Abstract
Background
The use of compression garments (CGs) during or after training and competition has gained popularity in the last few decades. However, the data concerning CGs’ beneficial effects on muscle strength-related outcomes after physical exercise remain inconclusive.
Objective
The aim was to determine whether wearing CGs during or after physical exercise would facilitate the recovery of muscle strength-related outcomes.
Methods
A systematic literature search was conducted across five databases (PubMed, SPORTDiscus, Web of Science, Scopus, and EBSCOhost). Data from 19 randomized controlled trials (RCTs) including 350 healthy participants were extracted and meta-analytically computed. Weighted between-study standardized mean differences (SMDs) with respect to their standard errors (SEs) were aggregated and corrected for sample size to compute overall SMDs. The type of physical exercise, the body area and timing of CG application, and the time interval between the end of the exercise and subsequent testing were assessed.
Results
CGs produced no strength-sparing effects (SMD [95% confidence interval]) at the following time points (t) after physical exercise: immediately ≤ t < 24 h: − 0.02 (− 0.22 to 0.19), p = 0.87; 24 ≤ t < 48 h: − 0.00 (− 0.22 to 0.21), p = 0.98; 48 ≤ t < 72 h: − 0.03 (− 0.43 to 0.37), p = 0.87; 72 ≤ t < 96 h: 0.14 (− 0.21 to 0.49), p = 0.43; 96 h ≤ t: 0.26 (− 0.33 to 0.85), p = 0.38. The body area where the CG was applied had no strength-sparing effects. CGs revealed weak strength-sparing effects after plyometric exercise.
Conclusion
Meta-analytical evidence suggests that wearing a CG during or after training does not seem to facilitate the recovery of muscle strength following physical exercise. Practitioners, athletes, coaches, and trainers should reconsider the use of CG as a tool to reduce the effects of physical exercise on muscle strength.
Trial Registration Number
PROSPERO CRD42021246753.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The present systematic review is the first to use the generic inverse variance model to calculate standardized mean differences (SMDs) and, therefore, might estimate the real effects of compression garments (CGs) on muscle strength more accurately than SMD estimates used previously. |
This review revealed that wearing a CG during or after training appears not to facilitate muscle strength recovery following physical exercise. |
Practitioners, athletes, coaches, and therapists should reconsider the use of CG as an adjuvant to reduce the deleterious effects of physical exercise on muscle strength and seek alternative methods. |
1 Introduction
Athletes train intensively and compete on consecutive days. Competition success necessitates rapid recovery from the aftereffects of physical exercise and the circumvention of overreaching [1, 2]. To this aim, athletes use a variety of post-exercise recovery strategies including cryotherapy [3], low-intensity exercise [4], and tissue compression [5] in an attempt to reduce the immediate and lasting effects of physical exercise on muscle strength. Compression garments (CGs) are form-fitted elastic garments that mechanically compress the limb surface to stabilize or support the underlying tissue [6]. Mechanical pressure is also thought to promote tissue regeneration following exercise [7], and hasten post-exercise recovery by attenuating muscle damage-induced inflammation [8]. The garments presumably facilitate post-exercise recovery by reducing muscle fatigue [9] through improved venous return when worn during or after exercise [10, 11]. Acute muscle fatigue has been defined as a reversible exercise-induced reduction in muscle performance, irrespective of task completion [12]. Previously, it has been postulated that wearing CG during exercise may (1) reduce muscle oscillations [13], (2) decrease post-exercise blood lactate accumulation [10], and (3) improve spinal cord excitability [6]. More specifically, CG-related reductions in muscle oscillation may result in optimized neurotransmission and mechanics at the molecular level [14], which may reduce myoelectric activity [15]. In addition, there is evidence that compression increases venous blood flow, which helps to clear blood lactate from the muscle and subsequently increases tissue oxidation [16]. This hypothesis has been supported by the finding that during recovery, blood lactate disappearance and maximal oxygen consumption significantly correlated [10]. Moreover, a previous study [6] found a CG-related decrease in H-reflex amplitude, indicating altered spinal cord excitability via the presynaptic inhibition of Ia afferent transmission and/or via postsynaptic inhibition of motoneuron excitability [17, 18]. These mechanisms are also supported by findings of a recently published study [19] indicating improved resting markers of venous return and muscle blood flow in male basketball players in response to the application of a CG. Furthermore, the results of this study also suggest that a CG covering larger compared with smaller body areas appear to be more effective [19]. In sum, a recently published scoping review [20] concluded that while CGs are likely to improve muscle oscillatory properties, increase arterial blood flow and local skin temperature, facilitate signal transmission in the sensorimotor system, and may reduce perception of exercise-induced muscle soreness and pain, it is unlikely that CGs can meaningfully alter tissue metabolism, blood pressure, heart rate, and cardiorespiratory function.
In 2019, worldwide sales of CGs reached 5 million pieces with an overall value of $45 million [21]. Although a study showed that swimmers generally preferred active recovery over the use of CGs as the most effective recovery strategy [22], another study in 512 athletes showed that athletes perceived positive effects from wearing CGs [23]. Still, there is no consensus as to whether CGs can increase athletic performance [16, 24,25,26]. Experimental setup, participants’ training status, exercise modality, body area where the garment was applied, design, exposure duration, timing of wear (during or after exercise), and compression levels are factors, which individually or in combination contribute to the observed inconsistencies [16, 24, 27,28,29,30,31,32]. For example, although some experimental studies have demonstrated potential beneficial effects of wearing a CG during exercise [16, 27, 28], others did not confirm CGs’ performance-enhancing effects during exercise [24, 29, 30]. On the other hand, CG may hasten recovery when worn during the post-exercise regeneration period [31, 32]. The application of CGs after exercise reduced the decline in muscle power and strength [8, 10, 33], reduced the metabolites and concentrations of serum muscle damage markers [34, 35], and improved perceptual measures of recovery, i.e., muscle soreness, vitality, and readiness to train [11, 36]. Previous systematic reviews and meta-analyses suggest that CGs may aid faster recovery of exercise-induced muscle damage [5, 37, 38] by reducing inflammation [39]. However, whilst the use of CGs improved running economy, biomechanical variables (i.e., ground contact time, step frequency, step length, swing time), and perceived body temperature [40], garment wear was not associated with improved sports performance during high-intensity exercise [41].
