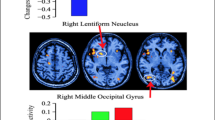
Abstract
Purpose of Review
Recent studies have linked fibromyalgia (nociplastic pain) and depression to neuroinflammation. Restless legs syndrome (RLS) is highly prevalent in CWP (chronic widespread pain) and FMS (fibromyalgia syndrome) as well as in depression. This review focuses on evidence in the relationship between CWP and RLS and possible common pathophysiological mechanisms.
Recent Findings
CWP is chronic pain spread over various locations of the body and is a cardinal sign of fibromyalgia. Poor sleep quality is a common symptom in fibromyalgia as well as high levels of fatigue, poor cognition, and other associated features which include depression, headache, and abdominal pain. Evidence for a bilateral relationship between pain and sleep deficit is robust. The latest research focus is not only on insomnia symptoms in chronic pain but also on other types of sleep disorders such as RLS which is characterized by complaints of an “urge to move” frequently associated with dysesthesias. These sensations can also have painful characteristics. Thus, there is a possible overlap between these two entities. The high prevalence of RLS (33–54%) in CWP has been shown in several studies. The pathophysiological mechanisms behind CWP and RLS are still not completely known, but there is a rising amount of evidence on proinflammatory and neuroinflammatory processes in CWP. In RLS, there are links to dopaminergic dysfunction and more generally to monoaminergic dysfunctional circuits, BID (brain iron deficiency), altered glutamatergic neurotransmission, and genetic traits. Depression is a common comorbidity as well in CWP as RLS, and proinflammatory mechanisms have also been demonstrated in this condition.
Summary
The association between CWP and RLS is shown to be high and the pathogenesis of both conditions is still not completely understood. Recent research is focusing on proinflammatory and neuroinflammatory processes, not uniquely emerging in fibromyalgia/CWP, but inflammatory features have been found in depression as well as in sleep deficit. Proinflammatory/neuroinflammatory processes may be an underlying, common factor in both CWP and RLS that future research should investigate further.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chronic Pain and CWP
Prevalence
Chronic pain is a common condition, and globally, it has been estimated that 1 in 5 adults suffer from pain and that another 1 in 10 adults are diagnosed with chronic pain (CP) each year [1]. A multicenter study carried out in 1998 by the World Health Organization (WHO) has shown a prevalence of CP in 22% of the world population [2] although the prevalence is higher in some studies [3]. Patients with chronic neck pain and chronic low back pain show a high prevalence of sleep deprivation (42.2%), even when analgesics had been taken. About 19.8% of the patients reported serious sleep impairment (i.e., < 4 h of sleep per night) [4]. The prevalence of sleep disturbances is even higher in chronic widespread pain (CWP) and in fibromyalgia (FM), and the prevalence of sleep complaints reaches 99% [5].
Pathogenesis
Both central sensitization and dysfunctional descending pain inhibition are recognized as pathological mechanisms in CWP, FM, and chronic pain with spreading of pain localizations, i.e., nociplastic pain. The relationship between pain and sleep is shown to be bidirectional. Painful conditions enhance or cause a cortical hyperalert state which compromises sleep architecture, and the risk of development of chronic pain rises with preexisting sleep disorders. One of the most common sleep disorders in chronic pain, and especially in CWP, is restless legs syndrome (RLS) and PLM (periodic limb movements), leading to sleep deprivation or fragmentation.
Restless Legs Syndrome
Prevalence
RLS or Willis-Ekbom disease (WED) is a common neurological sensorimotor condition often related to sleep disturbances, most frequently reported in the legs or arms. RLS creates a substantial negative impact on sleep, quality of life, and general health [6]. The prevalence of RLS/WED symptoms is about 2.4–10% in western countries [7,8,9,10] but less frequent in East Asia and sub-Saharan Africa [11,12,13]. Although RLS is a common condition, it is still under-diagnosed and under-treated at present. Despite 81% of patients with RLS having reported their complaints to their primary care physician, only 6% were given a precise diagnosis of RLS/WED [14], according to Allen et al. in the study dated 2008. RLS sensations are extremely variable and 24% of patients report painful symptoms [15]. The diagnostic criteria are described in Table 1.
Pathogenesis
The pathogenesis of RLS is incompletely elucidated, but there is robust evidence on BID (brain iron deficiency) and to date, links have been made to dopaminergic dysfunction, hypo-adenosinergic state, genetic traits, inflammatory and immunological mechanisms, altered hypocretin/orexin or endorphin activity, local hypoxic and vascular mechanisms, and increased thalamic glutamate/glutamine [16,17,18].
New evidence has been found of structural changes in the brain’s somatosensory cortex in RLS, and symptoms may be possibly related to the pathological changes in this area of the brain although cause versus effect cannot be proven. Subjects with RLS included in one study had a 7.5% decrease in the average thickness of brain tissue in the area of the brain that processes sensations compared to healthy participants [19].
