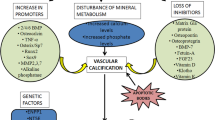
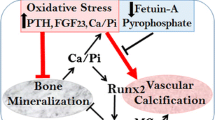
Abstract
Vascular calcification is a common complication in patients with chronic kidney disease (CKD) and is strongly associated with an increased risk of cardiovascular events and all-cause mortality. Calciphylaxis is a specific and life-threatening manifestation of vascular calcifications that usually affects individuals with advanced kidney function impairment or those undergoing dialysis. Currently, the treatment of vascular calcification and calciphylaxis in CKD lacks approved treatments and focuses on controlling risk factors. SNF472, the intravenous formulation of myo-inositol hexaphosphate, is a novel vascular calcification inhibitor currently undergoing phase 3 clinical trials, demonstrating its ability to directly inhibit the formation of calcium and phosphorus crystals, thereby blocking the production and deposition of ectopic calcium. The efficacy and safety of SNF472 in inhibiting vascular calcification have been confirmed in recent clinical studies. This review summarizes the results of studies related to SNF472 to provide a comprehensive overview of its mechanism of action, efficacy, safety, and ongoing clinical studies.
Graphical abstract


Similar content being viewed by others
References
GBD Chronic Kidney Disease Collaboration (2020) Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. The Lancet 395(10225):709–733. https://doi.org/10.1016/S0140-6736(20)30045-3
Jankowski J, Floege J, Fliser D, Böhm M, Marx N (2021) Cardiovascular disease in chronic kidney disease: pathophysiological insights and therapeutic options. Circulation 143(11):1157–1172. https://doi.org/10.1161/CIRCULATIONAHA.120.050686
Thompson S, James M, Wiebe N, Hemmelgarn B, Manns B, Klarenbach S et al (2015) Cause of death in patients with reduced kidney function. J Am Soc Nephrol 26(10):2504–2511. https://doi.org/10.1681/ASN.2014070714
Nigwekar SU, Thadhani R, Brandenburg VM (2018) Calciphylaxis. N Engl J Med 378(18):1704–1714. https://doi.org/10.1056/NEJMra1505292
Mccarthy JT, El-Azhary RA, Patzelt MT, Weaver AL, Albright RC, Bridges AD et al (2016) Survival, risk factors, and effect of treatment in 101 patients with calciphylaxis. Mayo Clin Proc 91(10):1384–1394. https://doi.org/10.1016/j.mayocp.2016.06.025
Nigwekar SU, Zhao S, Wenger J, Hymes JL, Maddux FW, Thadhani RI et al (2016) A nationally representative study of calcific uremic arteriolopathy risk factors. J Am Soc Nephrol 27(11):3421–3429. https://doi.org/10.1681/ASN.2015091065
Nelson AJ, Raggi P, Wolf M, Gold AM, Chertow GM, Roe MT (2020) Targeting vascular calcification in chronic kidney disease. JACC Basic Transl Sci 5(4):398–412. https://doi.org/10.1016/j.jacbts.2020.02.002
Wang L, Tang R, Zhang Y, Chen S, Guo Y, Wang X et al (2021) PTH-induced EndMT via miR-29a-5p/GSAP/Notch1 pathway contributed to valvular calcification in rats with CKD. Cell Proliferat 54(6):e13018. https://doi.org/10.1111/cpr.13018
Demer LL, Tintut Y (2008) Vascular calcification: pathobiology of a multifaceted disease. Circulation 117(22):2938–2948. https://doi.org/10.1161/CIRCULATIONAHA.107.743161
Moe SM, Chen NX (2008) Mechanisms of vascular calcification in chronic kidney disease. J Am Soc Nephrol 19(2):213–216. https://doi.org/10.1681/ASN.2007080854
Tesauro M, Mauriello A, Rovella V, Annicchiarico-Petruzzelli M, Cardillo C, Melino G et al (2017) Arterial ageing: from endothelial dysfunction to vascular calcification. J Intern Med 281(5):471–482. https://doi.org/10.1111/joim.12605