In terms of the exercise modality, it seems that eccentric versus concentric training preferentially leads to exercise-induced muscle damage [42, 43], characterized by delayed-onset muscle soreness [44] and impaired muscle function [45, 46]. Eccentric exercise can be performed in a laboratory using an isokinetic dynamometer [47], or by plyometric training involving the stretch–shortening cycle, during which lengthening (eccentric) contractions are quickly followed by shortening (concentric) contractions [48, 49]. Eccentrically compared with concentrically biased or isometric exercise is characterized by cellular [50] and myofibrillar [51,52,53] disruption. Myofiber disruption may release protein-associated ions, resulting in increased osmotic pressure and muscle edema [54]. CGs may enhance muscle strength recovery following eccentric or plyometric exercise (PLYO) by attenuating the inflammation associated with muscle damage [55]. It has been hypothesized that tissue compression creates an external pressure gradient that reduces the space available for swelling to spread, whereby inflammation is reduced [56]. Indeed, a previous review suggested that CGs might have the greatest beneficial effects on strength recovery from resistance exercise (RES), including PLYO [5]. However, no likely recovery benefits from CGs were demonstrated following running [57] most likely due to the lower levels of muscle damage caused by running compared with RES [58,59,60]. In addition to exercise type, it is of interest to examine whether other factors may contribute to the efficacy of CGs. For instance, the preponderance of the previous studies using lower-limb CGs [33, 61,62,63,64,65,66] showed reduced exercise-induced decrements in maximal voluntary isometric torque (MVIC) after different types of exercise modalities. In contrast, studies applying upper-body [67] or whole-body [68] garments revealed zero or even detrimental effects on muscle strength recovery, respectively. Moreover, a previous meta-analysis [5] detected large, likely beneficial effects of CGs 24 h following physical exercise, indicating that the timing and duration of CG application may also contribute to the efficacy of CG. That is, lower-limb CG compared with upper-limb or combined application of the garment might be more effective to reduce exercise-induced decrements in muscle strength [33, 61,62,63,64,65,66], especially 24 h following physical exercise [5]. Although some researchers suggested that experimentally induced muscle fatigue affects the generation of mechanical work and power in lower-limb joints even during gait [69, 70], a previous study found no effects of hundreds of sit-to-stand trials on knee MVIC in healthy younger and older adults [71]. A previous review discussed that muscle fatigue induced by repetitive single-joint muscle contractions such as knee extension-flexion is most probably due to a localized force impairment, while when a multi-joint fatiguing protocol is used (e.g., sit-to-stand task, 6-min walk test), any adaptation in gait after the task is the result of a combined physiological and cognitive effect [72]. Therefore, it might be worth investigating whether the application of CG would influence the exercise-induced decrease in muscle strength differently according to the time interval between the end of the exercise and subsequent testing.
The use of CGs to facilitate recovery of exercise-induced muscle damage is supported by encouraging scientific evidence (for reviews, see [5, 37, 38]); however, paralleling the inconsistencies in study designs, the results are also contradictory concerning how, if at all, CG-induced tissue compression would affect the recovery of muscle strength after physical exercise. Therefore, the purpose of this systematic review with meta-analyses was to determine if wearing a CG during or after physical exercise would reduce the deleterious effects of physical exercise on muscle strength-related outcomes. Based on the extant data [8, 63, 73], we hypothesized that wearing a CG would facilitate the recovery of muscle strength after physical exercise, but this effect may differ according to (1) the type of physical exercise; (2) the body area where the CG is applied; (3) the timing of CG application, and/or (4) the time interval between the end of the exercise and subsequent testing. In contrast to previous reviews, which were limited in their statistical approach and entered the mean and the standard deviation (SD) of the percent changes into the model and used correlation coefficient estimates to calculate or impute the missing SDs for changes [74, 75], we applied the generic inverse variance method that minimizes the imprecision of the pooled effect estimate by adjusting the weight of each study according to the sample size [76]. Therefore, the method used in the current meta-analysis appears to be more reliable for calculating standardized mean differences (SMDs) and, therefore, might estimate the effects of CGs on muscle strength more accurately [77].
2 Methods
A systematic review with meta-analysis was conducted according to the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [78]. This study was registered with the PROSPERO database on May 5th, 2021 (ID: CRD42021246753).
2.1 Data Sources and Search Strategy
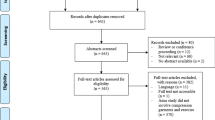
A systematic literature search was performed across five databases (PubMed, SPORTDiscus, Web of Science, Scopus, and EBSCOhost). A combination of keywords related to physical exercise, strength, training, exercise, rehabilitation, and compression were used with Boolean conjunctions AND, OR, and NOT. The detailed search strategy has been provided in Table 1. Publication time was not restricted, to ensure all relevant literature was identified for screening. Reference lists of all included studies were also screened to identify any further articles. The initial literature search was performed on April 12th, 2021 by two authors (JN and JH) and an alert for our syntax was installed in case of a relevant publication. No relevant publication was identified through this alert between April and August 2021. The selection process started with the removal of duplicate studies. Titles and abstracts of publications obtained by the search strategy were screened. Only peer-reviewed parallel and crossover randomized controlled trials (RCTs) written in English were classified as relevant. Based on the information within the full text, the inclusion and exclusion criteria were applied to select the trials eligible for inclusion in the review and meta-analysis. In the case of disagreement regarding study eligibility, co-authors UG, TH, and RN were consulted for clarification. Conference papers and case studies were excluded, as were reviews, but their references were manually screened to ensure all appropriate citations were also considered for inclusion.
2.2 Eligibility Criteria
A PICOS (Participants, Intervention, Comparators, Outcomes, and Study design) approach was used to rate studies for eligibility [79]. The respective inclusion/exclusion criteria are reported in Table 2. Briefly, both randomized crossover and parallel RCTs met the inclusion criteria if participants were healthy adults, irrespective of gender, training status, or age. With respect to the intervention, studies were included that compared CG with a no-CG or sham-CG control during or after physical exercise. Means and SDs of measures of isometric, isokinetic, or isotonic muscle strength had to be reported for all groups before and after intervention. Considering that the overall aim of the study was to determine if wearing a CG during or after physical exercise would reduce the deleterious effects of physical exercise on muscle strength-related outcomes, we only included studies that observed decrements in strength-related outcomes in the control (no-CG or sham-CG) condition. Exclusion criteria were (1) no physical exercise, (2) use of braces or orthoses, (3) measures other than strength, (4) control group showed no reduction in strength-related outcome after exercise, and (5) no control condition.
2.3 Data Extraction
Pre- and post-test means and SDs were extracted from studies that assessed the effects of CG compared to a control condition (e.g., sham) on muscle strength recovery after physical exercise. In some cases [8, 80, 81], the mean and SD data were extrapolated from the figures using a data extraction software (http://getdata-graph-digitizer.com/) [82]. In addition, we contacted authors to request the exact mean and SD values when they only reported the changes from baseline values in the article. When authors did not respond, the study was excluded from the meta-analyses due to the lack of crucial data. Using Cochrane decision rules, only one of multiple interventions or one of multiple strength outcomes were included in the analyses. For instance, MVIC of the quadriceps was preferred over MVIC of the hamstrings or plantar flexors for the sake of homogeneity.
