Abstract
Introduction
Patients with acute myeloid leukemia (AML), especially those with relapsed or refractory AML, have poor clinical prognosis and outcomes. Health-related quality of life (HRQoL) assessments have become increasingly important in oncology, aiding in identifying and informing supportive therapy needs during treatment and beyond; however, HRQoL in hematology, and AML in particular, has received relatively minor attention. The aim was to identify and summarize estimates of HRQoL in patients with AML, including patients with relapsed or refractory AML.
Methods
A systematic literature review was performed. MEDLINE and EMBASE databases were searched for peer-reviewed literature published between 2004 and 2014 in the US and Europe. Abstracts from four relevant conference proceedings between 2012 and 2014 were reviewed. Data from eligible studies were extracted describing the HRQoL instruments used, domains assessed, and HRQoL scores reported.
Results
Fourteen peer-reviewed studies met the eligibility criteria and were included in the review. Cancer- or leukemia-specific HRQoL measures were used in 78.6% of the studies. Overall, HRQoL was superior among AML survivors compared to individuals on active treatment. Fatigue was identified as the most problematic symptom domain in patients, irrespective of their treatment status. Reported HRQoL declined shortly after diagnosis or treatment initiation and recovered over time.
Conclusion
The included studies identified a decrease in HRQoL after treatment initiation and highlighted the role of fatigue in HRQoL in this patient population. Limited HRQoL data were identified among relapsed or refractory AML patients although they have worse prognostic outcomes. New treatment options that have less negative impact on HRQoL or health initiatives specifically targeting HRQoL of patients with AML are warranted. In addition, further studies exploring HRQoL in the relapsed or refractory patient population are needed to inform disease management and treatment decisions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute myeloid leukemia (AML) in adults is associated with poor clinical prognosis and outcomes with 26% of patients reaching 5-year survival after receiving an AML diagnosis [1]. Specific subgroups within the AML patient population have worse prognosis, including individuals with certain molecular and cytogenetic profiles; relapsed patients, who achieved complete remission after initial AML treatment only to have disease recurrence; or refractory patients, who fail to achieve remission after AML treatment [2]. For individuals diagnosed with AML, intensive chemotherapy and bone marrow transplantation are still the first-line treatment options, often requiring long hospitalizations and severe treatment side effects [3], thereby significantly impacting health-related quality of life (HRQoL).
HRQoL is conceptualized as a broad variety of domains underpinning individuals’ well-being; it is assessed through multidimensional evaluation of the included domains. Assessment of HRQoL has become increasingly important in oncology because of the recognition that traditional survival endpoints in clinical trials may overlook disease- or treatment-related events that have an impact on patients’ well-being [4–6]. In addition, understanding HRQoL can help identify and inform supportive therapy needs both during and beyond treatment cessation [7–9].
While HRQoL has been increasingly used as an outcome measure in oncology clinical trials, HRQoL in hematology and in AML in particular has received relatively minor attention. A review published in 2008 of HRQoL in leukemia randomized controlled trials (RCTs), including AML, highlighted the paucity of HRQoL data available in leukemia research [10]. Despite the importance of patient well-being, information on HRQoL of patients with AML and survivors of AML is lacking [3].
To our knowledge, a single review of HRQoL in the AML patient population was conducted by Redaelli et al. [3]; however, no updates on AML-related HRQoL are available. Thus, the primary objective of the present study was to provide an updated synthesis of studies published between 2004 and 2014 on HRQoL in patients with AML from the USA and Europe. An additional objective was to identify published estimates of HRQoL by AML subgroups, including patients who have relapsed or refractory AML.
Methods
A systematic literature review (SLR) of peer-reviewed publications published between 2004 and 2014 on AML patients from the US or EU was conducted in accordance with the Preferred Reporting Items for Systematic Reviews (PRISMA) reporting guideline [11]. Relevant publications were identified via the MEDLINE and EMBASE bibliographic databases; additionally, relevant abstracts between the years 2012 and 2014 from four conference proceedings—American Society of Clinical Oncology (ASCO), American Society of Hematology (ASH), European Society for Medical Oncology (ESMO), and the European Hematology Association (EHA)—were searched. The reference lists of all included publications were reviewed for additional eligible studies.
