Downloads/report.pdf
Krishnan RY, Manikandan S, Subbaiya R et al (2023) Recent approaches and advanced wastewater treatment technologies for mitigating emerging microplastics contamination – a critical review. Sci Total Environ 858:159681. https://doi.org/10.1016/J.SCITOTENV.2022.159681
Kumar R, Sharma P, Manna C, Jain M (2021) Abundance, interaction, ingestion, ecological concerns, and mitigation policies of microplastic pollution in riverine ecosystem: a review. Sci Total Environ 782:146695. https://doi.org/10.1016/J.SCITOTENV.2021.146695
Kutralam-Muniasamy G, Shruti VC, Pérez-Guevara F, Roy PD (2022) Microplastic diagnostics in humans: “The 3Ps” Progress, problems, and prospects. https://doi.org/10.1016/j.scitotenv.2022.159164
Lahiri SK, Azimi Dijvejin Z, Golovin K (2023) Polydimethylsiloxane-coated textiles with minimized microplastic pollution. Nat Sustain 2023:1–9. https://doi.org/10.1038/s41893-022-01059-4
Lam TWL, Fok L, Lin L et al (2020) Spatial variation of floatable plastic debris and microplastics in the Pearl River Estuary, South China. Mar Pollut Bull 158. https://doi.org/10.1016/j.marpolbul.2020.111383
Lee JH, Kim JH (2023) Toxic effects of microplastic (Polyethylene) on fish: accumulation, hematological parameters and antioxidant responses in Korean Bullhead, Pseudobagrus fulvidraco. Sci Total Environ 877:162874. https://doi.org/10.1016/J.SCITOTENV.2023.162874
Leslie HA, Brandsma SH, van Velzen MJM, Vethaak AD (2017) Microplastics en route: field measurements in the Dutch river delta and Amsterdam canals, wastewater treatment plants, North Sea sediments and biota. Environ Int 101:133–142. https://doi.org/10.1016/j.envint.2017.01.018
Li J, Shan E, Zhao J et al (2023a) The factors influencing the vertical transport of microplastics in marine environment: A review. Sci Total Environ 870:161893. https://doi.org/10.1016/J.SCITOTENV.2023.161893
Li X, Chen Y, Zhang S (2023b) From marine to freshwater environment: a review of the ecotoxicological effects of microplastics. https://doi.org/10.1016/j.ecoenv.2023.114564
Liang W, Li B, Jong M-C (2023) Process-oriented impacts of microplastic fibers on behavior and histology of fish. https://doi.org/10.1016/j.jhazmat.2023.130856
Liu J, Gheorghe S, Stoica C et al (2024) Laboratory assessment for determining microplastics in freshwater systems—characterization and identification along the Somesul Mic River. Water 16:233. https://doi.org/10.3390/W16020233
Manbohi A, Mehdinia A, Rahnama R et al (2023) Distribution of microplastics in upstream and downstream surface waters of the Iranian Rivers discharging to the southern Caspian Sea. Environ Sci Pollut Res 30:92359. https://doi.org/10.1007/s11356-023-28898-8
Marine Debris Program N (2015) Laboratory methods for the analysis of microplastics in the marine environment: recommendations for quantifying synthetic particles in waters and sediments. file:///Users/daliasaad/Downloads/noaa_10296_DS1.pdf
Mason SA, Garneau D, Sutton R et al (2016) Microplastic pollution is widely detected in US municipal wastewater treatment plant effluent. Environ Pollut 218:1045–1054. https://doi.org/10.1016/j.envpol.2016.08.056
Murphy L, Germaine K, Kakouli-Duarte T, Cleary J (2022) Assessment of microplastics in Irish river sediment. Heliyon 8:e09853. https://doi.org/10.1016/J.HELIYON.2022.E09853
Naidoo T, Glassom D, Smit AJ (2015) Plastic pollution in five urban estuaries of KwaZulu-Natal, South Africa. Mar Pollut Bull 101. https://doi.org/10.1016/j.marpolbul.2015.09.044
Naidoo T, Thompson RC, Rajkaran A (2019) Quantification and characterisation of microplastics ingested by selected juvenile fish species associated with mangroves in KwaZulu-Natal, South Africa *. https://doi.org/10.1016/j.envpol.2019.113635
Nan B, Su L, Kellar C et al (2020) Identification of microplastics in surface water and Australian freshwater shrimp Paratya australiensis in Victoria. Australia Environmental Pollution 259:113865. https://doi.org/10.1016/J.ENVPOL.2019.113865
