Abstract
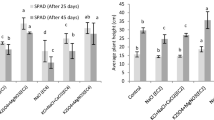
Crops are living in harsh condition ever since emergence such as high temperature, water shortage or excessive watering, high concentration of salt, heavy metals and among others described as abiotic stresses. The major plant nutrient Mg (magnesium), Ca (calcium) is involved in numerous physiological processes, and additional supplements can enhance plant growth and biomass under various environmental stresses such as salinity and heat. This study aimed to investigate, if Magnesium and Calcium sulfate applications on seedlings of Medicago sativa L., Trifolium pratense L., Festuca arundinacea Shreb. in practically relevant amounts can efficiently alleviate the effects of salinity and heat stress, namely reduced biomass, disturbed ion homeostasis and decreased growth. Seedlings were treated by fourteen treatments which include 100 mM NaCl, MgSO4 (0.5 mM, 1 mM, 2 mM); CaSO4 (5 mM, 7.5 mM, 10 mM); and salinity plus CaSO4 and MgSO4 under two temperatures (20 °C and 30 °C) with 16 h/8 h photoperiod in growth chamber. Results clearly demonstrated that combined salinity and heat stress had great effects on seedling growth but additional CaSO4 and MgSO4 have enhanced the growth specifically under stressed conditions. Significant increases in biomass (TB, root, shoot) and root length, as well as shoot height, has been achieved. The elemental analysis furthermore revealed an increment of Ca, Na (Sodium), K (Potassium) level when plants under salinity stress. Together, our results indicate that additional supply of MgSO4 and CaSO4 on seedlings turned to be efficient under abiotic stresses. It might be of practical relevance to add correct amount of CaSO4 and MgSO4 during the early growth phase of plants that are exposed to environmental stress.








Similar content being viewed by others
References
Abid, M., Ferchichi, M., & Haddad, A. (2008). Effect of magnesium sulphate on the first stage of development of Lucerne. In: Porqueddu, C., & Tavares-de-Sousa, M. M. (Eds.), Sustainable Mediterranean grasslands and their multi-functions (Vol. 408, Issue 79, pp. 405–408).
Acosta-Motos, J. R., Ortuño, M. F., Bernal-Vicente, A., Diaz-Vivancos, P., Sanchez-Blanco, M. J., & Hernandez, J. A. (2017). Plant responses to salt stress: Adaptive mechanisms. Agronomy, 7(1), 1–38. https://doi.org/10.3390/agronomy7010018
AL-Shoaibi, A. A. (2020). Combined effects of salinity and temperature on germination, growth and gas exchange in two cultivars of Sorghum bicolor. Journal of Taibah University for Science, 14(1), 812–822. https://doi.org/10.1080/16583655.2020.1777800
Balke, N. E., & Hodges, T. K. (1975). Plasma membrane adenosine triphosphatase of oat roots: Activation and inhibition by Mg2+ and ATP. Plant Physiology, 55(1), 83–86.
Bláha, L. (2019). Importance of root-shoot ratio for crops production. Agronomy & Agricultural Science, 2, 012. https://doi.org/10.24966/aas-8292/100012
Bloom, A. J., & Kameritsch, P. (2017). Relative association of Rubisco with manganese and magnesium as a regulatory mechanism in plants. Physiologia Plantarum, 161(4), 545–559. https://doi.org/10.1111/ppl.12616
Corte, A. P. D., da Silva, F., & Sanquetta, C. R. (2012). Biomass expansion factor and root-to-shoot ratio for Pinus spp. in southern Brazil. Floresta, 42(4), 755–768. https://doi.org/10.5380/rf.v42i4.17771
Del Río, L. A., Sandalio, L. M., Corpas, F. J., Palma, J. M., & Barroso, J. B. (2006). Reactive oxygen species and reactive nitrogen species in peroxisomes. Production, scavenging, and role in cell signaling. Plant Physiology, 141(2), 330–335. https://doi.org/10.1104/pp.106.078204
Dumont, S., & Rivoal, J. (2019). Consequences of oxidative stress on plant glycolytic and respiratory metabolism. Frontiers in Plant Science, 10, 166. https://doi.org/10.3389/fpls.2019.00166
Grieve, C., & Grattan, S. (1992). Mineral nutrient acquisition and response by plants grown in saline environments. Agriculture, Ecosystems and Environment, 38, 275–300. https://doi.org/10.1201/9780824746728.ch9
Guo, R., Shi, L. X., Ding, X. M., Hu, Y., Tian, S. Y., Yan, D. F., Shao, S., Gao, Y., Liu, R., & Yang, Y. F. (2010). Effects of saline and alkaline stress on germination, seedling growth, and ion balance in wheat. Agronomy Journal, 102(4), 1252–1260. https://doi.org/10.2134/agronj2010.0022
Guo, W., Chen, S., Hussain, N., Cong, Y., Liang, Z., & Chen, K. (2015). Magnesium stress signaling in plant. Plant Signaling and Behavior, 10(3), e992287.