Abstracts identified through the search strategy were evaluated for inclusion in the SLR using pre-defined eligibility criteria by two experienced reviewers. Both the search strategy and eligibility criteria were approved by two clinical experts in AML (FR and ML). Discrepancies between the reviewers were resolved through iterative reviews and discussions. The following eligibility criteria had to be fulfilled: (1) the target population comprised adults 18 years and older from the US or the EU; (2) a current or previous diagnosis of primary or secondary AML, including patients who had relapsed or refractory AML, or were identified as having an fms-like tyrosine kinase-internal tandem duplication (FLT3-ITD) or FLT3-tyrosine kinase domain (TKD) mutation; and (3) HRQoL measured using a validated quality of life instrument. Articles that met the eligibility criteria were extracted and included in the SLR.
For each eligible study, pre-defined outcomes were independently abstracted by two independent reviewers and a quality assessment was conducted to assess the robustness of HRQoL outcomes reporting as described by Jacobs et al. [12]. Outcomes of interest included HRQoL domain scores reported by leukemia-specific, cancer-specific, or general quality of life instruments in the target population. Reported domains typically included a global assessment of overall HRQoL; domains that measure perceptions of functionality, including physical, mental, emotional or social functioning; and symptom-specific domains that target symptoms associated with cancer or leukemia.
Due to the quantitative and qualitative heterogeneity in the HRQoL instruments and results, this review did not include a formal meta-analysis component. Results were presented as reported in the studies or were summarized in tabular or graphic form. This work is based on previously conducted studies; no new data were collected on human or animal subjects in relation to this work.
Results
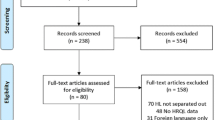
A summary of the number of articles identified from the search strategy, the reasons for publication exclusion, and total number of publications included is presented in Fig. 1. Publications and study design details of all included studies are presented in Appendix Table 1.
Key demographic and clinical characteristics of the included study populations can be found in Table 1. The sample size of included studies ranged from 8 to 481. The study populations primarily comprised of middle-aged adults; two studies reported mean age below 40 years of age [13, 14], and three studies had a mean age above 65 years of age [15–17]. Five studies reported the distribution of AML disease origin at baseline; study populations were primarily comprised of primary AML patients (range: 54.5–100.0%). No individuals with refractory AML were identified in the included studies.
The most frequently utilized HRQoL instrument was the cancer-specific EORTC QLQ-C30 questionnaire (50.0%, Table 1); generic HRQoL instruments (EQ-5D and SF-36 or SF-12) were utilized in only 35.7% of included studies. Descriptions of the included instruments, including measured domains and interpretations of scoring, can be found in Appendix Table 2.
Global Assessment of HRQoL
Eleven studies (78.6%) reported a global assessment of HRQoL. Ten studies (71.4%) utilized a cancer or leukemia-specific instrument; of these, six studies measured global HRQoL in patients who were survivors of AML, including four studies with participants who had achieved complete remission at the time of assessment. The mean global HRQoL scores in AML survivors and patients with active AML are presented in Tables 2 and 3.
An improvement in HRQoL was generally shown among survivors compared to active AML patients using the FACT-G and EORTC QLQ-C30 instruments: the mean EORTC QLQ-C30 scores ranged from 67.9 [18] to 80 [19] in survivors compared to 50 [16] to 77.2 [20] in patients with active AML, and mean FACT-G scores was 77.5 [18] in survivors compared to 56.1 [13] to 84 [17] in patients with active AML. An exception to this trend was the FACT-G scores reported by Sekeres et al., where superior HRQoL was reported among active AML patients [17]. The reason for this difference is unknown because of insufficient reporting of relevant clinical characteristics of the sample populations.
The EQ-5D instrument was utilized in two studies of AML survivor populations (Table 3). Leunis et al. reported an EQ-5D utility score of 0.82, and an EQ visual analog scale (VAS) score of 74.6, a significantly lower result compared to a general population sample (p = 0.0333). A separate study by Slovacek et al. reported the lowest EQ VAS score, 67.5, among individuals previously treated with autologous hematopoietic stem cell transplantation [24].
