Abstract
Purpose of Review
Interferon-induced transmembrane protein 3 (IFITM3) is a cellular restriction factor that blocks fusion between virus and host membranes. Here, we provide an introduction to IFITM3 and the biochemical regulation underlying its antiviral activity. Further, we analyze and summarize the published literature examining phenotypes of IFITM3 knockout mice upon infections with viral pathogens and discuss the controversial association between single nucleotide polymorphisms (SNPs) in the human IFITM3 gene and severe virus infections.
Recent Findings
Recent publications show that IFITM3 knockout mice experience more severe pathologies than wild-type mice in diverse virus infections, including infections with influenza A virus, West Nile virus, Chikungunya virus, Venezuelan equine encephalitis virus, respiratory syncytial virus, and cytomegalovirus. Likewise, numerous studies of humans of Chinese ancestry have associated the IFITM3 SNP rs12252-C with severe influenza virus infections, though examinations of other populations, such as Europeans, in which this SNP is rare, have largely failed to identify an association with severe infections. A second SNP, rs34481144-A, found in the human IFITM3 promoter has also recently been reported to be a risk allele for severe influenza virus infections.
Summary
There is significant evidence for a protective role of IFITM3 against virus infections in both mice and humans, though additional work is required to identify the range of pathogens restricted by IFITM3 and the mechanisms by which human SNPs affect IFITM3 levels or functionality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The interferon (IFN)-induced transmembrane proteins (IFITMs) 1, 2, and 3 are upregulated upon stimulation of cells by type I IFNs [1, 2], and these proteins subsequently block membrane fusion between virus and host membranes [3,4,5]. IFITM cDNAs were among the first IFN stimulated genes (ISGs) to be cloned and sequenced in the early 1980s [6]. However, the ability of IFITM3 to inhibit virus infections was not demonstrated until 2009 when researchers performing a genome-wide siRNA screen for influenza virus host dependency factors conversely discovered that IFITM3 knockdown resulted in increased influenza virus infection of cells [1]. Confirmatory reports were quickly published by several groups in which other screening studies converged upon the importance of IFITMs, and particularly IFITM3, in innate antiviral defense [2, 7,8,9]. Overexpressed IFITM3 potently inhibits influenza virus infection, and IFITM3 KO cells are highly susceptible to infection [1, 3, 10,11,12,13]. Remarkably, IFITM3 KO cells remain highly susceptible to influenza virus infection even after IFN treatments that make WT cells refractory to infection [13]. This in vitro work established that IFITM3 is an essential ISG for cellular defense against influenza virus and, further, that endogenous IFITMs 1 and 2, despite similar antiviral activities to IFITM3 when overexpressed [14], cannot compensate for loss of IFITM3.
IFITM3 is a 15-kDa membrane-associated protein that localizes primarily to endosomes and lysosomes. Evolutionary analysis indicates that an IFITM gene was acquired via horizontal gene transfer from bacteria to a single-celled mammalian ancestor [15]. Though the function of IFITMs in bacteria is entirely unexplored, expression of mycobacterial IFITMs in human cells provided mild resistance to influenza virus infection, suggesting that acquisition of an IFITM gene may have provided an evolutionary advantage against virus infections [16]. IFITM gene duplications are commonly observed in mammalian species [15, 17] and have been proposed to provide increased antiviral coverage of cell membranes by maintaining a highly conserved antiviral core amino acid region with divergent N- and C-termini that dictate protein localization and interactions [18]. Indeed, among the long list of viruses that have now been shown to be restricted by IFITM3, a commonality has emerged in that their membrane fusion reactions generally occur in endosomes where IFITM3 is most abundant [19, 20]. IFITM variants that show altered localization may restrict membrane fusion of a distinct subset of viruses, such as those that fuse at the plasma membrane [18, 21, 22].
IFITM3 Posttranslational Regulation
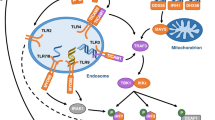
The study of posttranslational modifications of IFITM3 has provided numerous insights into its cellular trafficking, turnover/stability, and mechanism of action [23]. IFITM3 undergoes at least four distinct modifications on multiple amino acid residues (Fig. 1). (1) Phosphorylation at the IFITM3 N-terminal region on Y20 blocks the interaction of a YxxΦ trafficking motif (20-YEML-23 in human IFITM3) with the AP-2 endocytic adaptor protein complex that normally facilitates endocytosis of IFITM3 from the plasma membrane to endosomes and lysosomes (Fig. 1) [21, 24, 25]. Y20 phosphorylation is mediated by the Src family kinase Fyn and results in accumulation of IFITM3 at the plasma membrane and decreased antiviral activity against viruses, such as influenza virus, that enter cells through endocytosis and fuse with endosomes [24, 25]. While phosphorylation may decrease IFITM3 activity against influenza virus, the altered localization enhances its ability to restrict certain viruses that fuse at the plasma membrane as mentioned above [18, 21, 22]. Phosphorylation of IFITM3 is dynamic, involving currently unidentified phosphatases [24, 25]. Though its study led to a better understanding of IFITM3 trafficking, the relevant physiological triggers of phosphorylation and dephosphorylation remain to be identified. (2) Monomethylation of IFITM3 at K88 by the SET7 methyltransferase decreases its antiviral activity [26]. This methylation can also be removed by the histone demethylase LSD1, which is recruited to IFITM3 during IFN treatment of cells [27]. Additional studies will be required to determine the molecular mechanism by which K88 methylation inhibits IFITM3 and whether this inhibition is beneficial to cells. (3) Ubiquitination of IFITM3 can occur on each of four lysines within IFITM3 (K24, K83, K88, and K104) [11] and is primarily mediated by interaction with the E3 ubiquitin ligase NEDD4 via a conserved PPxY motif in IFITM3 (17-PPNY-20 in human IFITM3) (Fig. 1) [28]. Mutation of the IFITM3 ubiquitination sites or depletion of NEDD4 from cells results in increased stability of IFITM3 [11, 28]. As such, NEDD4 KO cells accumulate high levels of basal IFITM3 even in the absence of IFN stimulation and are thus resistant to infection with IFITM3-sensitive viruses [22, 28]. It is unknown whether IFITM3 is deubiquitinated in cells, and like methylation, it is also unclear whether the negative regulation of IFITM3 via ubiquitination is beneficial to cells given that these modifications limit the antiviral function of IFITM3. (4) S-palmitoylation of IFITM3 occurs on three highly conserved cysteine residues (C71, C72, and C105) and is the only modification discovered to date that is required for robust IFITM3 antiviral activity [2, 11, 29,30,31]. This lipid modification is constitutively added to IFITM3 in an irreversible manner [11] by at least three cellular palmitoyltransferase enzymes (ZDHHC3, ZDHHC7, and ZDHHC20) [13]. Nearly 100% of IFITM3 is palmitoylated on at least one cysteine, and examination of antiviral activity of IFITM3 mutants lacking specific cysteines suggests that C72 is particularly critical for inhibition of influenza virus infection [32]. The primary function of S-palmitoylation at C72 and the neighboring C71 is likely the targeting or anchoring of a highly conserved amphipathic helix (59-VWSLFNTLFM-68 in human IFITM3) that is required for inhibition of membrane fusion by IFITM3 and that is adjacent to these S-palmitoylation sites in the IFITM3 amino acid sequence (Fig. 1) [33•].
