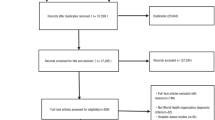
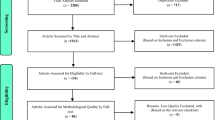
Abstract
Purpose
The association between aldehyde exposure and bone health in humans remains unclear. This study was to evaluate the association of serum aldehydes with bone mineral density (BMD) and osteopenia/osteoporosis.
Methods
We analyzed the US National Health and Nutrition Examination Survey cross-sectional data from 2013 to 2014. Weighted multivariate-adjusted linear regression and logistic regression models were used to assess the association between specific aldehydes and osteopenia/osteoporosis. Associations between aldehyde combinations and BMD were also evaluated using the restricted cubic spline (RCS) method.
Results
Compared with men in the first tertile, those in the third tertile of propanaldehyde concentration were negatively associated with proximal femur and lumbar spine BMD. Significant inverse associations were observed between benzaldehyde exposure and trochanter BMD in women. Benzaldehyde increased the risk of osteopenia/osteoporosis 2.75-fold [95% confidence interval (CI) = 1.06, 7.11] in the highest tertile in women compared to the lowest tertile concentration. In males, the prevalence of total femur, femur neck, and trochanter osteopenia/osteoporosis was significantly higher in the highest versus the lowest tertile of propanaldehyde exposure, with odds ratios (ORs) of 6.84 (95% CI = 2.33, 20.04), 2.72 (95% CI = 1.18, 6.27), and 3.26 (95% CI = 1.25, 8.56), respectively. RCS regression also showed decreased BMD continuously with increasing serum mixed aldehyde levels.
Conclusions
Serum aldehyde concentrations were associated with low BMD and high osteopenia/osteoporosis risk in adults, with propanaldehyde and benzaldehyde being the most critical. Co-exposure to aldehyde combinations was negatively correlated with BMD.


Similar content being viewed by others
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Camacho PM, Petak SM, Binkley N, Diab DL, Eldeiry LS, Farooki A et al (2020) American Association of Clinical Endocrinologists/American College of Endocrinology Clinical Practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis-2020 Update. Endocr Pract 26:1–46
Hendrickx G, Boudin E, Van Hul W (2015) A look behind the scenes: the risk and pathogenesis of primary osteoporosis. Nat Rev Rheumatol 11:462–474
Wright NC, Saag KG, Dawson-Hughes B, Khosla S, Siris ES (2017) The impact of the new National Bone Health Alliance (NBHA) diagnostic criteria on the prevalence of osteoporosis in the USA. Osteoporos Int 28:1225–1232
Greenblatt MB, Tsai JN, Wein MN (2017) Bone turnover markers in the diagnosis and monitoring of metabolic bone disease. Clin Chem 63:464–474
Liu J, Curtis EM, Cooper C, Harvey NC (2019) State of the art in osteoporosis risk assessment and treatment. J Endocrinol Invest 42:1149–1164
Elonheimo H, Lange R, Tolonen H, Kolossa-Gehring M (2021) Environmental substances associated with osteoporosis-a scoping review. Int J Environ Res Pub Health 18(2):738
Cai S, Zhu J, Sun L, Fan C, Zhong Y, Shen Q et al (2019) Association between urinary triclosan with bone mass density and osteoporosis in US adult women, 20052010. J Clin Endocrinol Metab 104:4531–4538
Ma Y, Ran D, Shi X, Zhao H, Liu Z (2021) Cadmium toxicity: a role in bone cell function and teeth development. Sci Total Environ 769:144646
Sinharoy P, McAllister SL, Vasu M, Gross ER (2019) Environmental aldehyde sources and the health implications of exposure. Adv Exp Med Biol 1193:35–52
Silva LK, Espenship MF, Newman CA, Zhang L, Zhu W, Blount BC et al (2021) Assessment of serum concentrations of 12 Aldehydes in the U.S. population from the 2013–2014 National Health and Nutrition Examination Survey. Environ Sci Technol 55:5076–5083
