Abstract
Purpose
Somatostatin analogs (SSAs) are considered one of the most effective medical treatments for patients with growth hormone-secreting pituitary adenomas (GH-PAs). The postoperative electron microscopy (EM) pathological subtype and SSTR2 expression in the tumor are the most established predictors of patient response to SSA therapy. The aim of this study was to evaluate how will magnetic resonance spectroscopy (MRS) measurements before surgery predict the EM pathological subtypes and SSTR2 expression of tumors, and thereby serve as an indicator for the therapeutic sensitivity to SSAs of patients with GH-PAs.
Methods
Eighteen patients with GH pituitary macroadenomas who underwent transsphenoidal surgery were included in this retrospective study. The preoperative MRS data and T2 signal intensity were obtained from patients by 1.5 T MR spectroscopy of the sellar mass. The EM pathological subtypes of tumors were determined after surgery through examination of cell granulations. The expressions of somatostatin receptor 2 (SSTR2), SSTR5, P21, P27, and Ki-67 were evaluated by immunohistochemistry.
Results
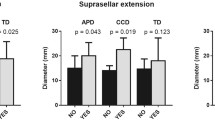
The MRS parameters that were found to significantly predict the EM pathological subtypes of tumors, as calculated by the receiver operating characteristic curve, were the choline (Ch) value at 3140.5 MR units (sensitivity 69.2%, specificity 100%) and the choline/creatine (Ch/Cr) ratio at 1.27 (sensitivity 92.3%, specificity 100%). Further, the Ch/Cr ratio, but not other MRS data, was shown to negatively correlate with the expression of SSTR2 (P = 0.02). The Ch/Cr ratio was also found to positively correlate with the Ki-67 value (P < 0.05) and T2 signal (P < 0.05), but not with other factors that were examined in this study. Moreover, the Ch/Cr ratio could predict the EM pathological subtypes of tumors with an accuracy of 83.3% (5/6) for patients with an isointense T2 signal.
Conclusion
The Ch/Cr ratio by MRS could effectively predict the tumor subtype and was significantly correlated with the expression of SSTR2, which was consistent with other predictors. It was also able to distinguish the patients with isointense T2 signals. Our results provide a potentially new and non-invasive method to predict the response to SSAs in patients with GH pituitary macroadenomas.



Similar content being viewed by others
References
Colao A, Ferone D, Marzullo P, Lombardi G (2004) Systemic complications of acromegaly: epidemiology, pathogenesis, and management. Endocr Rev 25(1):102–152. https://doi.org/10.1210/er.2002-0022
Chanson P, Salenave S (2008) Acromegaly. Orphanet J Rare Dis 3:17. https://doi.org/10.1186/1750-1172-3-17
Melmed S, Colao A, Barkan A, Molitch M, Grossman AB, Kleinberg D, Clemmons D, Chanson P, Laws E, Schlechte J, Vance ML, Ho K, Giustina A, Acromegaly Consensus G (2009) Guidelines for acromegaly management: an update. J Clin Endocrinol Metab 94(5):1509–1517. https://doi.org/10.1210/jc.2008-2421
Puig Domingo M (2015) Treatment of acromegaly in the era of personalized and predictive medicine. Clin Endocrinol (Oxf) 83(1):3–14. https://doi.org/10.1111/cen.12731
Nomikos P, Buchfelder M, Fahlbusch R (2005) The outcome of surgery in 668 patients with acromegaly using current criteria of biochemical ‘cure’. Eur J Endocrinol 152(3):379–387. https://doi.org/10.1530/eje.1.01863
Mercado M, Borges F, Bouterfa H, Chang TC, Chervin A, Farrall AJ, Patocs A, Petersenn S, Podoba J, Safari M, Wardlaw J, Group SBS (2007) A prospective, multicentre study to investigate the efficacy, safety and tolerability of octreotide LAR (long-acting repeatable octreotide) in the primary therapy of patients with acromegaly. Clin Endocrinol (Oxf) 66(6):859–868. https://doi.org/10.1111/j.1365-2265.2007.02825.x