Maher ER, Young G, Smyth-Walsh B, Pugh S, Curtis JR (1987) Aortic and mitral valve calcification in patients with end-stage renal disease. Lancet 2(8564):875–877. https://doi.org/10.1016/s0140-6736(87)91370-5
Rattazzi M, Bertacco E, Del Vecchio A, Puato M, Faggin E, Pauletto P (2013) Aortic valve calcification in chronic kidney disease. Nephrol Dial Transplant 28(12):2968–2976. https://doi.org/10.1093/ndt/gft310
Chang JJ (2019) Calciphylaxis: diagnosis, pathogenesis, and treatment. Adv Skin Wound Care 32(5):205–215. https://doi.org/10.1097/01.Asw.0000554443.14002.13
Tsuchiya K, Akihisa T (2021) the importance of phosphate control in chronic kidney disease. Nutrients. https://doi.org/10.3390/nu13051670
Floege J, Kubo Y, Floege A, Chertow GM, Parfrey PS (2015) The effect of cinacalcet on calcific uremic arteriolopathy events in patients receiving hemodialysis: the EVOLVE Trial. Clin J Am Soc Nephrol 10(5):800–807. https://doi.org/10.2215/CJN.10221014
O’Neill WC, Hardcastle KI (2012) The chemistry of thiosulfate and vascular calcification. Nephrol Dial Transplant 27(2):521–526. https://doi.org/10.1093/ndt/gfr375
Lau WL, Leaf EM, Hu MC, Takeno MM, Kuro-O M, Moe OW et al (2012) Vitamin D receptor agonists increase klotho and osteopontin while decreasing aortic calcification in mice with chronic kidney disease fed a high phosphate diet. Kidney Int 82(12):1261–1270. https://doi.org/10.1038/ki.2012.322
Hansen D, Rasmussen K, Rasmussen LM, Bruunsgaard H, Brandi L (2014) The influence of vitamin D analogs on calcification modulators, N-terminal pro-B-type natriuretic peptide and inflammatory markers in hemodialysis patients: a randomized crossover study. BMC Nephrol 15:130. https://doi.org/10.1186/1471-2369-15-130
Wang AY, Fang F, Chan J, Wen YY, Qing S, Chan IH et al (2014) Effect of paricalcitol on left ventricular mass and function in CKD–the OPERA trial. J Am Soc Nephrol 25(1):175–186. https://doi.org/10.1681/ASN.2013010103
El-Azhary RA, Arthur AK, Davis MDP, Mcevoy MT, Gibson LE, Weaver AL et al (2013) Retrospective analysis of tissue plasminogen activator as an adjuvant treatment for calciphylaxis. Jama Dermatol 149(1):63–67. https://doi.org/10.1001/2013.jamadermatol.5
Christiadi D, Singer RF (2018) Calciphylaxis in a dialysis patient successfully treated with high-dose vitamin K supplementation. Clin Kidney J 11(4):528–529. https://doi.org/10.1093/ckj/sfx126
Marietta M, Coluccio V, Boriani G, Luppi M (2020) Effects of Anti-vitamin k oral anticoagulants on bone and cardiovascular health. Eur J Intern Med 79:1–11. https://doi.org/10.1016/j.ejim.2020.05.032
Lomashvili KA, Monier-Faugere MC, Wang X, Malluche HH, O’Neill WC (2009) Effect of bisphosphonates on vascular calcification and bone metabolism in experimental renal failure. Kidney Int 75(6):617–625. https://doi.org/10.1038/ki.2008.646
Grases F (2006) Phytate (Myo-inositol hexakisphosphate) inhibits cardiovascular calcifications in rats. Front Biosci. https://doi.org/10.2741/1786
Grases F, Sanchis P, Costa-Bauzá A, Bonnin O, Isern B, Perelló J et al (2008) Phytate inhibits bovine pericardium calcification in vitro. Cardiovasc Pathol 17(3):139–145. https://doi.org/10.1016/j.carpath.2007.08.005
Sanchis P, Buades JM, Berga F, Gelabert MM, Molina M, íñigo MV et al (2016) Protective effect of myo-inositol hexaphosphate (Phytate) on abdominal aortic calcification in patients with chronic kidney disease. J Ren Nutr 26(4):226–236. https://doi.org/10.1053/j.jrn.2016.01.010