The symptoms of RLS were first described by Willis (1685) and then published by Ekbom (1960). Despite being introduced hundreds of years ago, this is still a poorly recognized disorder despite effects in patients with moderate to severe symptoms on quality of life and difficulty in their daily life including their jobs and social activities due to impaired sleep and sleep deficit. In a recent meta-analysis, the conclusion is that the prevalence of hypertension and possibly cardiovascular disease in patients with RLS is higher but the results remain to be confirmed in large prospective studies [20]. RLS seems also to relate to a higher risk of suicide in depressed patients and according to some new studies, there seems to be a significant relationship between RLS and multiple medical problems, especially cardiovascular disease as above [21, 22].
Painful RLS
The majority of RLS patients (80%) report symptoms other than pain such as crawling or creeping or discomfort in the legs or arms and an irresistible urge to move the limbs. However, a small subgroup of approximately 10–20% of RLS patients report painful RLS symptoms [23].
There is a high prevalence of painful RLS in neuropathy. RLS and neuropathy are two distinct neurological conditions sharing common features which include damage to or impairment of the nerves in the legs or feet caused by, for instance, diabetes mellitus, radiculopathy, and other forms of polyneuropathy. Some studies suggest that central nervous system pain processing may be amplified in RLS, perhaps partially as a consequence of sleep disruption [24]. RLS patients, even those whose symptoms are managed pharmacologically, may be at elevated long-term risk for the development or maintenance of persistent pain conditions.
RLS seems to be a common comorbidity in FMS according to multiple studies, and the prevalence varies between 33% and 64% according to recent studies [25,26,27,28]. Sleep disruption caused by RLS plausibly exacerbates the symptoms of FMS in a bidirectional manner and impairs the quality of life.
Sleep Disturbances, RLS, and Widespread Pain
There are robust and consistent findings on a negative influence of pain and RLS on sleep quality and daytime function. A recent study showed that multi-site pain reduces and fragments sleep in a dose-dependent manner [26]. As is well known, sleep is even affected by mild to moderate symptom frequency of RLS, and conversely, sleep is known to interact with RLS and pain. A recently published meta-analysis on chronic pain patients and sleep disturbance showed compelling co-prevalence rates. Overall, 44% of individuals with CP had some type of diagnosed sleep disorder. Both RLS and obstructive sleep apnea (OSA) are 16 times more prevalent in chronic pain patients compared to healthy peers. The individuals with CP were thirteen times more likely to be diagnosed with insomnia than their healthy peers [29]. Sleep fragmentation seems to affect pain processing and may contribute to the experience of reduced pain tolerance [30], and sleep deprivation induces hyperalgesia to experimental superficial and deep-muscle pain [31]. An important impact of sleep deficit on pain was found in the Norwegian HUNT study showing that sleep problems were dose-dependently associated with an increased risk of pain in the lower back and the neck/shoulder regions of both women and men. Those subjects reporting sleep problems “sometimes” or “always” had a 23–32% resp. 51–66% higher risk of CP [32]. Recent studies have revealed a complex and bidirectional relationship between pain and sleep [33,34,35], but there is a lack of large studies on sleep management and treatment of sleep disorders that may reduce pain intensity and eventually improve chronic pain syndromes [36, 37].
Sleep disturbance and short sleep duration are associated with increased production of IL-6, TNF-α, IL-1β, and related disorders including cardiovascular disease, arthritis, diabetes mellitus, and certain cancers [38]. However, very large cohorts are needed to better understand the possible relationship regarding inflammation and sleep duration in the general population [39].
Both periodic limb movement syndrome (PLMS) and RLS cause sleep disturbance and fragmentation [40] and are often present in pain syndromes, especially in CWP and FM. Edwards et al. showed that pain processing may be amplified in RLS, perhaps in part as a consequence of sleep disruption, sleep deprivation [24], and enhanced proinflammatory cytokines in the brain.
Altered HPA Axis and Sympathetic Activity in CWP and RLS
A large body of evidence is dedicated to elucidating the relationship between pain and stress. Numbers of studies demonstrate dysregulation of the hypothalamic pituitary adrenal (HPA) axis in CP and CWP, both hyper- and hypocortisolemia [38, 39, 41,42,43]. An ACTH hyper-responsiveness and a relative adrenal hypo-responsiveness were shown in studies on HPA axis dysfunction in FMS patients, indicating a tertiary (hypothalamic or supra-hypothalamic level) of hypoadrenalism [44]. Stress seems to increase the risk of FMS. The indication of stress response dysfunction is discussed in a review by Crofford and colleagues [45]. The normal circadian rhythm for plasma cortisol level is disrupted in FMS patients and both stress systems, HPA axis and sympathetic-adreno-medullary axis (SAM), are demonstrated to be altered [45]. Concentrations of corticotropin-releasing hormone (CRH) in the CSF [46] were elevated in patients with FMS and were associated with pain but not fatigue symptoms [47]. In a cross-sectional study by Stehlik et al., an almost 25% increase in the morning cortisol in CWP subjects was found. The result was consistent even after assessing confounders. Elevated fasting blood glucose in addition to HPA axis alteration also supports the hypothesis of an activated SAM system. The data suggest altered insulin sensitivity, a finding observed both in chronic stress [48] and in disturbed sleep [49]. Research on the HPA axis function in RLS patients is limited. A nocturnal activation of the HPA system was demonstrated in a study by Schiling C et al. [50]. When cortisol and fasting glucose were measured in the morning [51], there was however no evidence showing any effect of RLS. The possibility to measure sympathetic activity by a novel method of pulse wave amplitude (PWA) in assessment of overnight skin vascular reactivity was used in the Stehlik et al. study [52]. PWA is a technique that allows the accurate recording of peripheral pressure waveforms and generation of the corresponding central waveform from which the augmentation index and central pressure can be derived and measures finger vascular tone. Vascular tone is largely affected by skin sympathetic nerve activity via modulation of vascular alpha-receptors [53]. PWA is used both in the diagnosis of sleep, arousal from sleep, and sleep-disordered breathing. In the current context, PWA analysis mirrored overnight sympathetic activity and vascular function. The increased event index of strong vasoconstriction in the finger (PWA 50% events) was an interesting finding observed in CWP women and suggests an elevated sympathetic vascular tone during sleep. A study by Zou et al. demonstrated attenuation of PWA in response to obstructive breathing events during sleep and was reversed by the α-receptor antagonist phentolamine and doxazosin (pharmacological blockade of vascular alpha-receptors) [53, 54]. In the Stehlik et al. study, controls had a higher PWA event index in the lower attenuation range (PWA 10–50%) and an increase in small PWA attenuations in the range of 10–30 which in the literature is associated with better cardiovascular function and lower cardiovascular risk [55]. OSA affects sympathovagal modulation through complex mechanisms and interestingly, women with CWP showed significantly increased time in flow limitation indicating a mild degree of obstructive sleep-disordered breathing [51, 56, 57].
