Abstract
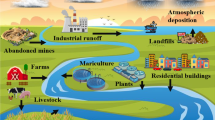
Chitosan, a well-known biopolymer, has been impregnated with tetrabutylammonium bromide using the reflux method. The synthesized adsorbent, tetrabutylammonium impregnated chitosan(C-TBA) was subjected to intense characterization using various spectral techniques which confirms impregnation. The pH point of zero charge was found to be 8.2 which indicate the suitability of the material for both anionic and cationic dyes. C-TBA was employed as adsorbent for the removal of two potent carcinogenic dyes, namely, Congo red (CR) and Crystal violet (CV). Batch experiments were carried out to study the effects of pH of dye solution, contact time, adsorbent dose, adsorbate concentration and temperature. A number of adsorption isotherm models namely, Langmuir, Freundlich, R-P, D-R, Temkin, and Harkin-Jura were studied. The best fit isotherm model was judged by plotting the non-linear plots. Maximum monolayer adsorption capacity of C-TBA towards adsorption of CR was found to be 12.66 mg g−1 and for CV 0.86 mg g−1. Adsorption kinetics was well fitted to pseudo-second-order model and thermodynamics show that the process of adsorption was spontaneous. Statistical analysis as well as column adsorption studies was also carried out.
Article Highlights
-
Impregnated chitosan for removal of carcinogenic dyes from wastewater.
-
Use of non-linear plots for identification of best-fit adsorption isotherm model.
-
Use of R2 and χ2 tests for analyzing statistical data.







Similar content being viewed by others
Availability of Data
All the necessary data has been included in the manuscript and supplementary files.
References
Alkizwini RS (2021) The Use of an Organo-Kaolinite Sorbent in a Permeable Reactive Barrier for Remediating Groundwater Contaminated with Methylene Blue Dye: Experimental and Theoretical Investigation. Environ Process 8:889–910. https://doi.org/10.1007/s40710-021-00515-1
Ardila-Leal LD, Poutou-Piñales RA, Pedroza-Rodríguez AM, Quevedo-Hidalgo BE (2021) A brief history of colour, the environmental impact of synthetic dyes and removal by using laccases. Molecules 26:3813. https://doi.org/10.3390/molecules26133813
Auta M, Hameed BH (2014) Chitosan-clay composite as highly effective and low-cost adsorbent for batch and fixed-bed adsorption of methylene blue. J Chem Eng 237:352–361. https://doi.org/10.1016/j.cej.2013.09.066
Balarak D, Zafariyan M, Igwegbe CA (2021) Adsorption of Acid Blue 92 Dye from Aqueous Solutions by Single-Walled Carbon Nanotubes: Isothermal, Kinetic, and Thermodynamic Studies. Environ Process 8:869–888. https://doi.org/10.1007/s40710-021-00505-3
Belala Z, Jeguirim M, Belhachemi M, Addoun F, Trouvé G (2011) Biosorption of copper from aqueous solutions by date stones and palm-trees waste. Environ Chem Lett 9:65–69. https://doi.org/10.1007/s10311-009-0247-5
Bharathi KS, Ramesh ST (2013) Removal of dyes using agricultural waste as low-cost adsorbents: a review. J Appl Water Sci 3:773–790. https://doi.org/10.1007/s13201-013-0117-y
Chiou MS, Li HY (2003) Adsorption behaviour of reactive dye in aqueous solution on chemical cross-linked chitosan beads. Chemosphere 50:1095–1105. https://doi.org/10.1016/S0045-6535(02)00636-7
Elgarahy AM, Elwakeel KZ, Elshoubaky GA, Mohammad SH (2019) Untapped sepia shell–based composite for the sorption of cationic and anionic dyes. Water AirSoil Pollut 230:217. https://doi.org/10.1007/s11270-019-4247-1
Elwakeel KZ, Elgarahy AM, Elshoubaky GA, Mohammad SH (2019a) Microwave assist sorption of crystal violet and congo red dyes onto amphoteric sorbent based on upcycled Sepia shells. J Environ Health Science Engineering. https://doi.org/10.1007/s40201-019-00435-1
Elwakeel KZ, Shahat A, Khan ZA, Alshitari W, Guibal E (2019b) Magnetic metal oxide organic framework material for ultrasonic-assisted sorption of Titan Yellow and Rose Bengal from aqueous solutions. Chem Eng J. https://doi.org/10.1016/j.cej.2019b.123635
Elwakeel KZ, El-Bindary AA, El-Sonbati AZ, Hawas AR (2017) Magnetic alginate beads with high basic dye removal potential and excellent regeneration ability. Can J Chem 95:807–815. https://doi.org/10.1139/cjc-2016-0641
