Abstract
Necrosis is a common feature of solid tumours that offers a unique opportunity for targeted cancer therapy as it is absent from normal healthy tissues. Tumour necrosis provides an ideal environment for germination of the anaerobic bacterium Clostridium from endospores, resulting in tumour-specific colonisation. Two main species, Clostridium novyi-NT and Clostridium sporogenes, are at the forefront of this therapy, showing promise in preclinical models. However, anti-tumour activity is modest when used as a single agent, encouraging development of Clostridium as a tumour-selective gene delivery system. Various methods, such as allele-coupled exchange and CRISPR–cas9 technology, can facilitate the genetic modification of Clostridium, allowing chromosomal integration of transgenes to ensure long-term stability of expression. Strains of Clostridium can be engineered to express prodrug-activating enzymes, resulting in the generation of active drug selectively in the tumour microenvironment (a concept termed Clostridium-directed enzyme prodrug therapy). More recently, Clostridium strains have been investigated in the context of cancer immunotherapy, either in combination with immune checkpoint inhibitors or with engineered strains expressing immunomodulatory molecules such as IL-2 and TNF-α. Localised expression of these molecules using tumour-targeting Clostridium strains has the potential to improve delivery and reduce systemic toxicity. In summary, Clostridium species represent a promising platform for cancer therapy, with potential for localised gene delivery and immunomodulation selectively within the tumour microenvironment. The ongoing clinical progress being made with C. novyi-NT, in addition to developments in genetic modification techniques and non-invasive imaging capabilities, are expected to further progress Clostridium as an option for cancer treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Clostridium are a genus of anaerobic bacteria that can colonise tumour necrosis. |
Strains of Clostridium can be engineered to express prodrug activating enzymes and immunomodulatory molecules selectively within the tumour microenvironment. |
1 Clostridium Species
Clostridium is a genus of Gram-positive obligate anaerobic bacteria [1]. In aerobic environments they exist as inert endospores, germinating into metabolically active vegetative cells in the absence of oxygen. This genus includes some well-known pathogenic species, such as Clostridium tetani and Clostridium botulinum, but most are generally non-pathogenic and reside in the subsoil [2], or the intestinal tract of animals and humans [3]. Clostridium species have been widely used in biotechnology to produce biofuels, such as ethanol [4] and butanol [5], but also have therapeutic applications due to their anaerobic nature. For example, areas of tumour necrosis are known to support the selective germination and growth of anaerobic bacteria such as Clostridium (Fig. 1) [6].
2 The Use of Clostridium in Cancer Therapy
Areas of tumour necrosis are commonly observed in the core regions of solid tumours due to insufficient vascularisation and subsequent metabolic stresses such as hypoxia and nutrient deprivation. Tumour necrosis is associated with poor prognosis in a wide range of cancer types and can be prevalent at end-stage disease [7]. Being completely absent from normal healthy tissues, tumour necrosis ensures that germination and growth of Clostridium is targeted selectively to the tumour microenvironment. Two main species of Clostridium are frequently used in this context, namely Clostridium novyi and Clostridium sporogenes.
The most clinically advanced Clostridium species used for cancer therapy is C. novyi-NT, an attenuated variant of C. novyi with the lethal α-toxin gene removed. C. novyi-NT was first identified as a promising species from a panel of 26 anaerobic bacteria that were screened for their ability to colonise and grow within necrotic tumours [8]. In pre-clinical mouse (CT26) and rabbit (VX2) models, intravenously injected endospores of C. novyi-NT were shown to germinate in necrotic tumour areas, producing complete responses in approximately 30% of treated animals [8,9]. This effect was immune-mediated, with cured animals developing long-term cellular immunity to the original tumours [9]. However, in a subsequent dose-escalation study in dogs with spontaneously occurring tumours, intravenous injection of C. novyi-NT endospores resulted in significant toxicities that necessitated the study to be discontinued [10]. This included anaemia, thought to be a result of tumour-related haemorrhage, and abscesses that required extensive surgical debridement. A follow-up study evaluated direct intratumoural injection, where 37.5% of treated dogs had objective responses (three complete and three partial responses in 16 animals) [11]. Based on the safety and efficacy observed in this study, a phase I clinical trial was initiated in patients with treatment-refractory solid tumours (NCT01924689). Radiological and clinical signs of tumour lysis were observed in 42% of treated patients with injected lesions, with stable disease being the best overall response [12]. However, this was associated with some significant toxicities, including sepsis and gas gangrene. All patients who experienced these dose-limiting toxicities eventually recovered.
Whilst promising, intratumoural injection of C. novyi-NT endospores limits the ability to target tumours beyond the reach of percutaneous injection. The use of C. sporogenes could allow for successful intravenous delivery of endospores. The first clinical trials for the use of C. sporogenes (then named C. butyricum M55) on cancer patients were initiated in the 1960s and 1970s [13,14]. In general, intravenous administration of endospores was well tolerated, with patients mainly experiencing fever and leukocytosis that was controllable by antibiotics [13]. Tumours in these patients frequently became liquified (indicative of oncolysis) and required drainage, but complete tumour regression was not observed [13]. More recently, C. sporogenes has been attenuated to enhance clinical safety, after identification of a nine gene cluster with high sequence similarity to the streptolysin S (SLS) operon of Streptococcus pyrogenes [15]. This haemolysis operon was deleted from the genome using CRISPR–cas9 technology, with the resulting strain (C. sporogenes-NT) now free from haemolytic activity and thus better positioned for successful clinical translation [16].