Halliwell, B. (1987). Oxidative damage, lipid peroxidation and antioxidant protection in chloroplasts. Chemistry and Physics of Lipids, 44, 327–340.
Hanley, M. E., Fenner, M., Whibley, H., & Darvill, B. (2004). Early plant growth: Identifying the end point of the seedling phase. New Phytologist, 163(1), 61–66. https://doi.org/10.1111/j.1469-8137.2004.01094.x
He, M., He, C. Q., & Ding, N. Z. (2018). Abiotic stresses: General defenses of land plants and chances for engineering multistress tolerance. Frontiers in Plant Science, 9, 1771. https://doi.org/10.3389/fpls.2018.01771
Heckathorn, S. A., Giri, A., Mishra, S., & Bista, D. (2013). Heat stress and roots. Climate Change and Plant Abiotic Stress Tolerance. https://doi.org/10.1002/9783527675265.ch05
Hu, L., Huang, Z., Liu, S., & Fu, J. (2012). Growth response and gene expression in antioxidant-related enzymes in two bermudagrass genotypes differing in salt tolerance. Journal of the American Society for Horticultural Science, 137(3), 134–143. https://doi.org/10.21273/jashs.137.3.134
Huang, B., & Xu, Q. (2008). Root growth and nutrient element status of creeping bentgrass cultivars differing in heat tolerance as influenced by supraoptimal shoot and root temperatures. Journal of Plant Nutrition, 23(7), 979–990. https://doi.org/10.1080/01904160009382075
Ibrahim, E. A. (2016). Seed priming to alleviate salinity stress in germinating seeds. Journal of Plant Physiology, 192, 38–46. https://doi.org/10.1016/j.jplph.2015.12.011
International Plant Nutrition Institute. (1999). Functions of phosphorus in plants. Better Crops, 83(1), 6–7.
Jezek, M., Geilfus, C. M., Bayer, A., & Muhling, K. H. (2015). Photosynthetic capacity, nutrient status, and growth of maize (Zea mays L.) upon MgSO4 leaf-application. Frontiers in Plant Science, 5, 781. https://doi.org/10.3389/fpls.2014.00781
Kent, L. M., & Lauchli, A. (1985). Germination and seedling growth of cotton: Salinity-calcium interactions. Plant, Cell & Environment, 8(2), 155–159. https://doi.org/10.1111/j.1365-3040.1985.tb01223.x
Knight, H., Trewavas, A. J., & Knight, M. R. (1997). Calcium signalling in Arabidopsis thaliana responding to drought and salinity. Plant Journal, 12(5), 1067–1078. https://doi.org/10.1046/j.1365-313X.1997.12051067.x
Kumari, V. V., Banerjee, P., Verma, V. C., Sukumaran, S., Alickal, M., Chandran, S., Gopinath, K. A., Venkatesh, G., Yadav, S. K., Singh, V. K., & Awasthi, N. K. (2022). Plant nutrition: An effective way to alleviate abiotic stress in agricultural crops. International Journal of Molecular Sciences, 23(8519), 1–30.
Langjun, C., Jianlong, L., Yamin, F., & Sheng, X. (2006). High temperature effects on photosynthesis, PSII functionality and antioxidant activity of two Festuca arundinacea cultivars with different heat susceptibility. Botanical Studies, 47, 61–69.
Li, R., Shi, F., Fukuda, K., & Yang, Y. (2010). Effects of salt and alkali stresses on germination, growth, photosynthesis and ion accumulation in alfalfa (Medicago sativa L.). Soil Science and Plant Nutrition, 56(5), 725–733. https://doi.org/10.1111/j.1747-0765.2010.00506.x
Lutts, S., Almansouri, M., & Kinet, J. M. (2004). Salinity and water stress have contrasting effects on the relationship between growth and cell viability during and after stress exposure in durum wheat callus. Plant Science, 167(1), 9–18. https://doi.org/10.1016/j.plantsci.2004.02.014
Manishankar, P., Wang, N., Köster, P., Alatar, A. A., & Kudla, J. (2018). Calcium signaling during salt stress and in the regulation of ion homeostasis. Journal of Experimental Botany, 69(17), 4215–4226. https://doi.org/10.1093/jxb/ery201
Mittler, R. (2006). Abiotic stress, the field environment and stress combination. Trends in Plant Science, 11(1), 15–19. https://doi.org/10.1016/j.tplants.2005.11.002
Monk, C. (1966). Ecological importance of root/shoot ratios. Bulletin of the Torrey Botanical Club, 93(6), 402–406.