2.4 Risk of Bias Assessment
Risk of bias was calculated in accordance with the Cochrane Collaboration Guidelines [83]. Systematic differences (heterogeneity) were assessed using an I2 statistic, which indicates the percentage of variability across studies due to heterogeneity [79]. Low, moderate, and high heterogeneity correspond to I2 outcomes of 25%, 50%, and 75%, respectively [84]. A value above 75% is rated as being considerably heterogeneous [76].
2.5 Statistical Analyses
To examine the effects of CG on strength-related outcomes during or after physical exercise, weighted between-study SMDs were computed for post-test mean values and corrected for sample size according to Hedges and Olkin [76] using the following equation:
where m1 is the mean post-value of the intervention group, m2 is the mean post-value of the control group, SDp is the pooled SD, and N represents the total sample size.
The inverse variance method is so named because the weight given to each study is chosen to be the inverse of the variance of the effect estimate (i.e., 1 over the square of its standard error [SE]). Thus, larger studies, which have smaller SEs, are given more weight than smaller studies, which have larger SEs. This choice of weights minimizes the imprecision (uncertainty) of the pooled effect estimate (see chapter 10.3 [76]).
Statistical analyses were carried out using Review Manager 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, 2011). All data were analyzed using a random-effect model. To examine whether wearing CG during or after physical exercise would reduce exercise-induced decrements in muscle strength-related outcomes, SMDs were weighted with respect to their SEs and aggregated to compute the overall SMDs, which are presented with 95% confidence intervals. Overall SMDs were set at < 0.2 = trivial, 0.2–0.49 = small, 0.5–0.79 = moderate, and ≥ 0.8 = large [85]. The χ2 was used to determine whether the differences in the results are due by chance, and in such a case, a low p value, or high χ2 statistic, relative to degrees of freedom would be apparent [76]. To assess the impact of different meta-analytic methods, a sensitivity analysis was completed. The significance level was set at p ≤ 0.05.
2.6 Subgroup Analyses
The Cochrane Handbook for Systematic Reviews of Interventions suggests a minimum of two data points to aggregate study findings [76]. If at least two studies were available, subgroup analyses were computed for the type of physical exercise. Studies were categorized with respect to whether they applied PLYO versus lab-based eccentric exercise protocols (ECC) versus RES versus running exercise protocols (RUN) versus combined physical exercise protocols (COMB), including sport-specific training or the combination of PLYO and RUN. Further subgroup analyses were computed with regard to the body area, where the CG was applied using the following categories: arm/upper body versus leg/lower body versus upper + lower body. Finally, subgroup analyses were also performed on the timing of CG application with the following categories: during exercise versus after exercise for ≤ 12 h versus after exercise for ≤ 48 h versus after exercise for ≤ 72 h. Two studies [68, 80] were excluded from this particular subgroup analysis because they applied the CG during and after the physical exercise in the same study cohort, which would bias the results of the analysis. To assess the relative benefits of CG on strength-related outcomes in relation to the time point (t) of subsequent testing, separated meta-analyses were performed for those taken at (a) immediately ≤ t < 24 h, (b) 24 ≤ t < 48 h, (c) 48 ≤ t < 72 h, (d) 72 ≤ t < 96 h, and (e) 96 h ≤ t.
3 Results
3.1 Search Results
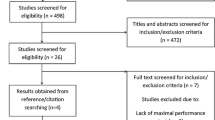
An overview of the search process is displayed in Fig. 1. Initially, 910 studies were identified. After removing duplicates, 803 records remained. We screened out 688 records based on titles and abstracts. From the remaining 115 records, 95 were excluded during the eligibility check based on the a priori defined exclusion criteria (Table 2). Two additional studies [81, 86] were identified through reference list searches; therefore, a total of 22 studies were included, from which 13 were parallel RCTs (n = 283 participants) [8, 9, 28, 33, 61, 66, 81, 87,88,89,90,91,92], while the remaining nine were crossover RCTs (n = 117) [62, 65, 67, 68, 73, 80, 86, 93, 94]. Three studies (n = 50) [73, 89, 93] were excluded from the meta-analysis due to insufficient data reporting.
3.2 Sample Characteristics
The training status of the participants in the studies included in the meta-analysis ranged from untrained adults to professional athletes (e.g., elite judoka [94], marathon runners [92], handball players from the professional French National Handball League [65]). The total number of participants in the data set was 350 (n = 251 men and n = 99 women), with a mean and SD age of 24.5 ± 5.5 years, height of 174.4 ± 5.3 cm, and body mass of 73.1 ± 7.5 kg. Two studies [90, 94] did not report participants’ body height, from which Pearce et al. [90] did not report the age and body mass of participants. Eleven studies solely enrolled male participants, two studies solely enrolled female participants, and six studies included both, males and females.
Participants performed the following physical exercise protocols: PLYO (four RCTs, n = 97), ECC (seven RCTs, n = 121), RES (three RCTs, n = 39), RUN (one RCT, n = 24), or COMB (four RCTs, n = 69). The garments were applied to the arm/upper body (six RCTs, n = 88), leg/lower body (ten RCTs, n = 217), or both on the upper and lower body (three RCTs, n = 38) during (seven RCTs, n = 116) or after exercise for ≤ 12 h (five RCTs, n = 205), ≤ 48 h (one RCT, n = 29), or ≤ 72 h (four RCTs, n = 106). CGs were used during and after physical exercise (two RCTs, n = 21). Strength outcomes included the following: MVIC of the quadriceps, hamstrings, elbow flexors, or plantar flexors; maximal concentric force/torque of quadriceps, hamstrings, or elbow flexors; grip strength; or bench/chest press as strength-related outcomes (Table 3).
Five studies [9, 63, 68, 80, 81] administered multiple interventions or measured multiple strength outcomes; therefore, we had to choose and include one representative outcome in the meta-analyses. In the case of three studies, MVIC of the quadriceps was preferred over MVIC of the hamstrings [68, 80] or plantar flexors [81]. The results of moderate-velocity (60°/s) plantar flexor torque were extracted and used in the meta-analyses. In addition, the CG providing 18 mmHg pressure at the ankle was chosen in the study of Miyamoto et al. [9] for the sake of homogeneity. Finally, because most of the included studies measured MVIC at 90° of knee extension, we extracted the MVIC data of 80° instead of 60° from the study of Négyesi et al. [63].