Human IFITM3 critical domains and posttranslational modification sites. The horizontal line represents the 133 amino acids of human IFITM3 from the N- to C-terminus. Colored boxes above the horizontal line represent known structural motifs or domains, and arrows point to characterized functions of these regions. Specific amino acid residues known to be posttranslationally modified are indicated below the horizontal line. Red lines indicate negative regulatory modifications and green lines indicate activating modifications. Phos, phosphorylation; Ub, ubiquitination; Palm, palmitoylation; Meth, monomethylation
IFITM3 Mechanisms of Action
The primary mechanism by which IFITM3 inhibits virus infection is via blockade of fusion pore formation between virus and host membranes [3, 5]. This prevents the entry of viral genomes into the cytosol and subsequent virus replication. Single particle imaging of fluorescently labeled viruses has revealed that hemifusion between virus and host membranes occurs in cells expressing IFITM3, but that the fusion process is stalled prior to formation of a membrane pore [4]. Insertion of the aforementioned IFITM3 S-palmitoylated amphipathic helix into the cytoplasm-facing leaflet of the host membrane bilayer has been proposed to stabilize a membrane intermediate of the fusion process, consistent with the well-characterized ability of amphipathic helices to induce or stabilize membrane curvature [33•].
The inhibition of fusion of incoming virus requires that IFITM3 is induced and present at inhibitory levels in the target cells. However, IFITMs may have additional functions subsequent to initial infection [34]. Groups studying HIV infections first showed that IFITMs induced in infected cells are incorporated into the membrane envelopes of nascent virions and that this presence of IFITMs limits virion infectivity [35, 36]. This effect was subsequently shown for numerous additional viruses and appears to be a wide-ranging additional function of IFITMs, including IFITM3 [37]. IFITMs in virions likely impair fusogenicity of the viruses, and this likely also involves the conserved amphipathic helix. Additional experiments are required to determine precisely how IFITM3 affects membrane fusion dynamics, and such experiments will not only be informative as to the mechanisms of action of IFITM3, but will also shed light on the fundamental fusion mechanisms used by enveloped viruses.
IFITM3 Limits Severity of Virus Infections in Mice
IFITM3 KO mice were initially generated to study a suspected role of IFITM3 in germ cell development in vivo [38]. However, neither IFITM3 KO mice nor mice lacking the entire IFITM locus (referred to as IFITMdel mice in the literature) possessed a defect in germ cells or reproduction [38]. Upon discovery of the antiviral activity of IFITM3 in vitro, these mice were re-examined for phenotypes during microbial infections (Table 1). IFITM3 KO mice were observed in two studies to experience increased weight loss and mortality from H1N1 and H3N2 influenza A virus infections, correlating with increased virus lung titers, systemic lymphopenia, and increased pro-inflammatory cytokine levels in the lungs [39, 40]. Remarkably, IFITMdel mice did not experience more severe infections than IFITM3 KO mice, suggesting that other IFITMs do not contribute significantly to influenza virus resistance in vivo [40]. In addition to the role of IFITM3 in the innate immune response, it also protects lung dendritic cells from productive infection with influenza virus, thus allowing their trafficking to draining lymph nodes and priming of CD8 T cells [46]. IFITM3 also protects resident memory CD8 T cells from infection and death in the lung during secondary influenza virus infections [47]. Thus, the importance of IFITM3 in primary and secondary influenza virus infections is multifactorial.
Additional viruses that show susceptibility to IFITM3 inhibition in vitro have also been examined in IFITM3 KO mice. Chikungunya virus infection in the footpad caused increased ankle joint swelling in IFITM3 KO versus WT mice, correlating with increased virus burden in the serum, spleen, and ankle early in infection [41•]. Similarly, IFITM3 KO mice experienced increased lethality upon footpad infection with Venezuelan equine encephalitis virus that was accompanied by increased virus titers in the spleen, liver, spinal cord, and, most significantly, brain [41•]. Together, these experiments demonstrate that IFITM3 is essential for limiting the severity of alphavirus infections in vivo.