3 Clostridium as a Vector for Solid Tumours
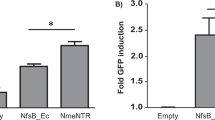
Initially, Clostridium species demonstrated modest anti-tumour activity when used as a single agent. However, tumour regrowth eventually occurred from an outer rim of viable oxygenated cells where Clostridium could neither germinate nor penetrate. This led to the idea that engineered strains of Clostridium could be used as a tumour-selective gene delivery system [17]. In early studies, the enzyme cytosine deaminase (CD) was cloned into a clostridial expression vector and transformed into Clostridium beijerinckii [17]. CD can be used to convert the non-toxic prodrug 5-fluorocytosine (5-FC) into the active chemotherapy agent 5-fluorouracil (5-FU) [18]. Significant levels of CD in the bacterial supernatant of transformed C. beijerinckii cultures increased the sensitivity of tumour cells to 5-fluorocytosine by up to 500-fold, indicating successful expression and secretion of functional CD from C. beijerinckii [17]. Later, these findings were confirmed in an in vivo setting using a strain of C. beijerinckii engineered to express a nitroreductase enzyme from Escherichia coli, NfsB [19]. Nitroreductase activity was detectable in ten out of ten tumours for 5 days after intravenous injection of NfsB-expressing C. beijerinckii endospores, with no nitroreductase protein observed in normal tissues, demonstrating proof-of-principle for selective gene delivery to the tumour using Clostridium [19].
There are many advantages to using Clostridium species as a vector for solid tumours over other vector options (Fig. 2). For example, Clostridium endospores are not immunogenic [20], allowing for repeated systemic administration of endospores without neutralisation by circulating antibodies or the complement system. The ability to perform repeated intravenous injections of Clostridium endospores was first demonstrated in early human studies [14]. These findings were later confirmed in preclinical models, where multiple consecutive treatment cycles of Clostridium infection were possible in between periods of antibiotic treatment to clear the infection [21]. Another advantage to the use of Clostridium as a vector is their genomic size and autonomy. Larger gene inserts at more than one loci are feasible, and infection and lysis of the host cells is not required for successful bacterial replication. As a consequence, there is minimal risk of recombination with the host genome and no loss of the transgene or any intracellular co-factors to the extracellular space. Germinated Clostridium are also flagellated, providing motility that can facilitate intratumoural spread. Importantly, control of proliferation is possible using antibiotics, either at the conclusion of treatment or if an adverse event occurs. Sensitivity to generic antibiotics such as metronidazole is an attractive safety feature that can help to alleviate safety concerns around the use of replicating biological vectors in humans.
Schematic representation of the key advantages in using Clostridium species as vectors for solid tumours. A Genomic autonomy minimises the risk of recombination with the host genome, B flagella provide motility to promote intratumoural spread, C genomic size allows for larger gene inserts at more than one loci, D control of proliferation is possible with generic antibiotics, and E Clostridium endospores are not immunogenic, allowing for repeated intravenous (IV) injections
4 Methods for Genetic Modification of Clostridium
To successfully develop Clostridium for use as a vector, methods that allow for construction of stable recombinant strains are essential. Initially, Clostridium species were successfully engineered to express genes from autonomous plasmids. Saccharolytic Clostridium strains such as C. beijerinckii were the first to be engineered for plasmid-based expression of various transgenes (covered in Sect. 3), but colonisation of tumours using these strains was low. Proteolytic Clostridium strains were much better at colonising tumour tissue but were initially overlooked due to their resistance to genetic modification. Eventually, a transformation method was developed for the proteolytic strain C. sporogenes that involved use of a polyethylene glycol based transfection buffer [22], but this method had low efficiency and reproducibility. Subsequent transformation protocols based on conjugation or electroporation were then developed for C. sporogenes that were highly successful [21,23], allowing for tumour-specific expression of a transgene following colonisation by Clostridium. However, plasmid-based expression of genes can result in segregational instability, in which the proportion of plasmid-containing cells within a population decreases over multiple cycles of replication [24,25].
More recently, a reliable method for the stable integration of expression constructs into the chromosome of Clostridium was developed, termed Allele-Coupled Exchange (ACE) [26]. ACE is based on a double-crossover homologous recombination event that transfers a region of a plasmid into a specific site in the Clostridium genome. Each gene sequence is usually codon optimised to ensure efficient expression in the Clostridium host. Formation of the desirable double-crossover cells is coupled to an associated selectable phenotype that allows these cells to be isolated. Clostridium strains generated using ACE have the transgene inserted into the pyrE locus. The pyrE gene is essential for Clostridium to synthesise uracil, a nucleobase that plays a role in the manufacture of proteins. By inactivating pyrE during integration, recombinant strains are rendered uracil auxotrophs, disabled from undesirable growth in the environment. Consequently, ACE does not require the use of antibiotic resistance markers. In addition, integration of the transgene into the chromosome provides complete segregational stability with no mechanism for horizontal gene transfer. Importantly, ACE allows for generation of Clostridium endospore preparations that are compatible with clinical regulatory requirements. Several recombinant strains of C. sporogenes have been generated using ACE technology, with functional expression of the desired transgene (a bacterial nitroreductase enzyme from Neisseria meningitides, NmeNTR) demonstrated [27,28]. However, ACE can be time consuming and large gene inserts (≥ 28 kb) can decrease transformation efficiency [26].
The development of CRISPR–Cas9 genome editing technology has revolutionised the ability to engineer recombinant strains of Clostridium [29,30]. CRISPR–Cas9 allows genetic material to be added, modified, or deleted at precise locations in the genome using short ‘guide’ sequences of RNA that direct the Cas nuclease to its target. Here, double-stranded breaks are created in the targeted DNA sequence and one of the two main DNA repair pathways can then be exploited to render genes non-functional (non-homologous end joining) or to insert new genes (homology-directed repair). CRISPR–Cas9 has the advantage of being simple, quick and cheap to perform in Clostridium compared with other techniques. It has been performed successfully in both C. novyi-NT (genomic modification) [31] and C. sporogenes (gene insertion and gene deletion) [16,32], although it appears that design of optimal constructs and guide RNAs is strain-specific. The use of CRISPR–Cas9 is not without its challenges, however. Expression of Cas9 in bacteria can be toxic when combined with guide RNAs that have specific motifs at their protospacer adjacent motif (PAM)-proximal end [33]. These Cas9 complexes have been shown to bind at off-target positions within the bacterial chromosome, silencing essential genes and causing toxicity [34]. Alternative endonucleases (e.g. Cas12a, also known as Cpf1) have since been developed to overcome limitations of Cas9 and may prove more advantageous for genomic modification of Clostridium in future [35,36,37].