The outcome of the risk of bias assessment revealed that sequence generation and allocation concealment were largely unclear, with only one study [66] reporting how participants were allocated to groups and one study with a high risk of selection bias (Fig. 2; Supplementary Fig. 1, see the electronic supplementary material).
Risk of bias percentile chart in accordance with the Cochrane Collaboration [83]. Horizontal axis denotes percent of studies
3.3 The Overall Effects of CG
Forests plots depict the meta-analytical comparisons of the overall effects of CG at different time points (Fig. 3a–e). Three additional RCTs were included in the qualitative analysis only [73, 89, 93]. After physical exercise, CG versus no CG revealed no effects on muscle strength at any time point: (a) immediately ≤ t < 24 h: SMD = − 0.02 (− 0.22 to 0.19), p = 0.87 (Fig. 3a); (b) 24 ≤ t < 48 h: SMD = − 0.00 (− 0.22 to 0.21), p = 0.98 (Fig. 3b); (c) 48 ≤ t < 72 h: SMD = − 0.03 (− 0.43 to 0.37), p = 0.87 (Fig. 3c); (d) 72 ≤ t < 96 h: SMD = 0.14 (− 0.21 to 0.49), p = 0.43 (Fig. 3d); (e) 96 h ≤ t: SMD = 0.26 (− 0.33 to 0.85), p = 0.38 (Fig. 3e). Values for I2 ranged between 0 and 49% and indicated low heterogeneity.
Effect of CG versus a control condition on measures of strength after physical exercise at each time point: a immediately ≤ t < 24 h; b 24 ≤ t < 48 h; c 48 ≤ t < 72 h; d 72 ≤ t < 96 h; e 96 h ≤ t. CG compression garment, CI confidence interval, df degrees of freedom, IV inverse variance, SE standard error
3.4 Subgroup Analyses
Table 4 shows time-point subgroup analyses. Considering that only one study applied RUN [92], we have excluded it from the subgroup analysis for the type of physical exercise. There was a small and unlikely beneficial dose–response effect of CG after PLYO (SMD = 0.42 [− 0.03 to 0.87], p = 0.07, three RCTs) (Supplementary Fig. 2, see the electronic supplementary material). Moreover, only one study [86] out of three applied the CG during exercise; therefore, this result does not represent the effects of CG on muscle strength. CGs applied during or after PLYO resulted in (all p > 0.05) small and not very likely effects on muscle strength at other time points (24 ≤ t < 48 h: SMD = 0.37 [− 0.43 to 1.17], p = 0.37, three studies; 48 ≤ t < 72 h: SMD = 0.44 [− 0.72 to 1.61], p = 0.46, three studies; 72 ≤ t < 96 h: SMD = 0.49 [− 0.21 to 1.18], p = 0.17, three studies) compared to other physical exercise types where non-significant trivial effects were observed (Supplementary Figs. 3–5).
The body area where the CG was applied did not affect strength-related outcomes after exercise (Supplementary Figs. 6–10). The timing and duration of CG application also had no effects on the exercise-induced decrements in muscle strength (Supplementary Figs. 11–15). In all subgroup analyses, heterogeneity was low to moderate (I2 ≤ 53%).
4 Discussion
Contrary to the hypothesis, meta-analytical evidence suggests that wearing a CG during or after training does not seem to facilitate the recovery of muscle strength after physical exercise. We discuss the overall and subgroup analyses with a perspective on the use of CGs to reduce physical exercise-induced strength loss.
4.1 Overall Effects of CGs on Muscle Strength
We hypothesized that wearing CGs would facilitate the recovery of muscle strength from physical exercise. Contrary to this expectation, the meta-analytical comparisons between CG versus control revealed no muscle strength-sparing effects from physical exercise. This somewhat unexpected result is not in line with findings of previous reviews [5, 37, 39] that reported beneficial effects of CGs on muscle strength and recovery of muscle function after exercise. The inconsistencies might be due to differences in the applied meta-analytical methods between this current and previous meta-analyses.
To our knowledge, the present systematic review is the first that used the generic inverse variance method. Previous reviews entered the mean and SD of the percent change into the model. When an RCT reported insufficient information, change-from-baseline SDs for the same outcome measure from other studies in the review can be used to calculate the SDs for the changes [95]. Another option is to calculate or impute the missing SDs for changes using a correlation coefficient [74, 75]. However, one can never be certain that an imputed correlation coefficient is accurate. On the other hand, the generic inverse variance method minimizes the imprecision of the pooled effect estimate by adjusting the weight of each study according to the sample size, as described in the Cochrane guidelines for meta-analyses [76]. Therefore, the method used in the meta-analyses of the present review appears to be more reliable for calculating SMDs and, might, therefore, estimate the real effects of CGs on muscle strength more accurately. However, for this method, pre and post mean and SD data are needed, which are often unreported. We received the necessary data for 19 RCTs (n = 350) (Fig. 1, Table 3), but not for others (three RCTs, n = 50) [73, 89, 93], which were excluded from the meta-analysis.
The second possible reason for the inconsistent results between the present and previous reviews might be related to the multiple imputations of the same RCT within one meta-analysis. Multiple outcome domains from single RCTs that are likely to be not independent or one single outcome at different time points included multiple times can also create bias. In such a case, all outcomes extracted from one study or subgroup must be combined to calculate one effect size using standard procedures [96, 97]. Nevertheless, measuring the impact of treatment across a broad array of measures through the calculation of an average effect across measures within an RCT may not accurately represent the effectiveness of this study’s intervention [97]. Previous reviews that aimed to identify the effects of CGs on muscle strength and muscle function after physical exercise included multiple outcomes or one outcome at different time points from one study, which most likely biased the results of the meta-analysis. We, therefore, only used the most relevant outcomes according to Cochrane decision rules and performed separate meta-analyses for each time point to objectively determine the effects of CG on muscle strength. Sensitivity analyses confirmed the current results (Supplementary Fig. 16, see the electronic supplementary material).
4.2 The Effects of CG Application During or After Different Exercise Types on Strength
An eccentric contraction-biased exercise i.e., PLYO or ECC, is associated with myofibrillar disruption and damage [50,51,52]. CGs may have the potential to reduce the increased osmotic pressure and the resultant edema in response to muscle damage arising from PLYO or ECC. This is achieved by producing external pressure to oppose the internal pressure created by the edematous fluid that could in turn reduce inflammation [54]. Thus, CGs may facilitate muscle recovery following eccentrically biased exercises. This is in line with a previous review that suggested the largest benefits resulting from CGs are for strength recovery from RES, including PLYO [5]. Yet, according to previous studies, CGs have little to no effects on exercise performance during prolonged exercise, including running [92, 98], hiking [99], or even repeated sprints [24, 36, 80, 100].