Several flaviviruses, such as Dengue virus, West Nile virus, Zika virus, and hepatitis C virus, are also inhibited by IFITM3 in cell culture models [1, 48,49,50,51]. In vivo, subcutaneous infection with West Nile virus induced increased lethality in IFITM3 KO mice compared to that in WT mice [42]. This enhanced pathogenicity correlated with increased virus titers in the serum, lymph node, brain, and spinal cord. Using bone marrow reconstitutions, it was determined that IFITM3 plays essential roles in both hematopoietic and non-hematopoietic cells to restrict West Nile virus infection severity. Interestingly, unlike subcutaneous infection, intracranial infection did not result in significant differences in virus burden in the brain or spinal cord, suggesting that IFITM3 does not directly inhibit West Nile virus in the central nervous system [42].
IFITM3 KO mice also showed modest weight loss during respiratory syncytial virus infection that was absent in WT mice [45], consistent with effects of IFITM3 on this virus in vitro [44]. This weight loss correlated with increased levels of virus in the lungs and increased lymphocyte counts in lung tissue and lung lavage fluid of KO mice [45]. In general, mice are considered a poor model for respiratory syncytial virus infection since high doses of virus are required to achieve measurable infection of lung cells and since weight loss indicative of illness is not usually observed. The small amount of weight loss observed in IFITM3 KO mice suggests that IFITM3 may contribute to respiratory syncytial virus susceptibility in vivo [45], but its loss does not overcome the significant species restriction barrier for this virus in mice.
IFITM3 may also provide a protective effect in certain infections independent of its inhibition of virus entry into cells. Examination of cytomegalovirus infection of several cell types in vitro indicated that this virus is not restricted by IFITM3 [43•]. However, IFITM3 KO mice experience more severe weight loss and increased mortality compared to WT mice following intraperitoneal cytomegalovirus infection [43•]. These effects correlated with a general lymphopenia in circulation and in the spleens of KO mice early in infection. Virus burden and IL-6, TNF, and IFNα were concurrently increased in these spleen homogenates. Stimulation of IFITM3 KO dendritic cells in vitro with irradiated cytomegalovirus or Toll-like receptor ligands resulted in increased secretion of IL-6 compared to that of WT cells. Further, treatment of infected WT or KO mice with anti-IL-6 neutralizing antibodies caused a reduction in weight loss during infection and also restored NK cells to WT levels in the KO mice [43•]. Interestingly, IL-6 was also among the cytokines increased in the lungs of influenza virus-infected IFITM3 KO mice [39]. Overall, these results suggest that IFITM3 may possess a mechanistically uncharacterized ability to dampen IL-6 production [43•] and also that IFITM3 may play underappreciated roles in preventing cytokine-driven immunopathologies, even in infections that are not directly restricted by IFITM3.
IFITM3 Polymorphisms in Humans Are Linked to Severe Virus Infections
The well-defined ability of IFITM3 to prevent infection of cells by influenza virus, along with the increased virus-induced morbidity and mortality of IFITM3 KO mice, has prompted numerous research groups to examine whether single nucleotide polymorphisms (SNPs) in the human IFITM3 gene associate with influenza virus infections or infection severity (all studies to date are summarized in Table 2). The pioneering study in this field sequenced the IFITM3 gene regions of 53 patients hospitalized with severe 2009 H1N1 pandemic influenza A virus infection [39]. Three of these patients were homozygous for a SNP, rs12252-C, where the T majority allele is substituted with a C. Three out of 53 was a significant overrepresentation of the C allele as compared to matched European controls from 1000 genomes in which the SNP is rare. The authors of this study hypothesized that the C substitution alters a splice acceptor site, resulting in a truncated IFITM3 protein lacking its first 21 amino acids. Overexpression of the truncated IFITM3 variant provided less inhibition of influenza virus as compared to WT IFITM3 [39], and subsequent studies demonstrated that this mutant is mislocalized due to loss of its YxxΦ endocytosis motif as discussed above [18, 21, 24, 25]. While altered splicing and mislocalization of IFITM3 provided a satisfying mechanistic explanation for effects of the rs12252-C SNP, the existence of truncated IFITM3 protein has not been observed in cells homozygous for the C allele [39, 66•]. Likewise, studies in which RNA sequencing was performed reported that the predicted alternatively spliced transcripts were not detected in cells from individuals homozygous for the C allele [58, 66•]. Thus, whether there is a direct effect of rs12252-C on IFITM3 function or influenza virus infections remains unclear.
Several follow-up studies examining Europeans and other populations in which rs12252-C is rare have failed to find an association between the SNP and severe influenza virus infection [57,58,59,60,61,62], although some studies support a link between the SNP and mild influenza [56, 57]. In contrast, a strong overrepresentation of homozygous rs12252-C genotypes in severe influenza patients has been repeatedly observed in studies examining Chinese individuals in which the allele frequency for this SNP is greater than 50% in the general population [52,53,54,55]. This association is consistent between studies examining 2009 H1N1 pandemic virus [53, 55], H7N9 virus [53, 54], and seasonal influenza virus [52] infections. Likewise, Chinese individuals who were infected with Hantaan virus and experienced severe hemorrhagic fever were also more likely to be carriers or homozygous for rs12252-C than the general Chinese population [63]. A study examining HIV-infected Chinese individuals also reported that carriage of rs12252-C was more common among rapid progressors with higher viral load set points and a faster decline of CD4 T cells than in those with slower progression of disease [64]. However, results from this study have been cautiously interpreted by the field and will require additional corroboration since rs12252-C homozygous individuals were surprisingly underrepresented among the rapid progressors [64]. In sum, there is generally a strong association between severe influenza virus infections and homozygosity of the IFITM3 rs12252-C SNP in Chinese individuals, while infrequency of the SNP in Europeans makes it difficult to confidently assess its effects in this population (Table 2).