Nel HA, Froneman PW (2015) A quantitative analysis of microplastic pollution along the south-eastern coastline of South Africa. Mar Pollut Bull 101:274. https://doi.org/10.1016/j.marpolbul.2015.09.043
Nielsen AS, Batchelder DN, Pyrz R (2002) Estimation of crystallinity of isotactic polypropylene using Raman spectroscopy. Polymer (guildf) 43:2671–2676. https://doi.org/10.1016/S0032-3861(02)00053-8
Nikiema J, Asiedu Z, Mateo-Sagasta J et al (2020a) Catalogue of technologies to address the risks of contamination of water bodies with plastics and microplastics. United Nations Environment. file:///Users/daliasaad/Downloads/CTWM.pdf Programme
Nikiema J, Mateo-Sagasta, Asiedu Z et al (2020b) Water pollution by plastics and microplastics- a review of technical solutions from source to sea. United Nations Environment Programme. file:///Users/daliasaad/Downloads/WPMM.pdf
Nuelle MT, Dekiff JH, Remy D, Fries E (2014) A new analytical approach for monitoring microplastics in marine sediments. Environ Pollut 184:161–169. https://doi.org/10.1016/j.envpol.2013.07.027
Onoja S, Nel HA, Abdallah MAE, Harrad S (2022) Microplastics in freshwater sediments: analytical methods, temporal trends, and risk of associated organophosphate esters as exemplar plastics additives. Environ Res 203. https://pubmed.ncbi.nlm.nih.gov/34358506/
Ory NC, Gallardo C, Lenz M, Thiel M (2018) Capture, swallowing, and egestion of microplastics by a planktivorous juvenile fish. Environ Pollut 240:566–573. https://doi.org/10.1016/J.ENVPOL.2018.04.093
Park H, Park BS (2021) Review of microplastic distribution, toxicity, analysis methods, and removal technologies. Water (Switzerland) 13. https://www.mdpi.com/2073-4441/13/19/2736
Pivokonsky M, Cermakova L, Novotna K et al (2018) Occurrence of microplastics in raw and treated drinking water. Sci Total Environ 643:1644. https://doi.org/10.1016/j.scitotenv.2018.08.102
Qiao R, Deng Y, Zhang S et al (2019) Accumulation of different shapes of microplastics initiates intestinal injury and gut microbiota dysbiosis in the gut of zebrafish. Chemosphere 236:124334. https://doi.org/10.1016/J.CHEMOSPHERE.2019.07.065
Qiu Y, Zhou S, Qin W et al (2023) Effects of land use on the distribution of soil microplastics in the Lihe River watershed. China Chemosphere 324:138292. https://doi.org/10.1016/J.CHEMOSPHERE.2023.138292
Ramaremisa G, Ndlovu M, Saad D (2022) Comparative assessment of microplastics in surface waters and sediments of the Vaal River, South Africa: abundance, composition, and sources. Environ Toxicol Chem 41:3029–3040. https://doi.org/10.1002/ETC.5482
Rochman CM (2013) Plastics and priority pollutants: a multiple stressor in aquatic habitats. Environ Sci Technol 47:2439–2440. https://doi.org/10.1021/ES400748B
Rodrigues dos Santos C, Pinheiro Drumond G, Rezende Moreira V et al (2023) Microplastics in surface water: occurrence, ecological implications, quantification methods and remediation technologies. Chem Eng J 474:144936. https://doi.org/10.1016/J.CEJ.2023.144936
Saad D (2022) Selective removal of Cr(III) from aqueous solution using cross-linked polyethylenimine: experimental optimization and modeling. Polym Bull 79:1583. https://doi.org/10.1007/s00289-021-03578-0
Saad D (2023) Why microplastics are exceptional contaminants? In: Advances and Challenges in Microplastics, El-Sayed Salama (ed), IntechOpen. https://doi.org/10.5772/intechopen.109173
Saad D, Alamin H (2024) The first evidence of microplastic presence in the River Nile in Khartoum, Sudan: Using Nile Tilapia fish as a bio-indicator. Heliyon 10:e23393. https://doi.org/10.1016/J.HELIYON.2023.E23393
Saad D, Chauke P, Cukrowska E et al (2022a) First biomonitoring of microplastic pollution in the Vaal river using Carp fish (Cyprinus carpio) “as a bio-indicator.” Sci Total Environ 836:155623. https://doi.org/10.1016/J.SCITOTENV.2022.155623