Elwakeel KZ, El-Bindary AA, Ismail A, Morshidy AM (2016a) Sorptive removal of Remazol Brilliant Blue R from aqueous solution by diethylenetriamine functionalized magnetic macro-reticular hybrid material. RSC Adv 6:22395–22410. https://doi.org/10.1039/c5ra26508h
Elwakeel KZ, El-Bindary AA, Ismail A, Morshidy AM (2016b) Magnetic chitosan grafted with polymerized thiourea for remazol brilliant blue r recovery: effects of uptake conditions. J Dispersion Science Technology. https://doi.org/10.1080/01932691.2016.1216436
Elwakeel KZ, Elgarahy AM, Mohammad SH (2016c) Use of beach bivalve shells located at Port Said coast (Egypt) as a green approach for methylene blue removal. J Environ Chem Eng 5:578–587. https://doi.org/10.1016/j.jece.2016.12.032
Elwakeel KZ (2009) Removal of Reactive Black 5 from aqueous solutions using magnetic chitosan resins. J Hazard Mater 167:383–392. https://doi.org/10.1016/j.jhazmat.2009.01.051
El Nemr A, Shoaib AGM, El Sikaily A (2021) Evaluation of Cationic Methylene Blue Dye Removal by High Surface Area Mesoporous Activated Carbon Derived from Ulva lactuca. Environ Process 8:311–332. https://doi.org/10.1007/s40710-020-00487-8
Farias R, Buarque H, Cruz M, Cardoso L, Gondim T, Paula V (2018) Adsorption of Congo red dye from aqueous solution onto amino-functionalized silica gel. Eng Sanit Ambient 23:1053–1060. https://doi.org/10.1590/S1413-41522018172982
Gao Q, Zhu H, Luo WJ, Wang S, Zhou CG (2014) Preparation, characterization, and adsorption evaluation of chitosan-functionalized mesoporous composites. Microporous Mesoporous Mater 193:15–26. https://doi.org/10.1016/j.micromeso.2014.02.025
Guo J, Chen S, Liu L, Li B, Yang P, Zhang L, Feng Y (2012) Adsorption of dye from wastewater using chitosan-ctab modified bentonites. J Colloid Interface Sci 382:61–66. https://doi.org/10.1016/j.jcis.2012.05.044
Hassan N, Shahat A, El-Didamony A, El-Desouky MG, El-Bindary AA (2020) Synthesis and characterization of ZnO nanoparticles via zeolitic imidazolate framework-8 and its application for removal of dyes. Journal of Molecular Structure
Ho YS, Chiu WT, Wang CC (2004) Regression analysis for the sorption isotherms of basic dyes on sugarcane dust. Bioresour Technol 96:1285–1291. https://doi.org/10.1016/j.biortech.2004.10.021
Igwegbe CA, Onukwuli OD, Ighalo JO (2020) Adsorption of Cationic Dyes on Dacryodes edulis Seeds Activated Carbon Modified Using Phosphoric Acid and Sodium Chloride. Environ Process 7:1151–1171. https://doi.org/10.1007/s40710-020-00467-y
Jabbar A, Hadi A, Sami F (2014) Removal of Azo dye from aqueous solutions using chitosan. Orient J Chem 30:571–575. https://doi.org/10.13005/ojc/300222
Jeyaseelan C, Chaudhary N, Jugade R (2018) Sulphate-crosslinked chitosan as an adsorbent for the removal of Congo red dye from aqueous solution. Air Soil and Water Research (ASW) 11:1–8. https://doi.org/10.1177/1178622118811680
Kahu S (2016) Logical approaches in the removal of toxic elements from wastewater using structurally modified biopolymers. Unpublished PhD thesis, R.T.M. Nagpur University, Nagpur, Maharashtra, India
Kahu S, Shekhawat A, Saravanan D, Jugade R (2017) Stannic chloride impregnated chitosan for defluoridation of water. Int J Biol Macromol 104:1528–1538. https://doi.org/10.1016/j.jbiomac.2017.02.101
Karaoǧlu MH, Doǧan M, Alkan M (2010) Removal of reactive blue 221 by kaolinite from aqueous solutions Indus. Eng Chem Res 49:1534–1540. https://doi.org/10.1021/ie9017258
Kiwaan HA, Atwee TM, Azab EA, El-Bindary AA (2019) Efficient photocatalytic degradation of Acid Red 57 using synthesized ZnO nanowires. J Chin Chem Soc 66:89–98. https://doi.org/10.1002/jccs.201800092
Khattab TA, Abdelrahman MS, Rehan M (2020) Textile dyeing industry: environmental impacts and remediation. Environ Sci Pollut Res 27:3803–3818
Khoshnamvand N, Jafari A, Kamarehie B (2019) Removal of Malachite Green Dye from Aqueous Solutions Using Zeolitic Imidazole Framework-8. Environ Process 6:757–772. https://doi.org/10.1007/s40710-019-00384-9
Kumaria HJ, Krishnamoorthy P, Arumugam TK, Radhakrishnan S, Vasudevan D (2012) An efficient removal of crystal violet dye from waste water by adsorption onto TLAC/Chitosan composite: A novel low cost adsorbent. Int J Biol Macromol 96:324–333. https://doi.org/10.1016/j.ijbiomac.2016.11.077