A recent landmark study examined a second IFITM3 SNP located within the IFITM3 gene promoter [65••]. A minor allele, rs34481144-A, was associated with severe influenza virus infections in a largely African American population and with death following influenza virus infection in a largely European population [65••]. Additionally, early replication of influenza virus in a third study group made up of individuals of European ancestry was also linked to carriage of this SNP [65••]. In the populations examined, this SNP ranged in allele frequency from 16.9 to 33.25%. Mechanistically, cells from individuals possessing the rs34481144-A SNP showed decreased IFITM3 mRNA and protein levels in a dose-responsive manner, correlating with the presence of one or two rs34481144-A alleles [65••]. This effect on IFITM3 gene expression was suggested to be due to differential occupancy of the IFITM3 promoter by activating versus inhibitory transcription factors [65••]. These intriguing new findings further support the importance of IFITM3 in antiviral defense in vivo, and indeed, SNPs in IFITM3 remain the only reproducibly observed human genetic associations with severe influenza virus infections [67].
Remaining Questions
We have only begun to understand the importance and functions of IFITM3 in antiviral defense in vivo. For example, only a limited number of the viruses restricted by IFITM3 in vitro have been studied in IFITM3 KO mice and even fewer infections have been examined in humans. To date, studies in humans have primarily focused on influenza virus infections. The discovery of the importance of the rs34481144-A SNP in controlling IFITM3 levels [65••] should facilitate the study of IFITM3 in additional human infections in diverse populations. Additionally, the discovery of a regulatory role for IFITM3 in IL-6 production [43•], along with a recent report that IFITM3 is a negative feedback inhibitor of the type I IFN induction pathway [68], may suggest that IFITM3 is a broad regulator of inflammation and that it may play important roles in infections in which the balance between pathogen control versus tissue pathology is determined by these cytokines. Interestingly, IFITM3 KO mice were not observed to be more susceptible to infections with Salmonella typhimurium, Citrobacter rodentium, Mycobacterium tuberculosis, or Plasmodium berghei [45] (Table 1). Thus, at present, the critical role of IFITM3 in antimicrobial defense appears to be limited to virus infections, though this remains an open question.
Given the strong and reproducible association between homozygosity of the IFITM3 rs12252-C SNP and severe influenza in the Chinese population, it will be important to determine the underlying mechanism by which this SNP influences disease. The initially proposed hypothesis that the SNP affects splicing [39] has not been supported by subsequent studies [58, 66•]. Whether rs12252-C influences IFITM3 gene splicing or protein levels in a cell type-specific manner and whether this SNP cosegregates with a different causative allele both remain unknown.
The antiviral effects of IFITM3 are reversed by the membrane-destabilizing drug amphotericin B, which is commonly prescribed to treat fungal infections in humans [69]. Mice treated with amphotericin B experience more severe influenza virus infections, similar to IFITM3 KO mice [69]. We recently determined that IFITM3 is the first known restriction factor for human metapneumovirus infection, and we confirmed that amphotericin B completely neutralizes the antiviral activity of IFITM3 against both influenza virus and human metapneumovirus [22]. Thus, it will be interesting and important to determine whether clinical use of amphotericin B has the unintended consequence of increasing susceptibility to or pathogenicity of these and other virus infections due to its neutralizing effects on IFITM3.
Antiviral restriction factors often drive the evolution of viruses to evade or inhibit virus restriction [70]. IFITM3 inhibits the membrane fusion of influenza virus, which is typically triggered by low pH in endosomes [3,4,5]. Given that endocytosis and membrane fusion are fundamental steps in the entry of all influenza viruses, all strains of the virus that have been tested to date are susceptible to IFITM3 restriction to varying degrees. The degree of susceptibility appears to be determined by the pH optimum at which the virus fuses. Viruses that fuse at low pH in late endosomes where IFITM3 is abundant are strongly restricted [71, 72]. Conversely, viruses that have evolved to fuse at a higher pH in early endosomes where IFITM3 is less abundant are partially resistant to IFITM3 restriction [71, 72]. Similarly, in vitro studies of HIV have shown that over time in culture the HIV env protein will evolve to evade restriction of the virus by IFITMs [73]. Lack of restriction by IFITMs is indeed a characteristic of transmitted founder HIV isolates derived from recently infected humans [74••, 75]. Interestingly, as HIV evolves its env protein in vivo to evade adaptive immune responses, restriction by IFITMs increases [74••, 75]. The ongoing evolutionary battle between viruses and IFITMs will be an exciting area for further investigation.
Conclusions
Several broad conclusions can be drawn from the research performed on IFITM3 over the past decade. First, IFITM3 is a broad inhibitor of membrane fusion between viruses and cell membranes and is particularly active against viruses that enter cells via endocytosis due to its primary localization at endosomes. Second, IFITM3 utilizes an amphipathic helix to block virus membrane fusion. Third, IFITM3 is regulated by multiple posttranslational modifications that determine its intracellular localization, activity, and abundance. Fourth, IFITM3 knockout mice experience increased susceptibility to a wide range of virus infections. Fifth, IFITM3 gene SNPs in the human population are correlated with severe virus infections. The continued study of IFITM3 and other IFITMs in terms of their precise mechanisms of action, their range of microbial restriction in vivo, effects of human SNPs, and possible viral countermeasures will continue to increase our understanding of virus-host interactions.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Brass AL, Huang IC, Benita Y, John SP, Krishnan MN, Feeley EM, et al. The IFITM proteins mediate cellular resistance to influenza A H1N1 virus, West Nile virus, and dengue virus. Cell. 2009;139(7):1243–54. https://doi.org/10.1016/j.cell.2009.12.017.
Yount JS, Moltedo B, Yang YY, Charron G, Moran TM, Lopez CB, et al. Palmitoylome profiling reveals S-palmitoylation-dependent antiviral activity of IFITM3. Nat Chem Biol. 2010;6(8):610–4. https://doi.org/10.1038/nchembio.405.