Three studies used the SF-12 or SF-36 scales to measure HRQoL, although one study did not report results for the AML population [21]. One study measured HRQoL in adult survivors of childhood AML and found no difference in physical or mental scales compared to population norms [14]. Among participants actively undergoing treatment for AML, mean SF-36 physical scale scores were approximately two standard deviations below population norms within the first 6 weeks from diagnosis, indicating a quick and sharp decline in HRQoL shortly after diagnosis; mental scores were comparable with general norm scores [17].
Functional Domains
Five of the included studies (35.7%) reported HRQoL measures of functional domains using the EORTC QLQ-C30. Scores for each of the domains ranged from 75.9 to 83 (cognitive), 64.1 to 83.1 (emotional), 27 to 74.8 (role), 67 to 86.7 (physical), and 56 to 83 (social) at baseline. Score ranges were similar between individuals with active AML and AML survivors in the cognitive, emotional, and social domains. Poorer scores were observed among individuals with active AML compared to AML survivors in the role domain—ranging from 27 to 67 vs. 72.5 to 74.8, respectively—and the physical domain—ranging from 67 to 73 vs. 80.4 to 86.7, respectively.
A comparison of mean scores between AML survivors and a general population sample found significantly lower scores reported across all functional domains in the AML population [22].
Symptom Domains and Financial Difficulties
Symptom-specific domains were included in six instruments, five of which were cancer or leukemia specific (Table 4). The most frequently used instrument to measure symptoms was the EORTC QLQ-C30, which includes three symptom scales—fatigue, nausea/vomiting, and pain—several single items assessing symptoms commonly reported by cancer patients, and perceived financial impact of the disease (Appendix Table 1).
Across studies, the most affected domains were fatigue, pain, dyspnea, insomnia, appetite loss, financial difficulties, and anemia (Table 4). Active AML patients reported appetite loss and fatigue symptoms as having the most detrimental impact on HRQoL, whereas fatigue and insomnia had the worst impact on HRQoL among AML survivors. One study comparing AML survivors to a general population sample identified a statistically significant negative impact on AML survivors in the fatigue, pain, dyspnea, and appetite loss domains (p value <0.05 for all comparisons) and a clinically relevant impact—defined as a difference of at least ten points—in the fatigue and dyspnea domains [22].
Perceived financial difficulties showed negative impact in both active AML patients and AML survivors and were reported to be significantly worse in AML survivors compared to a general population sample (p value <0.05) [22].
A single study utilized the leukemia-specific EORTC QLQ-Leu instrument among adult AML survivors; the subscales with the highest percentage of survivors reporting problems included graft-versus-host disease (GVHD; 86%) and infection susceptibility (51%), although the authors noted that few participants scored these symptoms as severe [19].
Prospective Studies
Four studies were prospective in design, providing a temporal perspective of symptoms during and after AML chemotherapy, with a follow-up period ranging from approximately 6 to 39 weeks. Of the four studies, three [13, 15, 17] assessed HRQoL beginning at diagnosis, while the fourth study measured HRQoL at the end of patients’ induction therapy and followed patients for 9 months post-induction [20].
In the majority of the studies, short-term trends—from diagnosis to week 10—depict a rapid worsening of HRQoL across all domains shortly after diagnosis, followed by a gradual increase in HRQoL from week 2 to 6 (Fig. 2; Appendix Figure 1). Exceptions to this trend were reported by Moller et al., where the poorest HRQoL scores were observed at baseline, followed by consistent improvements in functional and symptom domains over time. This observation is likely due to the lack of an assessment following induction through to week 12. Symptom and functional domains reported similar short- and long-term trends (Appendix Figure 1).
HRQoL by AML Subgroup
A single study reported HRQoL of relapsed AML survivors; no information of refractory participants was identified. HRQoL in relapsed participants was worse than in non-relapsed participants (EORTC QLQ-C30: 72 vs. 76; EQ-5D utility: 0.8 vs. 0.8; and EQ-VAS: 73 vs. 75) [22]. These differences were not reported to be statistically significant, although this could be due to the small sample size of relapsed participants (n = 17) [22].