5 Clostridium-Directed Enzyme Prodrug Therapy
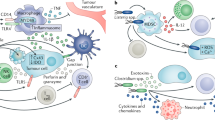
Clostridium has been used as a tumour-selective gene delivery vector primarily in the context of Clostridium-directed enzyme prodrug therapy (CDEPT). CDEPT is a cancer gene therapy approach whereby an exogenous therapeutic enzyme expressed by Clostridium can metabolise otherwise inert prodrugs into active, cytotoxic metabolites (Fig. 3). Localised activation of prodrug in this way can create an improved therapeutic index and, in principle, produce continuous active drug concentrations within the tumour microenvironment. Depending on the tissue penetration capacity of the active metabolite(s), cytotoxic products may diffuse into and kill adjacent cells within the tumour; a phenomenon termed the ’bystander effect’. A robust bystander effect would allow for cell killing outside of the necrotic boundaries, beyond where Clostridium germination is possible.
Schematic representation of Clostridium-directed enzyme prodrug therapy (CDEPT) using the enzyme/prodrug combination of nitroreductase/PR-104A as an example. (1) Inert prodrugs can be administered systemically and diffuse into the tumour. (2) Exogenous therapeutic enzyme expressed by Clostridium can metabolise the inert prodrug into active, cytotoxic metabolites. (3) These metabolites can be designed to diffuse out of necrotic regions into the outer rim of viable tumour (metabolite ‘bystander’ effect)
-
(i)
Enzyme-prodrug combinations
The therapeutic success of CDEPT relies partly on the properties of the enzyme/prodrug combination used. The prodrug activating enzyme should have a high kcat (catalytic rate) and low Km (substrate concentration required for effective catalysis to occur) to maximise production of the active metabolite. The prodrug itself should be a stable, water soluble, systemic agent that remains inert in normal tissues, with good extravascular penetration into the tumour. The physiochemical properties of the active prodrug metabolites should allow for good cellular uptake and the ability to diffuse throughout the tumour microenvironment to generate bystander cell killing. A range of enzyme/prodrug combinations have been studied in the context of CDEPT, although none have been evaluated in clinical trial to date.
Proteolytic Clostridium strains engineered for CDEPT were the first to demonstrate beneficial effects on tumour growth in vivo after administration of prodrug, likely due to their improved ability to colonise tumours. Initial studies were performed using the CD/5-FC enzyme/prodrug combination. Intravenous injection of a strain C. sporogenes engineered to express CD resulted in expression of active CD exclusively in the tumour [22]. The combination of these CD-expressing spores and prodrug 5-FC produced a larger tumour growth delay than that produced by 5-FU (the active metabolite of 5-FC) when given at maximum tolerated dose [22]. However, the cell cycle selectivity of 5-FU [38] is a major drawback to this particular enzyme/prodrug combination.
Bacterial nitroreductase (NTR) enzymes metabolise a diverse range of nitroheterocyclic substrates [39] and provide an ideal alternative to CD for expression in Clostridium. Traditionally, NTRs have been used to convert the non-toxic prodrug CB1954 to the active metabolites that cause DNA mono adducts and crosslinks [40]. The NTR/CB1954 combination is not cell cycle specific, as killing has been observed in both cycling and quiescent cells [41]. Clostridium has been engineered to express various bacterial NTRs for use in combination with CB1954. For example, intravenously injected spores of C. sporogenes expressing an NTR from Haemophilus influenzae produced significant in vivo anti-tumour efficacy when combined with administration of CB1954 (P < 0.0001 when compared with untreated control animals) [21]. Interestingly, bacteria were cleared from the animals in this study using antibiotics between treatment cycles, with no reduction in numbers of germinated bacteria occurring after repeated administration of spores. This was in line with previous observations suggesting that repeated spore administration is not immunogenic to the host [20,42]. More recently, a novel nitroreductase from Neisseria meningitidis (NmeNTR) was expressed from a strain of C. sporogenes that is potentially suitable for clinical trial due to the absence of antibiotic resistance markers [27]. NmeNTR-expressing C. sporogenes in combination with CB1954 produced a significant growth delay (P < 0.001 when compared with untreated control animals), with 4/16 tumours undergoing maintained complete regressions [27]. In contrast, CB1954 administered alone had no effect on tumour volume, and a small, non-significant growth delay was observed when NmeNTR-expressing C. sporogenes spores were administered as a single agent [27]. Whilst encouraging, the dose of CB154 used in these studies is in excess of that which can be safely achieved in humans [43]. In addition, the NTRs described produce the 4-hydroxylamine metabolite of CB1954 exclusively [44], resulting in a limited bystander cell killing.
As an alternative to CB1954, the prodrug PR-104A (or its phosphate ester ‘pre-prodrug’ PR-104) has been proposed for use in combination with NTR due to its improved potency and potential for bystander cell killing [45]. PR-104 has been used in pre-clinical models of E. coli NTR-armed CDEPT with considerable success [23]. This NTR/prodrug combination produced superior anti-tumour activity (tumour growth delay > 13 days, P < 0.05) compared with NTR-expressing spores or PR-104 alone [23]. The NmeNTR strain of C. sporogenes described above also demonstrated a significant improvement in survival in combination with PR-104, an increase of 20 days compared with untreated control animals (P < 0.001) [28]. The superior activity of the NmeNTR/PR-104 combination was confirmed by a 100% overall response rate as determined by RECIST 1.1 criteria [28]. However, PR-104A has been identified as a substrate for aldo-keto reductase 1C3 (AKR1C3) [46], a human enzyme involved in steroid hormone and prostaglandin regulation [47]. Reduction of PR-104A by AKR1C3 is thought to contribute to the dose-limiting toxicities observed in humans that significantly restricts exposure [48]. Fortunately, novel analogues of PR-104A are being developed that are resistant to metabolism by AKR1C3 but still have significant activity with NTR enzymes [49,50,51].