Feeley EM, Sims JS, John SP, Chin CR, Pertel T, Chen LM, et al. IFITM3 inhibits influenza A virus infection by preventing cytosolic entry. PLoS Pathog. 2011;7(10):e1002337. https://doi.org/10.1371/journal.ppat.1002337.
Desai TM, Marin M, Chin CR, Savidis G, Brass AL, Melikyan GB. IFITM3 restricts influenza A virus entry by blocking the formation of fusion pores following virus-endosome hemifusion. PLoS Pathog. 2014;10(4):e1004048. https://doi.org/10.1371/journal.ppat.1004048.
Li K, Markosyan RM, Zheng YM, Golfetto O, Bungart B, Li M, et al. IFITM proteins restrict viral membrane hemifusion. PLoS Pathog. 2013;9(1):e1003124. https://doi.org/10.1371/journal.ppat.1003124.
Friedman RL, Manly SP, McMahon M, Kerr IM, Stark GR. Transcriptional and posttranscriptional regulation of interferon-induced gene expression in human cells. Cell. 1984;38(3):745–55.
Schoggins JW, Wilson SJ, Panis M, Murphy MY, Jones CT, Bieniasz P, et al. A diverse range of gene products are effectors of the type I interferon antiviral response. Nature. 2011;472(7344):481–5. https://doi.org/10.1038/nature09907.
Lu J, Pan Q, Rong L, He W, Liu SL, Liang C. The IFITM proteins inhibit HIV-1 infection. J Virol. 2011;85(5):2126–37. https://doi.org/10.1128/JVI.01531-10.
Weidner JM, Jiang D, Pan XB, Chang J, Block TM, Guo JT. Interferon-induced cell membrane proteins, IFITM3 and tetherin, inhibit vesicular stomatitis virus infection via distinct mechanisms. J Virol. 2010;84(24):12646–57. https://doi.org/10.1128/JVI.01328-10.
John SP, Chin CR, Perreira JM, Feeley EM, Aker AM, Savidis G, et al. The CD225 domain of IFITM3 is required for both IFITM protein association and inhibition of influenza A virus and dengue virus replication. J Virol. 2013;87(14):7837–52. https://doi.org/10.1128/JVI.00481-13.
Yount JS, Karssemeijer RA, Hang HC. S-palmitoylation and ubiquitination differentially regulate interferon-induced transmembrane protein 3 (IFITM3)-mediated resistance to influenza virus. J Biol Chem. 2012;287(23):19631–41. https://doi.org/10.1074/jbc.M112.362095.
Hach JC, McMichael T, Chesarino NM, Yount JS. Palmitoylation on conserved and nonconserved cysteines of murine IFITM1 regulates its stability and anti-influenza A virus activity. J Virol. 2013;87(17):9923–7. https://doi.org/10.1128/JVI.00621-13.
McMichael TM, Zhang L, Chemudupati M, Hach JC, Kenney AD, Hang HC, et al. The palmitoyltransferase ZDHHC20 enhances interferon-induced transmembrane protein 3 (IFITM3) palmitoylation and antiviral activity. J Biol Chem. 2017;292(52):21517–26. https://doi.org/10.1074/jbc.M117.800482.
Huang IC, Bailey CC, Weyer JL, Radoshitzky SR, Becker MM, Chiang JJ, et al. Distinct patterns of IFITM-mediated restriction of filoviruses, SARS coronavirus, and influenza A virus. PLoS Pathog. 2011;7(1):e1001258. https://doi.org/10.1371/journal.ppat.1001258.
Almen MS, Bringeland N, Fredriksson R, Schioth HB. The Dispanins: a novel gene family of ancient origin that contains 14 human members. Plos One. 2012;7(2). https://doi.org/10.1371/journal.pone.0031961.
Melvin WJ, McMichael TM, Chesarino NM, Hach JC, Yount JS. IFITMs from mycobacteria confer resistance to influenza virus when expressed in human cells. Viruses. 2015;7(6):3035–52. https://doi.org/10.3390/v7062759.
Zhang Z, Liu J, Li M, Yang H, Zhang C. Evolutionary dynamics of the interferon-induced transmembrane gene family in vertebrates. PLoS One. 2012;7(11):e49265. https://doi.org/10.1371/journal.pone.0049265.
Compton AA, Roy N, Porrot F, Billet A, Casartelli N, Yount JS, et al. Natural mutations in IFITM3 modulate post-translational regulation and toggle antiviral specificity. EMBO Rep. 2016;17(11):1657–71. https://doi.org/10.15252/embr.201642771.
Perreira JM, Chin CR, Feeley EM, Brass AL. IFITMs restrict the replication of multiple pathogenic viruses. J Mol Biol. 2013;425(24):4937–55. https://doi.org/10.1016/j.jmb.2013.09.024.
Smith S, Weston S, Kellam P, Marsh M. IFITM proteins-cellular inhibitors of viral entry. Curr Opin Virol. 2014;4:71–7. https://doi.org/10.1016/j.coviro.2013.11.004.
Jia R, Xu F, Qian J, Yao Y, Miao C, Zheng YM, et al. Identification of an endocytic signal essential for the antiviral action of IFITM3. Cell Microbiol. 2014;16(7):1080–93. https://doi.org/10.1111/cmi.12262.
McMichael TM, Zhang Y, Kenney AD, Zhang L, Lu M, Chemudupati M et al. Opposing roles of endosomal innate immunity proteins IFITM3 and TLR7 in human metapneumovirus infection. bioRxiv. 2018. https://doi.org/10.1101/290957.
Chesarino NM, McMichael TM, Yount JS. Regulation of the trafficking and antiviral activity of IFITM3 by post-translational modifications. Future Microbiol. 2014;9(10):1151–63. https://doi.org/10.2217/fmb.14.65.