Saad D, Ndlovu M, Ramaremisa G, Tutu H (2022b) Microplastics in freshwater environment: the first evaluation in sediment of the Vaal River. South Africa Heliyon 8:e11118. https://doi.org/10.1016/J.HELIYON.2022.E11118
Saad D, Ndlovu M, Ramaremisa G (2023) Characteristics of microplastics in sediment of the Vaal River, South Africa: implications on bioavailability and toxicity. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-023-05168-1
Saad D, Ramaremisa G, Ndlovu M et al (2024) Microplastic abundance and sources in surface water samples of the Vaal River, South Africa. Bull Environ Contam Toxicol 112:1–7. https://doi.org/10.1007/S00128-023-03845-Y/TABLES/1
Sadan Z, de Kock L (2020) Plastics: facts and futures moving beyond pollution management towards a circular plastics economy in South Africa. https://wwfafrica.awsassets.panda.org/downloads/wwf_plastics_report_final_2nov2020.pdf
Samandra S, Johnston JM, Jaeger JE (2021) Microplastic contamination of an unconfined groundwater aquifer in Victoria Australia. https://doi.org/10.1016/j.scitotenv.2021.149727
Schwabl P, Köppel S, Königshofer P et al (2019) Detection of various microplastics in human stool. Ann Intern Med 171. https://doi.org/10.7326/m19-0618
Sequeira IF, Prata JC, da Costa JP et al (2020) Worldwide contamination of fish with microplastics: a brief global overview. Mar Pollut Bull 160:111681. https://doi.org/10.1016/j.marpolbul.2020.111681
Shamskhany A, Li Z, Patel P, Karimpour S (2021) Evidence of microplastic size impact on mobility and transport in the marine environment: a review and synthesis of recent research. Front Mar Sci 8:1869. https://doi.org/10.3389/FMARS.2021.760649/BIBTEX
Shu X, Xu L, Yang M et al (2023) Spatial distribution characteristics and migration of microplastics in surface water, groundwater and sediment in karst areas: the case of Yulong River in Guilin, Southwest China. Sci Total Environ 868:161578. https://doi.org/10.1016/J.SCITOTENV.2023.161578
Su L, Xue Y, Li L et al (2016) Microplastics in Taihu Lake, China. Environ Pollut 216:711–719. https://doi.org/10.1016/j.envpol.2016.06.036
Summers E, Du J, Park K, Kaiser K (2023) How does buoyancy behavior impact microplastic transport in an estuarine environment? Sci Total Environ 899:165687. https://doi.org/10.1016/J.SCITOTENV.2023.165687
Sun Q, Ren SY, Ni HG (2020) Incidence of microplastics in personal care products: an appreciable part of plastic pollution. Sci Total Environ 742:140218. https://doi.org/10.1016/J.SCITOTENV.2020.140218
Tamminga M, Stoewer SC, Fischer EK (2019) On the representativeness of pump water samples versus manta sampling in microplastic analysis. Environ Pollut 254:112970. https://doi.org/10.1016/j.envpol.2019.112970
Tekman MB, Wekerle C, Lorenz C et al (2020) Tying up loose ends of microplastic pollution in the arctic: Distribution from the Sea Surface through the Water Column to Deep-Sea Sediments at the HAUSGARTEN Observatory. Environ Sci Technol 54:4079–4090. https://doi.org/10.1021/acs.est.9b06981
Tomasini EP, Halac EB, Reinoso M et al (2012) Micro-Raman spectroscopy of carbon-based black pigments. J Raman Spectrosc. https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/jrs.4159
Tran-Nguyen QA, Le TM, Nguyen HNY et al (2024) Microplastics in the surface water of urban lakes in central Vietnam: pollution level, characteristics, and ecological risk assessment. Case Studies in Chemical and Environmental Engineering 9:100622. https://doi.org/10.1016/J.CSCEE.2024.100622
Verster C, Bouwman H (2020) Land-based sources and pathways of marine plastics in a South African context. S Afr J Sci 116. http://www.scielo.org.za/pdf/sajs/v116n5-6/02.pdf
Verster C, Minnaar K, Bouwman H (2017) Marine and freshwater microplastic research in South Africa. Integr Environ Assess Manag 13:533–535. https://pubmed.ncbi.nlm.nih.gov/28440929/