-
(i)
Non-invasive imaging capability
For decades, reporter genes such as β-galactosidase and Renilla luciferase have been used to monitor transgene expression in tissue biopsy samples. However, these techniques are invasive and time consuming and are therefore not suitable for repeated clinical evaluation. With tomographic scanning becoming more routine, the ability to non-invasively image certain reporter genes is now possible. This would provide a means to accurately determine Clostridium distribution and replication in real-time, particularly after systemic administration, and may assist with expediting clinical development of Clostridium vectors.
CD and NTR enzymes are most commonly imaged in vitro and in vivo using red or near-infrared fluorescent probes [52,53,54]. However, there are issues with light penetration through tissue, as it has been predicted that fluorescent light cannot penetrate beyond the depth of several centimetres [55]. The recent demonstration that NTR enzymes can metabolise positron emission tomography (PET) imaging agents already in clinical development for imaging of hypoxia is a significant advantage to the use of this enzyme in the CDEPT context over CD. 18F-labelled 2-nitroimidazole compounds can detect hypoxic cells following an oxygen-sensitive single electron reduction step, leading to further reduction to the reactive products that can covalently bind to cell components and accumulate inside the cell [56]. NTR was hypothesised to bypass the oxygen-sensitive step to produce the reactive products directly by two-electron reduction [39]. The hypoxia PET probe 18F-HX4 was first used to detect expression of an NTR from E. coli, where a significantly higher tumour-to-blood ratio was observed for tumours over-expressing NTR compared with corresponding parental tumour [57]. These results were confirmed in a separate study using 18F-FMISO to detect NTR expression, including in small metastatic lesions [58]. However, the innate background signal from hypoxic tissue may somewhat limit the utility of these probes for NTR imaging. NTR-selective analogues of these 2-NI PET probes are being developed to overcome this issue [59].
-
(iii)
Increased sensitivity to antibiotics
An advantage to NTR as a transgene is its catalytic activity across a range of antibiotic and anti-infective substrates, providing an additional safety feature which may be useful for clinical development. The hypersensitivity to these substrates that is provided by the insertion of NTR could, in theory, assist in eliminating Clostridium infection at the end of treatment or if an adverse event occurs. Metronidazole (FlagylTM) is most commonly associated with the treatment of anaerobic bacterial infections [60], and expression of NTR has been shown to render host cells more sensitive to this antibiotic [28,61]. NTR has also shown activity with other anti-infective substrates such as tinidazole, nitrofurantoin, nimorazole and misonidazole, offering some alternative options in case of antibiotic resistance [28].
6 Clostridium in the Context of Immunotherapy
Cancer immunotherapies have been widely hailed as a breakthrough for cancer treatment in the last decade, epitomised by the unprecedented results observed with immune checkpoint inhibitors (ICIs). This paradigm shift towards harnessing an anti-tumour immune response as a treatment strategy has led to a renaissance for tumour-targeting bacteria such as Clostridium. Strains of Clostridium can be engineered to express immunomodulatory molecules (Fig. 4), resulting in localised delivery to the tumour microenvironment whilst potentially minimising systemic toxicity, thus improving therapeutic index.
Schematic representation of how Clostridium can be utilised in the context of cancer immunotherapy. (1) Clostridium strains can be engineered to express immunomodulatory molecules selectively within the tumour microenvironment, improving therapeutic index. (2) Transgene-free Clostridium can be used to promote immune cell infiltration into the tumour microenvironment, potentially sensitising tumours to immunotherapy treatment
-
(i)
Interleukin 2 and Tumour Necrosis Factor alpha
Interleukin 2 (IL-2) is a key cytokine with pleiotropic effects on the immune system, including the differentiation and activation of T cells [62]. High doses of recombinant IL-2 can induce tumour regression in patients [63,64], leading to its approval by the FDA for the treatment of metastatic melanoma and metastatic renal cell carcinoma. However, systemic administration of this molecule can cause significant dose-limiting toxicities, including high serum cytokine levels that appear to be directly or indirectly toxic to multiple organs [65]. This makes IL-2 an ideal candidate for tumour selective Clostridium-mediated expression. Initial studies used C. acetobutylicum DSM792 for plasmid-based expression of rat IL-2 at therapeutically relevant concentrations [66]. More recently, C. sporogenes-NT was used for plasmid-based expression of mouse IL-2 [16].
Tumour necrosis factor alpha (TNF-α) is a major regular of inflammatory responses and can be a potent mediator of tumour regression 67. Similar to IL-2, systemic administration of this cytokine is not possible as it results in serious dose-limiting toxicities that are associated with sepsis [68]. Therefore, a strain of C. acetobutylicum DSM792 was engineered to express mouse TNF-α from a plasmid in an attempt to improve tumour selectivity [69]. Biological activity of the secreted TNF-α was confirmed in a bioassay using WEH164 clone 13 cells (which are highly sensitive to the cytotoxic effect of TNF-α), and the plasmid used to derive TNF-α expression was segregationally stable [69]. Whilst the above studies report expression and secretion of a biologically active cytokine from Clostridium, none of the engineered strains were tested for therapeutic efficacy in vivo. This next step will be crucial for progression of cytokine-expressing strains towards clinical trial.