The extant data gave rise to the hypothesis that the nature of strength recovery may differ according to the type of physical exercise so that CGs may spare decline in muscle strength after exercise. CGs produced no strength-sparing effects after any type of exercise, surprisingly not even after PLYO (Supplementary Figs. 2–5, see the electronic supplementary material), against the observation reported by a previous systematic review with meta-analysis [5]. Variation between PLYO RCTs in the magnitude of strength recovery might negate any potential CG effects. Quadriceps MVIC recovered at an accelerated rate over 72 h post-PLYO with the post-exercise application of a high-compression CG to the thigh (14.8 ± 2.2 mmHg) and calf (24.3 ± 3.7 mmHg) for 72 h [61]. In line with these data, CG worn for 12 h after PLYO also reduced the physical exercise-induced decrements of quadriceps MVIC as compared to a passive recovery group [33]. In contrast, CG did not reduce physical exercise-induced strength loss after exhaustive PLYO [81, 86]. The interpretation of these studies is complicated by considering the area of the leg where the CGs were applied (thigh vs. calf), implying the effects of anthropometric differences, which we were unable to systematically examine.
Quantitative analyses revealed no muscle strength-sparing effects of CGs after other exercise types. Notably, only one study that met the inclusion criteria [92] applied RUN; therefore, we could not include it in the subgroup analysis. Nevertheless, this study also reported no effects of lower-limb CG on MVIC when worn for 72 h after the marathon run. Qualitatively, the ‘beneficial’ effects of CGs on quadriceps MVIC after a 3 × 5.2-km trail run [93] or RES [73] were found to be in contrast with the no effects of compression tights or socks worn during 2 h uphill running on quadriceps or plantar flexor MVIC [89].
4.3 Effects of the Body Area and Timing of CG Application on Muscle Strength
We found no effects of body area and timing of CG application on muscle strength after physical exercise (Supplementary Figs. 11–15, see the electronic supplementary material). Based on the preponderance of the involved RCTs showing positive effects of lower-limb CGs on muscle strength recovery after PLYO [33, 61], ECC [62, 63], or even COMB [65, 66], we expected that lower-limb CGs compared with upper-limb or combined application of the garment might be more effective to reduce physical exercise-induced decrements in MVIC. However, when meta-analyzing the data with the generic inverse variance method, we found that CGs produced minimal strength-sparing effects. These effects were also independent of the body area where CGs were applied (Table 4). The high risk of heterogeneity at time points ≥ 48 h in most of the subgroups is perhaps related to differences in participants’ training status and exercise characteristics. While in one RCT, healthy young men performed 30 repetitions of eccentric actions with weighted dumbbells [88], others used an isokinetic dynamometer and examined its effect on muscle strength recovery in (1) non-trained women following two sets of 50 arm curls at 60° s−1 [8] or (2) healthy men following 35 maximal isokinetic eccentric extensions of the elbow flexors at 90° s−1 [90]. Nevertheless, only one of these RCTs reported beneficial effect of upper-limb CG on MVIC of elbow flexors. Overall, subgroup analyses revealed heterogeneity, preventing us from detecting any meaningful effects of the body area of CG application on muscle strength.
Regarding the timing and duration of CG application, a previous review detected large, likely beneficial effects of CG at 24 h following physical exercise, regardless of training status [5]. When worn immediately after the physical exercise is terminated, CG may have the potential to enhance metabolite clearance and reduce swelling and the perception of muscle soreness [101]. However, our analysis did not confirm this possible favorable post-exercise effect (Supplementary Figs. 11–15). The pressure exerted locally by a CG in the studies reviewed here ranged between 8.1 and 35 mmHg. However, most studies failed to report the level of pressure [62, 63, 67, 68, 88, 90, 91] or provided it as reported by the manufacturer or previous researches [9, 33, 65, 80, 81, 86], instead of measuring pressure directly [8, 61, 66, 87, 92, 94]. Although a previous review of studies with both trained and untrained participants reported no relationship between cuff pressure and CG-produced effects, differences in cuff pressure may explain the inconsistent results between studies [57]. Besides the limited data, the subgroup showed a moderate heterogeneity (I2 = 53%), which might be related to differences in the exercise modality (PLYO vs. ECC), the body area of CG application (arm vs. leg), and the outcome measure (elbow flexor vs. quadriceps MVIC).
4.4 Limitations and Future Direction
Based on the risk of bias assessment, the overall methodological quality of the included studies is limited, which is why more high-quality studies are needed in this area in the future.
Contrary to the hypothesis, our meta-analytical evidence suggests that wearing CGs during or after exercise does not seem to facilitate the recovery of muscle strength from physical exercise; this could be related to the low pressures (< 35 mmHg) exerted by the CG. Directly measuring and consistently reporting CG-induced pressure can help to elucidate the potential underlying mechanisms responsible for strength recovery benefits. Moreover, the levels of pressure applied by the garment may vary due to anthropometric differences of participants within and between studies. Therefore, future studies should carefully measure and report participants’ anthropometric characteristics. Finally, participant training status was not consistent across the included studies, as most enrolled healthy untrained individuals. Therefore, there is a need to examine the effects of CGs on strength recovery in highly trained athletes. Overall, future studies should clarify the potential benefits of CGs in relation to applied pressures, participants’ anthropometric characteristics, and training status so that the optimal conditions for CG use can be determined.
5 Conclusions
This systematic review with meta-analytical evidence suggests that wearing a CG during or after exercise does not seem to facilitate the recovery of muscle strength from physical exercise even when the type of exercise or the body area and timing of CG application is taken into account. Practitioners, athletes, coaches, and therapists should reconsider the use of CG as an adjuvant to reduce physical exercise-induced decrements in muscle strength and seek methods alternative to CG.
References
Bahnert A, Norton K, Lock P. Association between post-game recovery protocols, physical and perceived recovery, and performance in elite Australian Football League players. J Sci Med Sport. 2013;16(2):151–6.
Halson SL, Bridge MW, Meeusen R, Busschaert B, Gleeson M, Jones DA, et al. Time course of performance changes and fatigue markers during intensified training in trained cyclists. J Appl Physiol (1985). 2002;93(3):947–56.
Ferreira-Junior JB, Bottaro M, Vieira CA, Soares SR, Vieira A, Cleto VA, et al. Effects of partial-body cryotherapy (-110°C) on muscle recovery between high-intensity exercise bouts. Int J Sports Med. 2014;35(14):1155–60.
Suzuki M, Umeda T, Nakaji S, Shimoyama T, Mashiko T, Sugawara K. Effect of incorporating low intensity exercise into the recovery period after a rugby match. Br J Sports Med. 2004;38(4):436.