Kiseljak-Vassiliades K, Carlson NE, Borges MT, Kleinschmidt-DeMasters BK, Lillehei KO, Kerr JM, Wierman ME (2015) Growth hormone tumor histological subtypes predict response to surgical and medical therapy. Endocrine 49(1):231–241. https://doi.org/10.1007/s12020-014-0383-y
Gatto F, Feelders RA, van der Pas R, Kros JM, Waaijers M, Sprij-Mooij D, Neggers SJ, van der Lelij AJ, Minuto F, Lamberts SW, de Herder WW, Ferone D, Hofland LJ (2013) Immunoreactivity score using an anti-sst2A receptor monoclonal antibody strongly predicts the biochemical response to adjuvant treatment with somatostatin analogs in acromegaly. J Clin Endocrinol Metab 98(1):E66–E71. https://doi.org/10.1210/jc.2012-2609
Fougner SL, Bollerslev J, Svartberg J, Oksnes M, Cooper J, Carlsen SM (2014) Preoperative octreotide treatment of acromegaly: long-term results of a randomised controlled trial. Eur J Endocrinol 171(2):229–235. https://doi.org/10.1530/EJE-14-0249
Bandgar TR, Sarathi V, Shivane V, Bansode N, Menon PS, Shah NS (2010) The value of an acute octreotide suppression test in predicting response to long-term somatostatin analogue therapy in patients with acromegaly. J Postgrad Med 56(1):7–11. https://doi.org/10.4103/0022-3859.62421
Halperin I, Nicolau J, Casamitjana R, Sesmilo G, Serra-Prat M, Palomera E, Puig-Domingo M (2008) A short acute octreotide test for response prediction of long-term treatment with somatostatin analogues in acromegalic patients. Hormone Metab Res 40(6):422–426. https://doi.org/10.1055/s-2008-1065339
de Herder WW, Taal HR, Uitterlinden P, Feelders RA, Janssen JA, van der Lely AJ (2005) Limited predictive value of an acute test with subcutaneous octreotide for long-term IGF-I normalization with Sandostatin LAR in acromegaly. Eur J Endocrinol 153(1):67–71. https://doi.org/10.1530/eje.1.01935
Pokrajac A, Claridge AG, Shakoor SK, Trainer PJ (2006) The octreotide test dose is not a reliable predictor of the subsequent response to somatostatin analogue therapy in patients with acromegaly. Eur J Endocrinol 154(2):267–274. https://doi.org/10.1530/eje.1.02073
Heck A, Ringstad G, Fougner SL, Casar-Borota O, Nome T, Ramm-Pettersen J, Bollerslev J (2012) Intensity of pituitary adenoma on T2-weighted magnetic resonance imaging predicts the response to octreotide treatment in newly diagnosed acromegaly. Clin Endocrinol (Oxf) 77(1):72–78. https://doi.org/10.1111/j.1365-2265.2011.04286.x
Puig-Domingo M, Resmini E, Gomez-Anson B, Nicolau J, Mora M, Palomera E, Marti C, Halperin I, Webb SM (2010) Magnetic resonance imaging as a predictor of response to somatostatin analogs in acromegaly after surgical failure. J Clin Endocrinol Metab 95(11):4973–4978. https://doi.org/10.1210/jc.2010-0573
Heck A, Emblem KE, Casar-Borota O, Bollerslev J, Ringstad G (2016) Quantitative analyses of T2-weighted MRI as a potential marker for response to somatostatin analogs in newly diagnosed acromegaly. Endocrine 52(2):333–343. https://doi.org/10.1007/s12020-015-0766-8
Oz G, Alger JR, Barker PB, Bartha R, Bizzi A, Boesch C, Bolan PJ, Brindle KM, Cudalbu C, Dincer A, Dydak U, Emir UE, Frahm J, Gonzalez RG, Gruber S, Gruetter R, Gupta RK, Heerschap A, Henning A, Hetherington HP, Howe FA, Huppi PS, Hurd RE, Kantarci K, Klomp DW, Kreis R, Kruiskamp MJ, Leach MO, Lin AP, Luijten PR, Marjanska M, Maudsley AA, Meyerhoff DJ, Mountford CE, Nelson SJ, Pamir MN, Pan JW, Peet AC, Poptani H, Posse S, Pouwels PJ, Ratai EM, Ross BD, Scheenen TW, Schuster C, Smith IC, Soher BJ, Tkac I, Vigneron DB, Kauppinen RA, Group MRSC (2014) Clinical proton MR spectroscopy in central nervous system disorders. Radiology 270(3):658–679. https://doi.org/10.1148/radiol.13130531