-
(ii)
Nanobodies
ICIs target inhibitory receptors (e.g., CTLA-4, PD-1) and ligands (e.g., PD-L1) expressed on T-cells, antigen presenting cells and tumour cells to stimulate the immune system and elicit an anti-tumour response. Diseases with historically poor long-term survival (e.g., advanced melanoma, advanced non-small cell lung cancer) are now experiencing significantly improved therapeutic outcomes [70,71]. Unfortunately, ICIs can be associated with systemic autoimmune-related adverse events of varying severity, often requiring treatment cessation and clinical intervention, thereby limiting therapeutic utility [72]. In addition, some ICIs engage targets deep within the tumour microenvironment; these large macromolecules (146–149 kDa) may experience restricted diffusion into tumour tissue [73]. Clostridium strains offer a unique opportunity to generate high local concentrations of ICI, potentially improving the selectivity and therapeutic efficacy of ICIs in certain patients.
In the past, C. novyi-NT and C. sporogenes have been engineered to produce the variable domain of a heavy-chain antibody (VHH antibody or nanobody) specific for human HIF-1α [74]. The antibodies isolated from these strains retained their binding capacity and specificity for the HIF-1α antigen. It is reasonable to assume that production of VHH antibodies against more immunologically relevant molecules (e.g. CTLA-4, PD-1 and PD-L1) would be possible in the future, analogous to the strain of E. coli Nissle 1917 that produces VHH antibodies against PD-L1 and CTLA-4 [75].
-
(iii)
Clostridium in combination with immune checkpoint inhibitors
Whilst checkpoint blockade has revolutionised cancer therapy, only a minority of patients are able to achieve durable, long-term remissions. Responsive patients often have pre-existing immune cell infiltrate within the tumour microenvironment. As an alternative to immunomodulatory-expressing Clostridium vectors, selective colonisation of the tumour by transgene-free Clostridium strains could alter the local immune microenvironment to one that is more immunogenic through the innate and adaptive immune response to infection (Fig. 4). This approach has the potential to sensitise tumours to subsequent treatment with immunotherapy and improve rates of response, particularly in tumours that are inherently immunosuppressive and thus largely resistant to immunotherapy at present (e.g. pancreatic cancer and glioblastoma [76,77]).
Early clinical data with C. novyi-NT demonstrated that intratumoural injection of endospores produced a transient systemic cytokine response and tumour-specific T-cell responses [12]. These data supported an additional phase Ib dose escalation study to assess the safety and potential synergistic effects of C. novyi-NT in combination with pembrolizumab in advanced solid tumours (NCT03435952). Initial results indicate that this combination is well tolerated in the 16 treatment-refractory patients recruited thus far, with a confirmed overall objective response rate of 25% (made up of three partial responses and one complete response) [78]. Currently there is no confirmation of tumour colonisation in responding patients or information on host immune responses. This proof-of-principle study is ongoing to identify the optimal dose for phase II.
7 Conclusion
Clostridium species are a promising and versatile vector system for use in cancer therapy, with many advantages over traditional vector platforms. Recent developments in methods for the genetic modification of Clostridium have allowed for expression of prodrug-activating genes and immunotherapeutics selectively within the tumour microenvironment. However, clinical development has been slow thus far, likely due to challenging and complex regulatory requirements. The ongoing clinical progress being made with C. novyi-NT, in addition to the potential for non-invasive monitoring of transgene expression through PET imaging, should help to demonstrate the feasibility and relative safety of Clostridium vectors and hopefully lead to additional clinical studies in this area.
References
Cruz-Morales P, Orellana CA, Moutafis G, Moonen G, Rincon G, Nielsen LK, et al. Revisiting the evolution and taxonomy of Clostridia, a phylogenomic update. Genome Biol Evol. 2019;11:2035–44.
Pahalagedara ASNW, Jauregui R, Maclean P, Altermann E, Flint S, Palmer J, et al. Culture and genome-based analysis of four soil Clostridium isolates reveal their potential for antimicrobial production. BMC Genomics. 2021;22:686.
Lopetuso LR, Scaldaferri F, Petito V, Gasbarrini A. Commensal Clostridia: leading players in the maintenance of gut homeostasis. Gut Pathogens. 2013;5:23.
Fathima AA, Sanitha M, Kumar T, Iyappan S, Ramya M. Direct utilization of waste water algal biomass for ethanol production by cellulolytic Clostridium phytofermentans DSM1183. Bioresour Technol. 2016;202:253–6.
Nakas JP, Schaedle M, Parkinson CM, Coonley CE, Tanenbaum SW. System development for linked-fermentation production of solvents from algal biomass. Appl Environ Microbiol. 1983;46:1017–23.
Minton NP. Clostridia in cancer therapy. Nat Rev Microbiol. 2003;1:237–42.
Richards CH, Mohammed Z, Qayyum T, Horgan PG, McMillan DC. The prognostic value of histological tumor necrosis in solid organ malignant disease: a systematic review. Future Oncol. 2011;7:1223–35.
Dang LH, Bettegowda C, Huso DL, Kinzler KW, Vogelstein B. Combination bacteriolytic therapy for the treatment of experimental tumors. Proc Natl Acad Sci USA. 2001;98:15155–60.
Agrawal N, Bettegowda C, Cheong I, Geschwind J-F, Drake CG, Hipkiss EL, et al. Bacteriolytic therapy can generate a potent immune response against experimental tumors. Proc Natl Acad Sci USA. 2004;101:15172–7.
Krick EL, Sorenmo KU, Rankin SC, Cheong I, Kobrin B, Thornton K, et al. Evaluation of Clostridium novyi-NT spores in dogs with naturally occurring tumors. Am J Vet Res. 2012;73:112–8.
Roberts NJ, Zhang L, Janku F, Collins A, Bai R-Y, Staedtke V, et al. Intratumoral injection of Clostridium novyi-NT spores induces antitumor responses. Sci Transl Med. 2014;6:249ra111.