In summary, the data from several studies show an activation of the HPA axis and the SAM system in women with CWP. Sleep disturbances may contribute to the activation of the stress system. HPA hyperactivity and the inflammatory response system have been suggested to underlie depressive disorders and, similarly, chronic pain conditions. Conversely, anti-depressants have been shown to attenuate HPA axis activity and to normalize HPA dysfunction [58].
Psychiatric Comorbidity in CWP and RLS
The coexistence of chronic pain and anxiety and depression is well known and well described and further aggravates the severity of both disorders [59]. The relationship between pain, anxiety, and depression results not only from psychological but also from biological factors and pain shares some biological mechanisms with anxiety and depression. Psychiatric disease contributes not only to pain intensity but also to an increased risk of disability [60]. Research on depressive symptoms in chronic pain syndromes and comorbid RLS is very limited. Some research, however, shows higher prevalence of RLS in women with depressive symptoms [51] whereas anxiety symptoms were more strongly linked to CWP subjects. Comparable findings have been reported in other studies both for FMS [61] and for RLS [62]. Depression is more common as RLS severity worsens. The poor sleep in those with RLS and PLMS could explain the relationship between depressive symptoms and RLS. Sleep deficit is linked to a two times risk of developing depression and in two studies, psychiatric comorbidity was independently associated with disturbed sleep, increased daytime sleepiness, and body fatigue independent of pain and RLS [57, 63]. Depression seems to alter the brain’s response to pain, and possibly, the occurrence of RLS symptoms in depression has an inhibitory influence on sleep. Indeed, sleep deficit, depression, and chronic pain are known to reciprocally influence each other in a vicious circle of cause and effect. In Irwin et al.’s study on patients with rheumatoid arthritis, disturbed mood and pain ratings were increased after sleep loss when compared with controls [64]. Chiu et al. [65] investigated the role of psychological factors and sleep deficit and found that depressive symptoms and poor sleep were independently associated with a reduced pain threshold. Even after adjustment for confounders in the multivariate analysis, the results remained consistent. Non-restorative sleep seems to be the strongest link to the spreading of pain, and sleep problems are associated with depressive and anxiety symptoms [66, 67]. Insomnia is a significant factor for the prevalence of CWP, anxiety, and a range of both mental and physical conditions according to findings in the HUNT study [68]. Both depression and sleep deficit lead to an increase of proinflammatory markers and the increase seems to be related to the symptoms.
Proinflammatory and Neuroimmunoendocrine Mechanisms
Typical of acute inflammation is the accumulation of neutrophil granulocytes which are attracted to the site by mediators produced by tissue-bound macrophages. Furthermore, vasodilatation on the arterial side and permeability increase in the vascular walls of the venules are typical findings. Macrophages have a central role in most inflammatory processes and occur as several variants (microglia in the CNS, Kupffer cells in the liver, osteoclasts in the skeleton) which may have different functions. For example, macrophages also produce mediators of great importance for inflammation, ex. proinflammatory interleukins (IL) as IL-1β, TNF-α, PGE2, platelet-activating factor (PAF) and free oxygen radicals, bradykinin, prostaglandin E, substance P (SP), and neural growth factor (NGF). These signaling mediators are known to cause sensitization in acute pain [69]. FMS has been suggested hypothetically to be a neuroinflammatory disorder due to increased levels of CRH and SP that have been shown in the CSF of FMS patients, as well as increased SP, IL-6, and IL-8 in the serum [70]. Proinflammatory cytokines such as IL-1β, IL-6, and TNF-α, as well as increased release of CRH and SP, are also involved in inflammation-induced central sensitization [71]. Further, excitatory neurotransmitters released from nearby neurons can activate CNS glial cells and may cause neuroinflammation and promotion of maladaptive neural plasticity resulting in neuropathy. Indeed, a strong correlation has been established between inflammation and spinal glia cell activation on one hand and induction of neuropathic pain states on the other hand [63, 71,72,73]. There is accumulating evidence that in FMS, as well as in other pain syndromes, levels of pro- inflammatory cytokines like IL-6 and IL-8 are increased whereas anti-inflammatory cytokines are decreased and several chemokines appear to be increased in FMS patients [72, 74,75,76,77]. A recent study demonstrated high concentrations of cytokines in the cerebrospinal fluid in FM patients and provides the first in vivo evidence supporting a role for glial activation in FM pathophysiology [78••]. By measuring a glial marker, the study group observed extended glial activation and neuroinflammation in cortical areas. These data could be compared to a previous study on chronic low back pain in which a different spatial pattern of glial activation was observed. This was localized to the thalamus and areas of the somatosensory and motor cortices, consistent with the somatotopic representation of the back and leg regions in which those participants experienced pain.