Brown F, Gissane C, Howatson G, van Someren K, Pedlar C, Hill J. Compression garments and recovery from exercise: a meta-analysis. Sports Med. 2017;47(11):2245–67.
Barss TS, Pearcey GEP, Munro B, Bishop JL, Zehr EP. Effects of a compression garment on sensory feedback transmission in the human upper limb. J Neurophysiol. 2018;120(1):186–95.
Trenell MI, Rooney KB, Sue CM, Thomspon CH. Compression garments and recovery from eccentric exercise: a (31)P-MRS study. J Sports Sci Med. 2006;5(1):106–14.
Kraemer WJ, Bush JA, Wickham RB, Denegar CR, Gómez AL, Gotshalk LA, et al. Influence of compression therapy on symptoms following soft tissue injury from maximal eccentric exercise. J Orthop Sports Phys Ther. 2001;31(6):282–90.
Miyamoto N, Hirata K, Mitsukawa N, Yanai T, Kawakami Y. Effect of pressure intensity of graduated elastic compression stocking on muscle fatigue following calf-raise exercise. J Electromyogr Kinesiol. 2011;21(2):249–54.
Chatard JC, Atlaoui D, Farjanel J, Louisy F, Rastel D, Guézennec CY. Elastic stockings, performance and leg pain recovery in 63-year-old sportsmen. Eur J Appl Physiol. 2004;93(3):347–52.
Pruscino CL, Halson S, Hargreaves M. Effects of compression garments on recovery following intermittent exercise. Eur J Appl Physiol. 2013;113(6):1585–96.
Bigland-Ritchie B, Woods JJ. Changes in muscle contractile properties and neural control during human muscular fatigue. Muscle Nerve. 1984;7(9):691–9.
Bringard A, Perrey S, Belluye N. Aerobic energy cost and sensation responses during submaximal running exercise–positive effects of wearing compression tights. Int J Sports Med. 2006;27(5):373–8.
MacIntosh BRGPFMAJ. Skeletal muscle: form and function. 2006.
Nigg BM, Wakeling JM. Impact forces and muscle tuning: a new paradigm. Exerc Sport Sci Rev. 2001;29(1):37–41.
Berry MJ, McMurray RG. Effects of graduated compression stockings on blood lactate following an exhaustive bout of exercise. Am J Phys Med. 1987;66(3):121–32.
Knikou M. The H-reflex as a probe: pathways and pitfalls. J Neurosci Methods. 2008;171(1):1–12.
Zehr EP. Considerations for use of the Hoffmann reflex in exercise studies. Eur J Appl Physiol. 2002;86(6):455–68.
O'Riordan SF, McGregor R, Halson SL, Bishop DJ, Broatch JR. Sports compression garments improve resting markers of venous return and muscle blood flow in male basketball players. J Sport Health Sci. 2021.
Weakley J, Broatch J, O'Riordan S, Morrison M, Maniar N, Halson SL. Putting the squeeze on compression garments: current evidence and recommendations for future research: a systematic scoping review. Sports Med. 2021.
Compression Garments Market—Global Outlook and Forecast 2020–2025. 2020.
Shell SJ, Slattery K, Clark B, Broatch JR, Halson S, Kellmann M, et al. Perceptions and use of recovery strategies: do swimmers and coaches believe they are effective? J Sports Sci. 2020;38(18):2092–9.
Franke TPC, Backx FJG, Huisstede BMA. Lower extremity compression garments use by athletes: why, how often, and perceived benefit. BMC Sports Sci Med Rehabil. 2021;13(1):31.
MacRae BA, Cotter JD, Laing RM. Compression garments and exercise: garment considerations, physiology and performance. Sports Med. 2011;41(10):815–43.
Kraemer WJ, Bush JA, Bauer JA, Triplett-McBride NT, Paxton NJ, Clemson A, et al. Influence of compression garments on vertical jump performance in NCAA division I volleyball players. J Strength Cond Res. 1996;10(3):180–3.
Kraemer WJ, Bush JA, Newton RU, Duncan ND, Volek JS, Denegar CR, et al. Influence of a compression garment on repetitive power output production before and after different types of muscle fatigue. Sports Med Train Rehabil. 1998;8(2):163–84.
Scanlan AT, Dascombe BJ, Reaburn PR, Osborne M. The effects of wearing lower-body compression garments during endurance cycling. Int J Sports Physiol Perform. 2008;3(4):424–38.
Négyesi J, Zhang LY, Jin RN, Hortobágyi T, Nagatomi R. A below-knee compression garment reduces fatigue-induced strength loss but not knee joint position sense errors. Eur J Appl Physiol. 2021;121(1):219–29.
Burden RJ, Glaister M. The effects of ionized and nonionized compression garments on sprint and endurance cycling. J Strength Cond Res. 2012;26(10):2837–43.
Faulkner JA, Gleadon D, McLaren J, Jakeman JR. Effect of lower-limb compression clothing on 400-m sprint performance. J Strength Cond Res. 2013;27(3):669–76.
de Glanville KM, Hamlin MJ. Positive effect of lower body compression garments on subsequent 40-kM cycling time trial performance. J Strength Cond Res. 2012;26(2):480–6.
Hamlin MJ, Mitchell CJ, Ward FD, Draper N, Shearman JP, Kimber NE. Effect of compression garments on short-term recovery of repeated sprint and 3-km running performance in rugby union players. J Strength Cond Res. 2012;26(11):2975–82.
Jakeman JR, Byrne C, Eston RG. Lower limb compression garment improves recovery from exercise-induced muscle damage in young, active females. Eur J Appl Physiol. 2010;109(6):1137–44.
Gill ND, Beaven CM, Cook C. Effectiveness of post-match recovery strategies in rugby players. Br J Sports Med. 2006;40(3):260–3.
Kraemer WJ, Flanagan SD, Comstock BA, Fragala MS, Earp JE, Dunn-Lewis C, et al. Effects of a whole body compression garment on markers of recovery after a heavy resistance workout in men and women. J Strength Cond Res. 2010;24(3):804–14.
Duffield R, Portus M. Comparison of three types of full-body compression garments on throwing and repeat-sprint performance in cricket players. Br J Sports Med. 2007;41(7):409–14 (discussion 14).
Hill J, Howatson G, van Someren K, Leeder J, Pedlar C. Compression garments and recovery from exercise-induced muscle damage: a meta-analysis. Br J Sports Med. 2014;48(18):1340–6.
Mota GR, Simim MAM, Dos Santos IA, Sasaki JE, Marocolo M. Effects of wearing compression stockings on exercise performance and associated indicators: a systematic review. Open Access J Sports Med. 2020;11:29–42.