Janku F, Zhang HH, Pezeshki A, Goel S, Murthy R, Wang-Gillam A, et al. Intratumoral injection of Clostridium novyi-NT spores in patients with treatment-refractory advanced solid tumors. Clin Cancer Res. 2021;27:96–106.
Carey RW, Holland JF, Whang HY, Neter E, Bryant B. Clostridial oncolysis in man. Eur J Cancer. 1965;1967(3):37–42.
Heppner F, Möse JR. The liquefaction (oncolysis) of malignant gliomas by a non pathogenic Clostridium. Acta Neurochir (Wien). 1978;42:123–5.
Gonzalez DJ, Lee SW, Hensler ME, Markley AL, Dahesh S, Mitchell DA, et al. Clostridiolysin S, a post-translationally modified biotoxin from Clostridium botulinum. J Biol Chem. 2010;285:28220–8.
Kubiak AM, Bailey TS, Dubois LJ, Theys J, Lambin P. Efficient secretion of murine IL-2 from an attenuated strain of clostridium sporogenes, a novel delivery vehicle for cancer immunotherapy. Front Microbiol. 2021; 12.
Fox ME, Lemmon MJ, Mauchline ML, Davis TO, Giaccia AJ, Minton NP, et al. Anaerobic bacteria as a delivery system for cancer gene therapy: in vitro activation of 5-fluorocytosine by genetically engineered clostridia. Gene Ther. 1996;3:173–8.
Mullen CA, Kilstrup M, Blaese RM. Transfer of the bacterial gene for cytosine deaminase to mammalian cells confers lethal sensitivity to 5-fluorocytosine: a negative selection system. Proc Natl Acad Sci USA. 1992;89:33–7.
Lemmon MJ, van Zijl P, Fox ME, Mauchline ML, Giaccia AJ, Minton NP, et al. Anaerobic bacteria as a gene delivery system that is controlled by the tumor microenvironment. Gene Ther. 1997;4:791–6.
Fabricius EM, Schneeweiss U, Schau HP, Schmidt W, Benedix A. Quantitative investigations into the elimination of in vitro-obtained spores of the non-pathogenic Clostridium butyricum strain CNRZ 528, and their persistence in organs of different species following intravenous spore administration. Res Microbiol. 1993;144:741–53.
Theys J, Pennington O, Dubois L, Anlezark G, Vaughan T, Mengesha A, et al. Repeated cycles of Clostridium-directed enzyme prodrug therapy result in sustained antitumour effects in vivo. Br J Cancer. 2006;95:1212–9.
Liu SC, Minton NP, Giaccia AJ, Brown JM. Anticancer efficacy of systemically delivered anaerobic bacteria as gene therapy vectors targeting tumor hypoxia/necrosis. Gene Ther. 2002;9:291–6.
Liu S-C, Ahn G-O, Kioi M, Dorie M-J, Patterson AV, Brown JM. Optimized clostridium-directed enzyme prodrug therapy improves the antitumor activity of the novel DNA cross-linking agent PR-104. Cancer Res. 2008;68:7995–8003.
Popov M, Petrov S, Kirilov K, Nacheva G, Ivanov I. Segregational instability in E. Coli of expression plasmids carrying human interferon gamma gene and its 3’-end truncated variants. Biotechnol Biotechnol Equip. 2009;23:840–3.
Heap JT, Pennington OJ, Cartman ST, Minton NP. A modular system for Clostridium shuttle plasmids. J Microbiol Methods. 2009;78:79–85.
Heap JT, Ehsaan M, Cooksley CM, Ng Y-K, Cartman ST, Winzer K, et al. Integration of DNA into bacterial chromosomes from plasmids without a counter-selection marker. Nucleic Acids Res. 2012;40: e59.
Heap JT, Theys J, Ehsaan M, Kubiak AM, Dubois L, Paesmans K, et al. Spores of Clostridium engineered for clinical efficacy and safety cause regression and cure of tumors in vivo. Oncotarget. 2014;5:1761–9.
Mowday AM, Dubois LJ, Kubiak AM, Chan-Hyams JVE, Guise CP, Ashoorzadeh A, et al. Use of an optimised enzyme/prodrug combination for Clostridia directed enzyme prodrug therapy induces a significant growth delay in necrotic tumours. Cancer Gene Ther. 2022;29:178–88.
Joseph RC, Kim NM, Sandoval NR. Recent developments of the synthetic biology toolkit for Clostridium. Front Microbiol. 2018;9:154.
McAllister KN, Sorg JA. CRISPR genome editing systems in the genus clostridium: a timely advancement. J Bacteriol. 2019;201:e00219-e319.
Dailey KM, Jacobson RI, Johnson PR, Woolery TJ, Kim J, Jansen RJ et al. Methods and techniques to facilitate the development of Clostridium novyi NT as an effective, therapeutic oncolytic bacteria. Front Microbiol. 2021; 12.
Kubiak AM, Claessen L, Zhang Y, Khazaie K, Bailey TS. Refined control of CRISPR-Cas9 gene editing in Clostridium sporogenes: the creation of recombinant strains for therapeutic applications. Front Immunol. 2023; 14.
Cui L, Vigouroux A, Rousset F, Varet H, Khanna V, Bikard D. A CRISPRi screen in E. coli reveals sequence-specific toxicity of dCas9. Nat Commun. 2018;9:1912.
Rostain W, Grebert T, Vyhovskyi D, Pizarro PT, Tshinsele-Van Bellingen G, Cui L, et al. Cas9 off-target binding to the promoter of bacterial genes leads to silencing and toxicity. Nucleic Acids Res. 2023;51:3485–96.