Quality Assessment
Applying the quality assessment checklist outlined by Jacobs et al. [12], only two studies (14.3%) [15, 18] were deemed robust (Appendix Table 3). When scoring question four of the checklist—use of a validated, disease-specific questionnaire—only leukemia-specific tools were deemed acceptable to meet this criterion. Relaxing this requirement to include both leukemia- and cancer-specific tools resulted in five (35.7%) studies designated as robust [15, 18, 19, 22, 23]. The remaining studies were not deemed robust [13, 14, 16, 17, 20, 21, 24–26].
Discussion
Using a robust methodology, 14 studies reporting the HRQoL of individuals with AML were identified in the peer-reviewed literature between 2004 and 2014. HRQoL estimates varied across different subpopulations of AML: HRQoL was superior among AML survivors compared to individuals on active treatment for AML, and one study reported poorer HRQoL of relapsed AML survivors compared to non-relapsed survivors. One study provided comparisons with a general population sample and found individuals with AML had consistently lower HRQoL across all measured domains; however, the difference was not statistically different for all comparisons [22]. An assessment of the quality found few of the studies met the minimum requirements for robust reporting of HRQoL results.
The findings of our SLR are largely in line with the previous SLR published in 2004. Redaelli et al. observed that the largest HRQoL burden occurred shortly after diagnosis and during the course of treatment, and survivors appeared to recover HRQoL almost completely in multiple functional domains [3]. Studies included in the present SLR that measured HRQoL through the treatment period reported the poorest HRQoL shortly after initiating induction therapy followed by a rebound in HRQoL after induction therapy was completed. This observation suggests that AML treatment negatively affects patients’ HRQoL, although the impact of death rates during the study and their effect on the study sample may also be a factor in the observed increase in HRQoL over time.
While developments in identifying diagnostic and prognostic markers for AML have been achieved recently [27], treatment options still remain largely unchanged in the last decade, potentially explaining the agreement in HRQoL trends between the current work and the SLR published in 2004 [3]. A causal pathway linking HRQoL with hard clinical outcomes, such as survival, has not been established; however, HRQoL has been recognized as a predictor of clinical prognosis [28] and is a relevant consideration in disease management. Calls to increase the focus on the patient experience in oncology care further underline the importance of HRQoL [29]. This has led to supportive care initiatives being implemented to address the negative impacts of the disease and treatments on patients’ functional domains including emotional, social, and role functioning. Aside from mitigating the impact of aggressive treatment regimens, the development of novel therapeutic options for AML that are efficacious but less toxic would likely improve HRQoL in this patient population.
Across the included studies, fatigue was systematically identified as an important symptom negatively impacting HRQoL in patients with AML, irrespective of their treatment status. The impact of fatigue was reported to have the most significant impact on HRQoL and persisted over time [20]. These findings echo those of other studies assessing HRQoL of patients with AML [3], leukemia and hematology [4, 30], and in other cancer types [31, 32]. The impact of fatigue on HRQoL is not surprising, as cancer-related fatigue is commonly reported among cancer patients and may be attributable to several causes, including treatment-related side effects, comorbidities, psychosocial issues, or the disease itself [33, 34]. The reasons for the persistence of fatigue past treatment completion are unknown [34]; however, studies have found fatigue to be negatively correlated with global health scores and functional status and positively correlated with loss of appetite and depression in patients with AML [23, 35, 36].
Limited HRQoL data on patients with relapsed or refractory AML or with confirmed cytogenetic mutations were identified in this review. This seeming exclusion of reporting HRQoL by AML subgroups may partially be due to selection bias, as these populations have poorer prognosis, more intensive treatments, worse outcomes, and shorter survival when compared with non-relapsed or non-refractory AML patients. The few numbers of patients with AML may also limit the ability to conduct a robust analysis of these relapsed and refractory AML subgroups. The limited research on HRQoL in hematology, and AML in particular, compared to solid-tumor cancers has been highlighted in previous works [3, 4].