Kyzas GZ, Bikiaris DN (2015) Recent modifications of chitosan for adsorption applications: a critical and systematic review. Mar Drugs 13:312–337. https://doi.org/10.3390/md13010312
Ma JW, Wang H, Wang FY, Huang ZH (2010) Adsorption of 2, 4-dichlorophenol from aqueous solution by a new low-cost adsorbent—activated bamboo charcoal. Sep Sci Technol 45:2329–2336. https://doi.org/10.1080/01496395.2010.504482
Mohamed NA, Al-Harby NF, Almarshed MS (2019) Enhancement of adsorption of Congo red dye onto novel antimicrobial trimellitic anhydride isothiocyanate-cross-linked chitosan hydrogels. Polym Bull. https://doi.org/10.1007/s00289-019-03058-6
Mondal NK, Kar S (2018) Potentiality of banana peel for removal of Congo red dye from aqueous solution: isotherm, kinetics and thermodynamics studies. Appl Water Sci 8:157–168. https://doi.org/10.1007/s13201-018-0811-x
Ozturk G, Silah H (2020) Adsorptive Removal of Remazol Brilliant Blue R from Water by Using a Macroporous Polystyrene Resin: Isotherm and Kinetic Studies. Environ Process 7:479–492. https://doi.org/10.1007/s40710-020-00429-4
Saha TK, Bhoumik NC, Karmakar S, Ahmed MG, Ichikawa H, Fukumori Y (2010) Adsorption of methyl orange onto chitosan from aqueous solution. J Water Resour Protect 2:898–906. https://doi.org/10.4236/jwarp.2010.210107
Saini RD (2017) Textile organic dyes: polluting effects and elimination methods from textile waste water. Int J Chem Eng Res 9:975
Shouman M, Khedr S, Attia A (2012) Basic dye adsorption on low cost biopolymer: kinetic and equilibrium studies. IOSR J Appl Chem 2:27–36. https://www.iosrjournals.org
Sumanjit, Mahajan RK, Rani S, Walia TPS, Kaur R (2010) Adsorptive removal of five acid dyes using various unconventional adsorbents. J Surf Sci Technol 26:77–93. https://www.researchgate.net/publication/287934567
Travlou NA, Kyzas GZ, Lazaridis NK, Deliyanni EA (2013) Functionalization of graphite oxide with magnetic chitosan for the preparation of a nanocomposite dye adsorbent. Langmuir 29:1657–1668. https://doi.org/10.1021/la304696y
Vijayalakshmi P, Bala VSS, Thiruvengadaravi KS, Panneerselvam P, Palanichamy M, Sivanesan S (2010) Removal of acid violet 17 from aqueous solutions by adsorption onto activated carbon prepared from pistachio nut shell. Sep Sci Technol 46:155–163. https://doi.org/10.1080/01496395.2010.484006
Vital RK, Saibaba KVN, Shaik KB (2016) Dye removal by adsorption: a review. J Bioremediat Biodegrad 7:371. https://doi.org/10.4172/2155-6199.1000371
Wanassi B, Hariz IB, Ghimbeu CM et al (2017a) Carbonaceous adsorbents derived from textile cotton waste for the removal of Alizarin S dye from aqueous effluent: kinetic and equilibrium studies. Environ Sci Pollut Res 24:10041–10055. https://doi.org/10.1007/s11356-017-8410-1
Wanassi B, Hariz IB, Ghimbeu CM, Vaulot C, Jeguirim M (2017b) Green carbon composite-derived polymer resin and waste cotton fibers for the removal of alizarin red s dye. Energies 10(9):1321. https://doi.org/10.3390/en10091321
Yan Y, Xiang B, Li Y, Jia Q (2013) Preparation and adsorption properties of diethylenetriamine-modified chitosan beads for acid dyes J. Appl Polym Sci 130:4090–4098. https://doi.org/10.1002/app.39691
Acknowledgements
The authors are thankful to R.T.M. Nagpur University and P.G.T.D. Chemistry for providing all the necessary facilities required for the research work. Special thanks to Dr. S. Kahu and Dr. A. Shekhawat for their help in calculations and proof reading.
Funding
This work was supported by the Department of Science and Technology (DST), Ministry of Education, India, under the DST INSPIRE Fellowship programme [sanction order no. DST/INSPIRE Fellowship 2017/IF170496 dated October 21, 2020].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Maithili A. Khapre. The first draft of the manuscript was written by Ravin M. Jugade. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khapre, M.A., Jugade, R.M. Quaternary Ammonium Impregnated Chitosan for the Decontamination of Wastewater from Carcinogenic Dyes. Environ. Process. 9, 24 (2022). https://doi.org/10.1007/s40710-022-00565-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40710-022-00565-z