Abstract
Objective
The discrimination between recurrent glioma and radiation injury is often a challenge on conventional magnetic resonance imaging (MRI). We verified whether adding and combining proton MR spectroscopic imaging (1H-MRSI), diffusion-weighted imaging (DWI) and perfusion-weighted imaging (PWI) information at 3 Tesla facilitate such discrimination.
Materials and methods
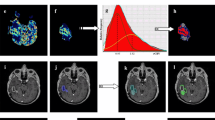
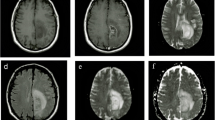
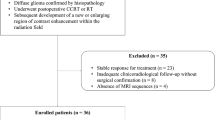
Twenty-nine patients with histologically verified high-grade gliomas, who had undergone surgical resection and radiotherapy, and had developed new contrast-enhancing lesions close to the treated tumour, underwent MRI, 1H-MRSI, DWI and PWI at regular time intervals. The metabolite ratios choline (Cho)/normal( n )Cho n , N-acetylaspartate (NAA)/NAA n , creatine (Cr)/Cr n , lactate/lipids (LL)/LL n , Cho/Cr n , NAA/Cr n , Cho/NAA, NAA/Cr and Cho/Cr were derived from 1H-MRSI; the apparent diffusion coefficient (ADC) from DWI; and the relative cerebral blood volume (rCBV) from PWI.
Results
In serial MRI, recurrent gliomas showed a progressive enlargement, and radiation injuries showed regression or no modification. Discriminant analysis showed that discrimination accuracy was 79.3 % when considering only the metabolite ratios (predictor, Cho/Cr n ), 86.2 % when considering ratios and ADC (predictors, Cho/Cr n and ADC), 89.7 % when considering ratios and rCBV (predictors, Cho/Cr n , Cho/Cr and rCBV), and 96.6 % when considering ratios, ADC and rCBV (predictors, Cho/Cho n , ADC and rCBV).
Conclusions
The multiparametric 3-T MR assessment based on 1H-MRSI, DWI and PWI in addition to MRI is a useful tool to discriminate tumour recurrence/progression from radiation effects.


Similar content being viewed by others
References
Siu A, Wind JJ, Iorgulescu JB et al (2012) Radiation necrosis following treatment of high grade glioma: a review of the literature and current understanding. Acta Neurochir 154:191–201
Alexiou GA, Tsiouris S, Kyritsis AP et al (2009) Glioma recurrence versus radiation necrosis: accuracy of current imaging modalities. J Neurooncol 95:1–11
McPherson CM, Warnick RE (2004) Results of contemporary surgical management of radiation necrosis using frameless stereotaxis and intraoperative magnetic resonance imaging. J Neurooncol 68:41–47
Scarabino T, Popolizio T, Trojsi F et al (2009) Role of advanced MR imaging modalities in diagnosing cerebral gliomas. Radiol Med 114:448–460
Jain R, Narang J, Sundgren PM et al (2010) Treatment induced necrosis versus recurrent/progressing brain tumor: going beyond the boundaries of conventional morphologic imaging. J Neurooncol 100:17–29
Fink JR, Carr RB, Matsusue E et al (2012) Comparison of 3 Tesla proton MR spectroscopy, MR perfusion and MR diffusion for distinguishing glioma recurrence from post treatment effects. J Magn Reson Imaging 35:56–63
Hu X, Wong KK, Young GS et al (2011) Support vector machine multiparametric MRI identification of pseudoprogression from tumor recurrence in patients with resected glioblastoma. J Magn Reson Imaging 33:296–305
Matsusue E, Fink JR, Rockhill JK et al (2010) Distinction between glioma progression and post-radiation change by combined physiologic MR imaging. Neuroradiology 52:297–306
Bobek-Billewicz B, Stasik-Pres G, Majchrzak H, Zarudzki L (2010) Differentiation between brain tumor recurrence and radiation injury using perfusion, diffusion-weighted imaging and MR spectroscopy. Folia Neuropathol 48:81–92