Di Costanzo A, Scarabino T, Trojsi F, Popolizio T, Bonavita S, de Cristofaro M, Conforti R, Cristofano A, Colonnese C, Salvolini U, Tedeschi G (2014) Recurrent glioblastoma multiforme versus radiation injury: a multiparametric 3-T MR approach. Radiol Med (Torino) 119(8):616–624. https://doi.org/10.1007/s11547-013-0371-y
Law M, Yang S, Wang H, Babb JS, Johnson G, Cha S, Knopp EA, Zagzag D (2003) Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. Am J Neuroradiol 24(10):1989–1998
Bluml S, Margol AS, Sposto R, Kennedy RJ, Robison NJ, Vali M, Hung LT, Muthugounder S, Finlay JL, Erdreich-Epstein A, Gilles FH, Judkins AR, Krieger MD, Dhall G, Nelson MD, Asgharzadeh S (2016) Molecular subgroups of medulloblastoma identification using noninvasive magnetic resonance spectroscopy. Neuro-oncology 18(1):126–131. https://doi.org/10.1093/neuonc/nov097
Chernov MF, Kawamata T, Amano K, Ono Y, Suzuki T, Nakamura R, Muragaki Y, Iseki H, Kubo O, Hori T, Takakura K (2009) Possible role of single-voxel (1)H-MRS in differential diagnosis of suprasellar tumors. J Neurooncol 91(2):191–198. https://doi.org/10.1007/s11060-008-9698-y
Alba M, Hall CM, Whatmore AJ, Clayton PE, Price DA, Salvatori R (2004) Variability in anterior pituitary size within members of a family with GH deficiency due to a new splice mutation in the GHRH receptor gene. Clin Endocrinol (Oxf) 60(4):470–475. https://doi.org/10.1111/j.1365-2265.2004.02003.x
Knosp E, Steiner E, Kitz K, Matula C (1993) Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery 33(4):610–617 (discussion 617–618)
Horvath E, Kovacs K (1976) Ultrastructural classification of pituitary adenomas. Can J Neurol Sci 3(1):9–21
Fougner SL, Casar-Borota O, Heck A, Berg JP, Bollerslev J (2012) Adenoma granulation pattern correlates with clinical variables and effect of somatostatin analogue treatment in a large series of patients with acromegaly. Clin Endocrinol (Oxf) 76(1):96–102. https://doi.org/10.1111/j.1365-2265.2011.04163.x
Bhayana S, Booth GL, Asa SL, Kovacs K, Ezzat S (2005) The implication of somatotroph adenoma phenotype to somatostatin analog responsiveness in acromegaly. J Clin Endocrinol Metab 90(11):6290–6295. https://doi.org/10.1210/jc.2005-0998
Fougner SL, Borota OC, Berg JP, Hald JK, Ramm-Pettersen J, Bollerslev J (2008) The clinical response to somatostatin analogues in acromegaly correlates to the somatostatin receptor subtype 2a protein expression of the adenoma. Clin Endocrinol (Oxf) 68(3):458–465. https://doi.org/10.1111/j.1365-2265.2007.03065.x
Wildemberg LE, Neto LV, Costa DF, Nasciuti LE, Takiya CM, Alves LM, Rebora A, Minuto F, Ferone D, Gadelha MR (2013) Low somatostatin receptor subtype 2, but not dopamine receptor subtype 2 expression predicts the lack of biochemical response of somatotropinomas to treatment with somatostatin analogs. J Endocrinol Invest 36(1):38–43. https://doi.org/10.3275/8305
Taboada GF, Luque RM, Neto LV, Machado Ede O, Sbaffi BC, Domingues RC, Marcondes JB, Chimelli LM, Fontes R, Niemeyer P, de Carvalho DP, Kineman RD, Gadelha MR (2008) Quantitative analysis of somatostatin receptor subtypes (1–5) gene expression levels in somatotropinomas and correlation to in vivo hormonal and tumor volume responses to treatment with octreotide LAR. Eur J Endocrinol 158(3):295–303. https://doi.org/10.1530/EJE-07-0562