Marqués-Jiménez D, Calleja-González J, Arratibel I, Delextrat A, Terrados N. Are compression garments effective for the recovery of exercise-induced muscle damage? A systematic review with meta-analysis. Physiol Behav. 2016;1(153):133–48.
Engel FA, Holmberg HC, Sperlich B. Is there evidence that runners can benefit from wearing compression clothing? Sports Med. 2016;46(12):1939–52.
da Silva CA, Helal L, da Silva RP, Belli KC, Umpierre D, Stein R. Association of lower limb compression garments during high-intensity exercise with performance and physiological responses: a systematic review and meta-analysis. Sports Med. 2018;48(8):1859–73.
Clarkson PM, Sayers SP. Etiology of exercise-induced muscle damage. Can J Appl Physiol. 1999;24(3):234–48.
Enoka RM. Eccentric contractions require unique activation strategies by the nervous system. J Appl Physiol (1985). 1996;81(6):2339–46.
Impellizzeri FM, Rampinini E, Castagna C, Martino F, Fiorini S, Wisloff U. Effect of plyometric training on sand versus grass on muscle soreness and jumping and sprinting ability in soccer players. Br J Sports Med. 2008;42(1):42–6.
Jakeman JR, Macrae R, Eston R. A single 10-min bout of cold-water immersion therapy after strenuous plyometric exercise has no beneficial effect on recovery from the symptoms of exercise-induced muscle damage. Ergonomics. 2009;52(4):456–60.
Nosaka K, Clarkson PM. Muscle damage following repeated bouts of high force eccentric exercise. Med Sci Sports Exerc. 1995;27(9):1263–9.
Kellis E, Baltzopoulos V. Isokinetic eccentric exercise. Sports Med. 1995;19(3):202–22.
Davies G, Riemann BL, Manske R. Current concepts of plyometric exercise. Int J Sports Phys Ther. 2015;10(6):760–86.
Komi PV. Stretch-shortening cycle: a powerful model to study normal and fatigued muscle. J Biomech. 2000;33(10):1197–206.
Croisier JL, Camus G, Deby-Dupont G, Bertrand F, Lhermerout C, Crielaard JM, et al. Myocellular enzyme leakage, polymorphonuclear neutrophil activation and delayed onset muscle soreness induced by isokinetic eccentric exercise. Arch Physiol Biochem. 1996;104(3):322–9.
Newham DJ, McPhail G, Mills KR, Edwards RH. Ultrastructural changes after concentric and eccentric contractions of human muscle. J Neurol Sci. 1983;61(1):109–22.
McHugh MP. Recent advances in the understanding of the repeated bout effect: the protective effect against muscle damage from a single bout of eccentric exercise. Scand J Med Sci Sports. 2003;13(2):88–97.
Hortobágyi T, Houmard J, Fraser D, Dudek R, Lambert J, Tracy J. Normal forces and myofibrillar disruption after repeated eccentric exercise. J Appl Physiol (1985). 1998;84(2):492–8.
Stauber WT, Clarkson PM, Fritz VK, Evans WJ. Extracellular matrix disruption and pain after eccentric muscle action. J Appl Physiol (1985). 1990;69(3):868–74.
Kraemer W, French D, Spiering BA. Compression in the treatment of acute muscle injuries in sport: review article. Int Sportmed J. 2004;5:200–8.
Davies V, Thompson KG, Cooper SM. The effects of compression garments on recovery. J Strength Cond Res. 2009;23(6):1786–94.
Beliard S, Chauveau M, Moscatiello T, Cros F, Ecarnot F, Becker F. Compression garments and exercise: no influence of pressure applied. J Sports Sci Med. 2014;14(1):75–83.
Baird MF, Graham SM, Baker JS, Bickerstaff GF. Creatine-kinase- and exercise-related muscle damage implications for muscle performance and recovery. J Nutr Metab. 2012;2012: 960363.
Paschalis V, Koutedakis Y, Jamurtas AZ, Mougios V, Baltzopoulos V. Equal volumes of high and low intensity of eccentric exercise in relation to muscle damage and performance. J Strength Cond Res. 2005;19(1):184–8.
Howatson G, van Someren KA. The prevention and treatment of exercise-induced muscle damage. Sports Med. 2008;38(6):483–503.
Hill J, Howatson G, van Someren K, Gaze D, Legg H, Lineham J, et al. The effects of compression-garment pressure on recovery after strenuous exercise. Int J Sports Physiol Perform. 2017;12(8):1078–84.
Shimokochi Y, Kuwano S, Yamaguchi T, Abutani H, Shima N. Effects of wearing a compression garment during night sleep on recovery from high-intensity eccentric-concentric quadriceps muscle fatigue. J Strength Cond Res. 2017;31(10):2816–24.
Negyesi J, Zhang LY, Jin RN, Hortobagyi T, Nagatomi R. A below-knee compression garment reduces fatigue-induced strength loss but not knee joint position sense errors. Eur J Appl Physiol. 2021;121(1):219–29.
Struhár I, Kumstát M, Králová DM. Effect of compression garments on physiological responses after uphill running. J Hum Kinet. 2018;61:119–29.
Ravier G, Bouzigon R, Beliard S, Tordi N, Grappe F. Benefits of compression garments worn during handball-specific circuit on short-term fatigue in professional players. J Strength Cond Res. 2018;32(12):3519–27.
Brown F, Jeffries O, Gissane C, Howatson G, van Someren K, Pedlar C, et al. Custom-fitted compression garments enhance recovery from muscle damage in rugby players. J Strength Cond Res. 2020.
Martorelli SS, Martorelli AS, Pereira MC, Rocha-Junior VA, Tan JG, Alvarenga JG, et al. Graduated compression sleeves: effects on metabolic removal and neuromuscular performance. J Strength Cond Res. 2015;29(5):1273–8.
Chan V, Duffield R, Watsford M. The effects of compression garments on performance of prolonged manual-labour exercise and recovery. Appl Physiol Nutr Metab. 2016;41(2):125–32.
Barbieri FA, Gobbi LT, Lee YJ, Pijnappels M, van Dieën JH. Effect of triceps surae and quadriceps muscle fatigue on the mechanics of landing in stepping down in ongoing gait. Ergonomics. 2014;57(6):934–42.
Riazati S, Caplan N, Matabuena M, Hayes PR. Fatigue induced changes in muscle strength and gait following two different intensity, energy expenditure matched runs. Front Bioeng Biotechnol. 2020.
Dos Santos PCR, Lamoth CJC, Gobbi LTB, Zijdewind I, Barbieri FA, Hortobágyi T. Older compared with younger adults performed 467 fewer sit-to-stand trials, accompanied by small changes in muscle activation and voluntary force. Front Aging Neurosci. 2021;13: 679282.