Ferrer MD, Pérez MM, Cànaves MM, Buades JM, Salcedo C, Perelló J (2017) A novel pharmacodynamic assay to evaluate the effects of crystallization inhibitors on calcium phosphate crystallization in human plasma. Sci Rep-UK 7(1):6858. https://doi.org/10.1038/s41598-017-07203-x
Perelló J, Joubert PH, Ferrer MD, Canals AZ, Sinha S, Salcedo C (2018) First-time-in-human randomized clinical trial in healthy volunteers and haemodialysis patients with SNF472, a novel inhibitor of vascular calcification. Br J Clin Pharmacol 84(12):2867–2876. https://doi.org/10.1111/bcp.13752
Perelló J, Ferrer MD, Del MPM, Kaesler N, Brandenburg VM, Behets GJ et al (2020) Mechanism of action of SNF472, a novel calcification inhibitor to treat vascular calcification and calciphylaxis. Br J Pharmacol 177(19):4400–4415. https://doi.org/10.1111/bph.15163
Ferrer MD, Ketteler M, Tur F, Tur E, Isern B, Salcedo C et al (2018) Characterization of SNF472 pharmacokinetics and efficacy in uremic and non-uremic rats models of cardiovascular calcification. PLoS One 13(5):e197061. https://doi.org/10.1371/journal.pone.0197061
Zabirnyk A, Ferrer MD, Bogdanova M, Pérez MM, Salcedo C, Kaljusto ML et al (2019) SNF472, a novel anti-crystallization agent, inhibits induced calcification in an in vitro model of human aortic valve calcification. Vascul Pharmacol 122–123:106583. https://doi.org/10.1016/j.vph.2019.106583
Zabirnyk A, Perez MDM, Blasco M, Stensløkken K, Ferrer MD, Salcedo C et al (2020) A novel ex vivo model of aortic valve calcification. A preliminary report. Front Pharmacol 11:568764. https://doi.org/10.3389/fphar.2020.568764
Salcedo C, Joubert PH, Ferrer MD, Canals AZ, Maduell F, Torregrosa V et al (2019) A phase 1b randomized, placebo-controlled clinical trial with SNF472 in haemodialysis patients. Br J Clin Pharmacol 85(4):796–806. https://doi.org/10.1111/bcp.13863
Nahapetian A, Young VR (1980) Metabolism of 14C-phytate in rats: effect of low and high dietary calcium intakes. J Nutr 110(7):1458–1472. https://doi.org/10.1093/jn/110.7.1458
Raggi P, Boulay A, Chasan-Taber S, Amin N, Dillon M, Burke SK et al (2002) Cardiac calcification in adult hemodialysis patients. A link between end-stage renal disease and cardiovascular disease? J Am Coll Cardiol 39(4):695–701. https://doi.org/10.1016/s0735-1097(01)01781-8
Alluri K, Joshi PH, Henry TS, Blumenthal RS, Nasir K, Blaha MJ (2015) Scoring of coronary artery calcium scans: history, assumptions, current limitations, and future directions. Atherosclerosis 239(1):109–117. https://doi.org/10.1016/j.atherosclerosis.2014.12.040
Bellasi A, Raggi P, Bover J, Bushinsky DA, Chertow GM, Ketteler M et al (2021) Trial design and baseline characteristics of CaLIPSO: a randomized, double-blind placebo-controlled trial of SNF472 in patients receiving haemodialysis with cardiovascular calcification. Clin Kidney J 14(1):366–374. https://doi.org/10.1093/ckj/sfz144
Raggi P, Bellasi A, Bushinsky D, Bover J, Rodriguez M, Ketteler M et al (2020) Slowing progression of cardiovascular calcification with SNF472 in patients on hemodialysis: results of a randomized phase 2b study. Circulation 141(9):728–739. https://doi.org/10.1161/CIRCULATIONAHA.119.044195
Raggi P, Bellasi A, Sinha S, Bover J, Rodriguez M, Ketteler M et al (2020) Effects of SNF472, a novel inhibitor of hydroxyapatite crystallization in patients receiving hemodialysis—subgroup analyses of the CALIPSO Trial. Kidney Int Rep 5(12):2178–2182. https://doi.org/10.1016/j.ekir.2020.09.032