Munns, R., & Termaat, A. (1986). Whole-plant responses to salinity. Australian Journal of Plant Physiology, 13(1), 143–160. https://doi.org/10.1071/PP9860143
Nadjafi, F., Shabahang, J., & Damghani, A. M. M. (2010). Effects of salinity and temperature on germination and seedling growth of nine medicinal plant species technologists. Seed Technology, 76(12), 96–107. https://doi.org/10.1055/s-0030-1264685
Plieth, C. (1999). Temperature sensing by plants: Calcium-permeable channels as primary sensors—A model. Membrane Biology, 172, 121–127. https://doi.org/10.1038/438577a
Rahman, A., Nahar, K., Hasanuzzaman, M., & Fujita, M. (2016). Calcium supplementation improves Na+/K+ ratio, antioxidant defense and glyoxalase systems in salt-stressed rice seedlings. Frontiers in Plant Science, 7, 609. https://doi.org/10.3389/fpls.2016.00609
Rezvani, M., Nadimi, S., Zaefarian, F., & Chauhan, B. S. (2021). Environmental factors affecting seed germination and seedling emergence of three Phalaris species. Crop Protection, 148(June), 105743. https://doi.org/10.1016/j.cropro.2021.105743
Rivero, R. M., Mestre, T. C., Mittler, R., Rubio, F., Garcia-Sanchez, F., & Martinez, V. (2014). The combined effect of salinity and heat reveals a specific physiological, biochemical and molecular response in tomato plants. Plant, Cell and Environment, 37(5), 1059–1073. https://doi.org/10.1111/pce.12199
SAS Institute Inc. (2014). SAS software 9.4 (Vol. 9.4, pp. 1–25). SAS Institute Inc.
Shabala, S., & Pottosin, I. (2014). Regulation of potassium transport in plants under hostile conditions: Implications for abiotic and biotic stress tolerance. Physiologia Plantarum, 151(3), 257–279. https://doi.org/10.1111/ppl.12165
Sharavdorj, K., Jang, Y., Byambadorj, S. O., & Cho, J. W. (2021). Understanding seed germination of forage crops under various salinity and temperature stress. Journal of Crop Science and Biotechnology. https://doi.org/10.1007/s12892-021-00101-9
Siddiqui, M. H., Alamri, S. A., Al-Khaishany, M. Y. Y., Al-Qutami, M. A., Ali, H. M., Al-Whaibi, M. H., Al-Wahibi, M. S., & Alharby, H. F. (2018). Mitigation of adverse effects of heat stress on Vicia faba by exogenous application of magnesium. Saudi Journal of Biological Sciences, 25(7), 1393–1401. https://doi.org/10.1016/j.sjbs.2016.09.022
Tang, R. J., & Luan, S. (2017). Regulation of calcium and magnesium homeostasis in plants: From transporters to signaling network. Current Opinion in Plant Biology, 39, 97–105. https://doi.org/10.1016/j.pbi.2017.06.009
Tobe, K., Li, X., & Omasa, K. (2002). Effects of sodium, magnesium and calcium salts on seed germination and radicle survival of a halophyte, Kalidium caspicum (Chenopodiaceae). Australian Journal of Botany, 50(2), 163–169. https://doi.org/10.1071/BT01065
Tuna, A. L., Kaya, C., Ashraf, M., Altunlu, H., Yokas, I., & Yagmur, B. (2007). The effects of calcium sulphate on growth, membrane stability and nutrient uptake of tomato plants grown under salt stress. Environmental and Experimental Botany, 59(2), 173–178. https://doi.org/10.1016/j.envexpbot.2005.12.007
Wang, Y., & Wu, W. H. (2013). Potassium transport and signaling in higher plants. Annual Review of Plant Biology, 64, 451–476. https://doi.org/10.1146/annurev-arplant-050312-120153
Waqas, M. A., Kaya, C., Riaz, A., Farooq, M., Nawaz, I., Wilkes, A., & Li, Y. (2019). Potential mechanisms of abiotic stress tolerance in crop plants induced by thiourea. Frontiers in Plant Science, 10, 01336. https://doi.org/10.3389/fpls.2019.01336
Waraich, E. A., Ahmad, R., Halim, A., & Aziz, T. (2012). Alleviation of temperature stress by nutrient management in crop plants: A review. Journal of Soil Science and Plant Nutrition, 12(2), 221–244. https://doi.org/10.4067/S0718-95162012000200003
Wilkinson, S., Clephan, A. L., & Davies, W. J. (2001). Rapid low temperature-induced stomatal closure occurs in cold-tolerant Commelina communis leaves but not in cold-sensitive tobacco leaves, via a mechanism that involves apoplastic calcium but not abscisic acid. Plant Physiology, 126(4), 1566–1578. https://doi.org/10.1104/pp.126.4.1566
Acknowledgements
This study was financially supported by research fund of National Research Foundation (No. 2021R1F1A1052323) of Korea in 2021.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharavdorj, K., Jang, Y., Byambadorj, SO. et al. The effect of MgSO4 and CaSO4 on seedlings of forage crops under environmental stress. Plant Physiol. Rep. 27, 702–716 (2022). https://doi.org/10.1007/s40502-022-00691-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-022-00691-8