Abstract
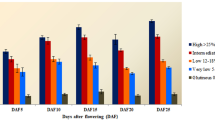
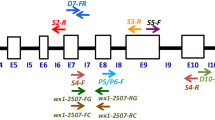
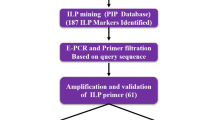
The Northeast India, being at the confluence of Indo-Malayan and Indo-Chinese biogeographic realms, represents one of the world “biodiversity hotspots”. With a rich diversity of cultivars, the rice gene pool of the region shows marked variations in various quality traits including aroma and starch quality. Since starch quality is primarily determined by its apparent amylose content and that the Waxy locus controls much of the variation in apparent amylose content, the linkage between sequence polymorphism in the Waxy locus and the apparent amylose content assumes significance. Our investigations on the relationship between SNPs in Waxy locus and variations in AAC of starch varieties of rice from North East India clearly reveal that in accessions having the sequence AGTTAT in 5′ leader intron splice site, “Ex6C” allele was associated with starches having 0–2% AAC and “Ex6A” allele was associated with starches having between 2 and 12% AAC. Based on combination of “G/T” polymorphism in 5′ leader intron splice site, length polymorphism in exon 2 and “A/C” polymorphism in exon 6, the Waxy locus of rice can have four allelic forms viz. “Int1T-Ex2(23 bp)2-Ex6C”, “Int1T-Ex2(23 bp)1-Ex6A”, “Int1G-Ex2(23 bp)1-Ex6C” and “Int1G-Ex2(23 bp)1-Ex6A” which correlate specifically with “waxy”, “very low”, “low/intermediate” and “high amylo-” cultivars, respectively. Phylogenetic tree on alignment matrix of nucleotide sequences of the p-SINEI element of the Waxy locus clustered the upland and japonica cultivars separately from the lowland and indica cultivars, thereby establishing the role of transposons in the Waxy locus in differentiation of upland/japonica and lowland/indica cultivars.






Similar content being viewed by others
Abbreviations
- SNP:
-
Single nucleotide polymorphism
- GBSS:
-
Granule bound starch synthase
- AAC:
-
Apparent amylose content
References
Ayres, N. M., McClung, A. M., Larkin, P. D., Bligh, H. F. J., Jones, C. A., & Park, W. D. (1997). Microsatellites and a single nucleotide polymorphism differentiate apparent amylose classes in an extended pedigree of US rice germplasm. Theoretical and Applied Genetics, 94, 773–781.
Biselli, C., Daniela, C., Rosaria, P., Alberto, G., Paolo, B., Simona, U., et al. (2014). Improvement of marker-based predictability of apparent amylose content in japonica rice through GBSSI allele mining. Rice. doi:10.1186/1939-8433-7-1.
Bligh, H. F. J., Larkin, P. D., Roach, P. S., Jonea, P. A., Fu, H., & Park, W. D. (1998). Use of alternate splicing sites in granule bound starch synthase mRNA from low amylose rice varieties. Plant Molecular Biology, 38, 407–415.
Cai, X. L., Wang, Z. Y., Xing, Y. Y., Zhang, J. L., & Hong, M. M. (1998). Aberrant splicing of intron 1 leads to the heterogeneous 5′ UTR and decreased expression of Waxy gene in rice cultivars of intermediate amylose content. The Plant Journal, 14, 459–465.
Chen, M. H., Bergman, C., Pinson, S., & Fjellstrom, R. (2008). Waxy gene haplotypes: Associations with apparent amylose content and the effect by the environment in an international rice germplasm collection. Journal of Cereal Science, 47, 536–545.
Cheng, J., Khan, M. A., Qiu, W. M., Li, J., Zhou, H., Zhang, Q., et al. (2012). Diversification of genes encoding granule-bound starch synthase in monocots and dicots is marked by multiple genome-wide duplication events. PLoS ONE, 7(e30088), 2012. doi:10.1371/journal.pone.0030088.
Cheng, Z. Q., Lui, Y. P., Chen, R., Peng, B., Xiong, H. B., & Zhang, C. (2010). Diversity of Waxy gene alleles in the wild rice species of the Oryza genus. Botanical Studies, 51, 403–411.
Cheng, C., Motohashi, R., Tsuchimoto, S., Fukuta, Y., Ohtsubo, H., & Ohtsubo, E. (2003). Polyphyletic origin of cultivated rice: Based on interspersion pattern of SINEs. Molecular Biology and Evolution, 20, 67–75.
Dobo, M., Ayers, M., Walker, G., & Park, W. D. (2010). Polymorphism in the GBSS gene affects amylose content in US and European rice germplasm. Journal of Cereal Science, 52, 450–456.
Hoai, T. T. T., Matsusaka, H., Toyosawa, Y., Suu, T. D., Satoh, H., & Kumamaru, T. (2014). Influence of single-nucleotide polymorphisms in the gene encoding granule-bound starch synthase I on amylose content in Vietnamese rice cultivars. Breeding Science, 64, 142–148.
Hoai, T. T., Nishi, A., & Satoh, H. (2008). Diversity of granule bound starch synthesis (GBSS) levels in North Vietnam local rice cultivars. Rice Genetics Newsletter, 24, 62–64.
Inukai, T., Sako, A., Hirano, H. Y., & Sano, Y. (2000). Analysis of intragenic recombination at wx in rice: correlation between the molecular and genetic maps within the locus. Genome, 43, 589–596.
Jayamani, P., Negrao, S., Brites, C., & Oliveira, M. M. (2007). Potential of Waxy gene microsatellite and single-nucleotide polymorphisms to develop japonica varieties with desired amylose levels in rice (Oryza sativa L.). Journal of Cereal Science, 46, 178–186.