Kasuki L, Wildemberg LE, Neto LV, Marcondes J, Takiya CM, Gadelha MR (2013) Ki-67 is a predictor of acromegaly control with octreotide LAR independent of SSTR2 status and relates to cytokeratin pattern. Eur J Endocrinol 169(2):217–223. https://doi.org/10.1530/EJE-13-0349
Kiseljak-Vassiliades K, Xu M, Mills TS, Smith EE, Silveira LJ, Lillehei KO, Kerr JM, Kleinschmidt-DeMasters BK, Wierman ME (2015) Differential somatostatin receptor (SSTR) 1-5 expression and downstream effectors in histologic subtypes of growth hormone pituitary tumors. Mol Cell Endocrinol 417:73–83. https://doi.org/10.1016/j.mce.2015.09.016
Soares DP, Law M (2009) Magnetic resonance spectroscopy of the brain: review of metabolites and clinical applications. Clin Radiol 64(1):12–21. https://doi.org/10.1016/j.crad.2008.07.002
Tamiya T, Kinoshita K, Ono Y, Matsumoto K, Furuta T, Ohmoto T (2000) Proton magnetic resonance spectroscopy reflects cellular proliferative activity in astrocytomas. Neuroradiology 42(5):333–338
Baltzer PA, Dietzel M (2013) Breast lesions: diagnosis by using proton MR spectroscopy at 1.5 and 3.0 T—systematic review and meta-analysis. Radiology 267(3):735–746. https://doi.org/10.1148/radiol.13121856
Sevcenco S, KrSSASsk M, Javor D, Ponhold L, Kuehhas FE, Fajkovic H, Haitel A, Shariat SF, Baltzer PA (2015) Diagnosis of renal tumors by in vivo proton magnetic resonance spectroscopy. World J Urol 33(1):17–23. https://doi.org/10.1007/s00345-014-1272-y
Acknowledgements
This work was supported by National Natural Science Foundation of China (no. 81528014) and New Clinical Technology Project of Xinqiao Hospital (no. 2015LCXJS034). The authors sincerely thank Prof. Qinglai Yang (Department of Microbiology and Immunology, Emory University School of Medicine) for helping to proofread for language and to format the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Ethical standards
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
40618_2018_939_MOESM1_ESM.tif
Supplemental Figure 1. The schematic images of 1H-MRSI data. Green point represent area of GH adenoma(A) and temporal lobe as self control(B). C: Oscillogram of 1H-MRSI, CH, choline-containing compound, value 6369; Cr, creatine, value 1969; NAA, N-acetylaspartate, value 816. D and E: The T2 intensity of GH-PAs hypointense and hyperintense respectively. F: Sagittal view was used to calculate tumor volume and help to locate the green point used in A and B (TIFF 2985 kb)
40618_2018_939_MOESM2_ESM.tif
Supplemental Figure 2. The ultrastructure of GH adenoma cells under electronic microscope. A: Densely granulated (DG), GH tumor with a large number of spherical secretory granules, the longest diameter varies between 300-600 nm (×8900). B: Sparsely granulated (SG), GH tumors with fibrous bodies and reduced size and number of secretory granules, the majority diameter were measured only 100-250 nm (×8900) (TIFF 2079 kb)
40618_2018_939_MOESM3_ESM.tif
Supplemental Figure 3. Immunohistochemical expression of SSTR2, SSTR5, P27 and P21 in GH-PAs. Representative examples of low—IRS(1-3), medium—IRS(4-9), and high—IRS(9-12) are demonstrated (magnification,×400) (TIFF 3462 kb)
Rights and permissions
About this article
Cite this article
Hu, J., Yan, J., Zheng, X. et al. Magnetic resonance spectroscopy may serve as a presurgical predictor of somatostatin analog therapy response in patients with growth hormone-secreting pituitary macroadenomas. J Endocrinol Invest 42, 443–451 (2019). https://doi.org/10.1007/s40618-018-0939-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-018-0939-4