Vilakati B, Sivasankar V, Mamba BB et al (2020) Characterization of plastic micro particles in the Atlantic Ocean seashore of Cape Town, South Africa and mass spectrometry analysis of pyrolyzate products. Environ Pollut 265:114859. https://doi.org/10.1016/j.envpol.2020.114859
Vilakati B, Sivasankar V, Nyoni H et al (2021) The Py – GC-TOF-MS analysis and characterization of microplastics (MPs) in a wastewater treatment plant in Gauteng Province, South Africa. Ecotoxicol Environ Saf 222. https://doi.org/10.1016/j.ecoenv.2021.112478
Vilakati B, Venkataraman S, Nyoni H et al (2022) Qualitative characterisation and identification of microplastics in a freshwater dam at Gauteng Province, South Africa, using pyrolysis–gas chromatography–time of flight–mass spectrometry (Py–GC–ToF–MS). Environ Sci Pollut Res 29:83452. https://doi.org/10.1007/s11356-022-21510-5
Weideman EA, Perold V, Ryan PG (2020) Limited long-distance transport of plastic pollution by the Orange-Vaal River system, South Africa. Sci Total Environ 727. https://doi.org/10.1016/j.scitotenv.2020.138653
Wepener V, van Dyk C, Bervoets L et al (2011) An assessment of the influence of multiple stressors on the Vaal River, South Africa. Phys Chem Earth 36:949–962. https://doi.org/10.1016/j.pce.2011.07.075
Yakes BJ, Michael TJ, Perez-Gonzalez M, Harp BP (2017) Investigation of tattoo pigments by Raman spectroscopy. J Raman Spectrosc 48. https://doi.org/10.1002/jrs.5095
Yang J, Monnot M, Sun Y et al (2023) Microplastics in different water samples (seawater, freshwater, and wastewater): Removal efficiency of membrane treatment processes. Water Res 232:119673. https://doi.org/10.1016/j.watres.2023.119673
Yin Z (2022) The sustainable management of microplastics pollution in sediments from China: promulgating relevant laws and implementing targeted management. https://doi.org/10.1016/j.scitotenv.2022.159262
Zhang J, Wang L, Kannan K (2021) Quantitative analysis of polyethylene terephthalate and polycarbonate microplastics in sediment collected from South Korea, Japan and the USA. Chemosphere 279. https://doi.org/10.1016/j.chemosphere.2021.130551
Zhang X, Chen Y, Li X et al (2022) Size/shape-dependent migration of microplastics in agricultural soil under simulative and natural rainfall. Sci Total Environ 815:152507. https://doi.org/10.1016/J.SCITOTENV.2021.152507
Zhao W, Li J, Liu M et al (2024) Seasonal variations of microplastics in surface water and sediment in an inland river drinking water source in southern China. Sci Total Environ 908:168241. https://doi.org/10.1016/J.SCITOTENV.2023.168241
Zhong Y, Ding Q, Huang Z et al (2023) Influence of ultraviolet-aging and adsorbed pollutants on toxicological effects of polyvinyl chloride microplastics to zebrafish. Environ Pollut 316:120617. https://doi.org/10.1016/J.ENVPOL.2022.120617
Zhou Y, Jing J, Yu R et al (2023) Microplastics in plateau agricultural areas: spatial changes reveal their source and distribution characteristics. Environ Pollut 319:121006. https://doi.org/10.1016/J.ENVPOL.2023.121006
Acknowledgements
The authors would like to thank Prof Rudolph Erasmus for the Raman Spectroscopy analysis.
Funding
Open access funding provided by University of the Witwatersrand. This project was supported by the Royal Society, UK [grant number: FLR\R1\201062].
Author information
Authors and Affiliations
Contributions
Dalia Saad: Conceptualization, Investigation, Supervision, Writing-original draft, Funding acquisition, Resources, and Project Administration; Gibbon Ramaremisa: Methodology, Investigation, Data Collection, Formal Analysis, Writing; Michelle Ndlovu: Methodology; Luke Chimuka: Supervision, Review and Editing.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saad, D., Ramaremisa, G., Ndlovu, M. et al. Morphological and Chemical Characteristics of Microplastics in Surface Water of the Vaal River, South Africa. Environ. Process. 11, 16 (2024). https://doi.org/10.1007/s40710-024-00693-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40710-024-00693-8