Bushinsky DA, Raggi P, Bover J, Ketteler M, Bellasi A, Rodriguez M et al (2021) Effects of myo-inositol hexaphosphate (SNF472) on bone mineral density in patients receiving hemodialysis: an analysis of the randomized, placebo-controlled CaLIPSO Study. Clin J Am Soc Nephrol 16(5):736–745. https://doi.org/10.2215/cjn.16931020
Nigwekar SU, Solid CA, Ankers E, Malhotra R, Eggert W, Turchin A et al (2014) Quantifying a rare disease in administrative data: the example of calciphylaxis. J Gen Intern Med 29 Suppl 3(Suppl 3):S724–S731. https://doi.org/10.1007/s11606-014-2910-1
Brandenburg VM, Kramann R, Rothe H, Kaesler N, Korbiel J, Specht P et al (2017) Calcific uraemic arteriolopathy (calciphylaxis): data from a large nationwide registry. Nephrol Dial Transplant 32(1):126–132. https://doi.org/10.1093/ndt/gfv438
Liu Y, Yang C, Yang X, Xie X, Liu H, Zhang L et al (2022) Prevalence and clinical characteristics of calciphylaxis in chinese hemodialysis patients. Front Med (Lausanne) 9:902171. https://doi.org/10.3389/fmed.2022.902171
Yang C, Liu Y, Ni H, Li X, Liu H, Zhang X (2021) Potential effect of sodium thiosulfate in calciphylaxis: remission of intractable pain. J Pak Med Assoc 71(1(B)):367–369. https://doi.org/10.47391/JPMA.1244
Rivera-Nieves J, Bamias G, Alfert J, Bickston SJ, Moskaluk CA, Cominelli F (2002) Intestinal ischemia and peripheral gangrene in a patient with chronic renal failure. Gastroenterology 122(2):495–499. https://doi.org/10.1053/gast.2002.31387
Shi W, Xie X, Liu Y, Yang C, Zhang X (2021) The mystery of black lungs in a patient with calciphylaxis. J Nephrol 34(5):1553–1555. https://doi.org/10.1007/s40620-021-01121-y
Yu Q, Liu Y, Xie X, Liu J, Huang S, Zhang X et al (2021) Radiomics-based method for diagnosis of calciphylaxis in patients with chronic kidney disease using computed tomography. Quant Imag Med Surg 11(11):4617–4626. https://doi.org/10.21037/qims-20-1211
Yang C, Zhang X, Xie X, Zhao Y (2022) Comments on “Ultrasound findings of calciphylaxis.” Kidney Int 101(5):1085. https://doi.org/10.1016/j.kint.2022.01.020
Zhu X, Liu Y, Yang X, Liu B, Zhang X (2022) Identifying subcutaneous tissue microcalcification by Fluo-3 AM imaging in cutaneous calciphylaxis. Exp Dermatol. https://doi.org/10.1111/exd.14579
Brandenburg VM, Sinha S, Torregrosa JV, Garg R, Miller S, Canals AZ et al (2019) Improvement in wound healing, pain, and quality of life after 12 weeks of SNF472 treatment: a phase 2 open-label study of patients with calciphylaxis. J Nephrol 32(5):811–821. https://doi.org/10.1007/s40620-019-00631-0
Harris C, Bates-Jensen B, Parslow N, Raizman R, Singh M, Ketchen R (2010) Bates-Jensen wound assessment tool: pictorial guide validation project. J Wound Ostomy Continence Nurs 37(3):253–259. https://doi.org/10.1097/WON.0b013e3181d73aab
Hjermstad MJ, Fayers PM, Haugen DF, Caraceni A, Hanks GW, Loge JH et al (2011) Studies comparing Numerical Rating Scales, Verbal Rating Scales, and Visual Analogue Scales for assessment of pain intensity in adults: a systematic literature review. J Pain Symptom Manage 41(6):1073–1093. https://doi.org/10.1016/j.jpainsymman.2010.08.016
Janke TM, Kozon V, Valiukeviciene S, Rackauskaite L, Reich A, Stępień K et al (2023) Validation of the Wound-QoL-17 and the Wound-QoL-14 in a European sample of 305 patients with chronic wounds. Int Wound J. https://doi.org/10.1111/iwj.14505