Evidence of the relationship between RLS and inflammatory processes is not fully established but inflammation or an immune component was found in approximately 95% of the 38 different health conditions that were strongly associated with RLS according to a review article that investigated health conditions reported to cause or exacerbate RLS symptoms [79]. Further evidence for a link between RLS and inflammation can be derived from the studies showing an elevated blood level of C-reactive protein (CRP) (a marker of systemic inflammation) which was associated with RLS severity and in patients with irritable bowel disease, CRP levels were higher in the RLS group [80]. Moreover, treatments reducing systemic inflammation such as infusion of hydrocortisone reduced RLS symptoms in a small crossover trial [81].
As is well known, iron deficiency is frequently concomitant with chronic inflammatory disease and the prevalence of iron deficiency is high in RLS. Sleep deficit due to sensorimotor symptoms in RLS may act as a proinflammatory process and has been associated with elevated IL-6 and TNF-a levels in RLS [80]. Finally, increased inflammation or immunological modulation in RLS may be also caused by genetic factors [79].
Conclusion
RLS is highly prevalent (33–56%) in chronic pain conditions, especially CWP, CMP, and fibromyalgia, named nociplastic pain in the nomenclature recently updated [25,26,27,28]. Also, depression and anxiety are frequently comorbid in both CWP/FMS and sleep deficit including RLS and there seems to be a triad of conditions (RLS, CWP, and depression) with similar, possibly common pathological mechanisms. The presence of inflammation could promote the spreading of pain [82]. Further, according to two studies, RLS prevalence increases with an increased number of pain areas [26, 83] and the higher the inflammatory load by spreading, the higher the occurrence of RLS, hypothetically. The pathological mechanisms are unknown but in the light of new findings on neuroinflammatory processes in FM [78••], possible involvement of inflammatory processes in RLS seems relevant to investigate further. As well, there is a need for more and larger studies verifying the hypotheses.
There seem to be multiple pathways involved in RLS pathogenesis, i.e., iron depletion, dopamine dysregulation, and genetic links, but also other pathophysiological changes are emerging such as altered HPA axis, altered sympathetic activity, proinflammatory processes, and neuroendocrine mechanisms. Patients with RLS have significantly greater anxiety and depression symptoms than control subjects [51, 59]. A multitude of studies have demonstrated increased mean concentrations of a variety of inflammatory markers in depressed patients compared with controls—including reproducible increases in the inflammatory cytokines TNF, IL-1β, IL-6, and CRP [84, 85•]. Inflammation may decrease DA synthesis and availability. Other findings have included inflammation-associated reductions in ventral striatal responses to hedonic reward, decreased dopamine and dopamine metabolites in cerebrospinal fluid, and decreased availability of striatal dopamine, all of which correlate with depressive symptoms [86•]. Inflammation reduces the availability of monoamines. Further, activating microglia by inflammation can lead to excessive glutamate. Excessive glutamate can lead to decreased brain-derived neurotrophic factor (BDNF) and excitotoxicity [87]. Increased CRP has been directly correlated with increased glutamate in the basal ganglia of patients with depression. In FMS, research shows abnormally high levels of glutamate in several brain areas [88] and in RLS increased glutamatergic activity in thalamus [16].
Chronic stress and traumatic events in childhood or adulthood are more prevalent in patients with chronic widespread pain and fibromyalgia. Chronic psychological stress is associated with the body losing its ability to regulate the inflammatory response [89]. In humans, stress-induced inflammation has been implicated in diseases such as osteoporosis, rheumatoid arthritis, myopathy, FM, chronic fatigue syndrome (CFS), chronic pelvic pain, temporo-mandibular dysfunction (TMD), chronic low back pain, sciatica, and more [41]. Cortisol is a potent anti-inflammatory, and dysregulation or its failure to function (as has been recognized in FMS and other chronic conditions) results in an unmodulated inflammatory response to pathogens, unrecognized foreign proteins, or to psychological stressors.
The etiology of RLS remains uncertain, but there is increasing evidence for proinflammatory and neuroimmunoendocrine mechanisms both in RLS, CWP, and comorbid conditions as depression.
In summary, RLS is found to be common in chronic pain conditions (CP) and possible common pathological mechanisms are still unknown but there seems to be a relationship between these two conditions. It is of importance to raise awareness of the high coexistence of RLS in CWP/FMS to investigate the comorbidity further with the aim to optimize treatment of these conditions.