Zhang Y, Kubiak AM, Bailey TS, Claessen L, Hittmeyer P, Dubois L, et al. Development of a CRISPR-Cas12a system for efficient genome engineering in clostridia. Microbiol Spectr. 2023;11: e0245923.
Hong W, Zhang J, Cui G, Wang L, Wang Y. Multiplexed CRISPR-Cpf1-mediated genome editing in Clostridium difficile toward the understanding of pathogenesis of C. difficile infection. ACS Synth Biol. 2018;7:1588–600.
Zhang J, Hong W, Zong W, Wang P, Wang Y. Markerless genome editing in Clostridium beijerinckii using the CRISPR-Cpf1 system. J Biotechnol. 2018;284:27–30.
Leichman CG. Schedule dependency of 5-fluorouracil. Oncology (Williston Park). 1999;13:26–32.
Williams EM, Little RF, Mowday AM, Rich MH, Chan-Hyams JVE, Copp JN, et al. Nitroreductase gene-directed enzyme prodrug therapy: insights and advances toward clinical utility. Biochem J. 2015;471:131–53.
Anlezark GM, Melton RG, Sherwood RF, Coles B, Friedlos F, Knox RJ. The bioactivation of 5-(aziridin-1-yl)-2,4-dinitrobenzamide (CB1954)–I. Purification and properties of a nitroreductase enzyme from Escherichia coli–a potential enzyme for antibody-directed enzyme prodrug therapy (ADEPT). Biochem Pharmacol. 1992;44:2289–95.
Bridgewater JA, Springer CJ, Knox RJ, Minton NP, Michael NP, Collins MK. Expression of the bacterial nitroreductase enzyme in mammalian cells renders them selectively sensitive to killing by the prodrug CB1954. Eur J Cancer. 1995;31:2362–70.
Gericke D, Dietzel F, Rüster I. Further progress with oncolysis due to local high frequency hyperthermia, local X-irradiation and apathogenic Clostridia*. J Microw Power. 1979;14:163–6.
Chung-Faye G, Palmer D, Anderson D, Clark J, Downes M, Baddeley J, et al. Virus-directed, enzyme prodrug therapy with nitroimidazole reductase: a phase I and pharmacokinetic study of its prodrug, CB1954. Clin Cancer Res. 2001;7:2662–8.
Helsby NA, Ferry DM, Patterson AV, Pullen SM, Wilson WR. 2-Amino metabolites are key mediators of CB 1954 and SN 23862 bystander effects in nitroreductase GDEPT. Br J Cancer. 2004;90:1084–92.
Patterson AV, Ferry DM, Edmunds SJ, Gu Y, Singleton RS, Patel K, et al. Mechanism of action and preclinical antitumor activity of the novel hypoxia-activated DNA cross-linking agent PR-104. Clin Cancer Res. 2007;13:3922–32.
Guise CP, Abbattista MR, Singleton RS, Holford SD, Connolly J, Dachs GU, et al. The bioreductive prodrug PR-104A is activated under aerobic conditions by human aldo-keto reductase 1C3. Cancer Res. 2010;70:1573–84.
Penning TM, Drury JE. Human aldo–keto reductases: Function, gene regulation, and single nucleotide polymorphisms. Arch Biochem Biophys. 2007;464:241–50.
Abbattista MR, Ashoorzadeh A, Guise CP, Mowday AM, Mittra R, Silva S, et al. Restoring tumour selectivity of the bioreductive prodrug PR-104 by developing an analogue resistant to aerobic metabolism by human aldo-keto reductase 1C3. Pharmaceuticals (Basel). 2021;14:1231.
Mowday AM, Ashoorzadeh A, Williams EM, Copp JN, Silva S, Bull MR, et al. Rational design of an AKR1C3-resistant analog of PR-104 for enzyme-prodrug therapy. Biochem Pharmacol. 2016;116:176–87.
Ashoorzadeh A, Mowday AM, Guise CP, Silva S, Bull MR, Abbattista MR, et al. Interrogation of the structure–activity relationship of a lipophilic nitroaromatic prodrug series designed for cancer gene therapy applications. Pharmaceuticals. 2022;15:185.
Ashoorzadeh A, Mowday AM, Abbattista MR, Guise CP, Bull MR, Silva S, et al. Design and biological evaluation of piperazine-bearing nitrobenzamide hypoxia/GDEPT prodrugs: the discovery of CP-506. ACS Med Chem Lett. 2023. https://doi.org/10.1021/acsmedchemlett.3c00321.
Thorne SH, Barak Y, Liang W, Bachmann MH, Rao J, Contag CH, et al. CNOB/ChrR6, a new prodrug enzyme cancer chemotherapy. Mol Cancer Ther. 2009;8:333–41.
Xing L, Deng X, Kotedia K, Ackerstaff E, Ponomarev V, Ling CC, et al. Non-invasive molecular and functional imaging of cytosine deaminase and uracil phosphoribosyltransferase fused with red fluorescence protein. Acta Oncol. 2008;47:1211–20.
McCormack E, Silden E, West RM, Pavlin T, Micklem DR, Lorens JB, et al. Nitroreductase, a near-infrared reporter platform for in vivo time-domain optical imaging of metastatic cancer. Cancer Res. 2013;73:1276–86.
Ntziachristos V, Bremer C, Weissleder R. Fluorescence imaging with near-infrared light: new technological advances that enable in vivo molecular imaging. Eur Radiol. 2003;13:195–208.
Nunn A, Linder K, Strauss HW. Nitroimidazoles and imaging hypoxia. Eur J Nucl Med. 1995;22:265–80.
Mowday AM, Copp JN, Syddall SP, Dubois LJ, Wang J, Lieuwes NG, et al. E. coli nitroreductase NfsA is a reporter gene for non-invasive PET imaging in cancer gene therapy applications. Theranostics. 2020;10:10548–62.
