Abstract
Introduction
Vitamin D is involved in the regulatory mechanisms of ovarian function and is frequently low in PCOS patients. Since obesity and hyperinsulinemic state negatively influenced vitamin D levels, therefore, we evaluated the production of vitamin D at the ovarian level only in lean and normoinsulinemic PCOS subjects. Basal, GnRH analogue-induced ovarian production of 25OH-vitamin D (VitD) and a direct sampling at ovarian vein level were investigated.
Methods
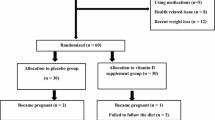
Basal and GnRH analogue-induced hormone levels were evaluated at peripheral level in 45 subjects, aged 18–39 years, and in 22 healthy women with age- and BMI-matched as controls. In 12 PCOS patients, undergoing laparoscopy, a venous sampling at both peripheral and ovarian level was further done. All subjects presented low VitD levels, appropriate to the season and with no difference between PCOS and control subjects.
Results
GnRH analogue significantly stimulated plasma LH, FSH, 17-OHP and estradiol secretion (p from < 0.05 to < 0.001 vs basal levels), whereas no effect was observed on both serum AMH and VitD concentrations in all groups. A significant difference (p < 0.006), between peripheral and ovarian veins, was observed in both AMH and estradiol levels in PCOS subjects, while no gradient of VitD was detected.
Conclusions
All patients presented with low VitD levels. The absence of any VitD variation, both at basal and after GnRH analogue administration, or at peripheral-ovarian vein gradient, suggests no pituitary–ovarian axis involvement in VitD production or its direct ovarian production in lean and normoinsulinemic PCOS subjects.




Similar content being viewed by others
References
DeLuca HF (1689S) Overview of general physiologic features and functions of vitamin D. Am J Clin Nutr 80:1689S–S1696
Zhu J, DeLuca HF (2012) Vitamin D 25-hydroxylase—four decades of searching, are we there yet? Arch Biochem Biophys 523:30–36
Cheng JB, Motola DL, Mangelsdorf DJ et al (2003) De-orphanization of cytochrome P450 2R1: a microsomal vitamin D 25-hydroxilase. J Biol Chem 278:38084–38093
Foresta C, Strapazzon G, De Toni L et al (2011) Bone mineral density and testicular failure: evidence for a role of vitamin D 25-hydroxylase in human testis. J Clin Endocrinol Metab 96:646E–E652
Jensen MB (2014) Vitamin D and male reproduction. Nat Rev Endocrinol 10(3):175–186
Karras S, Anagnostis P, Kotsa K et al (2016) Vitamin D and gonadal function in man: a potential inverse U-shaped association. Andrology 4(3):542–544
Bièche I, Narjoz C, Asselah T et al (2007) Reverse transcriptase-PCR quantification of mRNA levels from cytochrome (CYP)1, CYP2 and CYP3 families in 22 different human tissues. Pharmacogenet Genom 17(9):731–742
Pérez-Fernández R, Alonso M, Segura C et al (1997) Vitamin D receptor gene expression in human pituitary gland. Life Sci 60:35–42
Wang Y, Zhu J, DeLuca HF (2012) Where is the vitamin D receptor? Arch Biochem Biophys 523:123–133
Irani M, Merhi Z (2014) Role of vitamin D in ovarian physiology and its implication in reproduction: a systematic review. Fertil Steril 102(2):460–468
Shahrokhi SZ, Ghaffari F, Kazerouni F (2016) Role of vitamin D in female. Reprod Clin Chim Acta 455:33–38
Heyden EL, Wimalawansa SJ (2018) Vitamin D: effects on human reproduction, pregnancy, and fetal well-being. J Steroid Biochem Mol Biol 180(41):50
Zhao J, Liu S, Wang Y et al (2019) Vitamin D improves in-vitro fertilization outcomes in infertile women with polycystic ovary syndrome and insulin resistance. Minerva Med 110(3):199–208. https://doi.org/10.23736/S0026-4806.18.05946-3
Bakhshalizadeh S, Amidi F, Alleyassin A et al (2017) Modulation of steroidogenesis by vitamin D3 in granulosa cells of the mouse model of polycystic ovarian syndrome. Syst Biol Reprod Med 63(3):150–161
Thomson RL, Spedding S, Buckley JD (2012) Vitamin D in etiology and management of polycystic ovary syndrome. Clin Endocrinol 77:343–350
Menichini D, Facchinetti F (2019) Effects of vitamin D supplementation in women with polycystic ovary syndrome: a review. Gynecol Endocrinol 12:1–5. https://doi.org/10.1080/09513590.2019.1625881
The Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group (2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 81:19–25
Ferriman D, Gallwey JD (1961) Clinical assessment of body hair growth in women. J Clin Endocrinol Metab 21:1440–1447
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22:1462–1470
Mozzanega B, Mioni R, Granzotto M et al (2004) Obesity reduces the expression of GLUT4 in the endometrium of normoinsulinemic women affected by the polycystic ovary syndrome. Ann NY Acad Sci 1034:364–374
Mioni R, Chiarelli S, Xamin N et al (2004) Evidence for the presence of glucose transporter 4 in the endometrium and its regulation in polycystic ovary syndrome patients. J Clin Endocrinol Metab 89:4089–4096
Turra J, Granzotto M, Gallea M et al (2015) Sertoli–Leydig cell tumors: hormonal profile after dynamic test with GnRH analogue: triptorelin represents a useful tool to evaluate tumoral hyperandrogenism. Gynecol Endocrinol 31:18–21. https://doi.org/10.3109/09513590.2014.958993(Epub 2014 Oct 9PMID: 25299229)
Gallea M, Granzotto M, Azzolini S et al (2014) Insulin and body weight but not hyperandrogenism seem involved in seasonal serum 25-OH-vitamin D3 levels in subjects affected by PCOS. Gynecol Endocrinol 13:1–7
Kinuta K, Tanaka H, Moriwake T et al (2000) Vitamin D is an important factor in estrogen biosynthesis of both female and male gonads. Endocrinology 141:1317–1324
Yoshizawa T, Handa Y, Uematsu Y (1997) Mice lacking the vitamin D receptor exhibit impaired bone formation, uterine hypoplasia and growth retardation after weaning. Nat Genet 16:391–396
Panda DK, Miao D, Tremblay ML et al (2001) Targeted ablation of the 25-hydroxy vitamin D 1a-hydroxylase enzyme: evidence for skeletal, reproductive and immune dysfunction. Proc Natl Acad Sci 98:7498–75031
Ferlin A, Selice R, Carraro U et al (2013) Testicular function and bone metabolism—beyond testosterone. Nat Rev Endocrinol 9:548–554
Rosenfield RL, Burstein S, Cuttler L et al (1989) Use of nafarelin for testing pituitary-ovarian function. J Reprod Med 34:1044–1050
Fulghesu M, Angioni S, Belosi C et al (2006) Pituitary-ovarian response to the gonadotrophin-releasing hormone-agonist test in anovulatory patients with polycystic ovary syndrome: predictive role of ovarian stroma. Clin Endocrinol 65:396–401
Wojtusik J, Johnson PA (2012) Vitamin D regulates anti-Mullerian hormone expression in granulosa cells of the hen. Biol Reprod 86:91
Qi X, Pang Y, Qiao J (2016) The role of anti-Müllerian hormone in the pathogenesis and pathophysiological characteristics of polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol 199:82–87
Mioni R, Zuliani L, Azzolini S et al (2011) Anti-Müllerian hormone concentrations in hyperinsulinemic women with polycystic ovary syndrome before and after metformin therapy. J Endocrinol Investig 34:16
Hattori Y, Sato T, Okada H et al (2013) Comparison of follicular fluid and serum anti-Mullerian hormone levels as predictors of the outcome of assisted reproductive treatment. Eur J Obstet Gynecol Reprod Biol 169:252–256
Acknowledgements
We kindly thank Dr.ssa Barbara Martin, Mrs Mariangela Auleta, Mrs Sonia Leandri, and Mr Flaviano Favaro for their expert nursing and technical assistance. We are also grateful to Dr.ssa Patricia Padova for her help with scientific English.
Funding
The study was partially supported by a Grant (n.A00000D150 SICDE PPR/08) awarded to Prof. Roberto Vettor by the University of Padua, Italy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
The procedures used were in accordance with the guidelines of the Helsinki Declaration on human experimentation. The investigational nature of the study was explained to all participants.
Informed consent
Informed consent was obtained, according to the Local Ethical Committee (Protocol number: AO: 0431, October 2014).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mioni, R., Gallea, M., Granzotto, M. et al. Ovarian 25OH-vitamin D production in young women affected by polycystic ovary syndrome. J Endocrinol Invest 43, 1623–1630 (2020). https://doi.org/10.1007/s40618-020-01247-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-020-01247-z