Abstract
Background
Methadone use and methadone-associated sudden cardiac death have increased dramatically. Prolongation of the QT interval of the cardiac cycle predisposes to arrhythmia and is common among methadone users.
Objective
We studied the relationship between pharmacogenetic variables and methadone metabolites and QT prolongation.
Methods
Blood was obtained on days 1, 7, and 21 from consenting individuals initiating methadone treatment. Plasma methadone and ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidene (EDDP) were measured using liquid chromatographic–electrospray ionization–tandem mass spectrometry. The corrected QT interval (QTc) from 12-lead electrocardiograms (ECGs) was obtained at baseline and at 21 days.
Results
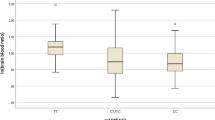
Total plasma EDDP, (S)-EDDP, and (R)-EDDP concentrations were significantly higher for individuals carrying the CYP2C19*2 variant (n = 8) than in 17 subjects carrying the CYP2C19 wild-type allele (p < 0.004). Seventeen (68 %) of 25 subjects experienced a QTc (Bazett) of 39.9 ± 28.4 ms (mean ± standard deviation). The methadone dose and the plasma EDDP concentration corrected for dose were both significantly associated with QTc at study termination and with change in QTc interval from baseline (∆QTc) (p < 0.03 to p < 0.0003). Based on a QTc increase, five subjects had a potentially increased arrhythmia risk. Compared with other participants, the mean dose for those individuals was higher (50.8 vs. 42.5 mg/day; p < 0.04) as was the total plasma EDDP (36.8 vs. 21.0 ng/mL; p < 0.002) and dose-corrected EDDP (0.27 vs. 0.16 ng/mL/mg; p < 0.003).
Conclusions
These results suggest that a notable change in the QTc interval was associated with both oral dose and increased methadone metabolism, as indicated by the higher plasma concentration of the principal methadone metabolite. The oral dose and plasma EDDP concentration may be useful in identifying individuals at risk for methadone-related arrhythmia.



Similar content being viewed by others
References
Zador D, Sunjic S. Deaths in methadone maintenance treatment in New South Wales, Australia 1990–1995. Addiction. 2000;95:77–84.
Wagner-Servais D, Erkens M. Methadone-related deaths associated with faulty induction procedures. J Maint Addic. 2003;2:57–67.
Krantz MJ, Lewkowiez L, Hays H, Woodroffe MA, Robertson AD, Mehler PS. Torsade de pointes associated with very-high-dose methadone. Ann Intern Med. 2002;137:501–4.
Fanoe S, Hvidt C, Ege P, Jensen GB. Syncope and QT prolongation among patients treated with methadone for heroin dependence in the city of Copenhagen. Heart. 2007;93:1051–5.
Maremmani I, Pacini M, Cesaroni C, Lovrecic M, Perugi G, Tagliamonte A. QTc interval prolongation in patients on long-term methadone maintenance therapy. Eur Addict Res. 2005;11:44–9.
Ehret GB, Voide C, Gex-Fabry M, Chabert J, Shah D, Broers B, et al. Drug-induced long QT syndrome in injection drug users receiving methadone: high frequency in hospitalized patients and risk factors. Arch Intern Med. 2006;166:1280–7.
Straus SM, Kors JA, De Bruin ML, van der Hooft CS, Hofman A, Heeringa J, et al. Prolonged QTc interval and risk of sudden cardiac death in a population of older adults. J Am Coll Cardiol. 2006;47:362–7.
Krantz MJ, Martin J, Stimmel B, Mehta D, Haigney MC. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387–95.
Eap CB, Crettol S, Rougier JS, Schlapfer J, Sintra Grilo L, Deglon JJ, et al. Stereoselective block of hERG channel by (S)-methadone and QT interval prolongation in CYP2B6 slow metabolizers. Clin Pharmacol Ther. 2007;81:719–28.
Chiang CE, Roden DM. The long QT syndromes: genetic basis and clinical implications. J Am Coll Cardiol. 2000;36:1–12.
Ansermot N, Albayrak O, Schlapfer J, Crettol S, Croquette-Krokar M, Bourquin M, et al. Substitution of (R, S)-methadone by (R)-methadone: impact on QTc interval. Arch Intern Med. 2010;170:529–36.
Eap CB, Buclin T, Baumann P. Interindividual variability of the clinical pharmacokinetics of methadone: implications for the treatment of opioid dependence. Clin Pharmacokinet. 2002;41:1153–93.
Atkinson D, Dunne A, Parker M. Torsades de pointes and self-terminating ventricular fibrillation in a prescription methadone user. Anaesthesia. 2007;62:952–5.
Peles E, Bodner G, Kreek MJ, Rados V, Adelson M. Corrected-QT intervals as related to methadone dose and serum level in methadone maintenance treatment (MMT) patients: a cross-sectional study. Addiction. 2007;102:289–300.
Pearson EC, Woosley RL. QT prolongation and torsades de pointes among methadone users: reports to the FDA spontaneous reporting system. Pharmacoepidemiol Drug Saf. 2005;14:747–53.