Jia R, Pan Q, Ding S, Rong L, Liu SL, Geng Y, et al. The N-terminal region of IFITM3 modulates its antiviral activity by regulating IFITM3 cellular localization. J Virol. 2012;86(24):13697–707. https://doi.org/10.1128/JVI.01828-12.
Chesarino NM, McMichael TM, Hach JC, Yount JS. Phosphorylation of the antiviral protein interferon-inducible transmembrane protein 3 (IFITM3) dually regulates its endocytosis and ubiquitination. J Biol Chem. 2014;289(17):11986–92. https://doi.org/10.1074/jbc.M114.557694.
Shan Z, Han Q, Nie J, Cao X, Chen Z, Yin S, et al. Negative regulation of interferon-induced transmembrane protein 3 by SET7-mediated lysine monomethylation. J Biol Chem. 2013;288(49):35093–103. https://doi.org/10.1074/jbc.M113.511949.
Shan J, Zhao B, Shan Z, Nie J, Deng R, Xiong R, et al. Histone demethylase LSD1 restricts influenza A virus infection by erasing IFITM3-K88 monomethylation. PLoS Pathog. 2017;13(12):e1006773. https://doi.org/10.1371/journal.ppat.1006773.
Chesarino NM, McMichael TM, Yount JS. E3 ubiquitin ligase NEDD4 promotes influenza virus infection by decreasing levels of the antiviral protein IFITM3. PLoS Pathog. 2015;11(8):e1005095. https://doi.org/10.1371/journal.ppat.1005095.
Yount JS, Zhang MM, Hang HC. Emerging roles for protein S-palmitoylation in immunity from chemical proteomics. Curr Opin Chem Biol. 2013;17(1):27–33. https://doi.org/10.1016/j.cbpa.2012.11.008.
Yount JS, Charron G, Hang HC. Bioorthogonal proteomics of 15-hexadecynyloxyacetic acid chemical reporter reveals preferential targeting of fatty acid modified proteins and biosynthetic enzymes. Bioorg Med Chem. 2012;20(2):650–4. https://doi.org/10.1016/j.bmc.2011.03.062.
Chesarino NM, Hach JC, Chen JL, Zaro BW, Rajaram MV, Turner J, et al. Chemoproteomics reveals toll-like receptor fatty acylation. BMC Biol. 2014;12:91. https://doi.org/10.1186/s12915-014-0091-3.
Percher A, Ramakrishnan S, Thinon E, Yuan X, Yount JS, Hang HC. Mass-tag labeling reveals site-specific and endogenous levels of protein S-fatty acylation. Proc Natl Acad Sci U S A. 2016;113(16):4302–7. https://doi.org/10.1073/pnas.1602244113.
• Chesarino NM, Compton AA, McMichael TM, Kenney AD, Zhang L, Soewarna V, et al. IFITM3 requires an amphipathic helix for antiviral activity. EMBO Rep. 2017;18(10):1740–51. https://doi.org/10.15252/embr.201744100. This study identifies a short, highly conserved amphipathic helix within the IFITM3 amino acid sequence as a structural element required for inhibition of virus membrane fusion. The proximity of this helix to critical S-palmitoylation sites suggests that this modification may be involved in membrane anchoring of the helix. Further, the known function of amphipathic helices in inducing membrane curvature provides a new mechanistic model for inhibition of virus infections by IFITM3.
Shi G, Schwartz O, Compton AA. More than meets the I: the diverse antiviral and cellular functions of interferon-induced transmembrane proteins. Retrovirology. 2017;14(1):53. https://doi.org/10.1186/s12977-017-0377-y.
Tartour K, Appourchaux R, Gaillard J, Nguyen XN, Durand S, Turpin J, et al. IFITM proteins are incorporated onto HIV-1 virion particles and negatively imprint their infectivity. Retrovirology. 2014;11:103. https://doi.org/10.1186/s12977-014-0103-y.
Compton AA, Bruel T, Porrot F, Mallet A, Sachse M, Euvrard M, et al. IFITM proteins incorporated into HIV-1 virions impair viral fusion and spread. Cell Host Microbe. 2014;16(6):736–47. https://doi.org/10.1016/j.chom.2014.11.001.
Tartour K, Nguyen XN, Appourchaux R, Assil S, Barateau V, Bloyet LM, et al. Interference with the production of infectious viral particles and bimodal inhibition of replication are broadly conserved antiviral properties of IFITMs. PLoS Pathog. 2017;13(9):e1006610. https://doi.org/10.1371/journal.ppat.1006610.
Lange UC, Adams DJ, Lee C, Barton S, Schneider R, Bradley A, et al. Normal germ line establishment in mice carrying a deletion of the Ifitm/Fragilis gene family cluster. Mol Cell Biol. 2008;28(15):4688–96. https://doi.org/10.1128/MCB.00272-08.
Everitt AR, Clare S, Pertel T, John SP, Wash RS, Smith SE, et al. IFITM3 restricts the morbidity and mortality associated with influenza. Nature. 2012;484(7395):519–23. https://doi.org/10.1038/nature10921.
Bailey CC, Huang IC, Kam C, Farzan M. Ifitm3 limits the severity of acute influenza in mice. PLoS Pathog. 2012;8(9):e1002909. https://doi.org/10.1371/journal.ppat.1002909.
• Poddar S, Hyde JL, Gorman MJ, Farzan M, Diamond MS. The interferon-stimulated gene IFITM3 restricts infection and pathogenesis of arthritogenic and encephalitic alphaviruses. J Virol. 2016;90(19):8780–94. https://doi.org/10.1128/JVI.00655-16. This study demonstrates that alphavirus infections induce more severe pathologies in IFITM3 KO mice than in WT mice. The work extends the known range of pathogens restricted by IFITM3 in vivo.