Abbreviations
- ACTH:
-
Adrenocorticotropic hormone
- BID:
-
Brain iron deficiency
- CNS:
-
Central nervous system
- CWP:
-
Chronic widespread pain
- CP:
-
Chronic pain
- FMS:
-
Fibromyalgia syndrome
- HPA:
-
Hypothalamic pituitary adrenal
- IRRLS:
-
International Restless Legs Group Rating Scale
- OSA:
-
Obstructive sleep apnea
- RLS:
-
Restless legs syndrome
- PLM:
-
Periodic limb movements
- PWA:
-
Pulse wave amplitude
- SAM:
-
Sympathetic adrenal medullary
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Brennan F, Carr DB, Cousins M. Pain management: a fundamental human right. Anesth Analg. 2007;105(1):205–21.
Gureje O, Von Korff M, Simon GE, Gater R. Persistent pain and well-being: a World Health Organization study in primary care. Jama. 1998;280(2):147–51.
Leao Ferreira KA, Bastos TR, Andrade DC, Silva AM, Appolinario JC, Teixeira MJ, et al. Prevalence of chronic pain in a metropolitan area of a developing country: a population-based study. Arq Neuropsiquiatr. 2016;74(12):990–8.
Artner J, Cakir B, Spiekermann JA, Kurz S, Leucht F, Reichel H, et al. Prevalence of sleep deprivation in patients with chronic neck and back pain: a retrospective evaluation of 1016 patients. J Pain Res. 2013;6:1–6.
Theadom A, Cropley M, Humphrey KL. Exploring the role of sleep and coping in quality of life in fibromyalgia. J Psychosom Res. 2007;62(2):145–51.
Earley CJ, Silber MH. Restless legs syndrome: understanding its consequences and the need for better treatment. Sleep Med. 2010;11(9):807–15.
Allen RP, Picchietti DL, Garcia-Borreguero D, Ondo WG, Walters AS, Winkelman JW, et al. Restless legs syndrome/Willis-Ekbom disease diagnostic criteria: updated international restless legs syndrome study group (IRLSSG) consensus criteria--history, rationale, description, and significance. Sleep Med. 2014;15(8):860–73.
Eckeli AL, Gitai LL, Dach F, Ceretta H, Sander HH, Passos AD, et al. Prevalence of restless legs syndrome in the rural town of Cassia dos Coqueiros in Brazil. Sleep Med. 2011;12(8):762–7.
Garcia-Borreguero D, Williams AM. An update on restless legs syndrome (Willis-Ekbom disease): clinical features, pathogenesis and treatment. Curr Opin Neurol. 2014;27(4):493–501.
Ohayon MM, O'Hara R, Vitiello MV. Epidemiology of restless legs syndrome: a synthesis of the literature. Sleep Med Rev. 2012;16(4):283–95.
Burtscher C, Baxmann A, Kassubek J, Hornyak M, Matuja W, Schmutzhard E, et al. Prevalence of restless legs syndrome in an urban population of eastern Africa (Tanzania). J Neurol Sci. 2014;346(1–2):121–7.
Chen NH, Chuang LP, Yang CT, Kushida CA, Hsu SC, Wang PC, et al. The prevalence of restless legs syndrome in Taiwanese adults. Psychiatry Clin Neurosci. 2010;64(2):170–8.
Nomura T, Inoue Y, Kusumi M, Uemura Y, Nakashima K. Prevalence of restless legs syndrome in a rural community in Japan. Mov Disord. 2008;23(16):2363–9.
Allen RP, Walters AS, Montplaisir J, Hening W, Myers A, Bell TJ, et al. Restless legs syndrome prevalence and impact: REST general population study. Arch Intern Med. 2005;165(11):1286–92.
Cho YW, Song ML, Earley CJ, Allen RP. Prevalence and clinical characteristics of patients with restless legs syndrome with painful symptoms. Sleep Med. 2015;16(6):775–8.
Allen RP, Barker PB, Horska A, Earley CJ. Thalamic glutamate/glutamine in restless legs syndrome: increased and related to disturbed sleep. Neurology. 2013;80(22):2028–34.
Allen RP, Mignot E, Ripley B, Nishino S, Earley CJ. Increased CSF hypocretin-1 (orexin-A) in restless legs syndrome. Neurology. 2002;59(4):639–41.
Nagandla K, De S. Restless legs syndrome: pathophysiology and modern management. Postgrad Med J. 2013;89(1053):402–10.
Unrath A, Juengling FD, Schork M, Kassubek J. Cortical grey matter alterations in idiopathic restless legs syndrome: an optimized voxel-based morphometry study. Mov Disord. 2007;22(12):1751–6.
Hwang IC, Na KS, Lee YJ, Kang SG. Higher prevalence of hypertension among individuals with restless legs syndrome: a meta-analysis. Psychiatry Investig. 2018;15(7):701–9.
Ramirez JO, Cabrera SA, Hidalgo H, Cabrera SG, Linnebank M, Bassetti CL, et al. Is preeclampsia associated with restless legs syndrome? Sleep Med. 2013;14(9):894–6.
Trenkwalder C, Allen R, Hogl B, Paulus W, Winkelmann J. Restless legs syndrome associated with major diseases: a systematic review and new concept. Neurology. 2016;86(14):1336–43.