Zeng QS, Li CF, Liu H et al (2007) Distinction between recurrent glioma and radiation injury using magnetic resonance spectroscopy in combination with diffusion-weighted imaging. Int J Radiat Oncol Biol Phys 68:151–158
Rock JP, Scarpace L, Hearshen D et al (2004) Associations among magnetic resonance spectroscopy, apparent diffusion coefficients, and image-guided histopathology with special attention to radiation necrosis. Neurosurgery 54:1111–1117
Henry RG, Vigneron DB, Fischbein NJ et al (2000) Comparison of relative cerebral blood volume and proton spectroscopy in patients with treated gliomas. AJNR Am J Neuroradiol 21:357–366
Di Costanzo A, Trojsi F, Tosetti M et al (2007) Proton MR spectroscopy of the brain at 3 T: an update. Eur Radiol 17:1651–1662
Scarabino T, Giannatempo GM, Popolizio T et al (2007) 3.0-T functional brain imaging: a 5-year experience. Radiol Med 112:97–112
Di Costanzo A, Pollice S, Trojsi F et al (2008) Role of perfusion-weighted imaging at 3 Tesla in the assessment of malignancy of cerebral gliomas. Radiol Med 113:134–143
Elias AE, Carlos RC, Smith EA et al (2011) MR spectroscopy using normalized and non-normalized metabolite ratios for differentiating recurrent brain tumor from radiation injury. Acad Radiol 18:1101–1108
Smith EA, Carlos RC, Junck LR et al (2009) Developing a clinical decision model: MR spectroscopy to differentiate between recurrent tumor and radiation change in patients with new contrast-enhancing lesions. AJR Am J Roentgenol 192:45–52
Zeng QS, Li CF, Zhang K et al (2007) Multivoxel 3D proton MR spectroscopy in the distinction of recurrent glioma from radiation injury. J Neurooncol 84:63–69
Rabinov JD, Lee PL, Barker FG et al (2002) In vivo 3-T MR spectroscopy in the distinction of recurrent glioma versus radiation effects: initial experience. Radiology 225:871–879
Rock JP, Hearshen D, Scarpace L et al (2002) Correlations between magnetic resonance spectroscopy and image-guided histopathology, with special attention to radiation necrosis. Neurosurgery 51:912–919
Chong VF, Rumpel H, Fan YF, Mukherji SK (2001) Temporal lobe changes following radiation therapy: imaging and proton MR spectroscopic findings. Eur Radiol 11:317–324
Dowling C, Bollen AW, Noworolski SM et al (2001) Preoperative proton MR spectroscopic imaging of brain tumors: correlation with histopathologic analysis of resection specimens. AJNR Am J Neuroradiol 22:604–612
Graves EE, Nelson SJ, Vigneron DB et al (2001) Serial proton MR spectroscopic imaging of recurrent malignant gliomas after gamma knife radiosurgery. AJNR Am J Neuroradiol 22:613–624
Kimura T, Sako K, Gotoh T et al (2001) In vivo single-voxel proton MR spectroscopy in brain lesions with ring-like enhancement. NMR Biomed 14:339–349
Movsas B, Li BS, Babb JS et al (2001) Quantifying radiation therapy-induced brain injury with whole-brain proton MR spectroscopy: initial observations. Radiology 221:327–331
Schlemmer HP, Bachert P, Herfarth KK et al (2001) Proton MR spectroscopic evaluation of suspicious brain lesions after stereotactic radiotherapy. AJNR Am J Neuroradiol 22:1316–1324
Taylor JS, Langston JW, Reddick WE et al (1996) Clinical value of proton magnetic resonance spectroscopy for differentiating recurrent or residual brain tumor from delayed cerebral necrosis. Int J Radiat Oncol Biol Phys 36:1251–1261