Gorman MJ, Poddar S, Farzan M, Diamond MS. The interferon-stimulated gene Ifitm3 restricts West Nile virus infection and pathogenesis. J Virol. 2016;90(18):8212–25. https://doi.org/10.1128/JVI.00581-16.
• Stacey MA, Clare S, Clement M, Marsden M, Abdul-Karim J, Kane L, et al. The antiviral restriction factor IFN-induced transmembrane protein 3 prevents cytokine-driven CMV pathogenesis. J Clin Invest. 2017;127(4):1463–74. https://doi.org/10.1172/JCI84889. This study shows that IFITM3 KO mice experience more severe pathologies than WT mice in cytomegalovirus infections. This is the first work to suggest that IFITM3 plays a role beyond direct virus restriction in vivo since cytomegalovirus infection of cells is not inhibited by IFITM3. The authors provide evidence that their observed effects are due to overproduction of IL-6 in IFITM3 KO mice.
Zhang W, Zhang L, Zan YL, Du N, Yang Y, Tien P. Human respiratory syncytial virus infection is inhibited by IFN-induced transmembrane proteins. J Gen Virol. 2015;96:170–82. https://doi.org/10.1099/vir.0.066555-0.
Everitt AR, Clare S, McDonald JU, Kane L, Harcourt K, Ahras M, et al. Defining the range of pathogens susceptible to Ifitm3 restriction using a knockout mouse model. PLoS One. 2013;8(11):e80723. https://doi.org/10.1371/journal.pone.0080723.
Infusini G, Smith JM, Yuan H, Pizzolla A, Ng WC, Londrigan SL, et al. Respiratory DC use IFITM3 to avoid direct viral infection and safeguard virus-specific CD8+ T cell priming. PLoS One. 2015;10(11):e0143539. https://doi.org/10.1371/journal.pone.0143539.
Wakim LM, Gupta N, Mintern JD, Villadangos JA. Enhanced survival of lung tissue-resident memory CD8(+) T cells during infection with influenza virus due to selective expression of IFITM3. Nat Immunol. 2013;14(3):238–45. https://doi.org/10.1038/ni.2525.
Savidis G, Perreira JM, Portmann JM, Meraner P, Guo Z, Green S, et al. The IFITMs inhibit Zika virus replication. Cell Rep. 2016;15(11):2323–30. https://doi.org/10.1016/j.celrep.2016.05.074.
Narayana SK, Helbig KJ, McCartney EM, Eyre NS, Bull RA, Eltahla A, et al. The interferon-induced transmembrane proteins, IFITM1, IFITM2, and IFITM3 inhibit hepatitis C virus entry. J Biol Chem. 2015;290(43):25946–59. https://doi.org/10.1074/jbc.M115.657346.
Yao L, Dong H, Zhu H, Nelson D, Liu C, Lambiase L, et al. Identification of the IFITM3 gene as an inhibitor of hepatitis C viral translation in a stable STAT1 cell line. J Viral Hepat. 2011;18(10):e523–9. https://doi.org/10.1111/j.1365-2893.2011.01452.x.
Monel B, Compton AA, Bruel T, Amraoui S, Burlaud-Gaillard J, Roy N, et al. Zika virus induces massive cytoplasmic vacuolization and paraptosis-like death in infected cells. EMBO J. 2017;36(12):1653–68. https://doi.org/10.15252/embj.201695597.
Pan Y, Yang P, Dong T, Zhang Y, Shi W, Peng X, et al. IFITM3 Rs12252-C variant increases potential risk for severe influenza virus infection in Chinese population. Front Cell Infect Microbiol. 2017;7:294. https://doi.org/10.3389/fcimb.2017.00294.
Lee N, Cao B, Ke C, Lu H, Hu Y, Tam CHT, et al. IFITM3, TLR3, and CD55 gene SNPs and cumulative genetic risks for severe outcomes in Chinese patients with H7N9/H1N1pdm09 influenza. J Infect Dis. 2017;216(1):97–104. https://doi.org/10.1093/infdis/jix235.
Wang Z, Zhang A, Wan Y, Liu X, Qiu C, Xi X, et al. Early hypercytokinemia is associated with interferon-induced transmembrane protein-3 dysfunction and predictive of fatal H7N9 infection. Proc Natl Acad Sci U S A. 2014;111(2):769–74. https://doi.org/10.1073/pnas.1321748111.
Zhang YH, Zhao Y, Li N, Peng YC, Giannoulatou E, Jin RH, et al. Interferon-induced transmembrane protein-3 genetic variant rs12252-C is associated with severe influenza in Chinese individuals. Nat Commun. 2013;4:1418. https://doi.org/10.1038/ncomms2433.
Mehrbod P, Eybpoosh S, Fotouhi F, Shokouhi Targhi H, Mazaheri V, Farahmand B. Association of IFITM3 rs12252 polymorphisms, BMI, diabetes, and hypercholesterolemia with mild flu in an Iranian population. Virol J. 2017;14(1):218. https://doi.org/10.1186/s12985-017-0884-4.
Mills TC, Rautanen A, Elliott KS, Parks T, Naranbhai V, Ieven MM, et al. IFITM3 and susceptibility to respiratory viral infections in the community. J Infect Dis. 2014;209(7):1028–31. https://doi.org/10.1093/infdis/jit468.
Randolph AG, Yip WK, Allen EK, Rosenberger CM, Agan AA, Ash SA, et al. Evaluation of IFITM3 rs12252 association with severe pediatric influenza infection. J Infect Dis. 2017;216(1):14–21. https://doi.org/10.1093/infdis/jix242.
Carter TC, Hebbring SJ, Liu J, Mosley JD, Shaffer CM, Ivacic LC, et al. Pilot screening study of targeted genetic polymorphisms for association with seasonal influenza hospital admission. J Med Virol. 2018;90(3):436–46. https://doi.org/10.1002/jmv.24975.