Stiasny-Kolster K, Magerl W, Oertel WH, Moller JC, Treede RD. Static mechanical hyperalgesia without dynamic tactile allodynia in patients with restless legs syndrome. Brain. 2004;127(Pt 4):773–82.
Edwards RR, Quartana PJ, Allen RP, Greenbaum S, Earley CJ, Smith MT. Alterations in pain responses in treated and untreated patients with restless legs syndrome: associations with sleep disruption. Sleep Med. 2011;12(6):603–9.
Stehlik R, Arvidsson L, Ulfberg J. Restless legs syndrome is common among female patients with fibromyalgia. Eur Neurol. 2009;61(2):107–11.
Stehlik R, Ulfberg J, Hedner J, Grote L. High prevalence of restless legs syndrome among women with multi-site pain: a population-based study in Dalarna, Sweden. Eur J Pain (London, England). 2014;18(10):1402–9.
Viola-Saltzman M, Watson NF, Bogart A, Goldberg J, Buchwald D. High prevalence of restless legs syndrome among patients with fibromyalgia: a controlled cross-sectional study. J Clin Sleep Med. 2010;6(5):423–7.
Yunus MB, Aldag JC. Restless legs syndrome and leg cramps in fibromyalgia syndrome: a controlled study. BMJ (Clin Res Ed). 1996;312(7042):1339.
Mathias JL, Cant ML, Burke ALJ. Sleep disturbances and sleep disorders in adults living with chronic pain: a meta-analysis. Sleep Med. 2018;52:198–210.
Onen SH, Alloui A, Gross A, Eschallier A, Dubray C. The effects of total sleep deprivation, selective sleep interruption and sleep recovery on pain tolerance thresholds in healthy subjects. J Sleep Res. 2001;10(1):35–42.
Lautenbacher S, Kundermann B, Krieg JC. Sleep deprivation and pain perception. Sleep Med Rev. 2006;10(5):357–69.
Mork PJ, Vik KL, Moe B, Lier R, Bardal EM, Nilsen TI. Sleep problems, exercise and obesity and risk of chronic musculoskeletal pain: the Norwegian HUNT study. Eur J Pub Health. 2014;24(6):924–9.
Alsaadi SM, McAuley JH, Hush JM, Lo S, Bartlett DJ, Grunstein RR, et al. The bidirectional relationship between pain intensity and sleep disturbance/quality in patients with low back pain. Clin J Pain. 2014;30(9):755–65.
Koffel E, Kroenke K, Bair MJ, Leverty D, Polusny MA, Krebs EE. The bidirectional relationship between sleep complaints and pain: analysis of data from a randomized trial. Health Psychol. 2016;35(1):41–9.
Okifuji A, Hare BD. Do sleep disorders contribute to pain sensitivity? Curr Rheumatol Rep. 2011;13(6):528–34.
Davies KA, Macfarlane GJ, Nicholl BI, Dickens C, Morriss R, Ray D, et al. Restorative sleep predicts the resolution of chronic widespread pain: results from the EPIFUND study. Rheumatology (Oxford, England). 2008;47(12):1809–13.
Roehrs TA, Harris E, Randall S, Roth T. Pain sensitivity and recovery from mild chronic sleep loss. Sleep. 2012;35(12):1667–72.
McBeth J, Silman AJ, Gupta A, Chiu YH, Ray D, Morriss R, et al. Moderation of psychosocial risk factors through dysfunction of the hypothalamic-pituitary-adrenal stress axis in the onset of chronic widespread musculoskeletal pain: findings of a population-based prospective cohort study. Arthritis Rheum. 2007;56(1):360–71.
Nees F, Loffler M, Usai K, Flor H. Hypothalamic-pituitary-adrenal axis feedback sensitivity in different states of back pain. Psychoneuroendocrinology. 2019;101:60–6.
Aurora RN, Kristo DA, Bista SR, Rowley JA, Zak RS, Casey KR, et al. The treatment of restless legs syndrome and periodic limb movement disorder in adults--an update for 2012: practice parameters with an evidence-based systematic review and meta-analyses: an American Academy of Sleep Medicine clinical practice guideline. Sleep. 2012;35(8):1039–62.
Hannibal KE, Bishop MD. Chronic stress, cortisol dysfunction, and pain: a psychoneuroendocrine rationale for stress management in pain rehabilitation. Phys Ther. 2014;94(12):1816–25.
Griep EN, Boersma JW, Lentjes EG, Prins AP, van der Korst JK, de Kloet ER. Function of the hypothalamic-pituitary-adrenal axis in patients with fibromyalgia and low back pain. J Rheumatol. 1998;25(7):1374–81.
Holtorf K. Diagnosis and treatment of hypothalamic-pituitary-adrenal (HPA) axis dysfunction in patients with chronic fatigue syndrome (CFS) and fibromyalgia (FM). J Chronic Fatigue Syndr. 2007;14(3):59–88.
Okifuji A, Turk DC. Stress and psychophysiological dysregulation in patients with fibromyalgia syndrome. Appl Psychophysiol Biofeedback. 2002;27(2):129–41.