This study has several notable strengths. Robust methodology, including transparent and reproducible methods, was used in accordance with the standard practice [37] to identify and select relevant studies for inclusion using a pre-defined eligibility criteria vetted by clinical experts in AML, and results were reported in accordance with PRISMA guidelines [38]. To our knowledge, this is the only systematic review in the past 10 years reporting HRQoL in patients with AML in the US and Europe. Moreover, the present review aimed to identify published estimates of HRQoL in relapsed or refractory AML patients, which has not been previously summarized in the existing literature. Recognizing that the aim of the study was to present the current state of knowledge regarding HRQoL in AML, the results were presented by meaningful core variables that impact HRQoL.
Limitations to the study must be acknowledged. The SLR design allows for limited interpretation of findings outside of those presented by study authors. Coupled with the wide variety of heterogeneity in key study design and clinical factors—including the number of different instruments utilized to measure HRQoL, the different time points after AML diagnosis when HRQoL was measured, and the different AML subpopulations—the present SLR can offer only broad conclusions on HRQoL in AML in the US and the EU. As this study supports previously identified broad trends in HRQoL of individuals with AML [10], future research may focus on reducing the heterogeneity of included studies to allow for robust methods to arrive at more precise estimates of HRQoL or expand the geographic scope to include countries outside of the US and EU. Finally, we recognize that selection and publication biases are inherent in SLRs. Recognizing the limitations to the study design, we adhered to methods meant to minimize the impact of selection bias by conducting a double, independent review and extraction of identified publications. While limited methods exist for reducing the impact of publication bias in SLRs, including gray-literature sources, or requesting individual participant data from clinical trials where HRQoL outcomes were collected but not reported can help minimize publication bias in future reviews.
Conclusions
Our results demonstrate that the impact of HRQoL on AML patients is significant, with deterioration occurring quickly at the time of diagnosis and treatment start. AML treatment options have been noted as being aggressive and requiring long hospitalizations, which has been touted as the reason for the lack of studies in HRQoL in this disease area [4]. However, a deeper understanding of the relationship between HRQoL in AML, particularly in poor prognosis AML subgroups with limited treatment options, may offer crucial insights into patient management options, including supportive care, and survival. Our findings also highlight the need for high-quality, robust studies assessing HRQoL to quantify the burden on patient HRQoL in these patient populations. Further examinations of the role of HRQoL in AML, particularly by AML subgroups, are warranted.
References
Surveillance Epidemiology and End Results (SEER). SEER Stat Fact Sheets: Acute Myeloid Leukemia (AML). 2015.
National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology. Acute myeloid leukemia version 2.2014. 2014.
Redaelli A, Stephens JM, Brandt S, Botteman MF, Pashos CL. Short- and long-term effects of acute myeloid leukemia on patient health-related quality of life. Cancer Treat Rev. 2004;30:103–17.
Efficace F, Novik A, Vignetti M, Mandelli F, Cleeland CS. Health-related quality of life and symptom assessment in clinical research of patients with hematologic malignancies: where are we now and where do we go from here? Haematologica. 2007;92:1596–8.
Calvert M, Brundage M, Jacobsen PB, Schunemann HJ, Efficace F. The CONSORT patient-reported outcome (PRO) extension: implications for clinical trials and practice. Health Qual Life Outcomes. 2013;11:184.
U.S. Department of Health and Human Services FDA Center for Drug Evaluation and Research, U.S. Department of Health and Human Services FDA Center for Biologics Evaluation and Research, U.S. Department of Health and Human Services FDA Center for Devices and Radiological Health. Guidance for industry: patient-reported outcome measures: use in medical product development to support labeling claims: draft guidance. Health Qual Life Outcomes. 2006;4:79.
Kornblith AB, Herndon JE, Weiss RB, Zhang C, Zuckerman EL, Rosenberg S, et al. Long-term adjustment of survivors of early-stage breast carcinoma, 20 years after adjuvant chemotherapy. Cancer. 2003;98:679–89.
Kent EE, Ambs A, Mitchell SA, Clauser SB, Smith AW, Hays RD. Health-related quality of life in older adult survivors of selected cancers: data from the SEER-MHOS linkage. Cancer. 2015;121:758–65.
Mehnert A, Koch U. Psychological comorbidity and health-related quality of life and its association with awareness, utilization, and need for psychosocial support in a cancer register-based sample of long-term breast cancer survivors. J Psychosom Res. 2008;64:383–91.