Lopez-Rodriguez M, Herrera-Ramos E, Sole-Violan J, Ruiz-Hernandez JJ, Borderias L, Horcajada JP, et al. IFITM3 and severe influenza virus infection. No evidence of genetic association. Eur J Clin Microbiol Infect Dis. 2016;35(11):1811–7. https://doi.org/10.1007/s10096-016-2732-7.
Gaio V, Nunes B, Pechirra P, Conde P, Guiomar R, Dias CM, et al. Hospitalization risk due to respiratory illness associated with genetic variation at IFITM3 in patients with influenza A(H1N1)pdm09 infection: a case-control study. PLoS One. 2016;11(6):e0158181. https://doi.org/10.1371/journal.pone.0158181.
David S, Correia V, Antunes L, Faria R, Ferrao J, Faustino P, et al. Population genetics of IFITM3 in Portugal and Central Africa reveals a potential modifier of influenza severity. Immunogenetics. 2017;70:169–77. https://doi.org/10.1007/s00251-017-1026-2.
Xu-Yang Z, Pei-Yu B, Chuan-Tao Y, Wei Y, Hong-Wei M, Kang T, et al. Interferon-induced transmembrane protein 3 inhibits Hantaan virus infection, and its single nucleotide polymorphism rs12252 influences the severity of hemorrhagic fever with renal syndrome. Front Immunol. 2016;7:535. https://doi.org/10.3389/fimmu.2016.00535.
Zhang YH, Makvandi-Nejad S, Qin L, Zhao Y, Zhang T, Wang LL, et al. Interferon-induced transmembrane protein-3 rs12252-C is associated with rapid progression of acute HIV-1 infection in Chinese MSM cohort. AIDS. 2015;29(8):889–94. https://doi.org/10.1097/Qad.0000000000000632.
•• Allen EK, Randolph AG, Bhangale T, Dogra P, Ohlson M, Oshansky CM, et al. SNP-mediated disruption of CTCF binding at the IFITM3 promoter is associated with risk of severe influenza in humans. Nat Med. 2017;23(8):975–83. https://doi.org/10.1038/nm.4370. This study identifies a new SNP in the IFITM3 gene, rs34411844-A, associated with severe influenza virus infections in humans. This SNP correlates with decreased IFITM3 protein and mRNA levels.
• Makvandi-Nejad S, Laurenson-Schafer H, Wang L, Wellington D, Zhao Y, Jin B, et al. Lack of truncated IFITM3 transcripts in cells homozygous for the rs12252-C variant that is associated with severe influenza infection. J Infect Dis. 2018;217(2):257–62. https://doi.org/10.1093/infdis/jix512. This study provides evidence that refutes the predominant hypothesis in the IFITM3 literature that the rs12252-C polymorphism alters IFITM3 splicing. Alternatively, spliced transcripts were not detected via RNA sequencing.
Kenney AD, Dowdle JA, Bozzacco L, McMichael TM, St Gelais C, Panfil AR, et al. Human genetic determinants of viral diseases. Annu Rev Genet. 2017;51:241–63. https://doi.org/10.1146/annurev-genet-120116-023425.
Jiang LQ, Xia T, Hu YH, Sun MS, Yan S, Lei CQ, et al. IFITM3 inhibits virus-triggered induction of type I interferon by mediating autophagosome-dependent degradation of IRF3. Cell Mol Immunol. 2017; https://doi.org/10.1038/cmi.2017.15.
Lin TY, Chin CR, Everitt AR, Clare S, Perreira JM, Savidis G, et al. Amphotericin B increases influenza A virus infection by preventing IFITM3-mediated restriction. Cell Rep. 2013;5(4):895–908. https://doi.org/10.1016/j.celrep.2013.10.033.
Daugherty MD, Malik HS. Rules of engagement: molecular insights from host-virus arms races. Annu Rev Genet. 2012;46:677–700. https://doi.org/10.1146/annurev-genet-110711-155522.
Sun X, Zeng H, Kumar A, Belser JA, Maines TR, Tumpey TM. Constitutively expressed IFITM3 protein in human endothelial cells poses an early infection block to human influenza viruses. J Virol. 2016;90(24):11157–67. https://doi.org/10.1128/JVI.01254-16.
Gerlach T, Hensen L, Matrosovich T, Bergmann J, Winkler M, Peteranderl C, et al. pH optimum of hemagglutinin-mediated membrane fusion determines sensitivity of influenza A viruses to the interferon-induced antiviral state and IFITMs. J Virol. 2017;91(11) https://doi.org/10.1128/JVI.00246-17.
Ding S, Pan Q, Liu SL, Liang C. HIV-1 mutates to evade IFITM1 restriction. Virology. 2014;454–455:11–24. https://doi.org/10.1016/j.virol.2014.01.020.
•• Foster TL, Wilson H, Iyer SS, Coss K, Doores K, Smith S, et al. Resistance of transmitted founder HIV-1 to IFITM-mediated restriction. Cell Host Microbe. 2016;20(4):429–42. https://doi.org/10.1016/j.chom.2016.08.006. This study suggests that HIV-1 evolves to evade restriction by IFITMs in humans. They provide evidence that transmitted founder viruses are resistant to IFITMs and that susceptibility to IFITMs increases as the virus mutates to evade adaptive immune mechanisms, such as antibodies.
Wang Y, Pan Q, Ding S, Wang Z, Yu J, Finzi A, et al. The V3 Loop of HIV-1 Env Determines Viral Susceptibility to IFITM3 Impairment of Viral Infectivity. J Virol. 2017;91(7) https://doi.org/10.1128/JVI.02441-16.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Virology
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zani, A., Yount, J.S. Antiviral Protection by IFITM3 In Vivo. Curr Clin Micro Rpt 5, 229–237 (2018). https://doi.org/10.1007/s40588-018-0103-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40588-018-0103-0