Weng X, Chen J, Fei Q, Guo X, Liu S, Wen L et al (2021) The association of aldehydes exposure with diabetes mellitus in US population: NHANES 2013–2014. Chemosphere 291:133019
Liao S, Zhang J, Shi S, Gong D, Lu X, Cheang I et al (2020) Association of aldehyde exposure with cardiovascular disease. Ecotoxicol Environ Saf 206:111385
Wojciechowska C, Jachec W, Romuk E, Ciszek A, Bodnar P, Chwalba T et al (2021) Serum sulfhydryl groups malondialdehyde, uric acid, and bilirubin as predictors of adverse outcome in heart failure patients due to ischemic or nonischemic cardiomyopathy. Oxid Med Cell Longev 2021:6693405
Srivastava S, Chandrasekar B, Bhatnagar A, Prabhu SD (2002) Lipid peroxidation-derived aldehydes and oxidative stress in the failing heart: role of aldose reductase. Am J Physiol Heart Circ Physiol 283:H2612-2619
Tsai HY, Hsu YJ, Lu CY, Tsai MC, Hung WC, Chen PC et al (2021) Pharmacological activation of aldehyde dehydrogenase 2 protects against heatstroke-induced acute lung injury by modulating oxidative stress and endothelial dysfunction. Front Immunol 12:740562
Romagnoli C, Marcucci G, Favilli F, Zonefrati R, Mavilia C, Galli G et al (2013) Role of GSH/GSSG redox couple in osteogenic activity and osteoclastogenic markers of human osteoblast-like SaOS-2 cells. FEBS J 280:867–879
Bonewald LF (2011) The amazing osteocyte. J Bone Miner Res 26:229–238
Maggio D, Barabani M, Pierandrei M, Polidori MC, Catani M, Mecocci P et al (2003) Marked decrease in plasma antioxidants in aged osteoporotic women: results of a cross-sectional study. J Clin Endocrinol Metab 88:1523–1527
Cervellati C, Bonaccorsi G, Cremonini E, Bergamini CM, Patella A, Castaldini C et al (2013) Bone mass density selectively correlates with serum markers of oxidative damage in post-menopausal women. Clin Chem Lab Med 51:333–338
Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey., Available from: https://wwwn.cdc.gov/nchs/nhanes/ContinuousNhanes/Default.aspx?BeginYear=2013. (accessed 9 May 2022). (2013–2014).
Silva LK, Hile GA, Capella KM, Espenship MF, Smith MM, De Jesus VR et al (2018) Quantification of 19 Aldehydes in Human Serum by Headspace SPME/GC/High-Resolution Mass Spectrometry. Environ Sci Technol 52:10571–10579
Centers for Disease Control and Prevention. 2012 National Health and Nutrition Examination Survey: 2013–2014 data documentation, codebook, and frequencies: dual energy X-ray absorptiometry-femur (DXXFEM_H). Available from: https://wwwn.cdc.gov/Nchs/Nhanes/2013-2014/DXXFEM_H.htm. (accessed 9 May 2022).
Looker AC, Orwoll ES, Johnston CC Jr, R.L. (1997) Lindsay, H.W. Wahner, W.L. Dunn, et al., Prevalence of low femoral bone density in older U.S. adults from NHANES III. J Bone Miner Res 12:1761–1768
Looker AC, Borrud LG, Hughes JP, Fan B, Shepherd JA, Melton LJ 3rd (2012) Lumbar spine and proximal femur bone mineral density, bone mineral content, and bone area: United States, 2005–2008, Vital Health. Stat 11:1–132
Phipps AI, Ichikawa L, Bowles EJ, Carney PA, Kerlikowske K, Miglioretti DL et al (2010) Defining menopausal status in epidemiologic studies: a comparison of multiple approaches and their effects on breast cancer rates. Maturitas 67:60–66
Qu CJ, Teng LQ, Liu XN, Zhang YB, Fang J, Shen CY (2021) Dose-response relationship between physical activity and the incidence of peripheral artery disease in general population: insights from the National Health and Nutrition Examination Survey 1999–2004. Front Cardiovasc Med 8:730508
Guo J, Huang Y, Bian S, Zhao C, Jin Y, Yu D et al (2018) Associations of urinary polycyclic aromatic hydrocarbons with bone mass density and osteoporosis in U.S. adults, NHANES 2005–2010. Environ Pollut 240:209–218
Hu Y, Chiu YH, Hauser R, Chavarro J, Sun Q (2016) Overall and class-specific scores of pesticide residues from fruits and vegetables as a tool to rank intake of pesticide residues in United States: a validation study. Environ Int 92–93:294–300