Crofford LJ. The hypothalamic-pituitary-adrenal stress axis in fibromyalgia and chronic fatigue syndrome. Z Rheumatol. 1998;57(Suppl 2):67–71.
McLean SA, Williams DA, Stein PK, Harris RE, Lyden AK, Whalen G, et al. Cerebrospinal fluid corticotropin-releasing factor concentration is associated with pain but not fatigue symptoms in patients with fibromyalgia. Neuropsychopharmacology. 2006;31(12):2776–82.
McLean SA, Williams DA, Harris RE, Kop WJ, Groner KH, Ambrose K, et al. Momentary relationship between cortisol secretion and symptoms in patients with fibromyalgia. Arthritis Rheum. 2005;52(11):3660–9.
Li L, Li X, Zhou W, Messina JL. Acute psychological stress results in the rapid development of insulin resistance. J Endocrinol. 2013;217(2):175–84.
Tanno S, Tanigawa T, Saito I, Nishida W, Maruyama K, Eguchi E, et al. Sleep-related intermittent hypoxemia and glucose intolerance: a community-based study. Sleep Med. 2014;15(10):1212–8.
Schilling C, Schredl M, Strobl P, Deuschle M. Restless legs syndrome: evidence for nocturnal hypothalamic-pituitary-adrenal system activation. Mov Disord. 2010;25(8):1047–52.
Stehlik R, Ulfberg J, Zou D, Hedner J, Grote L. Morning cortisol and fasting glucose are elevated in women with chronic widespread pain independent of comorbid restless legs syndrome. Scand J Pain. 2018;18(2):187–94.
Sommermeyer D, Zou D, Eder DN, Hedner J, Ficker JH, Randerath W, et al. The use of overnight pulse wave analysis for recognition of cardiovascular risk factors and risk: a multicentric evaluation. J Hypertens. 2014;32(2):276–85.
Zou D, Grote L, Eder DN, Peker Y, Hedner J. Obstructive apneic events induce alpha-receptor mediated digital vasoconstriction. Sleep. 2004;27(3):485–9.
Zou D, Grote L, Eder DN, Radlinski J, Hedner J. A double-blind, crossover study of doxazosin and enalapril on peripheral vascular tone and nocturnal blood pressure in sleep apnea patients. Sleep Med. 2010;11(3):325–8.
Grote L, Sommermeyer D, Zou D, Eder DN, Hedner J. Oximeter-based autonomic state indicator algorithm for cardiovascular risk assessment. Chest. 2011;139(2):253–9.
Broderick JE, Gold MS, Amin MM, Gold AR. The association of somatic arousal with the symptoms of upper airway resistance syndrome. Sleep Med. 2014;15(4):436–43.
Hedner J, Ejnell H, Sellgren J, Hedner T, Wallin G. Is high and fluctuating muscle nerve sympathetic activity in the sleep apnoea syndrome of pathogenetic importance for the development of hypertension? J Hypertens Supplement. 1988;6(4):S529–31.
Blackburn-Munro G. Hypothalamo-pituitary-adrenal axis dysfunction as a contributory factor to chronic pain and depression. Curr Pain Headache Rep. 2004;8(2):116–24.
Understanding fibromyalgia and its related disorders. Prim Care Companion J Clin Psychiatry. 2008;10(2):133–44.
Woo AK. Depression and anxiety in pain. Rev Pain. 2010;4(1):8–12.
Kurtze N, Svebak S. Fatigue and patterns of pain in fibromyalgia: correlations with anxiety, depression and co-morbidity in a female county sample. Br J Med Psychol. 2001;74(Pt 4):523–37.
Sevim S, Dogu O, Kaleagasi H, Aral M, Metin O, Camdeviren H. Correlation of anxiety and depression symptoms in patients with restless legs syndrome: a population based survey. J Neurol Neurosurg Psychiatry. 2004;75(2):226–30.
Watkins LR, Maier SF. Beyond neurons: evidence that immune and glial cells contribute to pathological pain states. Physiol Rev. 2002;82(4):981–1011.
Irwin MR, Olmstead R, Carrillo C, Sadeghi N, Fitzgerald JD, Ranganath VK, et al. Sleep loss exacerbates fatigue, depression, and pain in rheumatoid arthritis. Sleep. 2012;35(4):537–43.
Chiu YH, Silman AJ, Macfarlane GJ, Ray D, Gupta A, Dickens C, et al. Poor sleep and depression are independently associated with a reduced pain threshold. Results of a population based study. Pain. 2005;115(3):316–21.
Affleck G, Urrows S, Tennen H, Higgins P, Abeles M. Sequential daily relations of sleep, pain intensity, and attention to pain among women with fibromyalgia. Pain. 1996;68(2–3):363–8.
Bigatti SM, Hernandez AM, Cronan TA, Rand KL. Sleep disturbances in fibromyalgia syndrome: relationship to pain and depression. Arthritis Rheum. 2008;59(7):961–7.
Mundal I, Grawe RW, Bjorngaard JH, Linaker OM, Fors EA. Prevalence and long-term predictors of persistent chronic widespread pain in the general population in an 11-year prospective study: the HUNT study. BMC Musculoskelet Disord. 2014;15:213.