Efficace F, Kemmler G, Vignetti M, Mandelli F, Molica S, Holzner B. Health-related quality of life assessment and reported outcomes in leukaemia randomised controlled trials—a systematic review to evaluate the added value in supporting clinical decision making. Eur J Cancer. 2008;44:1497–506.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8:336–41.
Jacobs M, Macefield RC, Blazeby JM, Korfage IJ, van Berge Henegouwen MI, de Haes HC, et al. Systematic review reveals limitations of studies evaluating health-related quality of life after potentially curative treatment for esophageal cancer. Qual Life Res. 2013;22:1787–803.
Battaglini CL, Hackney AC, Garcia R, Groff D, Evans E, Shea T. The effects of an exercise program in leukemia patients. Integr Cancer Ther. 2009;8:130–8.
Schultz KAP, Chen L, Chen Z, Kawashima T, Oeffinger KC, Woods WG, et al. Health conditions and quality of life in survivors of childhood acute myeloid leukemia comparing post remission chemotherapy to BMT: a report from the children’s oncology group. Pediatr Blood Cancer. 2014;61:729–36.
Klepin HD, Danhauer SC, Tooze JA, Stott K, Daley K, Vishnevsky T, et al. Exercise for older adult inpatients with acute myelogenous leukemia: a pilot study. J Geriatr Oncol. 2011;2:11–7.
Oliva EN, Nobile F, Alimena G, Ronco F, Specchia G, Impera S, et al. Quality of life in elderly patients with acute myeloid leukemia: patients may be more accurate than physicians. Haematologica. 2011;96:696–702.
Sekeres MA, Stone RM, Zahrieh D, Neuberg D, Morrison V, De Angelo DJ, et al. Decision-making and quality of life in older adults with acute myeloid leukemia or advanced myelodysplastic syndrome. Leukemia. 2004;18:809–16.
Bieri S, Roosnek E, Helg C, Verholen F, Robert D, Chapuis B, et al. Quality of life and social integration after allogeneic hematopoietic SCT. Bone Marrow Transpl. 2008;42:819–27.
Watson M, Buck G, Wheatley K, Homewood JR, Goldstone AH, Rees JK, et al. Adverse impact of bone marrow transplantation on quality of life in acute myeloid leukaemia patients; analysis of the UK Medical Research Council AML 10 Trial. Eur J Cancer. 2004;40:971–8.
Moller T, Adamsen L, Appel C, Welinder P, Stage M, Jarden M, et al. Health related quality of life and impact of infectious comorbidity in outpatient management of patients with acute leukemia. Leuk Lymphoma. 2012;53:1896–904.
Johannsdottir IM, Hjermstad MJ, Moum T, Wesenberg F, Hjorth L, Schroder H, et al. Increased prevalence of chronic fatigue among survivors of childhood cancers: a population-based study. Pediatr Blood Cancer. 2012;58:415–20.
Leunis A, Redekop WK, Uyl-de Groot CA, Lowenberg B. Impaired health-related quality of life in acute myeloid leukemia survivors: a single-center study. Eur J Haematol. 2014;93:198–206.
Messerer D, Engel J, Hasford J, Schaich M, Ehninger G, Sauerland C, et al. Impact of different post-remission strategies on quality of life in patients with acute myeloid leukemia. Haematologica. 2008;93:826–33.
Slovacek L, Slovakova B, Jebavy L. Health and psychosocial aspects of quality of life in patients undergoing autologous progenitor stem cell transplantation [Czech]. Cas Lek Cesk. 2006;145:949–952.
Lopez-Jimenez J, Martin-Ballesteros E, Sureda A, Uralburu C, Lorenzo I, del CR et al. Chemotherapy-induced nausea and vomiting in acute leukemia and stem cell transplant patients: results of a multicenter, observational study. Haematologica. 2006;91:84–91.
Sherman AE, Motyckova G, Fega KR, DeAngelo DJ, Abel GA, Steensma D, Wadleigh M, Stone RM, Driver JA. Geriatric assessment in older patients with acute myeloid leukemia: A retrospective study of associated treatment and outcomes. Leuk Res. 2013;37(9):998–1003.