Santos PCR, Barbieri FA, Zijdewind I, Gobbi LTB, Lamoth C, Hortobágyi T. Effects of experimentally induced fatigue on healthy older adults’ gait: A systematic review. PLoS ONE. 2020;14(12): e0226939.
Goto K, Morishima T. Compression garment promotes muscular strength recovery after resistance exercise. Med Sci Sports Exerc. 2014;46(12):2265–70.
Abrams KR, Gillies CL, Lambert PC. Meta-analysis of heterogeneously reported trials assessing change from baseline. Stat Med. 2005;24(24):3823–44.
Follmann D, Elliott P, Suh I, Cutler J. Variance imputation for overviews of clinical trials with continuous response. J Clin Epidemiol. 1992;45(7):769–73.
Deeks JJ, Higgins JP, Altman DG, Group obotCSM. Analysing data and undertaking meta-analyses. Cochrane Handbook for Systematic Reviews of Interventions. 2019. p. 241–84.
Faraone SV. Interpreting estimates of treatment effects: implications for managed care. P T. 2008;33(12):700–11.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9, W64.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–34.
Duffield R, Cannon J, King M. The effects of compression garments on recovery of muscle performance following high-intensity sprint and plyometric exercise. J Sci Med Sport. 2010;13(1):136–40.
Terbizan DJ, Barry AS, Stone KA, Talaski JL, Bennett T, Hackney KJ. Compression garments do not improve recovery from plypmetric exercise. Mo J Health, Physical Education, Recreation, and Dance Addendum. 2018;28:1–11.
Wojtyniak J-G, Britz H, Selzer D, Schwab M, Lehr T. Data digitizing: accurate and precise data extraction for quantitative systems pharmacology and physiologically-based pharmacokinetic modeling. CPT Pharmacomet Syst Pharmacol. 2020;9(6):322–31.
Higgins JP, Savović J, Page MJ, Elbers RG, Sterne JA. Assessing risk of bias in a randomized trial. Cochrane Handbook for Systematic Reviews of Interventions. 2019. p. 205–28.
Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. 2011.
Cohen J. Statistical power analysis for the behavioral-sciences. Percept Motor Skill. 1988;67(3):1007.
Šambaher N, Aboodarda SJ, Silvey DB, Button DC, Behm DG. Effect of an ankle compression garment on fatigue and performance. J Strength Cond Res. 2016;30(2):326–35.
Carling J, Francis K, Lorish C. The effects of continuous external compression on delayed-onset muscle soreness (DOMS). Int J Rehabil Health. 1995;1(4):223–35.
Cerqueira MS, Santos Borges L, dos Santos Rocha JA, Brito Andrade H, Silva Pirôpo U, Lupato Conrado LA, et al. Twelve hours of a compression sleeve is not enough to improve the muscle recovery of an exercise-damaged upper arm. Apunts Medicina de l'Esport. 2015;50(185):23–8.
Mizuno S, Arai M, Todoko F, Yamada E, Goto K. Wearing compression tights on the thigh during prolonged running attenuated exercise-induced increase in muscle damage marker in blood. Front Physiol. 2017.
Pearce AJ, Kidgell DJ, Grikepelis LA, Carlson JS. Wearing a sports compression garment on the performance of visuomotor tracking following eccentric exercise: a pilot study. J Sci Med Sport. 2009;12(4):500–2.
Pereira MC, Bottaro M, Brown LE, Rocha-Junior VA, Martorelli SS, Nóbrega OT, et al. Do compression sleeves worn during exercise affect muscle recovery? Isokinet Exerc Sci. 2014;22:265–71.
Hill JA, Howatson G, van Someren KA, Walshe I, Pedlar CR. Influence of compression garments on recovery after marathon running. J Strength Cond Res. 2014;28(8):2228–35.
Bieuzen F, Brisswalter J, Easthope C, Vercruyssen F, Bernard T, Hausswirth C. Effect of wearing compression stockings on recovery after mild exercise-induced muscle damage. Int J Sports Physiol Perform. 2014;9(2):256–64.
Brown FCW, Hill JA, van Someren K, Howatson G, Pedlar CR. The effect of custom-fitted compression garments worn overnight for recovery from judo training in elite athletes. Eur J Sport Sci. 2021;14:1–9.
Higgins JP, Li T, Deeks JJ. Choosing effect measures and computing estimates of effect. Cochrane Handbook for Systematic Reviews of Interventions. 2019. p. 143–76.
Borenstein M, Hedges LV, Higgins JP, Rothstein HR. Introduction to meta-analysis. Wiley; 2021.
Scammacca N, Roberts G, Stuebing KK. Meta-analysis with complex research designs: dealing with dependence from multiple measures and multiple group comparisons. Rev Educ Res. 2014;84(3):328–64.
Vercruyssen F, Easthope C, Bernard T, Hausswirth C, Bieuzen F, Gruet M, et al. The influence of wearing compression stockings on performance indicators and physiological responses following a prolonged trail running exercise. Eur J Sport Sci. 2014;14(2):144–50.
McDonnell AC, Cooper D, Mlinar T, Mekjavic IB. The effect of post-exercise application of either graduated or uniform compression socks on the mitigation of delayed onset muscle soreness. Text Res J. 2019;89(9):1792–806.
Doan BK, Kwon YH, Newton RU, Shim J, Popper EM, Rogers RA, et al. Evaluation of a lower-body compression garment. J Sports Sci. 2003;21(8):601–10.
Born DP, Sperlich B, Holmberg HC. Bringing light into the dark: effects of compression clothing on performance and recovery. Int J Sports Physiol Perform. 2013;8(1):4–18.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
JN is supported by JSPS KAKENHI (Grant Number: 21K17600).
Conflict of interest
János Négyesi, Tibor Hortobágyi, Jessica Hill, Urs Granacher and Ryoichi Nagatomi declare that they have no conflict of interest to be disclosed.
Availability of data and material
The datasets used and/or analyzed during the current study are presented within the article and/or additional supporting files, and are also available from the corresponding author on reasonable request.
Author contributions
JN, TH, and RN developed the research question and concept. JN and JH performed the search. JN and JH performed risk of bias assessment. JN performed data extraction. JN performed data analyses. All authors contributed to drafting of manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Négyesi, J., Hortobágyi, T., Hill, J. et al. Can Compression Garments Reduce the Deleterious Effects of Physical Exercise on Muscle Strength? A Systematic Review and Meta-Analyses. Sports Med 52, 2159–2175 (2022). https://doi.org/10.1007/s40279-022-01681-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-022-01681-4