Jimenez, R. R., Resurreccion, A. P., & Fitzgerald, M. A. (2010). Moving from apparent to actual amylose in rice. In Proceedinds 28th International Rice Research Conference in Hanoi, Vietnam. OP10: Quality Grain, Health, and Nutrition, Nov. 8012.
Juliano, B. O. (1971). A simplified assay for milled-rice amylose. Cereal Science Today, 16, 334–338.
Juliano, B. O., & Pascual, C. G. (1980). Quality characteristics of milled rice grown in different countries. IRRI Research Paper Series 48, 1–25. International Rice Research Institute, Los Banos, Laguna, Philippines.
Kumar, S., Dudley, J., Nei, M., & Tamura, K. (2008). MRGA: A biologist–centric software for evolutionary analysis of DNA and protein sequences. Briefings in Bioinformatics, 9, 299–306.
Larkin, P. D., & Park, W. D. (1999). Transcript accumulation and utilization of alternate and non-consensus splice sites in rice granule-bound starch synthase are temperature-sensitive and controlled by a single-nucleotide polymorphism. Plant Molecular Biology, 40, 719–727.
Larkin, P. D., & Park, W. D. (2003). Association of waxy gene single nucleotide polymorphisms with starch characteristics in rice (Oryza sativa L.). Molecular Breeding, 12, 335–339.
Lawal, O. S., Lapasin, R., Bellich, B., Olayiwola, T. O., Cesaro, A., Yoshimura, M., et al. (2011). Rheology and functional properties of starches isolated from five improved rice varieties from West Africa. Food Hydrocolloids, 25, 1785–1792.
Momma, M., & Fujimoto, Z. (2012). Inter domain disulphide bridges in the rice granule bound starch synthase I catalytic domains as elucidated by X-ray structure analysis. Bioscience, Biotechnology, and Biochemistry, 76, 1591–1595.
Murray, M. G., & Thomson, W. F. (1980). Rapid isolation of high molecular weight plant DNA. Nucleic Acid Research, 8, 4321–4325.
Mutters, R. G., & Thomson, J. F. (2009). Rice Quality Handbook—University of California Agriculture and Natural Resources Publication number 3514.
Pace, C. N., Hebert, E. J., Bechert, J., Shaw, K., Sevcik, J., Urbanikova, L., et al. (2001). Tyrosine hydrogen bonds make a large contribution to protein stability. Journal of Molecular Biology, 312, 393–404.
Peroni, F., Rocha, T., & Franco, C. (2006). Some structural and physicochemical characteristics of tuber and root starches. Food Science and Technology International, 12, 505–513.
Preiss, J. (1991). Biology and molecular biology of starch synthesis and its regulation. In B. J. Miflin (Ed.), Oxford surveys of cellular and molecular biology (pp. 59–114). Oxford: Oxford University Press.
Rozen, S., & Skatelsky, H. J. (2000). In: Krawetz, S., Misener, S. (eds.) Bioinformatics methods and protocols: Methods in molecular biology (pp. 365–386). Humana Press Totowa New Jersey.
Sano, Y., Katsumata, M., & Omura, K. (1986). Genetic studies of speciation in cultivated rice. Inter- and intraspecific differentiation in the Waxy gene expression of rice. Euphytica, 35, 1–9.
Smith, A. M., Denyer, K., & Martin, C. (1977). The synthesis of the starch granule. Annual Review Plant Physiology and Plant Molecular Biology, 48, 67–87.
Takaoka, M., Wantanabe, S., Sassa, H., Yamamori, M., Nakamura, T., Sasakuma, T., et al. (1997). Structural characterization of high molecular weight starch granule-bound proteins in wheat (Triticum aestivum L.). Journal of Agriculture and Food Chemistry, 45, 2929–2934.
Thongbam, P. D., Raychaudhury, M., Tarentoshi, Durai, M., Das, S. P., Ramesh, T., et al. (2012). Studies on grain and food quality traits of some indigenous rice cultivars of North-eastern Hill region of India. Journal of Agricultural Science, 4, 259–270.
Umeda, M., Ohtsubo, H., & Ohtsubo, E. (1991). Diversification of the rice Waxy gene by insertion of mobile DNA elements into introns. Japanese Journal of Genetics, 66, 569–586.
Wanchana, S., Toojinda, T., Tragoonrung, S., & Vanavichit, A. (2003). Duplicated coding sequence in the Waxy allele of tropical glutinous rice (Oryza sativa L.). Plant Science, 165, 1193–1199.
Wang, Z. Y., Zheng, F. Q., Shen, G. Z., Gao, J. P., Snustad, D. P., Li, M. G., et al. (1995). The amylose content in rice endosperm is related to the post-transcriptional regulation of the Waxy gene. The Plant Journal, 7, 613–622.
Yamanaka, S., Nakamura, I., Nakai, H., & Sato, Yo-I. (2003). Dual origin of the cultivated rice based on molecular markers of newly collected annual and perennial strains of wild rice species, Oryza nivara and O. rufipogon. Genetic Resources and Crop Evolution, 50, 529–538.
Acknowledgements
The authors acknowledge partial financial support from Department of Biotechnology, Govt of India, New Delhi against grant nos. BT/HRD/01/002/2007 and BT/04/NE/2009.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chrungoo, N.K., Devi, A.G. Sequence polymorphism in the Waxy locus and its relationship with apparent amylose content of endosperm starch in cultivars of rice (Oryza sativa L.) from Northeast India. Ind J Plant Physiol. 21, 556–568 (2016). https://doi.org/10.1007/s40502-016-0262-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-016-0262-z