Ruiz de Garibay G, García de Jalón E, Stigen E, Lund KB, Popa M, Davidson B, et al. Repurposing 18F-FMISO as a PET tracer for translational imaging of nitroreductase-based gene directed enzyme prodrug therapy. Theranostics. 2021;11:6044–57.
Williams EM, Rich MH, Mowday AM, Ashoorzadeh A, Copp JN, Guise CP, et al. Engineering Escherichia coli NfsB to activate a hypoxia-resistant analogue of the PET probe EF5 to enable non-invasive imaging during enzyme prodrug therapy. Biochemistry. 2019;58:3700–10.
Löfmark S, Edlund C, Nord CE. Metronidazole is still the drug of choice for treatment of anaerobic infections. Clin Infect Dis. 2010;50(Suppl 1):S16-23.
Copp JN, Mowday AM, Williams EM, Guise CP, Ashoorzadeh A, Sharrock AV, et al. Engineering a multifunctional nitroreductase for improved activation of prodrugs and PET probes for cancer gene therapy. Cell Chem Biol. 2017;24:391–403.
Ross SH, Cantrell DA. Signaling and function of interleukin-2 in T lymphocytes. Annu Rev Immunol. 2018;36:411–33.
Rosenberg SA, Lotze MT, Muul LM, Leitman S, Chang AE, Ettinghausen SE, et al. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. New Engl J Med. 1985;313:1485–92.
Lotze MT, Chang AE, Seipp CA, Simpson C, Vetto JT, Rosenberg SA. High-dose recombinant interleukin 2 in the treatment of patients with disseminated cancer: responses, treatment-related morbidity, and histologic findings. JAMA. 1986;256:3117–24.
Panelli MC, White R, Foster M, Martin B, Wang E, Smith K, et al. Forecasting the cytokine storm following systemic interleukin (IL)-2 administration. J Transl Med. 2004;2:17.
Barbé S, Van Mellaert L, Theys J, Geukens N, Lammertyn E, Lambin P, et al. Secretory production of biologically active rat interleukin-2 by Clostridium acetobutylicum DSM792 as a tool for anti-tumor treatment. FEMS Microbiol Lett. 2005;246:67–73.
Bartsch HH, Pfizenmaier K, Schroeder M, Nagel GA. Intralesional application of recombinant human tumor necrosis factor alpha induces local tumor regression in patients with advanced malignancies. Eur J Cancer Clin Oncol. 1989;25:287–91.
Kimura K, Taguchi T, Urushizaki I, Ohno R, Abe O, Furue H, et al. Phase I study of recombinant human tumor necrosis factor. Cancer Chemother Pharmacol. 1987;20:223–9.
Theys J, Nuyts S, Landuyt W, Van Mellaert L, Dillen C, Böhringer M, et al. Stable Escherichia coli-Clostridium acetobutylicum shuttle vector for secretion of murine tumor necrosis factor alpha. Appl Environ Microbiol. 1999;65:4295–300.
Garon EB, Hellmann MD, Rizvi NA, Carcereny E, Leighl NB, Ahn M-J, et al. Five-year overall survival for patients with advanced non-small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 study. J Clin Oncol. 2019;37:2518–27.
Hamid O, Robert C, Daud A, Hodi FS, Hwu WJ, Kefford R, et al. Five-year survival outcomes for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. Ann Oncol. 2019;30:582–8.
Wang DY, Salem J-E, Cohen JV, Chandra S, Menzer C, Ye F, et al. Fatal Toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4:1721–8.
Wang W, Wang E, Balthasar J. Monoclonal antibody pharmacokinetics and pharmacodynamics. Clin Pharmacol Ther. 2008;84:548–58.
Groot AJ, Mengesha A, van der Wall E, van Diest PJ, Theys J, Vooijs M. Functional antibodies produced by oncolytic clostridia. Biochem Biophys Res Commun. 2007;364:985–9.
Gurbatri CR, Lia I, Vincent R, Coker C, Castro S, Treuting PM, et al. Engineered probiotics for local tumor delivery of checkpoint blockade nanobodies. Sci Transl Med. 2020;12:eaax0876.
Winograd R, Byrne KT, Evans RA, Odorizzi PM, Meyer ARL, Bajor DL, et al. Induction of T-cell immunity overcomes complete resistance to PD-1 and CTLA-4 blockade and improves survival in pancreatic carcinoma. Cancer Immunol Res. 2015;3:399–411.
Chuntova P, Chow F, Watchmaker PB, Galvez M, Heimberger AB, Newell EW, et al. Unique challenges for glioblastoma immunotherapy-discussions across neuro-oncology and non-neuro-oncology experts in cancer immunology. Meeting Report from the 2019 SNO Immuno-Oncology Think Tank. Neuro Oncol. 2019;2021(23):356–75.
Nelson BE, Janku F, Fu S, Dumbrava EI, Hong DS, Karp D, et al. Abstract CT107: Phase Ib study of pembrolizumab in combination with intratumoral injection of Clostridium novyi-NT in patients with advanced solid tumors. Cancer Res. 2023;83:CT107.
Acknowledgements
Figures were created using Biorender.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. The authors acknowledge support from the Marsden Fund Council, managed by Royal Society Te Apārangi (MFP-UOA2204) and the Auckland Medical Research Foundation (1122007).
Conflicts of Interest
JT, AVP and AMM declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Ethics Approval
Not applicable.
Consent
Not applicable.
Author Contributions
Literature review and initial draft of manuscript was performed by AMM. Critical review and editing of manuscript was performed by JT, AVP and AMM. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
Not applicable to this article as no datasets were generated or analysed.
Code Availability
Not applicable.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Theys, J., Patterson, A.V. & Mowday, A.M. Clostridium Bacteria: Harnessing Tumour Necrosis for Targeted Gene Delivery. Mol Diagn Ther 28, 141–151 (2024). https://doi.org/10.1007/s40291-024-00695-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-024-00695-0