Dohner H, Estey EH, Amadori S, Appelbaum FR, Buchner T, Burnett AK, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 2010;115:453–74.
Montazeri A. Quality of life data as prognostic indicators of survival in cancer patients: an overview of the literature from 1982 to 2008. Health Qual Life Outcomes. 2009;7:102.
Institute of Medicine. Delivering high-quality cancer care: charting a new course for a system in crisis. Washington, DC: The National Academies Press; 2013.
Allart-Vorelli P, Porro B, Baguet F, Michel A, Cousson-Gelie F. Haematological cancer and quality of life: a systematic literature review. Blood Cancer J. 2015;5:e305.
Bower JE, Ganz PA, Desmond KA, Bernaards C, Rowland JH, Meyerowitz BE, et al. Fatigue in long-term breast carcinoma survivors: a longitudinal investigation. Cancer. 2006;106:751–8.
Baker F, Denniston M, Smith T, West MM. Adult cancer survivors: how are they faring? Cancer. 2005;104:2565–76.
Wagner LI, Cella D. Fatigue and cancer: causes, prevalence and treatment approaches. Br J Cancer. 2004;91:822–8.
Hofman M, Ryan JL, Figueroa-Moseley CD, Jean-Pierre P, Morrow GR. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12(Suppl 1):4–10.
Schumacher A, Wewers D, Heinecke A, Sauerland C, Koch OM, van de Loo J, et al. Fatigue as an important aspect of quality of life in patients with acute myeloid leukemia. Leuk Res. 2002;26:355–62.
Alibhai SM, Leach M, Kowgier ME, Tomlinson GA, Brandwein JM, Minden MD. Fatigue in older adults with acute myeloid leukemia: predictors and associations with quality of life and functional status. Leukemia. 2007;21:845–8.
The Cochrane Collaboration. Cochrane handbook for systematic reviews of interventions. Version 5.1.0 (updated March 2011), 2011.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6:e1000100.
Acknowledgements
The study and article processing charges were funded by Astellas Pharma, Inc., Northbrook, IL, USA. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published. All co-authors contributed to study design. Ellen E. Korol and Sisi Wang performed the data abstract and synthesis, the results of which were reviewed and interpreted by all co-authors. Ellen E. Korol and Sisi Wang were responsible for drafting of the manuscript, and all co-authors provided critical review and editing of the manuscript draft. All co-authors have provided final approval of and take responsibility for the version to be published. No co-authors received an honorarium or grant for participation in the manuscript. The authors wish to acknowledge the efforts of Manpreet Sidhu, Aleksandra Gara, and Nathalie Waser for their contributions to this research project. The authors further wish to acknowledge Milana Zivkov and Cat Bui for their helpful feedback and review of the manuscript and Regina Switzer for her assistance in preparing the manuscript for publication and assisted with the formatting required by the journal. This was funded by Astellas Pharma, Inc. Data informing this manuscript were obtained from peer-reviewed literature published in the data sources described and conference abstracts; specific articles and abstracts are listed in the reference list.
Disclosures
Floortje van Nooten was an employee of Astellas Pharma, Inc., during the course of this research and manuscript development. Floortje van Nooten is currently an employee of Dompé Farmaceutici S.p.A. Karissa Johnston was an employee of ICON Commercialisation & Outcomes during the course of this research and manuscript development). Karissa Johnston is currently an employee of Broadstreet Health Economics and Outcomes Research. Ellen E. Korol is an employee of ICON Commercialisation and Outcomes. Sisi Wang is an employee of ICON Commercialisation and Outcomes. Mark Levis is a paid consultant to ICON plc. Farhad Ravandi-Kashani is a paid consultant to ICON plc.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no data sets were generated or analyzed during the current study. All data were taken from original publications available in peer-reviewed literature sources that are publically available.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/F957F0605D35BF94.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Korol, E.E., Wang, S., Johnston, K. et al. Health-Related Quality of Life of Patients with Acute Myeloid Leukemia: A Systematic Literature Review. Oncol Ther 5, 1–16 (2017). https://doi.org/10.1007/s40487-016-0039-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40487-016-0039-6