Kharasch ED, Walker A, Isoherranen N, Hoffer C, Sheffels P, Thummel K, et al. Influence of CYP3A5 genotype on the pharmacokinetics and pharmacodynamics of the cytochrome P4503A probes alfentanil and midazolam. Clin Pharmacol Ther. 2007;82:410–26.
Chang Y, Fang WB, Lin SN, Moody DE. Stereo-selective metabolism of methadone by human liver microsomes and cDNA-expressed cytochrome P450s: a reconciliation. Basic Clin Pharmacol Toxicol. 2011;108:55–62.
Totah RA, Allen KE, Sheffels P, Whittington D, Kharasch ED. Enantiomeric metabolic interactions and stereoselective human methadone metabolism. J Pharmacol Exp Ther. 2007;321:389–99.
Crettol S, Deglon JJ, Besson J, Croquette-Krokkar M, Gothuey I, Hammig R, et al. Methadone enantiomer plasma levels, CYP2B6, CYP2C19, and CYP2C9 genotypes, and response to treatment. Clin Pharmacol Ther. 2005;78:593–604.
Crettol S, Deglon JJ, Besson J, Croquette-Krokar M, Hammig R, Gothuey I, et al. ABCB1 and cytochrome P450 genotypes and phenotypes: influence on methadone plasma levels and response to treatment. Clin Pharmacol Ther. 2006;80:668–81.
He P, Court MH, Greenblatt DJ, von Moltke LL. Factors influencing midazolam hydroxylation activity in human liver microsomes. Drug Metab Dispos. 2006;34:1198–207.
Fonseca F, de la Torre R, Diaz L, Pastor A, Cuyas E, Pizarro N, et al. Contribution of cytochrome P450 and ABCB1 genetic variability on methadone pharmacokinetics, dose requirements, and response. PloS One. 2011;6:e19527.
Wang SC, Ho IK, Tsou HH, Liu SW, Hsiao CF, Chen CH, et al. Functional genetic polymorphisms in CYP2C19 Gene in relation to cardiac side effects and treatment dose in a methadone maintenance cohort. OMICS. 2013;17:519–26.
Pfeufer A, Sanna S, Arking DE, Muller M, Gateva V, Fuchsberger C, et al. Common variants at ten loci modulate the QT interval duration in the QTSCD study. Nat Genet. 2009;41:407–14.
Newton-Cheh C, Johnson T, Gateva V, Tobin MD, Bochud M, Coin L, et al. Genome-wide association study identifies eight loci associated with blood pressure. Nat Genet. 2009;41:666–76.
Moody DE. Metabolic and toxicological considerations of the opioid replacement therapy and analgesic drugs: methadone and buprenorphine. Expert Opin Drug Metab Toxicol. 2013;9:675–97.
Intermountain Health Care, Inc. Methadone monitoring for insights into adverse events (MEMORIES) [ClinicalTrials.gov identifier NCT01191242]. US National Institutes of Health, ClinicalTrials.gov. http://www.clinicaltrials.gov. Accessed 25 Mar 2015.
CredibleMeds®. Combined list of drugs that prolong QT and/or cause torsades de pointes (TDP). http://www.crediblemeds.org/pdftemp/pdf/CompositeList.pdf. Accessed 25 Mar 2015.
Acknowledgments
MEMORIES (ClinicalTrials.gov identifier NCT01191242) was funded in part by Grants from the Intermountain Healthcare Research and Medical Foundation and the Del Loy Hansen Heart Foundation. The authors would like to acknowledge Mark Morgan and Dawn Neimann and Discovery House Addiction Treatment Center for their invaluable contributions to the conduct of this study.
Conflict of interest
There are no financial relationships on the part of any author that could potentially bias the findings reported in the manuscript.
Author contributions
John F. Carlquist: Obtained funding, study design and analysis, manuscript preparation (guarantor of content). David E. Moody: Study design, metabolite analysis, manuscript preparation. Stacey Knight: Study design, statistical design, manuscript preparation. Eric G. Johnson: Study design and coordination. Wenfang B. Fang: Methadone and methadone metabolite analyses, manuscript preparation. John A. Huntinghouse: Designed and validated single nucleotide polymorphism (SNP) assays, manuscript preparation. Jeffrey S. Rollo: Designed and validated SNP assays, manuscript preparation. Lynn R. Webster: Study design, methadone maintenance treatment consultant, manuscript preparation. Jeffrey L. Anderson: Study design, ECG interpretation, manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carlquist, J.F., Moody, D.E., Knight, S. et al. A Possible Mechanistic Link Between the CYP2C19 Genotype, the Methadone Metabolite Ethylidene-1,5-Dimethyl-3,3-Diphenylpyrrolidene (EDDP), and Methadone-Induced Corrected QT Interval Prolongation in a Pilot Study. Mol Diagn Ther 19, 131–138 (2015). https://doi.org/10.1007/s40291-015-0137-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-015-0137-4