LoPachin RM, Gavin T (2014) Molecular mechanisms of aldehyde toxicity: a chemical perspective. Chem Res Toxicol 27:1081–1091
Xu C, Liang J, Xu S, Liu Q, Xu J, Gu A (2020) Increased serum levels of aldehydes are associated with cardiovascular disease and cardiovascular risk factors in adults. J Hazard Mater 400:123134
Huang CX, Lv B, Wang Y (2015) Protein phosphatase 2A mediates oxidative stress induced apoptosis in Osteoblasts. Mediat Inflamm 2015:804260
Almeida M, Martin-Millan M, Ambrogini E, Bradsher R 3rd, Han L, Chen XD et al (2010) Estrogens attenuate oxidative stress and the differentiation and apoptosis of osteoblasts by DNA-binding-independent actions of the ERalpha. J Bone Miner Res 25:769–781
Agidigbi TS, Kim C (2019) Reactive oxygen species in osteoclast differentiation and possible pharmaceutical targets of ros-mediated osteoclast diseases. Int J Mol Sci 20:3576
Chen L, Wang G, Wang Q, Liu Q, Sun Q, Chen L (2019) N-acetylcysteine prevents orchiectomy-induced osteoporosis by inhibiting oxidative stress and osteocyte senescence. Am J Transl Res 11:4337–4347
Ahmed Laskar A, Younus H (2019) Aldehyde toxicity and metabolism: the role of aldehyde dehydrogenases in detoxification, drug resistance and carcinogenesis. Drug Metab Rev 51:42–64
Hoshi H, Hao W, Fujita Y, Funayama A, Miyauchi Y, Hashimoto K et al (2012) Aldehyde-stress resulting from Aldh2 mutation promotes osteoporosis due to impaired osteoblastogenesis. J Bone Miner Res 27:2015–2023
Liao S, Wu N, Gong D, Tang X, Yin T, Zhang H et al (2020) Association of aldehydes exposure with obesity in adults. Ecotoxicol Environ Saf 201:110785
Barrera G, Pizzimenti S, Daga M, Dianzani C, Arcaro A, Cetrangolo GP et al (2018) Lipid peroxidation-derived aldehydes, 4-hydroxynonenal and malondialdehyde in aging-related disorders. Antioxidants (Basel) 7:102
Chrostek L, Jelski W, Szmitkowski M, Puchalski Z (2003) Gender-related differences in hepatic activity of alcohol dehydrogenase isoenzymes and aldehyde dehydrogenase in humans. J Clin Lab Anal 17:93–96
Acknowledgements
We thank members of the National Center for Health Statistics of the Centers for Disease Control and Prevention and the participants who enrolled in the National Health and Nutrition Examination Survey.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
PZ, XFX, and CPC designed the study and collected the data. PZ, XFX, and JMR analyzed the data. PZ, CPC and JMR wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethic approval
The National Center for Health Statistics institutional review board approved the study, and written informed consent was obtained from all participants included in the study.
Research involving human participants
NHANES was performed by the National Center for Health Statistics (NCHS) of the Centers for Disease Control and Prevention (CDC), and was approved by the institutional review board of the NCHS.
Informed consent
All participants provided written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
40618_2022_1840_MOESM2_ESM.tif
Supplementary Fig. 2. Correlation between exposure to different aldehydes. Pearson’s correlation coefficients were determined between serum concentrations of six aldehydes in US adults from NHANES 2013-2014. Darker color represents higher levels of correlation coefficients. BNA, benzaldehyde; BTA, butyraldehyde; HPA, heptanaldehyde; HXA, hexanaldehyde; IOA, isopentanaldehyde; POA, propanaldehyde; * p < 0.05, ** p < 0.01.
Rights and permissions
About this article
Cite this article
Zhu, P., Xiong, X., Chen, C. et al. Association of aldehyde exposure with bone mineral density in the national health and nutrition examination survey (NHANES 2013–2014). J Endocrinol Invest 45, 2085–2096 (2022). https://doi.org/10.1007/s40618-022-01840-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-022-01840-4