Mantyh PW, Koltzenburg M, Mendell LM, Tive L, Shelton DL. Antagonism of nerve growth factor-TrkA signaling and the relief of pain. Anesthesiology. 2011;115(1):189–204.
Wang H, Moser M, Schiltenwolf M, Buchner M. Circulating cytokine levels compared to pain in patients with fibromyalgia -- a prospective longitudinal study over 6 months. J Rheumatol. 2008;35(7):1366–70.
Watkins LR, Milligan ED, Maier SF. Glial proinflammatory cytokines mediate exaggerated pain states: implications for clinical pain. Adv Exp Med Biol. 2003;521:1–21.
Kosek E, Altawil R, Kadetoff D, Finn A, Westman M, Le Maitre E, et al. Evidence of different mediators of central inflammation in dysfunctional and inflammatory pain--interleukin-8 in fibromyalgia and interleukin-1 beta in rheumatoid arthritis. J Neuroimmunol. 2015;280:49–55.
Zhang JM, An J. Cytokines, inflammation, and pain. Int Anesthesiol Clin. 2007;45(2):27–37.
Gur A, Karakoc M, Nas K, Remzi, Cevik, Denli A, et al. Cytokines and depression in cases with fibromyalgia. J Rheumatol. 2002;29(2):358–61.
Maes M, Libbrecht I, Van Hunsel F, Lin AH, De Clerck L, Stevens W, et al. The immune-inflammatory pathophysiology of fibromyalgia: increased serum soluble gp130, the common signal transducer protein of various neurotrophic cytokines. Psychoneuroendocrinology. 1999;24(4):371–83.
Pay S, Calguneri M, Caliskaner Z, Dinc A, Apras S, Ertenli I, et al. Evaluation of vascular injury with proinflammatory cytokines, thrombomodulin and fibronectin in patients with primary fibromyalgia. Nagoya J Med Sci. 2000;63(3–4):115–22.
Wallace DJ, Linker-Israeli M, Hallegua D, Silverman S, Silver D, Weisman MH. Cytokines play an aetiopathogenetic role in fibromyalgia: a hypothesis and pilot study. Rheumatology (Oxford, England). 2001;40(7):743–9.
•• Albrecht DS, Forsberg A, Sandstrom A, Bergan C, Kadetoff D, Protsenko E, et al. Brain glial activation in fibromyalgia - a multi-site positron emission tomography investigation. Brain Behav Immunity. 2019;75:72–83. Evidence for brain glial activation and neuroinflammation.
Weinstock LB, Walters AS, Paueksakon P. Restless legs syndrome--theoretical roles of inflammatory and immune mechanisms. Sleep Med Rev. 2012;16(4):341–54.
Trotti LM, Rye DB, De Staercke C, Hooper WC, Quyyumi A, Bliwise DL. Elevated C-reactive protein is associated with severe periodic leg movements of sleep in patients with restless legs syndrome. Brain Behav Immun. 2012;26(8):1239–43.
Hornyak M, Rupp A, Riemann D, Feige B, Berger M, Voderholzer U. Low-dose hydrocortisone in the evening modulates symptom severity in restless legs syndrome. Neurology. 2008;70(18):1620–2.
Gerdle B, Ghafouri B, Ghafouri N, Backryd E, Gordh T. Signs of ongoing inflammation in female patients with chronic widespread pain: a multivariate, explorative, cross-sectional study of blood samples. Medicine. 2017;96(9):e6130.
Hoogwout SJ, Paananen MV, Smith AJ, Beales DJ, O'Sullivan PB, Straker LM, et al. Musculoskeletal pain is associated with restless legs syndrome in young adults. BMC Musculoskelet Disord. 2015;16:294.
Berk M, Williams LJ, Jacka FN, O'Neil A, Pasco JA, Moylan S, et al. So depression is an inflammatory disease, but where does the inflammation come from? BMC Med. 2013;11:200.
• Jeon SW, Kim YK. Inflammation-induced depression: its pathophysiology and therapeutic implications. J Neuroimmunol. 2017;313:92–8. Inflammation links to depression as one possible mechanism.
• Felger JC. The role of dopamine in inflammation-associated depression: mechanisms and therapeutic implications. Curr Top Behav Neurosci. 2017;31:199–219. Inflammation and its imapct on dopamin neurotransmission.
Haroon E, Miller AH, Sanacora G. Inflammation, glutamate, and glia: a trio of trouble in mood disorders. Neuropsychopharmacology. 2017;42(1):193–215.
Pyke TL, Osmotherly PG, Baines S. Measuring glutamate levels in the brains of fibromyalgia patients and a potential role for glutamate in the pathophysiology of fibromyalgia symptoms: a systematic review. Clin J Pain. 2017;33(10):944–54.
Cohen S, Janicki-Deverts D, Doyle WJ, Miller GE, Frank E, Rabin BS, et al. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proc Natl Acad Sci U S A. 2012;109(16):5995–9.
Funding
Open access funding provided by Uppsala University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None declared.
Human and Animal Rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Sleep and Pain
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stehlik, R., Ulfberg, J. (Neuro)Inflammatory Component May Be a Common Factor in Chronic Widespread Pain and Restless Legs Syndrome. Curr Sleep Medicine Rep 6, 121–128 (2020). https://doi.org/10.1007/s40675-020-00180-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40675-020-00180-0