Sinha S, Gould LJ, Nigwekar SU, Serena TE, Brandenburg V, Moe SM et al (2022) The CALCIPHYX study: a randomized, double-blind, placebo-controlled, Phase 3 clinical trial of SNF472 for the treatment of calciphylaxis. Clin Kidney J 15(1):136–144. https://doi.org/10.1093/ckj/sfab117
Gould LJ, Serena TE, Sinha S (2021) Development of the BWAT-CUA scale to assess wounds in patients with calciphylaxis. Diagnostics (Basel). https://doi.org/10.3390/diagnostics11040730
Leskinen Y, Salenius JP, Lehtimäki T, Huhtala H, Saha H (2002) The prevalence of peripheral arterial disease and medial arterial calcification in patients with chronic renal failure: requirements for diagnostics. Am J Kidney Dis 40(3):472–479. https://doi.org/10.1053/ajkd.2002.34885
Ho CY, Shanahan CM (2016) Medial arterial calcification: an overlooked player in peripheral arterial disease. Arterioscler Thromb Vasc Biol 36(8):1475–1482. https://doi.org/10.1161/ATVBAHA.116.306717
Bassissi F, Reynes MDF, Pérez MM, Perelló J, Salcedo C (2020) SNF472 improves limb blood perfusion and walking ability in a peripheral artery disease vascular calcification rat model. Nephrol Dial Transpl 35(SUPPL 3):i265. https://doi.org/10.1093/ndt/gfaa144.P1234
Pimentel A, Ureña-Torres P, Zillikens MC, Bover J, Cohen-Solal M (2017) Fractures in patients with CKD-diagnosis, treatment, and prevention: a review by members of the European Calcified Tissue Society and the European Renal Association of Nephrology Dialysis and Transplantation. Kidney Int 92(6):1343–1355. https://doi.org/10.1016/j.kint.2017.07.021
Arriero MM, Ramis JM, Perelló J, Monjo M (2012) Differential response of MC3T3-E1 and human mesenchymal stem cells to inositol hexakisphosphate. Cell Physiol Biochem 30(4):974–986. https://doi.org/10.1159/000341474
Addison WN, Mckee MD (2010) Inositol hexakisphosphate inhibits mineralization of MC3T3-E1 osteoblast cultures. Bone 46(4):1100–1107. https://doi.org/10.1016/j.bone.2010.01.367
Grases F, Sanchis P, Prieto RM, Perelló J, López-González ÁA (2010) Effect of tetracalcium dimagnesium phytate on bone characteristics in ovariectomized rats. J Med Food 13(6):1301–1306. https://doi.org/10.1089/jmf.2009.0152
López-González AA, Grases F, Monroy N, Marí B, Vicente-Herrero MT, Tur F et al (2013) Protective effect of myo-inositol hexaphosphate (phytate) on bone mass loss in postmenopausal women. Eur J Nutr 52(2):717–726. https://doi.org/10.1007/s00394-012-0377-6
López-González AA, Grases F, Roca P, Mari B, Vicente-Herrero MT, Costa-Bauzá A (2008) Phytate (myo-inositol hexaphosphate) and risk factors for osteoporosis. J Med Food 11(4):747–752. https://doi.org/10.1089/jmf.2008.0087
Funding
This work was supported by 1. National Natural Science Foundation of China (No. 81570612 and No. 81870497); 2. Jiangsu Province Key Research and Development Program-Social Development (No. BE2021737); 3. Nanjing Health and Scientific Technology Development Program (No. YKK20237 and No. YKK21268).
Author information
Authors and Affiliations
Contributions
YCL and ZXL designed the study. YCL, and WZY prepared material and collected study data. The first draft of the manuscript was written by YCL and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no competing interests.
Ethical approval
Not applicable.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, C., Wei, Z., Shi, W. et al. SNF472: a novel therapeutic agent for vascular calcification and calciphylaxis. J Nephrol (2024). https://doi.org/10.1007/s40620-024-01909-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40620-024-01909-8