Usenius T, Usenius JP, Tenhunen M et al (1995) Radiation-induced changes in human brain metabolites as studied by 1H nuclear magnetic resonance spectroscopy in vivo. Int J Radiat Oncol Biol Phys 33:719–724
Xu JL, Li YL, Lian JM et al (2010) Distinction between postoperative recurrent glioma and radiation injury using MR diffusion tensor imaging. Neuroradiology 52:1193–1199
Hein PA, Eskey CJ, Dunn JF, Hug EB (2004) Diffusion-weighted imaging in the follow-up of treated high-grade gliomas: tumor recurrence versus radiation injury. AJNR Am J Neuroradiol 25:201–209
Sundgren PC, Fan X, Weybright P et al (2006) Differentiation of recurrent brain tumor versus radiation injury using diffusion tensor imaging in patients with new contrast-enhancing lesions. Magn Reson Imaging 24:1131–1142
Asao C, Korogi Y, Kitajima M et al (2005) Diffusion-weighted imaging of radiation-induced brain injury for differentiation from tumor recurrence. AJNR Am J Neuroradiol 26:1455–1460
Fatterpekar GM, Galheigo D, Narayana A et al (2012) Treatment-related change versus tumor recurrence in high-grade gliomas: a diagnostic conundrum: use of dynamic susceptibility contrast-enhanced (DSC) perfusion MRI. AJR Am J Roentgenol 198:19–26
Romano A, Rossi Espagnet MC, Calabria LFA et al (2012) Clinical applications of dynamic susceptibility contrast perfusion-weighted MR imaging in brain tumours. Radiol Med 117:445–460
Xu JL, Shi DP, Dou SW et al (2011) Distinction between postoperative recurrent glioma and delayed radiation injury using MR perfusion weighted imaging. J Med Imaging Radiat Oncol 55:587–594
Sugahara T, Korogi Y, Tomiguchi S et al (2000) Posttherapeutic intraaxial brain tumor: the value of perfusion-sensitive contrast-enhanced MR imaging for differentiating tumor recurrence from non neoplastic contrast-enhancing tissue. AJNR Am J Neuroradiol 21:901–909
Hu LS, Baxter LC, Smith KA et al (2009) Relative cerebral blood volume values to differentiate high-grade glioma recurrence from posttreatment radiation effect: direct correlation between image-guided tissue histopathology and localized dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging measurements. AJNR Am J Neuroradiol 30:552–558
Zheng S, Chheda MG, Verhaak RG (2012) Studying a complex tumor: potential and pitfalls. Cancer J 18:107–114
Perry A, Schmidt RE (2006) Cancer therapy-associated CNS neuropathology: an update and review of the literature. Acta Neuropathol 111:197–212
Di Costanzo A, ScarabinoT Trojsi F et al (2008) Proton MR spectroscopy of cerebral gliomas at 3 T: spatial heterogeneity, and tumour grade and extent. Eur Radiol 18:1727–1735
Di Costanzo A, Scarabino T, Trojsi F et al (2006) Multiparametric 3T MR approach to the assessment of cerebral gliomas: tumor extent and malignancy. Neuroradiology 48:622–631
Acknowledgments
The authors are grateful to Italia Di Maggio, Giovanni Miscio and Piero Ghedin for expert technical assistance.
Conflict of interest
Alfonso Di Costanzo, Tommaso Scarabino, Francesca Trojsi, Teresa Popolizio, Simona Bonavita, Mario de Cristofaro, Renata Conforti, Adriana Cristofano, Claudio Colonnese, Ugo Salvolini, Gioacchino Tedeschi declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Di Costanzo, A., Scarabino, T., Trojsi, F. et al. Recurrent glioblastoma multiforme versus radiation injury: a multiparametric 3-T MR approach. Radiol med 119, 616–624 (2014). https://doi.org/10.1007/s11547-013-0371-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-013-0371-y